New Benzo[c]phenanthridine and Benzenoid Derivatives, and Other Constituents from Zanthoxylum ailanthoides: Effects on Neutrophil Pro-Inflammatory Responses
Abstract
:1. Introduction
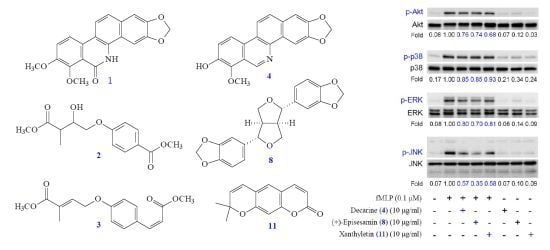
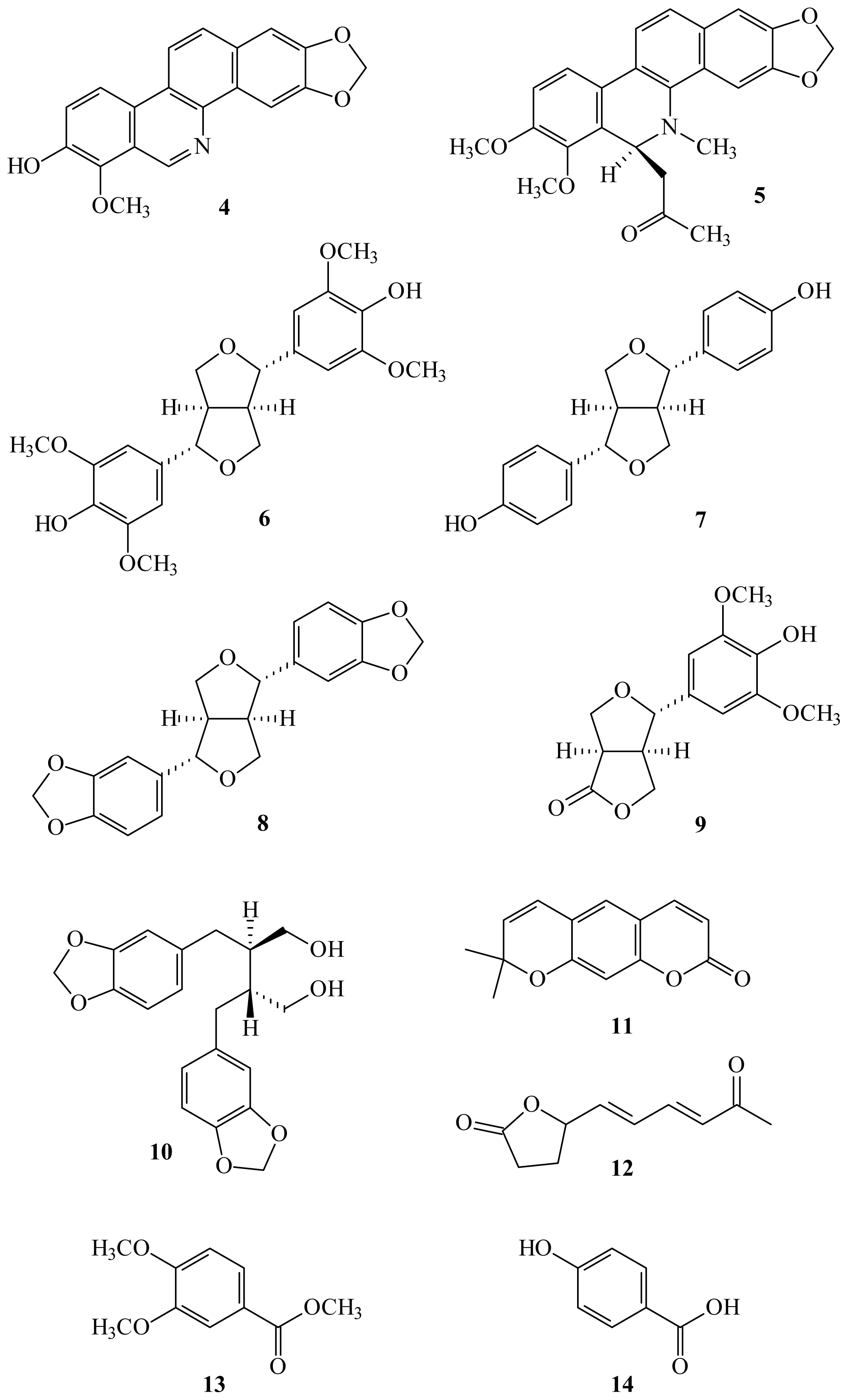
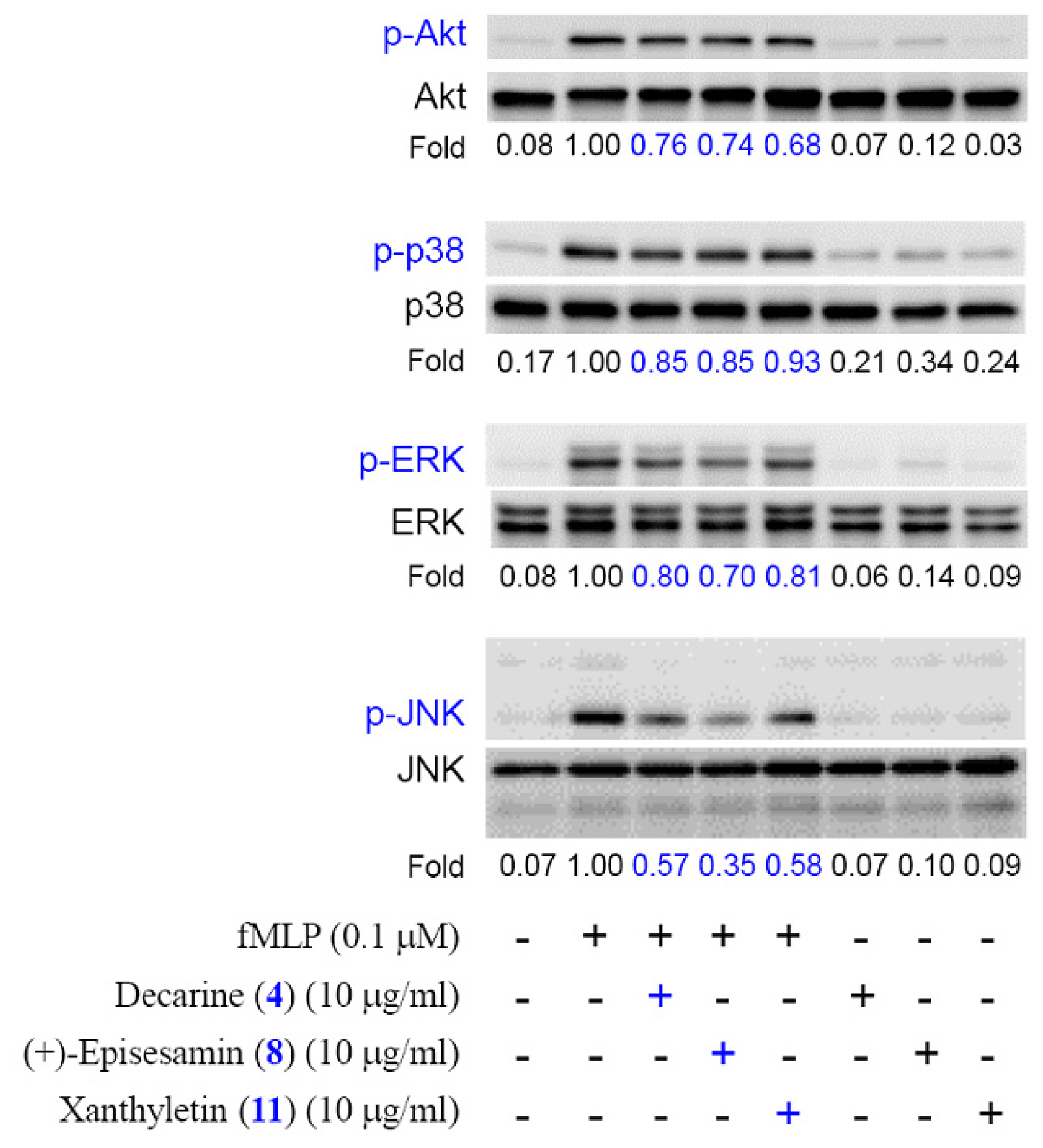
2. Results and Discussion
3. Experimental Section
3.1. General Experimental Procedures
3.2. Plant Material
3.3. Extraction and Isolation
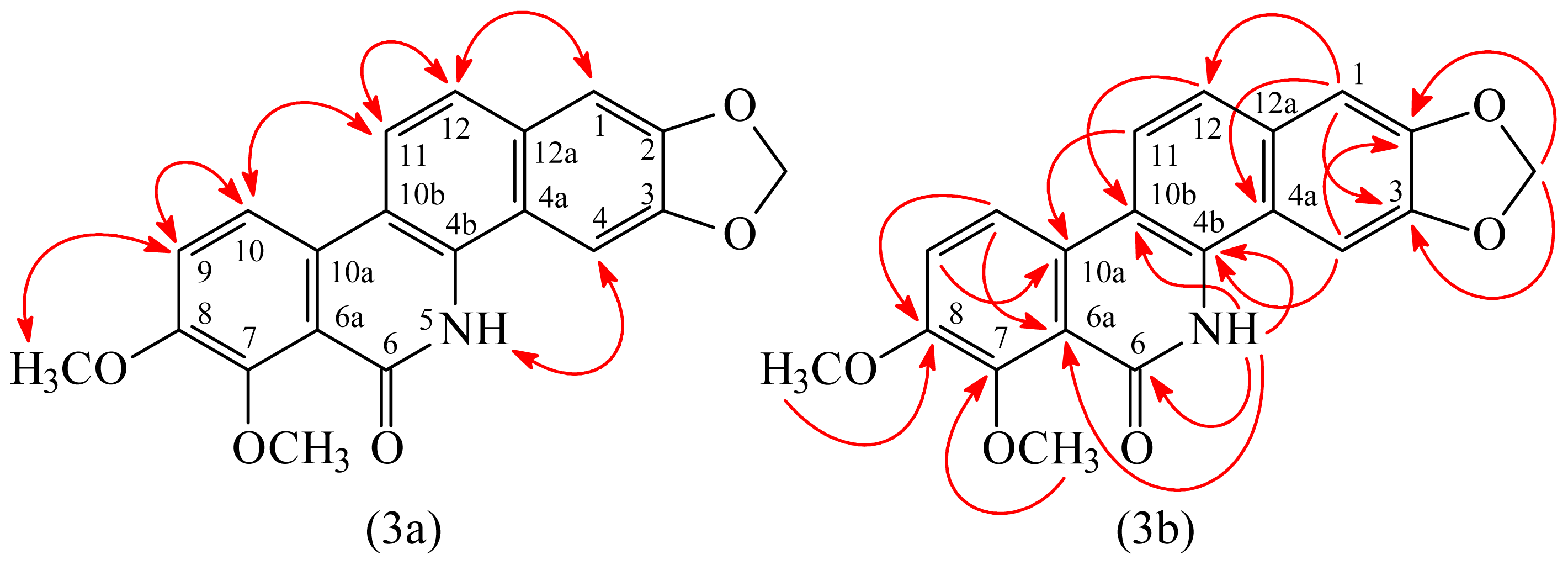
3.3.1. Oxynorchelerythrine (1)
3.3.2. Methyl 4-(2-Hydroxy-4-methoxy-3-methyl-4-oxobutoxy)benzoate (2)
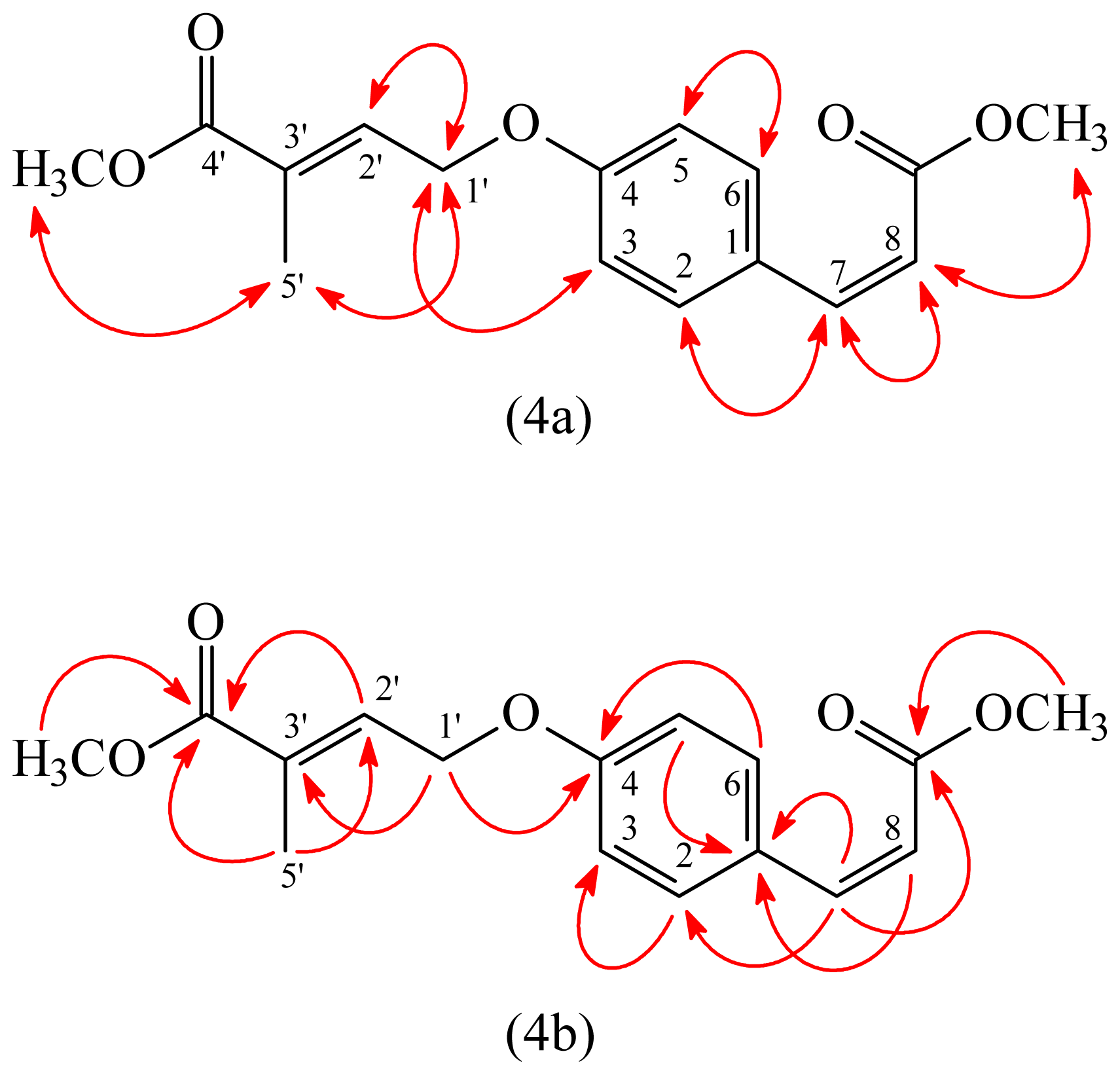
3.3.3. (E)-Methyl 4-(4-((Z)-3-methoxy-3-oxoprop-1-enyl)phenoxy)-2-methylbut-2-enoate (3)
3.4. Biological Assay
3.4.1. Preparation of Human Neutrophils
3.4.2. Measurement of Superoxide Anion Generation
3.4.3. Measurement of Elastase Release
3.4.4. Western Analysis
3.4.5. Statistical Analysis
4. Conclusions
Supplementary Information
ijms-14-22395-s001.pdf
| Position | δC | δH | NOESY | HMBC a |
|---|---|---|---|---|
| 1 | 123.1 | |||
| 2 | 131.6 | 7.99 (d, J = 9.0) | 3, MeOCO-1 | 3, 4, 6, MeOCO-1 |
| 3 | 114.1 | 6.93 (d, J = 9.0) | 2, 1′ | 1, 4, 5 |
| 4 | 162.1 | |||
| 5 | 114.1 | 6.93 (d, J = 9.0) | 6, 1′ | 1, 3, 4 |
| 6 | 131.6 | 7.99 (d, J = 9.0) | 5, MeOCO-1 | 2, 4, 5, MeOCO-1 |
| 1′ | 69.8 | 4.11 (m) | 3, 5, 3′, 5′ | 4, 2′, 3′ |
| 2′ | 71.8 | 4.11 (m) | 3′, 5′, OH-2′ | 1′, 4′, 5′ |
| 3′ | 42.0 | 2.88 (m) | 2′, 5′ | 1′, 2′, 4′ |
