The Effect of Different Intensities of Treadmill Exercise on Cognitive Function Deficit Following a Severe Controlled Cortical Impact in Rats
Abstract
:1. Introduction
2. Results and Discussion
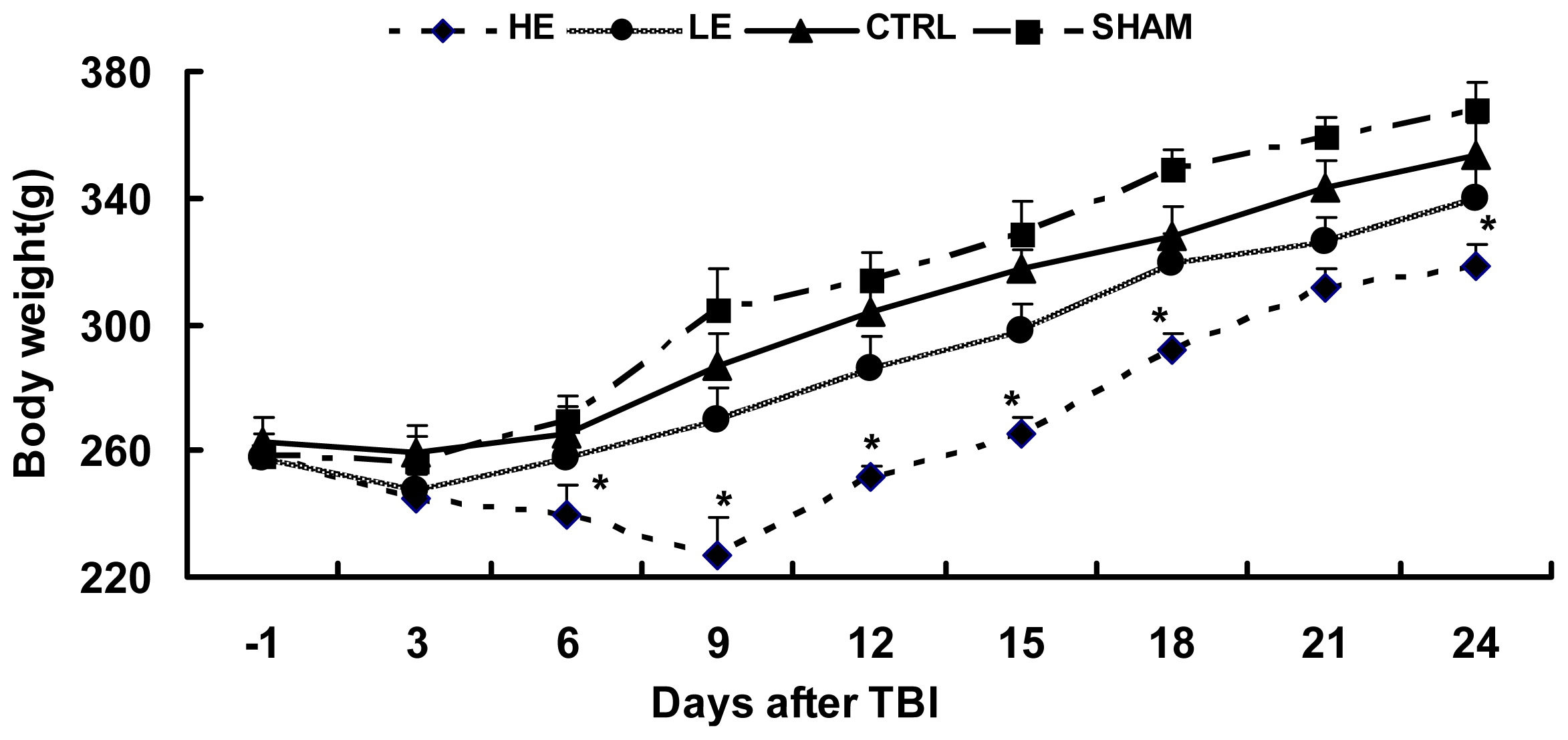
2.1. Effects of Treadmill Exercise on Weight Loss after CCI
2.2. Neurologic Deficit Scores in Each Group
2.3. Spatial Learning and Memory Performance
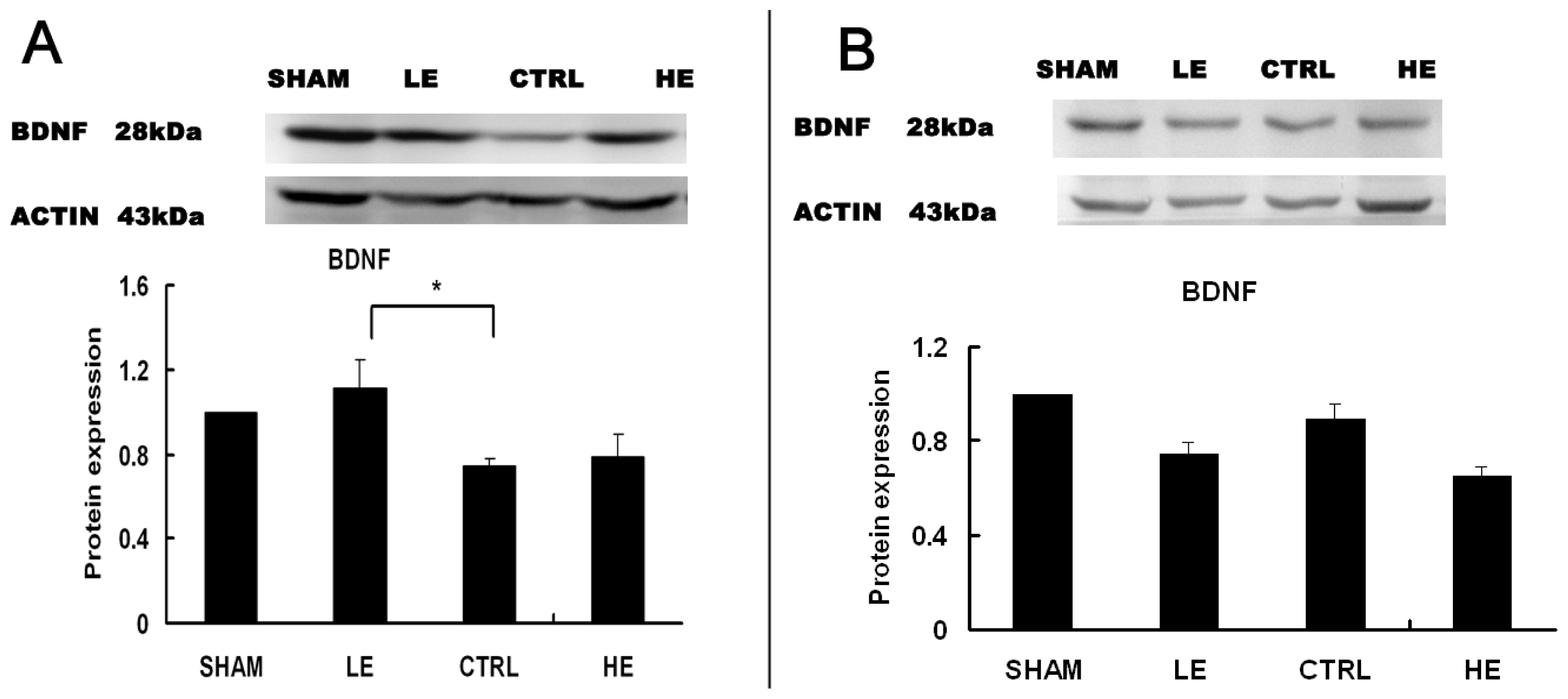
2.4. Effects of Treadmill Exercise on Hippocampal BDNF Protein
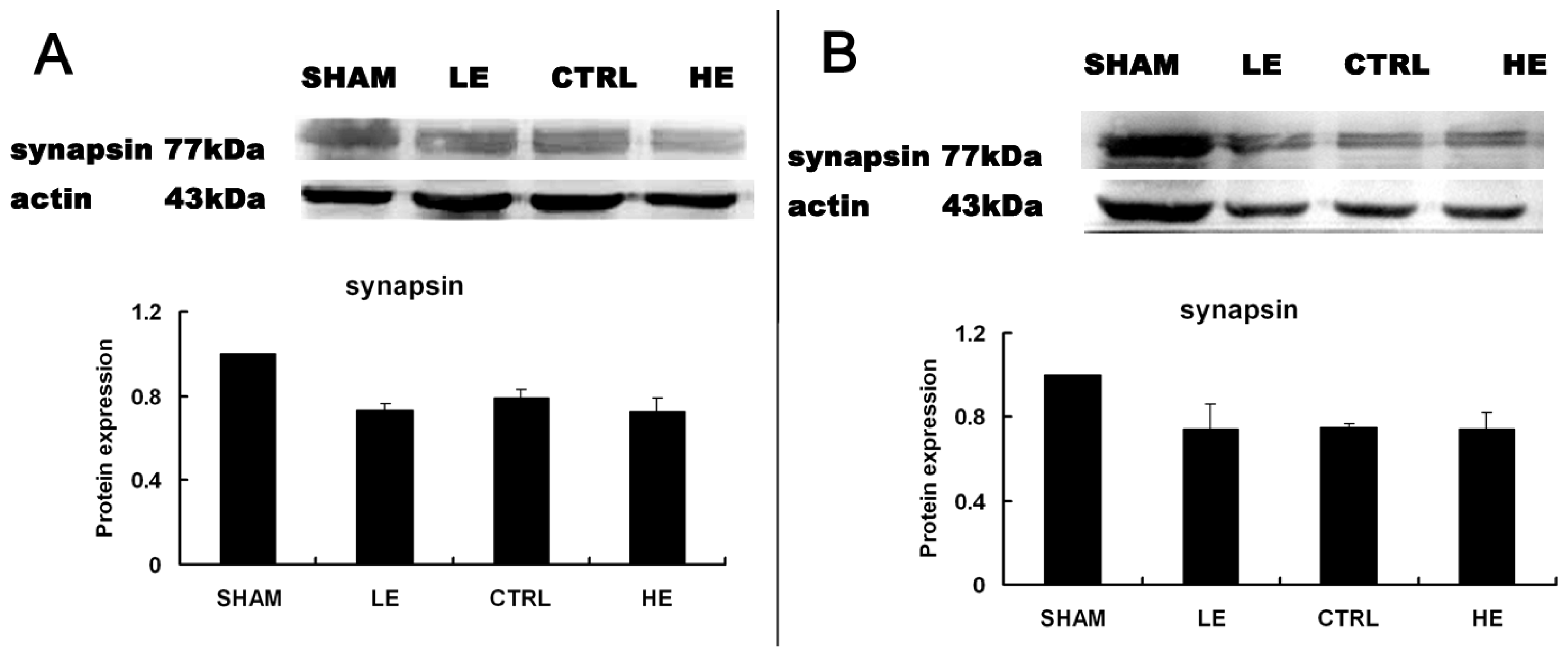
2.5. Effects of Treadmill Exercise on Hippocampal Synapsin I Protein
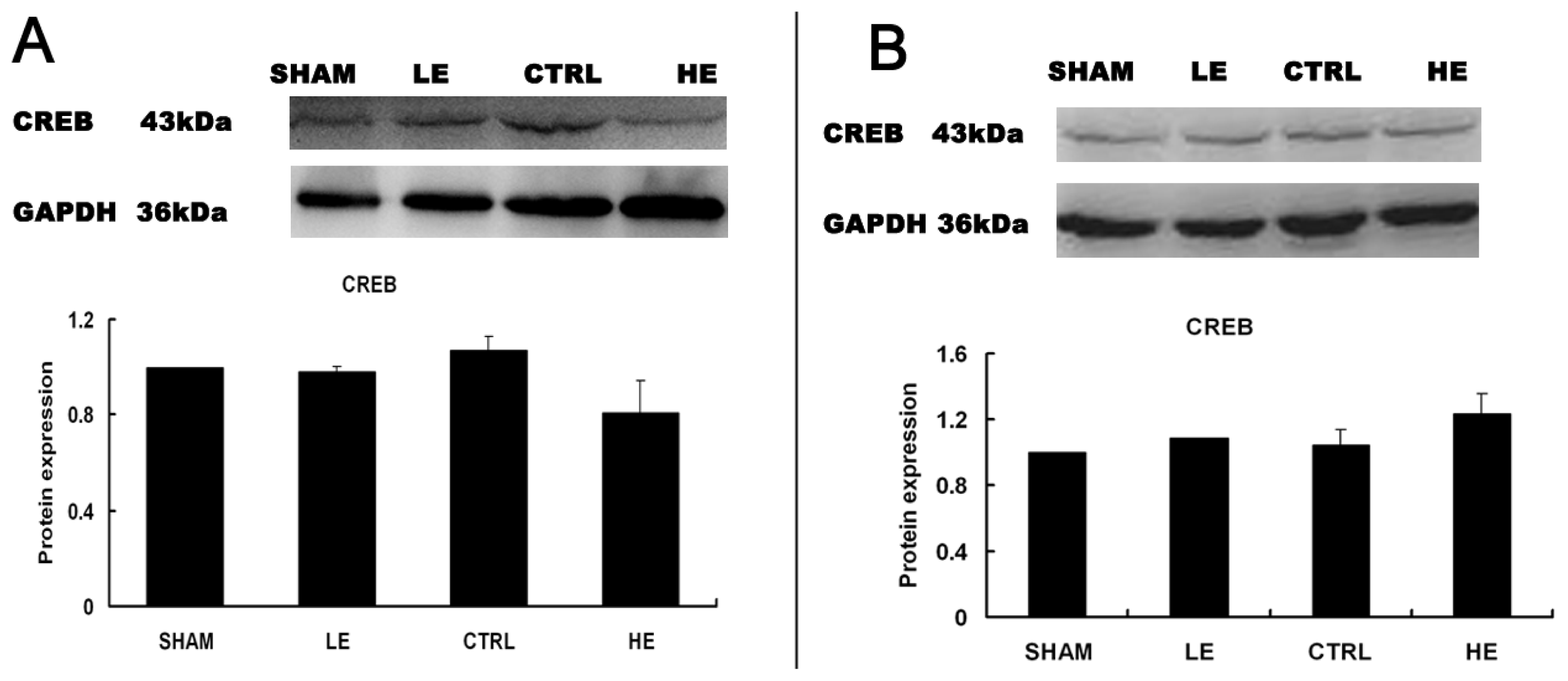
2.6. Effects of Treadmill Exercise on Hippocampal CREB Protein
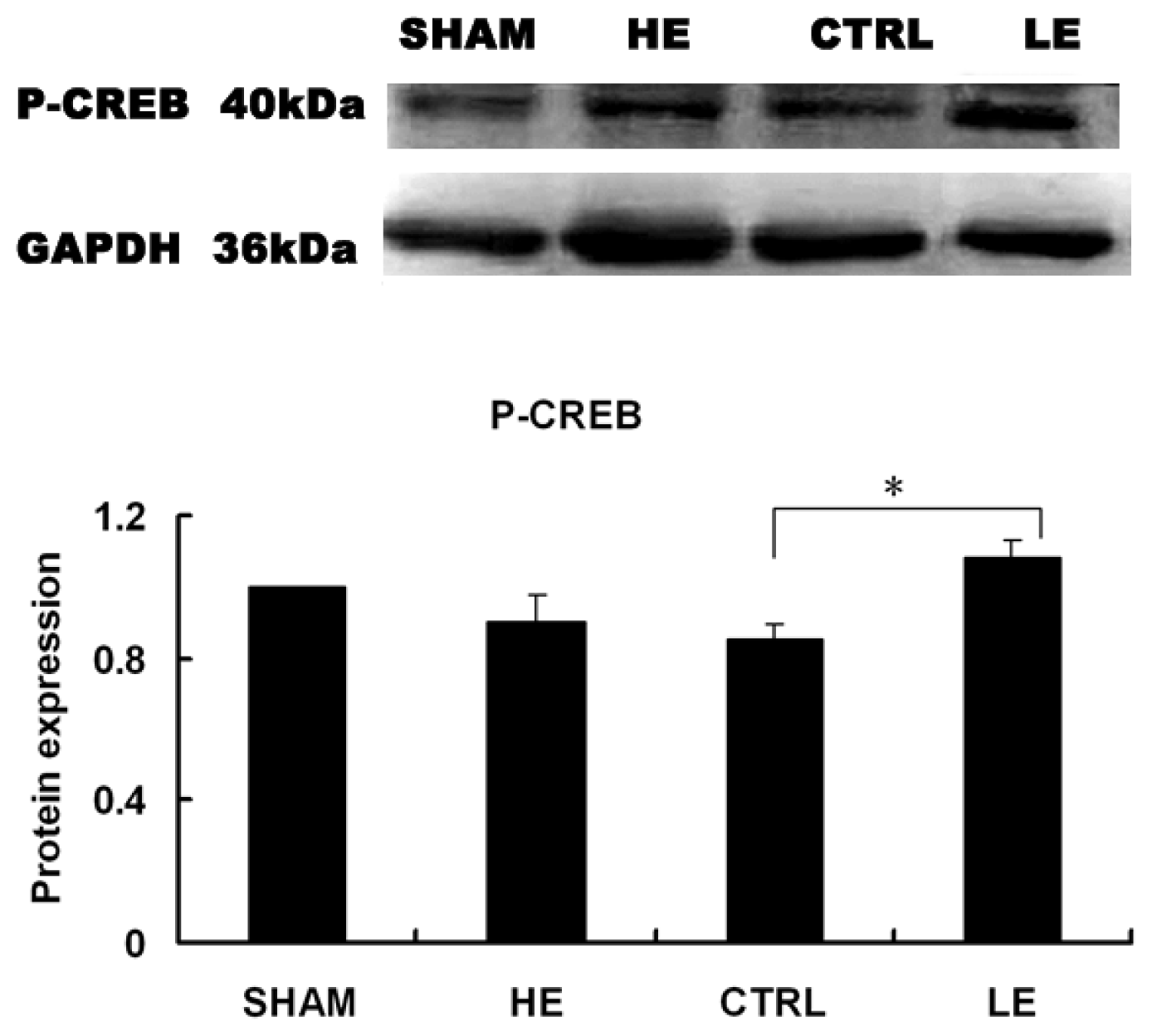
2.7. Effects of Treadmill Exercise on Hippocampal Phosphorylated CREB Protein
2.8. Discussion
3. Experimental Section
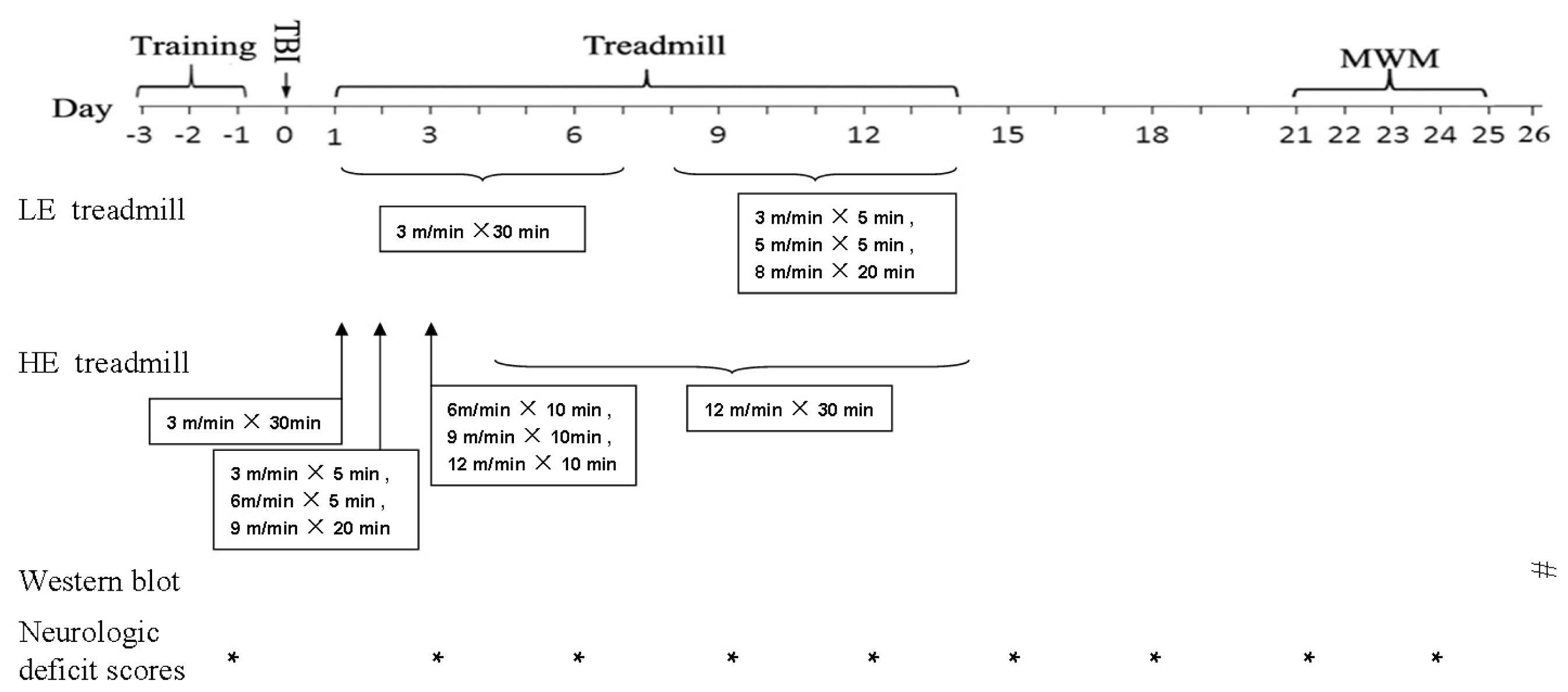
3.1. An Animal Model of Traumatic Brain Injury
3.2. Treadmill Training
3.3. Neurological Deficits and Cognitive Evaluations
3.3.1. Neurological Deficits
3.3.2. The Morris Water Maze Task
3.4. Western Blot
3.5. Statistical Analysis
4. Conclusions


Acknowledgments
Conflicts of Interest
References
- Hillman, C.H.; Erickson, K.I.; Kramer, A.F. Be smart, exercise your heart: Exercise effects on brain and cognition. Nat. Rev. Neurosci 2008, 9, 58–65. [Google Scholar]
- Carek, P.J.; Laibstain, S.E.; Carek, S.M. Exercise for the treatment of depression and anxiety. Int. J. Psychiatry Med 2011, 41, 15–28. [Google Scholar]
- Abbott, R.D.; White, L.R.; Ross, G.W.; Masaki, K.H.; Curb, J.D.; Petrovitch, H. Walking and dementia in physically capable elderly men. JAMA 2004, 292, 1447–1453. [Google Scholar]
- Dik, M.G.; Deeg, D.J.H.; Visser, M.; Jonker, C. Early life physical activity and cognition at old age. J. Clin. Exp. Neuropsychol 2003, 25, 643–653. [Google Scholar]
