Angiotensin-Converting Enzymes Play a Dominant Role in Fertility
Abstract
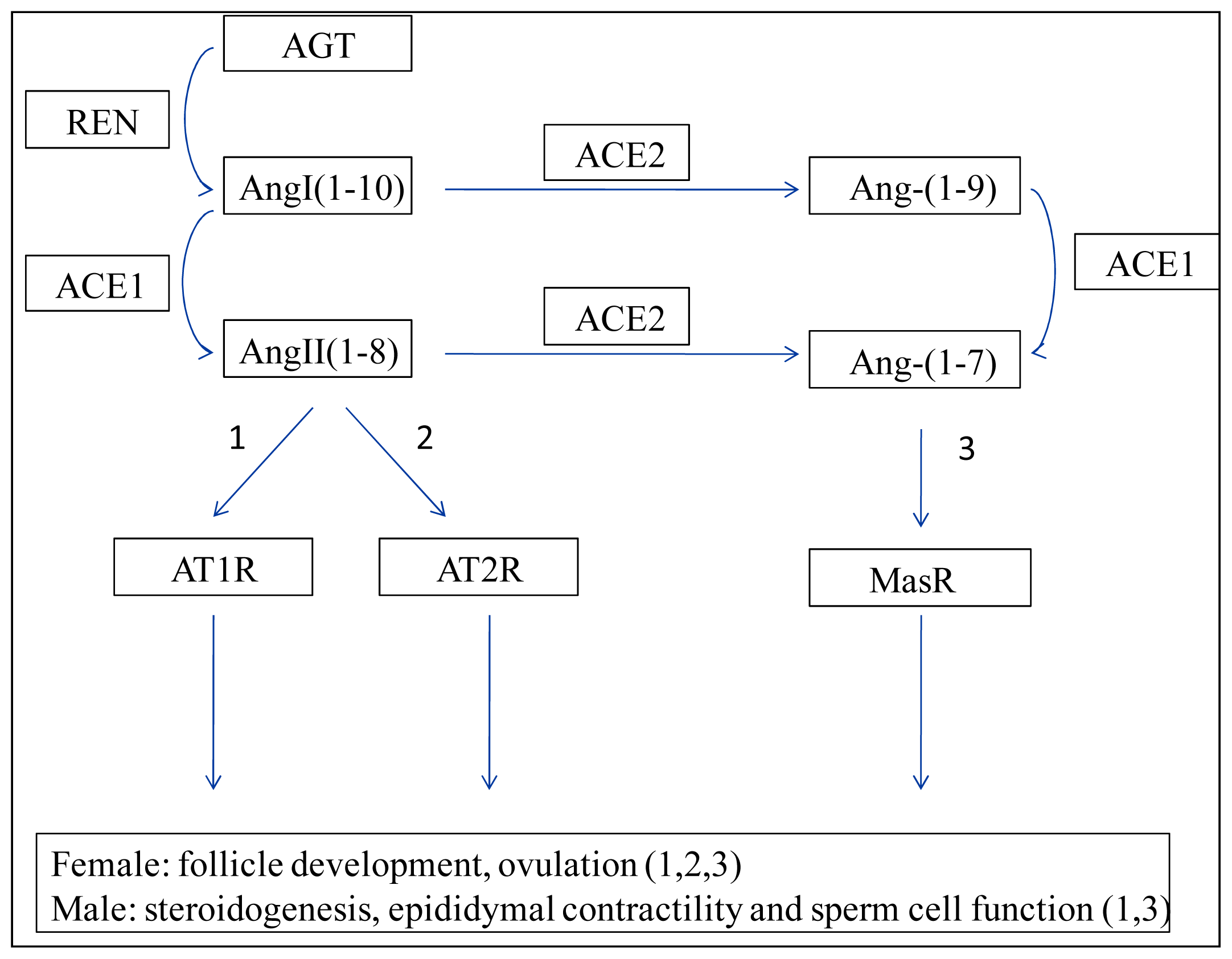
:1. Introduction
2. Renin–Angiotensin System (RAS)
3. Ovary ACEs
3.1. Ovary ACE1
3.2. Ovary ACE2
4. Testis ACEs
4.1. Testis ACE1
4.2. Testis ACE2
4.3. Testis ACE3
5. Sterility of MetS
6. Therapy for Infertility
7. Conclusions

Acknowledgments
Conflicts of Interest
References
- Corona, G.; Rastrelli, G.; Morelli, A.; Vignozzi, L.; Mannucci, E.; Maggi, M. Hypogonadism and metabolic syndrome. J. Endocrinol. Invest 2011, 34, 557–567. [Google Scholar]
- Corona, G.; Rastrelli, G.; Vignozzi, L.; Mannucci, E.; Maggi, M. Testosterone, cardiovascular disease and the metabolic syndrome. Best Pract. Res. Clin. Endocrinol. Metab 2011, 25, 337–353. [Google Scholar]
- Lotti, F.; Corona, G.; Degli Innocenti, S.; Filimberti, E.; Scognamiglio, V.; Vignozzi, L.; Forti, G.; Maggi, M. Seminal, ultrasound and psychobiological parameters correlate with metabolic syndrome in male members of infertile couples. Andrology 2013, 1, 229–239. [Google Scholar]
- Orio, F.; Palomba, S.; Di Biase, S.; Colao, A.; Tauchmanova, L.; Savastano, S.; Labella, D.; Russo, T.; Zullo, F.; Lombardi, G. Homocysteine levels and C677T polymorphism of methylenetetrahydrofolate reductase in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab 2003, 88, 673–679. [Google Scholar]
- Unsal, T.; Konac, E.; Yesilkaya, E.; Yilmaz, A.; Bideci, A.; Onen, H.I.; Cinaz, P.; Menevse, A. Genetic polymorphisms of FSHR, CYP17, CYP1A1, CAPN10, INSR, SERPINE1 genes in adolescent girls with polycystic ovary syndrome. J. Assist. Reprod Genet 2009, 26, 205–216. [Google Scholar]
- Boivin, J.; Bunting, L.; Collins, J.A.; Nygren, K.G. International estimates of infertility prevalence and treatment-seeking: Potential need and demand for infertility medical care. Hum. Reprod 2007, 22, 1506–1512. [Google Scholar]
- Farhi, J.; Ben-Haroush, A. Distribution of causes of infertility in patients attending primary fertility clinics in Israel. Isr. Med. Assoc. J 2011, 13, 51–54. [Google Scholar]
- Dorjpurev, U.; Kuwahara, A.; Yano, Y.; Taniguchi, T.; Yamamoto, Y.; Suto, A.; Tanaka, Y.; Matsuzaki, T.; Yasui, T.; Irahara, M. Effect of semen characteristics on pregnancy rate following intrauterine insemination. J. Med. Invest 2011, 58, 127–133. [Google Scholar]
- Azziz, R.; Marin, C.; Hoq, L.; Badamgarav, E.; Song, P. Health care-related economic burden of the polycystic ovary syndrome during the reproductive life span. J. Clin. Endocrinol. Metab 2005, 90, 4650–4658. [Google Scholar]
- Wu, X.; Lu, K.; Su, Y. Renin-angiotensin system: Involvement in polycystic ovarian syndrome. Zhonghua Fu Chan Ke Za Zhi 1997, 32, 428–431. [Google Scholar]
- Arefi, S.; Mottaghi, S.; Sharifi, A.M. Studying the correlation of renin-angiotensin-system (RAS) components and insulin resistance in polycystic ovary syndrome (PCOs). Gynecol. Endocrinol 2013, 29, 1–4. [Google Scholar]