| 4′ | 175.7 | |||
| 5′ | 14.1 | 1.30 (d, J = 7.0) | 1′, 2′, 3′, OMe-4′ | 2′, 3′, 4′ |
| MeOCO-1 | 51.9 | 3.89 (s) | 2, 6 | MeOCO-1 |
| MeOCO-1 | 166.7 | |||
| OH-2′ | 3.09 (br s) | 2′ | ||
| OMe-4′ | 52.0 | 3.74 (s) | 5′ | 4′ |
| Compounds | Superoxide anion | Elastase |
|---|---|---|
| IC50 [μg/mL] b or (Inh %) c | ||
| Oxynorchelerythrine (1) | (29.69 ± 1.29) g | (20.28 ± 5.20) f |
| Methyl 4-(2-hydroxy-4-methoxy-3-methyl-4-oxobutoxy)benzoate (2) | (19.46 ± 4.19) f | (8.32 ± 2.49) e |
| (E)-methyl 4-(4-((Z)-3-methoxy-3-oxoprop-1-enyl)phenoxy)-2-methylbut-2-enoate (3) | (33.42 ± 4.53) f | (24.15 ± 3.22) e |
| Decarine (4) | 1.31 ± 0.18 g | 1.95 ± 0.28 g |
| 6-Acetonyldihydrochelerythrine (5) | (48.36 ± 4.85) f | 7.12 ± 0.31 e |
| (−)-Syringaresinol (6) | 4.79 ± 0.39 g | (7.66 ± 3.71) |
| 5′,5″-Didemethoxypinoresinol (7) | (45.22 ± 3.31) g | (23.91 ± 5.75) e |
| (+)-Episesamin (8) | 4.33 ± 0.56 g | 1.42 ± 0.16 g |
| Glaberide I (9) | 3.98 ± 0.44 g | (23.00 ± 2.92) f |
| (−)-Dihydrocubebin (10) | 2.42 ± 0.47 f | (32.78 ± 4.94) f |
| Xanthyletin (11) | 4.16 ± 0.35 g | 5.48 ± 0.27 g |
| Lanyulactone (12) | (36.03 ± 5.00) f | (34.55 ± 6.14) f |
| Methyl 3,4-dimethoxybenzoate (13) | (40.21 ± 6.27) e | (29.96 ± 6.18) e |
| p-Hydroxybenzoic acid (14) | (17.30 ± 9.77) g | (32.79 ± 1.48) g |
| LY294002 d | 1.14 ± 0.12 f | 1.94 ± 0.23 f |
Acknowledgments
Conflicts of Interest
References