- Kramer, A.F.; Hahn, S.; Cohen, N.J.; Banich, M.T.; McAuley, E.; Harrison, C.R.; Chason, J.; Vakil, E.; Bardell, L.; Boileau, R.A.; et al. Ageing, fitness and neurocognitive function. Nature 1999, 400, 418–419. [Google Scholar]
- Van Praag, H.; Kempermann, G.; Gage, F.H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci 1999, 2, 266–270. [Google Scholar]
- Griesbach, G.S.; Hovda, D.A.; Gomez-Pinilla, F. Exercise-induced improvement in cognitive performance after traumatic brain injury in rats is dependent on BDNF activation. Brain Res 2009, 1288, 105–115. [Google Scholar]
- Dikmen, S.S.; Corrigan, J.D.; Levin, H.S.; Machamer, J.; Stiers, W.; Weisskopf, M.G. Cognitive outcome following traumatic brain injury. J. Head Trauma Rehabil 2009, 24, 430–438. [Google Scholar]
- Millis, S.R.; Rosenthal, M.; Novack, T.A.; Sherer, M.; Nick, T.G.; Kreutzer, J.S.; High, W.M., Jr.; Ricker, J.H. Long-term neuropsychological outcome after traumatic brain injury. J. Head Trauma Rehabil 2001, 16, 343–355. [Google Scholar]
- Hicks, R.R.; Boggs, A.; Leider, D.; Kraemer, P.; Brown, R.; Scheff, S.W.; Seroogy, K.B. Effects of exercise following lateral fluid percussion brain injury in rats. Restor. Neurol. Neurosci 1998, 12, 41–47. [Google Scholar]
- Wilde, M.C.; Castriotta, R.J.; Lai, J.M.; Atanasov, S.; Masel, B.E.; Kuna, S.T. Cognitive impairment in patients with traumatic brain injury and obstructive sleep apnea. Arch. Phys. Med. Rehabil 2007, 88, 1284–1288. [Google Scholar]
- Griesbach, G.S.; Sutton, R.L.; Hovda, D.A.; Ying, Z.; Gomez-Pinilla, F. Controlled contusion injury alters molecular systems associated with cognitive performance. J. Neurosci. Res 2009, 87, 795–805. [Google Scholar]