- Kessler, S.P.; Rowe, T.M.; Gomos, J.B.; Kessler, P.M.; Sen, G.C. Physiological non-equivalence of the two isoforms of angiotensin-converting enzyme. J. Biol. Chem 2000, 275, 26259–26264. [Google Scholar]
- Crackower, M.A.; Sarao, R.; Oudit, G.Y.; Yagil, C.; Kozieradzki, I.; Scanga, S.E.; Oliveira-dos-Santos, A.J.; da Costa, J.; Zhang, L.; Pei, Y. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature 2002, 417, 822–828. [Google Scholar]
- Inoue, N.; Kasahara, T.; Ikawa, M.; Okabe, M. Identification and disruption of sperm-specific angiotensin converting enzyme-3 (ACE3) in mouse. PLoS One 2010, 5, e10301. [Google Scholar]
- De Kloet, A.D.; Krause, E.G.; Kim, D.-H.; Sakai, R.R.; Seeley, R.J.; Woods, S.C. The effect of angiotensin-converting enzyme inhibition using captopril on energy balance and glucose homeostasis. Endocrinology 2009, 150, 4114–4123. [Google Scholar]
- Jayasooriya, A.P.; Mathai, M.L.; Walker, L.L.; Begg, D.P.; Denton, D.A.; Cameron-Smith, D.; Egan, G.F.; McKinley, M.J.; Rodger, P.D.; Sinclair, A.J. Mice lacking angiotensin-converting enzyme have increased energy expenditure, with reduced fat mass and improved glucose clearance. Proc. Natl. Acad. Sci. USA 2008, 105, 6531–6536. [Google Scholar]
- De Kloet, A.D.; Krause, E.G.; Woods, S.C. The renin angiotensin system and the metabolic syndrome. Physiol. Behav 2010, 100, 525–534. [Google Scholar]
- Mbah, A.; Ndukwu, G.; Ghasi, S.; Shu, E.; Ozoemena, F.; Mbah, J.; Onodugo, O.; Ejim, E.; Eze, M.; Nkwo, P. Low-dose lisinopril in normotensive men with idiopathic oligospermia and infertility: A 5-year randomized, controlled, crossover pilot study. Clin. Pharmacol. Ther 2012, 91, 582–589. [Google Scholar]
- Shiuchi, T.; Cui, T.-X.; Wu, L.; Nakagami, H.; Takeda-Matsubara, Y.; Iwai, M.; Horiuchi, M. ACE inhibitor improves insulin resistance in diabetic mouse via bradykinin and NO. Hypertension 2002, 40, 329–334. [Google Scholar]
- Carey, R.M.; Siragy, H.M. Newly recognized components of the renin-angiotensin system: Potential roles in cardiovascular and renal regulation. Endocr. Rev 2003, 24, 261–271. [Google Scholar]
- Gonçalves, P.B.; Ferreira, R.; Gasperin, B.; Oliveira, J.F. Role of angiotensin in ovarian follicular development and ovulation in mammals: A review of recent advances. Reproduction 2012, 143, 11–20. [Google Scholar]
- Speth, R.; Daubert, D.; Grove, K; Angiotensin, II. A reproductive hormone too? Regul. Pept 1999, 79, 25–40. [Google Scholar]
- Mitsube, K.; Mikuni, M.; Matousek, M.; Zackrisson, U.; Brannstrom, M. Role of the angiotensin II system in regulation of ovulation and blood flow in the rat ovary. Reproduction 2003, 125, 425–435. [Google Scholar]
- Reis, A.; Viana, G.; Pereira, V.; Santos, R. Angiotensin-(1–7) in the rabbit ovary: A novel local regulator of ovulation. Biol. Reprod 2009, 81, 566–566. [Google Scholar]
- Ferreira, R.; Gasperin, B.; Santos, J.; Rovani, M.; Santos, R.A.; Gutierrez, K.; Oliveira, J.F.; Reis, A.M.; Gonçalves, P.B. Angiotensin II profile and mRNA encoding RAS proteins during bovine follicular wave. J. Renin Angiotensin Aldosterone Syst 2011, 12, 475–482. [Google Scholar]