- Chang, C.E.; Hartley, T.G. Rutaceae. In Flora of Taiwan, 2nd ed; Huang, T.C., Ed.; Editorial Committee of the Flora of Taiwan: Taipei, Taiwan, 1993; Volume 3, pp. 510–544. [Google Scholar]
- Morita, N.; Shimizu, M. Studies on the medical resources. XXV. The components in the root bark of Zanthoxylum ailanthoides Engl. Yakugaku Zasshi 1966, 86, 732–734. [Google Scholar]
- Morita, N.; Fukuta, M. Studies on medicinal resources. XXVII. Components of Fagara plants (Rutaceae). Yakugaku Zasshi 1967, 87, 319–320. [Google Scholar]
- Nakaoki, T.; Morita, N. Constituents of the fruits of Fagara species of Japan. Yakugaku Zasshi 1953, 73, 770–771. [Google Scholar]
- Tomita, M.; Ishii, H. Alkaloids of Rutaceaous plants. IV. Alkaloids ofXanthoxylum ailanthoides. Yakugaku Zasshi 1958, 78, 1441–1443. [Google Scholar]
- Ishii, H.; Ishikawa, T.; Mihara, M.; Akaike, M. The chemical constituents of Xantoxylum ailanthoides. Yakugaku Zasshi 1983, 103, 279–292. [Google Scholar]
- Aburano, S.; Kurono, G.; Morimoto, M.; Nishikawa, Y. Studies on fatty acids from fruit and seed oils. 3. The fatty acid composition of some species of Rutaceae plants. Yakugaku Zasshi 1972, 92, 1298–1299. [Google Scholar]
- Yasuda, I.; Takeya, K.; Itokawa, H. Two new pungent principles isolated from the pericarps of Zanthoxylum ailanthoides. Chem. Pharm. Bull 1981, 29, 1791–1793. [Google Scholar]
- Cheng, M.J.; Tsai, I.L.; Chen, I.S. Chemical constituents from the root bark of Formosan Zanthoxylum ailanthoides. J. Chin. Chem. Soc 2003, 50, 1241–1246. [Google Scholar]