- Lighthall, J.W. Controlled cortical impact: A new experimental brain injury model. J. Neurotrauma 1988, 5, 1–15. [Google Scholar]
- Tehranian, R.; Rose, M.E.; Vagni, V.; Pickrell, A.M.; Griffith, R.P.; Liu, H.; Clark, R.S.; Dixon, C.E.; Kochanek, P.M.; Graham, S.H. Disruption of Bax protein prevents neuronal cell death but produces cognitive impairment in mice following traumatic brain injury. J. Neurotrauma 2008, 25, 755–767. [Google Scholar]
- Andelic, N.; Bautz-Holter, E.; Ronning, P.A.; Olafsen, K.; Sigurdardottir, S.; Schanke, A.K.; Sveen, U.; Tornas, S.; Sandhaug, M.; Roe, C. Does an early onset and continuous chain of rehabilitation improve the long-term functional outcome of patients with severe traumatic brain injury? J. Neurotrauma 2012, 29, 66–74. [Google Scholar]
- Franckeviciute, E.; Krisciunas, A. Evaluation of factors influencing effectiveness of kinesitherapy in patients after traumatic brain injury. Medicina 2006, 42, 732–737. [Google Scholar]
- Kim, D.H.; Ko, I.G.; Kim, B.K.; Kim, T.W.; Kim, S.E.; Shin, M.S.; Kim, C.J.; Kim, H.; Kim, K.M.; Baek, S.S. Treadmill exercise inhibits traumatic brain injury-induced hippocampal apoptosis. Physiol. Behav 2010, 101, 660–665. [Google Scholar]
- Griesbach, G.S.; Gomez-Pinilla, F.; Hovda, D.A. Time window for voluntary exercise-induced increases in hippocampal neuroplasticity molecules after traumatic brain injury is severity dependent. J. Neurotrauma 2007, 24, 1161–1171. [Google Scholar]
- Seo, T.B.; Kim, B.K.; Ko, I.G.; Kim, D.H.; Shin, M.S.; Kim, C.J.; Yoon, J.H.; Kim, H. Effect of treadmill exercise on Purkinje cell loss and astrocytic reaction in the cerebellum after traumatic brain injury. Neurosci. Lett 2010, 481, 178–182. [Google Scholar]