- Reis, A.B.; Araújo, F.C.; Pereira, V.M.; Dos Reis, A.M.; Santos, R.A.; Reis, F.M. Angiotensin (1–7) and its receptor Mas are expressed in the human testis: Implications for male infertility. J. Mol. Histol 2010, 41, 75–80. [Google Scholar]
- Reis, F.M.; Bouissou, D.R.; Pereira, V.M.; Camargos, A.F.; dos Reis, A.M.; Santos, R.A. Angiotensin-(1–7) its receptor Mas, and the angiotensin-converting enzyme type 2 are expressed in the human ovary. Fertil. Steril 2011, 95, 176–181. [Google Scholar]
- Rella, M.; Elliot, J.L.; Revett, T.J.; Lanfear, J.; Phelan, A.; Jackson, R.M.; Turner, A.J.; Hooper, N.M. Identification and characterisation of the angiotensin converting enzyme-3 (ACE3) gene: A novel mammalian homologue of ACE. BMC Genomics 2007. [Google Scholar] [CrossRef]
- Acosta, E.; Peña, Ó.; Naftolin, F.; Ávila, J.; Palumbo, A. Angiotensin II induces apoptosis in human mural granulosa-lutein cells, but not in cumulus cells. Fertil. Steril 2009, 91, 1984–1989. [Google Scholar]
- Peña, Ó.; Palumbo, A.; González-Fernández, R.; Hernández, J.; Naftolin, F.; Ávila, J. Expression of angiotensin II type 1 (AT1) and angiotensin II type 2 (AT2) receptors in human granulosa-lutein (GL) cells: Correlation with infertility diagnoses. Fertil. Steril 2010, 93, 1601–1608. [Google Scholar]
- De Gooyer, T.; Skinner, S.; Wlodek, M.; Kelly, D.; Wilkinson-Berka, J. Angiotensin II influences ovarian follicle development in the transgenic (mRen-2) 27 and Sprague-Dawley rat. J. Endocrinol 2004, 180, 311–324. [Google Scholar]
- Pereira, V.M.; Reis, F.M.; Santos, R.A.; Cassali, G.D.; Santos, S.H.; Honorato-Sampaio, K.; dos Reis, A.M. Gonadotropin stimulation increases the expression of angiotensin-(1–7) and Mas receptor in the rat ovary. Reprod. Sci 2009, 16, 1165–1174. [Google Scholar]
- Leung, P.S.; Sernia, C. The renin–angiotensin system and male reproduction: New functions for old hormones. J. Mol. Endocrinol 2003, 30, 263–270. [Google Scholar]
- Leung, P.; Wong, T.; Chung, Y.; Chan, H. Androgen dependent expression of AT1 receptor and its regulation of anion secretion in rat epididymis. Cell Biol. Int 2002, 26, 117–122. [Google Scholar]
- Ehlers, M.R.; Riordan, J.F. Angiotensin-converting enzyme: New concepts concerning its biological role. Biochemistry 1989, 28, 5311–5318. [Google Scholar]
- Donoghue, M.; Hsieh, F.; Baronas, E.; Godbout, K.; Gosselin, M.; Stagliano, N.; Donovan, M.; Woolf, B.; Robison, K.; Jeyaseelan, R. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ. Res 2000, 87, e1–e9. [Google Scholar]
- Hubert, C.; Houot, A.-M.; Corvol, P.; Soubrier, F. Structure of the angiotensin I-converting enzyme gene. Two alternate promoters correspond to evolutionary steps of a duplicated gene. J. Biol. Chem 1991, 266, 15377–15383. [Google Scholar]
- Xu, J.; Baulding, J.; Palli, S.R. Proteomics of Tribolium castaneum seminal fluid proteins: Identification of an angiotensin-converting enzyme as a key player in regulation of reproduction. J. Proteomics 2012, 78, 83–93. [Google Scholar]