- Sheen, W.S.; Tsai, I.L.; Teng, C.M.; Chen, I.S. Nor-neolignan and phenyl propanoid from Zanthoxylum ailanthoides. Phytochemistry 1994, 36, 213–215. [Google Scholar]
- Cheng, M.J.; Lee, K.H.; Tsai, I.L.; Chen, I.S. Two new sesquiterpenoids and anti-HIV principles from the root bark of Zanthoxylum ailanthoides. Bioorg. Med. Chem 2005, 13, 5915–5920. [Google Scholar]
- Chen, J.J.; Chung, C.Y.; Hwang, T.L.; Chen, J.F. Amides and benzenoids from Zanthoxylum ailanthoides with inhibitory activity on superoxide generation and elastase release by neutrophils. J. Nat. Prod 2009, 72, 107–111. [Google Scholar]
- Hanaoka, M.; Motonishi, T.; Mukai, C. Chemical transformation of protoberberines. Part 9. A biomimetic synthesis of oxychelerythrine, dihydrochelerythrine, and chelerythrine from berberine. J. Chem. Soc., Perkin Trans. 1 1986, 2253–2256. [Google Scholar]
- Lü, P.; Huang, K.L.; Xie, L.G.; Xu, X.H. Palladium-catalyzed tandem reaction to construct benzo[c]phenanthridine: application to the total synthesis of benzo[c]phenanthridine alkaloids. Org. Biomol. Chem 2011, 9, 3133–3135. [Google Scholar]
- Zhu, C.; Wang, R.; Falck, J.R. Mild and rapid hydroxylation of aryl/heteroaryl boronic acids and boronate esters with N-oxides. Org. Lett 2012, 14, 3494–3497. [Google Scholar]
- Vaquette, J.; Pousset, J.L.; Paris, R.R.; Cavé, A. Alcaloïdes de Zanthoxylum decaryi: la decarine, nouvel alcaloide dérivé de la benzophénanthridine. Phytochemistry 1974, 13, 1257–1259. [Google Scholar]