- Lima, F.D.; Oliveira, M.S.; Furian, A.F.; Souza, M.A.; Rambo, L.M.; Ribeiro, L.R.; Silva, L.F.; Retamoso, L.T.; Hoffmann, M.S.; Magni, D.V.; et al. Adaptation to oxidative challenge induced by chronic physical exercise prevents Na+,K+-ATPase activity inhibition after traumatic brain injury. Brain Res 2009, 1279, 147–155. [Google Scholar]
- Itoh, T.; Imano, M.; Nishida, S.; Tsubaki, M.; Hashimoto, S.; Ito, A.; Satou, T. Exercise inhibits neuronal apoptosis and improves cerebral function following rat traumatic brain injury. J. Neural Transm 2011, 118, 1263–1272. [Google Scholar]
- Szabo, Z.; Ying, Z.; Radak, Z.; Gomez-Pinilla, F. Voluntary exercise may engage proteasome function to benefit the brain after trauma. Brain Res 2010, 1341, 25–31. [Google Scholar]
- Itoh, T.; Imano, M.; Nishida, S.; Tsubaki, M.; Hashimoto, S.; Ito, A.; Satou, T. Exercise increases neural stem cell proliferation surrounding the area of damage following rat traumatic brain injury. J. Neural Transm 2010, 118, 193–202. [Google Scholar]
- Chytrova, G.; Ying, Z.; Gomez-Pinilla, F. Exercise normalizes levels of MAG and Nogo-A growth inhibitors after brain trauma. Eur. J. Neurosci 2008, 27, 1–11. [Google Scholar]
- Kozlowski, D.A.; James, D.C.; Schallert, T. Use-dependent exaggeration of neuronal injury after unilateral sensorimotor cortex lesions. J. Neurosci 1996, 16, 4776–4786. [Google Scholar]
- Miles, S.C.; Chun-Chung, C.; Hsin-Fu, L.; Hunter, S.D.; Dhindsa, M.; Nualnim, N.; Tanaka, H. Arterial blood pressure and cardiovascular responses to yoga practice. Altern Ther. Health Med 2013, 19, 38–45. [Google Scholar]
- Kurayama, T.; Tadokoro, Y.; Fujimoto, S.; Komiya, Z.; Yoshida, S.; Chakraborty, S.; Matsuzawa, D.; Shimizu, E.; Kondo, K.; Otaka, Y. A comparison of the movement characteristics between the kneeling gait and the normal gait in healthy adults. Gait Posture 2013, 37, 402–407. [Google Scholar]