- Shibahara, H.; Kamata, M.; Hu, J.; Nakagawa, H.; Obara, H.; Kondoh, N.; Shima, H.; Sato, I. Activity of testis angiotensin converting enzyme (ACE) in ejaculated human spermatozoa. Int. J. Androl 2001, 24, 295–299. [Google Scholar]
- Hagaman, J.R.; Moyer, J.S.; Bachman, E.S.; Sibony, M.; Magyar, P.L.; Welch, J.E.; Smithies, O.; Krege, J.H.; O’Brien, D.A. Angiotensin-converting enzyme and male fertility. Proc. Natl. Acad. Sci. USA 1998, 95, 2552–2557. [Google Scholar]
- Pauls, K.; Fink, L.; Franke, F. Angiotensin-converting enzyme (CD143) in neoplastic germ cells. Lab. Invest 1999, 79, 1425–1435. [Google Scholar]
- Franke, F.E.; Pauls, K.; Metzger, R.; Danilov, S.M. Angiotensin I-converting enzyme and potential substrates in human testis and testicular tumours. APMIS 2003, 111, 234–244. [Google Scholar]
- Pauls, K.; Metzger, R.; Steger, K.; Klonisch, T.; Danilov, S.; Franke, F. Isoforms of angiotensin I-converting enzyme in the development and differentiation of human testis and epididymis. Andrologia 2003, 35, 32–43. [Google Scholar]
- Tipnis, S.R.; Hooper, N.M.; Hyde, R.; Karran, E.; Christie, G.; Turner, A.J. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J. Biol. Chem 2000, 275, 33238–33243. [Google Scholar]
- Zhang, H.; Wada, J.; Hida, K.; Tsuchiyama, Y.; Hiragushi, K.; Shikata, K.; Wang, H.; Lin, S.; Kanwar, Y.S.; Makino, H. Collectrin, a collecting duct-specific transmembrane glycoprotein, is a novel homolog of ACE2 and is developmentally regulated in embryonic kidneys. J. Biol. Chem 2001, 276, 17132–17139. [Google Scholar]
- Brentjens, J.; Matsuo, S.; Andres, G.; Caldwell, P.; Zamboni, L. Gametes contain angiotensin converting enzyme (kininase II). Experientia 1986, 42, 399–402. [Google Scholar]
- Ferreira, R.; Gasperin, B.; Rovani, M.; Santos, J.; Barreta, M.; Bohrer, R.; Price, C.; Gonçalves, P.B.D. Angiotensin II signaling promotes follicle growth and dominance in cattle. Endocrinology 2011, 152, 4957–4965. [Google Scholar]
- Feral, C.; le Gall, S.; Leymarie, P. Angiotensin II modulates steroidogenesis in granulosa and theca in the rabbit ovary: Its possible involvement in atresia. Eur. J. Endocrinol 1995, 133, 747–753. [Google Scholar]
- Li, Y.; Jiao, L.; Liu, R.; Chen, X.; Wang, H.; Wang, W. Localization of angiotensin II in pig ovary and its effects on oocyte maturation in vitro. Theriogenology 2004, 61, 447–459. [Google Scholar]
- Stefanello, J.R.; Barreta, M.H.; Porciuncula, P.M.; Arruda, J.N.; Oliveira, J.F.; Oliveira, M.A.; Gonçalves, P.B. Effect of angiotensin II with follicle cells and insulin-like growth factor-I or insulin on bovine oocyte maturation and embryo development. Theriogenology 2006, 66, 2068–2076. [Google Scholar]
- Yoshimura, Y.; Karube, M.; Aoki, H.; Oda, T.; Koyama, N.; Nagai, A.; Akimoto, Y.; Hirano, H.; Nakamura, Y. Angiotensin II induces ovulation and oocyte maturation in rabbit ovaries via the AT2 receptor subtype. Endocrinology 1996, 137, 1204–1211. [Google Scholar]
- Agarwal, A.; Gupta, S.; Sharma, R.K. Role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol 2005, 3, 1–21. [Google Scholar]