- Chen, J.J.; Lin, Y.H.; Day, S.H.; Hwang, T.L.; Chen, I.S. New benzenoids and anti-inflammatory constituents from Zanthoxylum nitidum. Food Chem 2011, 125, 282–287. [Google Scholar]
- Chou, T.H.; Chen, I.S.; Peng, C.F.; Sung, P.J.; Chen, J.J. A new dihydroagarofuranoid sesquiterpene and antituberculosis constituents from the root of Microtropis japonica. Chem. Biodivers 2008, 5, 1412–1418. [Google Scholar]
- Kobayashi, M.; Ohta, Y. Induction of stress metabolite formation in suspension cultures of Vigna angularis. Phytochemistry 1983, 22, 1257–1261. [Google Scholar]
- Kinjo, J.; Higuchi, H.; Fukui, K.; Nohara, T. Lignoids from Albizziae Cortex II. A biodegradation pathway of syringaresinol. Chem. Pharm. Bull 1991, 39, 2952–2955. [Google Scholar]
- Tillekeratne, L.M.V.; Jayamanne, D.T.; Weerasuria, K.D.V.; Gunatilaka, A.A.L. Lignans of Horsfieldia iryaghedhi. Phytochemistry 1982, 21, 476–478. [Google Scholar]
- Tsai, I.L.; Lin, W.Y.; Huang, M.W.; Chen, T.L.; Chen, I.S. N-Isobutylamides and butyrolactone from Zanthoxylum integrifoliolum. Helv. Chim. Acta 2001, 84, 830–833. [Google Scholar]
- Narasimhan, B.; Ohlan, S.; Ohlan, R.; Judge, V.; Narang, R. Hansch analysis of veratric acid derivatives as antimicrobial agents. Eur. J. Med. Chem 2009, 44, 689–700. [Google Scholar]