- Burnfield, J.M.; McCrory, B.; Shu, Y.; Buster, T.W.; Taylor, A.P.; Goldman, A.J. Comparative kinematic and electromyographic assessment of clinician- and device-assisted sit-to-stand transfers in patients with stroke. Phys. Ther 2013, 93, 1331–1341. [Google Scholar]
- Triolo, R.J.; Bailey, S.N.; Lombardo, L.M.; Miller, M.E.; Foglyano, K.; Audu, M.L. Effects of intramuscular trunk stimulation on manual wheelchair propulsion mechanics in 6 subjects with spinal cord injury. Arch. Phys. Med. Rehabil 2013, 94, 1997–2005. [Google Scholar]
- St Clair Gibson, A.; de Koning, J.J.; Thompson, K.G.; Roberts, W.O.; Micklewright, D.; Raglin, J.; Foster, C. Crawling to the finish line: Why do endurance runners collapse? Implications for understanding of mechanisms underlying pacing and fatigue. Sports Med 2013, 43, 413–424. [Google Scholar]
- Vaynman, S.; Ying, Z.; Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci 2004, 20, 2580–2590. [Google Scholar]
- Cassilhas, R.C.; Lee, K.S.; Fernandes, J.; Oliveira, M.G.; Tufik, S.; Meeusen, R.; de Mello, M.T. Spatial memory is improved by aerobic and resistance exercise through divergent molecular mechanisms. Neuroscience 2012, 202, 309–317. [Google Scholar]
- Barde, Y.A. Trophic factors and neuronal survival. Neuron 1989, 2, 1525–1534. [Google Scholar]
- Lee, J.; Duan, W.; Mattson, M.P. Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J. Neurochem 2002, 82, 1367–1375. [Google Scholar]
- Poo, M.M. Neurotrophins as synaptic modulators. Nat. Rev. Neurosci 2001, 2, 24–32. [Google Scholar]
- Neeper, S.A.; Gomez-Pinilla, F.; Choi, J.; Cotman, C. Exercise and brain neurotrophins. Nature 1995, 373, 109. [Google Scholar]