- Gupta, S.; Goldberg, J.M.; Aziz, N.; Goldberg, E.; Krajcir, N.; Agarwal, A. Pathogenic mechanisms in endometriosis-associated infertility. Fertil. Steril 2008, 90, 247–257. [Google Scholar]
- Ruder, E.H.; Hartman, T.J.; Goldman, M.B. Impact of oxidative stress on female fertility. Curr. Opin. Obstet Gynecol 2009, 21, 219–222. [Google Scholar]
- Ruder, E.H.; Hartman, T.J.; Blumberg, J.; Goldman, M.B. Oxidative stress and antioxidants: Exposure and impact on female fertility. Hum. Reprod. Update 2008, 14, 345–357. [Google Scholar]
- Benicky, J.; Sánchez-Lemus, E.; Pavel, J.; Saavedra, J.M. Anti-inflammatory effects of angiotensin receptor blockers in the brain and the periphery. Cell. Mol. Neurobiol 2009, 29, 781–792. [Google Scholar]
- Inserra, F.; Martínez-Maldonado, M. Inflammation and the metabolic syndrome: Role of angiotensin II and oxidative stress. Curr. Hypertens. Rep 2006, 8, 191–198. [Google Scholar]
- Yoshimura, Y.; Koyama, N.; Karube, M.; Oda, T.; Akiba, M.; Yoshinaga, A.; Shiokawa, S.; Jinno, M.; Nakamura, Y. Gonadotropin stimulates ovarian renin–angiotensin system in the rabbit. J. Clin. Invest 1994, 93, 180–187. [Google Scholar]
- Daud, A.I.; Bumpus, F.M.; Husain, A. Characterization of angiotensin I-converting enzyme (ACE)-containing follicles in the rat ovary during the estrous cycle and effects of ACE inhibitor on ovulation. Endocrinology 1990, 126, 2927–2935. [Google Scholar]
- Costa, A.P.; Fagundes-Moura, C.R.; Pereira, V.M.; Silva, L.F.; Vieira, M.A.R.; Santos, R.A.; Dos Reis, A.M. Angiotensin-(1–7): A novel peptide in the ovary. Endocrinology 2003, 144, 1942–1948. [Google Scholar]
- Honorato-Sampaio, K.; Pereira, V.M.; Santos, R.A.; Reis, A.M. Evidence that angiotensin-(1–7) is an intermediate of gonadotrophin-induced oocyte maturation in the rat preovulatory follicle. Exp. Physiol 2012, 97, 642–650. [Google Scholar]
- Cushman, D.; Cheung, H. Spectrophotometric assay and properties of the angiotensin-converting enzyme of rabbit lung. Biochem. Pharmacol 1971, 20, 1637–1648. [Google Scholar]
- Langford, K.G.; Zhou, Y.; Russell, L.D.; Wilcox, J.N.; Bernstein, K.E. Regulated expression of testis angiotensin-converting enzyme during spermatogenesis in mice. Biol. Reprod 1993, 48, 1210–1218. [Google Scholar]
- Köhn, F.-M.; Dammshäuser, I.; Neukamm, C.; Renneberg, H.; Siems, W.-E.; Schill, W.-B.; Aumüller, G. Ultrastructural localization of angiotensin-converting enzyme in ejaculated human spermatozoa. Hum. Reprod 1998, 13, 604–610. [Google Scholar]
- Nikolaeva, M.A.; Balyasnikova, I.V.; Alexinskaya, M.A.; Metzger, R.; Franke, F.E.; Albrecht, R.F.; Kulakov, V.I.; Sukhikh, G.T.; Danilov, S.M. Testicular isoform of angiotensin I-converting enzyme (ACE, CD143) on the surface of human spermatozoa: Revelation and quantification using monoclonal antibodies. Am. J. Reprod. Immunol 2006, 55, 54–68. [Google Scholar]