- Tan, J.; Bednarek, P.; Liu, J.; Schneider, B.; Svatoš, A.; Hahlbrock, K. Universally occurring phenylpropanoid and species-specific indolic metabolites in infected and uninfected Arabidopsis thaliana roots and leaves. Phytochemistry 2004, 65, 691–699. [Google Scholar]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Invest 2000, 80, 617–653. [Google Scholar]
- Borregaard, N. The human neutrophil. Function and dysfunction. Eur. J. Haematol 1998, 41, 401–413. [Google Scholar]
- Roos, D.; van Bruggen, R.; Meischl, C. Oxidative killing of microbes by neutrophils. Microbes Infect 2003, 5, 1307–1315. [Google Scholar]
- Faurschou, M.; Borregaard, N. Neutrophil granules and secretory vesicles in inflammation. Microbes Infect 2003, 5, 1317–1327. [Google Scholar]
- Chen, Q.; Powell, D.W.; Rane, M.J.; Singh, S.; Butt, W.; Klein, J.B.; McLeish, K.R. Akt phosphorylates p47phox and mediates respiratory burst activity in human neutrophils. J. Immunol 2003, 170, 5302–5308. [Google Scholar]
- Sadhu, C.; Dick, K.; Tino, W.T.; Staunton, D.E. Selective role of PI3K delta in neutrophil inflammatory responses. Biochem. Biophys. Res. Commun 2003, 308, 764–769. [Google Scholar]
- Yang, S.C.; Chung, P.J.; Ho, C.M.; Kuo, C.Y.; Hung, M.F.; Huang, Y.T.; Chang, W.Y.; Chang, Y.W.; Chan, K.H.; Hwang, T.L. Propofol inhibits superoxide production, elastase release, and chemotaxis in formyl peptide-activated human neutrophils by blocking formyl peptide receptor 1. J. Immunol 2013, 190, 6511–6519. [Google Scholar]
- Boyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J. Clin. Lab. Invest 1968, 97, 77–89. [Google Scholar]
- Jauregui, H.O.; Hayner, N.T.; Driscoll, J.L.; Williams-Holland, R.; Lipsky, M.H.; Galletti, P.M. Trypan blue dye uptake and lactate dehydrogenase in adult rat hepatocytes-freshly isolated cells, cell suspensions, and primary monolayer cultures. In Vitro 1981, 17, 1100–1110. [Google Scholar]
- Babior, B.M.; Kipnes, R.S.; Curnutte, J.T. Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J. Clin. Invest 1973, 52, 741–744. [Google Scholar]
- Hwang, T.L.; Leu, Y.L.; Kao, S.H.; Tang, M.C.; Chang, H.L. Viscolin, a new chalcone from Viscum coloratum, inhibits human neutrophil superoxide anion and elastase release via a cAMP-dependent pathway. Free Radic. Biol. Med 2006, 41, 1433–1441. [Google Scholar]
- Gilbert, C.; Rollet-Labelle, E.; Caon, A.C.; Naccache, P.H. Immunoblotting and sequential lysis protocols for the analysis of tyrosine phosphorylation-dependent signaling. J. Immunol. Methods 2002, 271, 185–201. [Google Scholar]
- Yang, S.C.; Lin, C.F.; Chang, W.Y.; Kuo, J.; Huang, Y.T.; Chung, P.J.; Hwang, T.L. Bioactive secondary metabolites of a marine Bacillus sp. inhibit superoxide generation and elastase release in human neutrophils by blocking formyl peptide receptor 1. Molecules 2013, 18, 6455–6468. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chung, C.-Y.; Hwang, T.-L.; Kuo, L.-M.; Kuo, W.-L.; Cheng, M.-J.; Wu, Y.-H.; Sung, P.