- Griesbach, G.S.; Gomez-Pinilla, F.; Hovda, D.A. The upregulation of plasticity-related proteins following TBI is disrupted with acute voluntary exercise. Brain Res 2004, 1016, 154–162. [Google Scholar]
- Griesbach, G.S.; Hovda, D.A.; Molteni, R.; Wu, A.; Gomez-Pinilla, F. Voluntary exercise following traumatic brain injury: Brain-derived neurotrophic factor upregulation and recovery of function. Neuroscience 2004, 125, 129–139. [Google Scholar]
- Bruunsgaard, H.; Hartkopp, A.; Mohr, T.; Konradsen, H.; Heron, I.; Mordhorst, C.H.; Pedersen, B.K. In vivo cell-mediated immunity and vaccination response following prolonged, intense exercise. Med. Sci. Sports Exerc 1997, 29, 1176–1181. [Google Scholar]
- Hoffman-Goetz, L.; Spagnuolo, P.A.; Guan, J. Repeated exercise in mice alters expression of IL-10 and TNF-α in intestinal lymphocytes. Brain Behav. Immun 2008, 22, 195–199. [Google Scholar]
- Ostrowski, K.; Hermann, C.; Bangash, A.; Schjerling, P.; Nielsen, J.N.; Pedersen, B.K. A trauma-like elevation in plasma cytokines in humans in response to tread-mill running. J. Physiol 1998, 508, 949–953. [Google Scholar]
- McEwen, B.S.; Magarinos, A.M. Stress and hippocampal plasticity: Implications for the pathophysiology of affective disorders. Hum. Psychopharmacol 2001, 16, S7–S19. [Google Scholar]
- Sapolsky, R.M. Glucocorticoids and hippocampal atrophy in neu-ropsychiatric disorders. Arch. Gen. Psychiatry 2000, 57, 925–935. [Google Scholar]
- Hansson, A.C.; Sommer, W.H.; Metsis, M.; Stromberg, I.; Agnati, L.F.; Fuxe, K. Corticosterone actions on the hippocampal brain-derived neurotrophic factor expression are mediated by exon IV promoter. J. Neuroendocrinol 2006, 18, 104–114. [Google Scholar]
- Schaaf, M.J.; de Kloet, E.R.; Vreugdenhil, E. Corticosterone effects on BDNF expression in the hippocampus. Implications for memory formation. Stress 2000, 3, 201–208. [Google Scholar]