- Yokoyama, M.; Takada, Y.; Iwata, H.; Ochi, K.; Takeuchi, M.; Hiwada, K.; Kokubu, T. Correlation between angiotensin-converting enzyme activity and histologic patterns in benign prostatic hypertrophy tissue. J. Urol 1982, 127, 368–379. [Google Scholar]
- Dacheux, J.-L.; Belleannée, C.; Guyonnet, B.; Labas, V.; Teixeira-Gomes, A.-P.; Ecroyd, H.; Druart, X.; Gatti, J.-L.; Dacheux, F. The contribution of proteomics to understanding epididymal maturation of mammalian spermatozoa. Syst. Biol. Reprod. Med 2012, 58, 197–210. [Google Scholar]
- Cesari, A.; de Monclus, M. L.; Tejón, G.P.; Clementi, M.; Fornes, M.W. Regulated serine proteinase lytic system on mammalian sperm surface: There must be a role. Theriogenology 2010, 74, 699–711. [Google Scholar]
- Yamaguchi, R.; Yamagata, K.; Ikawa, M.; Moss, S.B.; Okabe, M. Aberrant distribution of ADAM3 in sperm from both angiotensin-converting enzyme (Ace)-and calmegin (Clgn)-deficient mice. Biol. Reprod 2006, 75, 760–766. [Google Scholar]
- Zhu, G.Z.; Gupta, S.; Myles, D.G.; Primakoff, P. Testase 1 (ADAM 24) a sperm surface metalloprotease is required for normal fertility in mice. Mol. Reprod. Dev 2009, 76, 1106–1114. [Google Scholar]
- Köhn, F.M.; Miska, W.; Schill, W.B. Release of angiotensin-converting enzyme (ACE) from human spermatozoa during capacitation and acrosome reaction. J. Androl 1995, 16, 259–265. [Google Scholar]
- Kamata, M.; Hu, J.; Shibahara, H.; Nakagawa, H. Assay of testicular angiotensin-converting enzyme activity in human spermatozoa. Int. J. Androl 2001, 24, 225–231. [Google Scholar]
- Kondoh, G.; Tojo, H.; Nakatani, Y.; Komazawa, N.; Murata, C.; Yamagata, K.; Maeda, Y.; Kinoshita, T.; Okabe, M.; Taguchi, R. Angiotensin-converting enzyme is a GPI-anchored protein releasing factor crucial for fertilization. Nat. Med 2005, 11, 160–166. [Google Scholar]
- Deguchi, E.; Tani, T.; Watanabe, H.; Yamada, S.; Kondoh, G. Dipeptidase-inactivated tACE action in vivo: Selective inhibition of sperm-zona pellucida binding in the mouse. Biol. Reprod 2007, 77, 794–802. [Google Scholar]
- Fuchs, S.; Frenzel, K.; Hubert, C.; Lyng, R.; Muller, L.; Michaud, A.; Xiao, H.D.; Adams, J.W.; Capecchi, M.R.; Corvol, P. Male fertility is dependent on dipeptidase activity of testis ACE. Nat. Med 2005, 11, 1140–1142. [Google Scholar]
- Leisle, L.; Parkin, E.T.; Turner, A.J.; Hooper, N.M. Angiotensin-converting enzyme as a GPIase: A critical reevaluation. Nat. Med 2005, 11, 1139–1140. [Google Scholar]
- Leal, M.C.; Pinheiro, S.V.; Ferreira, A.J.; Santos, R.A.; Bordoni, L.S.; Alenina, N.; Bader, M.; França, L.R. The role of angiotensin-(1–7) receptor Mas in spermatogenesis in mice and rats. J. Anat 2009, 214, 736–743. [Google Scholar]
- Kondoh, G.; Watanabe, H.; Tashima, Y.; Maeda, Y.; Kinoshita, T. Testicular angiotensin-converting enzyme with different glycan modification: Characterization on glycosylphosphatidylinositol-anchored protein releasing and dipeptidase activities. J. Biochem. 2009, 145, 115–121. [Google Scholar]
- Inoue, N.; Ikawa, M.; Isotani, A.; Okabe, M. The immunoglobulin superfamily protein Izumo is required for sperm to fuse with eggs. Nature 2005, 434, 234–238. [Google Scholar]