-J.; Chung, M.-I.; Chen, J.-J. New Benzo[c]phenanthridine and Benzenoid Derivatives, and Other Constituents from Zanthoxylum ailanthoides: Effects on Neutrophil Pro-Inflammatory Responses. Int. J. Mol. Sci. 2013, 14, 22395-22408. https://doi.org/10.3390/ijms141122395
Chung C-Y, Hwang T-L, Kuo L-M, Kuo W-L, Cheng M-J, Wu Y-H, Sung P-J, Chung M-I, Chen J-J. New Benzo[c]phenanthridine and Benzenoid Derivatives, and Other Constituents from Zanthoxylum ailanthoides: Effects on Neutrophil Pro-Inflammatory Responses. International Journal of Molecular Sciences. 2013; 14(11):22395-22408. https://doi.org/10.3390/ijms141122395
Chicago/Turabian StyleChung, Ching-Yi, Tsong-Long Hwang, Liang-Mou Kuo, Wen-Lung Kuo, Ming-Jen Cheng, Yi-Hsiu Wu, Ping-Jyun Sung, Mei-Ing Chung, and Jih-Jung Chen. 2013. "New Benzo[c]phenanthridine and Benzenoid Derivatives, and Other Constituents from Zanthoxylum ailanthoides: Effects on Neutrophil Pro-Inflammatory Responses" International Journal of Molecular Sciences 14, no. 11: 22395-22408. https://doi.org/10.3390/ijms141122395
APA StyleChung, C.-Y., Hwang, T.-L., Kuo, L.-M., Kuo, W.-L., Cheng, M.-J., Wu, Y.-H., Sung, P.-J., Chung, M.-I., & Chen, J.-J. (2013). New Benzo[c]phenanthridine and Benzenoid Derivatives, and Other Constituents from Zanthoxylum ailanthoides: Effects on Neutrophil Pro-Inflammatory Responses. International Journal of Molecular Sciences, 14(11), 22395-22408. https://doi.org/10.3390/ijms141122395