- Woolf, P.D.; Cox, C.; Kelly, M.; Nichols, D.; McDonald, J.V.; Hamill, R.W. The adrenocortical response to brain injury: Correlation with the severity of neurologic dysfunction, effects of intoxication, and patient outcome. Alcohol. Clin. Exp. Res 1990, 14, 917–921. [Google Scholar]
- Taylor, A.N.; Rahman, S.U.; Tio, D.L.; Gardner, S.M.; Kim, C.J.; Sutton, R.L. Injury severity differentially alters sensitivity to dexamethasone after traumatic brain injury. J. Neurotrauma 2010, 27, 1081–1089. [Google Scholar]
- Griesbach, G.S.; Hovda, D.A.; Tio, D.L.; Taylor, A.N. Heightening of the stress response during the first weeks after a mild traumatic brain injury. Neuroscience 2011, 178, 147–158. [Google Scholar]
- Blustein, J.E.; McLaughlin, M.; Hoffman, J.R. Exercise effects stress-induced analgesia and spatial learning in rats. Physiol. Behav 2006, 89, 582–586. [Google Scholar]
- Moraska, A.; Deak, T.; Spencer, R.L.; Roth, D.; Fleshner, M. Treadmill running produces both positive and negative physiological adaptations in Sprague-Dawley rats. Am. J. Physiol. Regul. Integr. Comp. Physiol 2000, 279, R1321–R1329. [Google Scholar]
- Sutton, R.L.; Lescaudron, L.; Stein, D.G. Unilateral cortical contusion injury in the rat: Vascular disruption and temporal development of cortical necrosis. J. Neurotrauma 1993, 10, 135–149. [Google Scholar]
- Golding, E.M.; Contant, C.F.; Robertson, C.S.; Bryan, R.M. Temporal effect of severe controlled cortical impact injury in the rat on the myogenic response of the middle cerebral artery. J. Neurotrauma 1998, 15, 973–984. [Google Scholar]
- Kochanek, P.M.; Marion, D.W.; Zhang, W.; Schiding, J.K.; White, M.; Palmer, A.M.; Clark, R.S.; O’Malley, M.E.; Styren, S.D.; Ho, C.; et al. Severe controlled cortical impact in rats: Assessment of cerebral edema, blood flow, and contusion volume. J. Neurotrauma 1995, 12, 1015–1025. [Google Scholar]