- Hudecova, M.; Holte, J.; Olovsson, M.; Larsson, A.; Berne, C.; Sundstrom-Poromaa, I. Prevalence of the metabolic syndrome in women with a previous diagnosis of polycystic ovary syndrome: Long-term follow-up. Fertil. Steril 2011, 96, 1271–1274. [Google Scholar]
- Jia, H.; Wang, B.; Yu, L.; Jiang, Z. Association of angiotensin-converting enzyme gene insertion/deletion polymorphism with polycystic ovary syndrome: A meta-analysis. J. Renin Angiotensin Aldosterone Syst 2012, 14, 255–262. [Google Scholar]
- Gard, P.R. Implications of the angiotensin converting enzyme gene insertion/deletion polymorphism in health and disease: A snapshot review. Int. J. Mol. Epidemiol. Genet 2010, 1, 145–157. [Google Scholar]
- Celik, O.; Yesilada, E.; Hascalik, S.; Celik, N.; Sahin, I.; Keskin, L.; Ozerol, E. Angiotensin-converting enzyme gene polymorphism and risk of insulin resistance in PCOS. Reprod. Biomed. Online 2010, 20, 492–498. [Google Scholar]
- Che, Y.; Cao, Y.; Wu, X.; Sun, H.-X.; Liang, F.; Yi, L.; Wang, Y. Association between ACE gene I/D polymorphisms and hyperandrogenism in women with polycystic ovary syndrome (PCOS) and controls. BMC Med. Genet 2009. [Google Scholar] [CrossRef]
- Nandi, A.; Wang, X.; Accili, D.; Wolgemuth, D.J. The effect of insulin signaling on female reproductive function independent of adiposity and hyperglycemia. Endocrinology 2010, 151, 1863–1871. [Google Scholar]
- Vandermolen, D.T.; Ratts, V.S.; Evans, W.S.; Stovall, D.W.; Kauma, S.W.; Nestler, J.E. Metformin increases the ovulatory rate and pregnancy rate from clomiphene citrate in patients with polycystic ovary syndrome who are resistant to clomiphene citrate alone. Fertil. Steril 2001, 75, 310–315. [Google Scholar]
- Du Plessis, S.S.; Cabler, S.; McAlister, D.A.; Sabanegh, E.; Agarwal, A. The effect of obesity on sperm disorders and male infertility. Nat. Rev. Urol 2010, 7, 153–161. [Google Scholar]
- Sermondade, N.; Faure, C.; Fezeu, L.; Lévy, R.; Czernichow, S. Obesity and increased risk for oligozoospermia and azoospermia. Arch. Intern. Med 2012, 172, 440–442. [Google Scholar]
- Riera-Fortuny, C.; Real, J.T.; Chaves, F.J.; Morales-Suarez-Varela, M.; Martinez-Triguero, M.L.; Morillas-Arino, C.; Hernández-Mijares, A. The relation between obesity, abdominal fat deposit and the angiotensin-converting enzyme gene I/D polymorphism and its association with coronary heart disease. Int. J. Obes 2005, 29, 78–84. [Google Scholar]
- Muciaccia, B.; Pensini, S.; Culasso, F.; Padula, F.; Paoli, D.; Gandini, L.; Di Veroli, C.; Bianchini, G.; Stefanini, M.; D’Agostino, A. Higher clusterin immunolabeling and sperm DNA damage levels in hypertensive men compared with controls. Hum. Reprod 2012, 27, 2267–2276. [Google Scholar]
- Ramalho-Santos, J.; Amaral, S.; Oliveira, P.J. Diabetes and the impairment of reproductive function: Possible role of mitochondria and reactive oxygen species. Curr. Diabetes Rev 2008, 4, 46–54. [Google Scholar]