- Zhang, P.Y.; Zhang, Q.; Pu, H.J.; Wu, Y.; Bai, Y.L.; Vosler, P.S.; Chen, J.; Shi, H.; Gao, Y.Q.; Hu, Y.S. Very early-initiated physical rehabilitation protects against ischemic brain injury. Front. Biosci 2012, 4, 2476–2489. [Google Scholar]
- Rogers, D.C.; Campbell, C.A.; Stretton, J.L. Correlation between motor impairment and infarct volume after permanent and transient middle cerebral artery occlusion in the rat. Stroke 1997, 28, 2060–2066. [Google Scholar]
- Vorhees, C.V.; Williams, M.T. Morris water maze: Procedures for assessing spatial and related forms of learning and memory. Nat. Protoc 2006, 1, 848–858. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shen, X.; Li, A.; Zhang, Y.; Dong, X.; Shan, T.; Wu, Y.; Jia, J.; Hu, Y. The Effect of Different Intensities of Treadmill Exercise on Cognitive Function Deficit Following a Severe Controlled Cortical Impact in Rats. Int. J. Mol. Sci. 2013, 14, 21598-21612. https://doi.org/10.3390/ijms141121598
Shen X, Li A, Zhang Y, Dong X, Shan T, Wu Y, Jia J, Hu Y. The Effect of Different Intensities of Treadmill Exercise on Cognitive Function Deficit Following a Severe Controlled Cortical Impact in Rats. International Journal of Molecular Sciences. 2013; 14(11):21598-21612. https://doi.org/10.3390/ijms141121598
Chicago/Turabian StyleShen, Xiafeng, Aiping Li, Yuling Zhang, XiaoMin Dong, Tian Shan, Yi Wu, Jie Jia, and Yongshan Hu. 2013. "The Effect of Different Intensities of Treadmill Exercise on Cognitive Function Deficit Following a Severe Controlled Cortical Impact in Rats" International Journal of Molecular Sciences 14, no. 11: 21598-21612. https://doi.org/10.3390/ijms141121598