- Gwathmey, T.M.; Pendergrass, K.D.; Reid, S.D.; Rose, J.C.; Diz, D.I.; Chappell, M.C. Angiotensin-(1–7)-angiotensin-converting enzyme 2 attenuates reactive oxygen species formation to angiotensin II within the cell nucleus. Hypertension 2010, 55, 166–171. [Google Scholar]
- Dikalov, S.I.; Nazarewicz, R.R. Angiotensin II-induced production of mitochondrial reactive oxygen species: Potential mechanisms and relevance for cardiovascular disease. Antioxid. Redox Signal 2012, 19, 1085–1094. [Google Scholar]
- Hacıhanefioglu, B.; Seyisoglu, H.; Karsıdag, K.; Elter, K.; Aksu, F.; Yılmaz, T.; Gurol, A.O. Influence of insulin resistance on total renin level in normotensive women with polycystic ovary syndrome. Fertil. Steril 2000, 73, 261–265. [Google Scholar]
- Franks, S. Genetic and environmental origins of obesity relevant to reproduction. Reprod. Biomed. Online 2006, 12, 526–531. [Google Scholar]
- Nawano, M.; Anai, M.; Funaki, M.; Kobayashi, H.; Kanda, A.; Fukushima, Y.; Inukai, K.; Ogihara, T.; Sakoda, H.; Onishi, Y. Imidapril, an angiotensin-converting enzyme inhibitor, improves insulin sensitivity by enhancing signal transduction via insulin receptor substrate proteins and improving vascular resistance in the Zucker fatty rat. Metabolism 1999, 48, 1248–1255. [Google Scholar]
- Muscogiuri, G.; Chavez, A.O.; Gastaldelli, A.; Perego, L.; Tripathy, D.; Saad, M.J.; Velloso, L.; Folli, F. The crosstalk between insulin and renin-angiotensin-aldosterone signaling systems and its effect on glucose metabolism and diabetes prevention. Curr. Vasc. Pharmacol 2008, 6, 301–312. [Google Scholar]
- Okeahialam, B.; Amadi, K.; Ameh, A. Effect of lisnopril, an angiotensin converting enzyme (ACE) inhibitor on spermatogenesis in rats. Syst. Biol. Reprod. Med 2006, 52, 209–213. [Google Scholar]
- Siems, W.E.; Maul, B.; Wiesner, B.; Becker, M.; Walther, T.; Rothe, L.; Winkler, A. Effects of kinins on mammalian spermatozoa and the impact of peptidolytic enzymes. Andrologia 2003, 35, 44–54. [Google Scholar]
- Monsees, T.K.; Miska, W.; Schill, W.-B. Characterization of kininases in testicular cells. Immunopharmacology 1996, 32, 169–171. [Google Scholar]
- Saha, L.; Garg, S.K.; Bhargava, V.K.; Mazumdar, S. Role of angiotensin-converting enzyme inhibitor, lisinopril, on spermatozoal functions in rats. Method Find. Exp. Clin. Pharmacol 2000, 22, 159–162. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pan, P.-P.; Zhan, Q.-T.; Le, F.; Zheng, Y.-M.; Jin, F. Angiotensin-Converting Enzymes Play a Dominant Role in Fertility. Int. J. Mol. Sci. 2013, 14, 21071-21086. https://doi.org/10.3390/ijms141021071
Pan P-P, Zhan Q-T, Le F, Zheng Y-M, Jin F. Angiotensin-Converting Enzymes Play a Dominant Role in Fertility. International Journal of Molecular Sciences. 2013; 14(10):21071-21086. https://doi.org/10.3390/ijms141021071
Chicago/Turabian StylePan, Pei-Pei, Qi-Tao Zhan, Fang Le, Ying-Ming Zheng, and Fan Jin. 2013. "Angiotensin-Converting Enzymes Play a Dominant Role in Fertility" International Journal of Molecular Sciences 14, no. 10: 21071-21086. https://doi.org/10.3390/ijms141021071



