Acute Effect of Ghrelin on Ischemia/Reperfusion Injury in the Rat Spinal Cord
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Methods
2.2. Animals Surgery
2.3. Drug Administration
2.4. Evaluation of Motor Function of Hind Limbs
2.5. Enzyme-Linked Immunosorbent Assay Measurement of Serum TNF-α
2.6. Myeloperoxidase (MPO) Activity Assay with Spinal Cord Tissues
2.7. Terminal Deoxynucleotidyl Transferase (TdT)-Mediated dUTP Nick End Labeling (TUNEL) Staining
2.8. RNA Preparation and RT-PCR
2.9. Western Blot Analysis
2.10. Statistical Analysis
3. Results
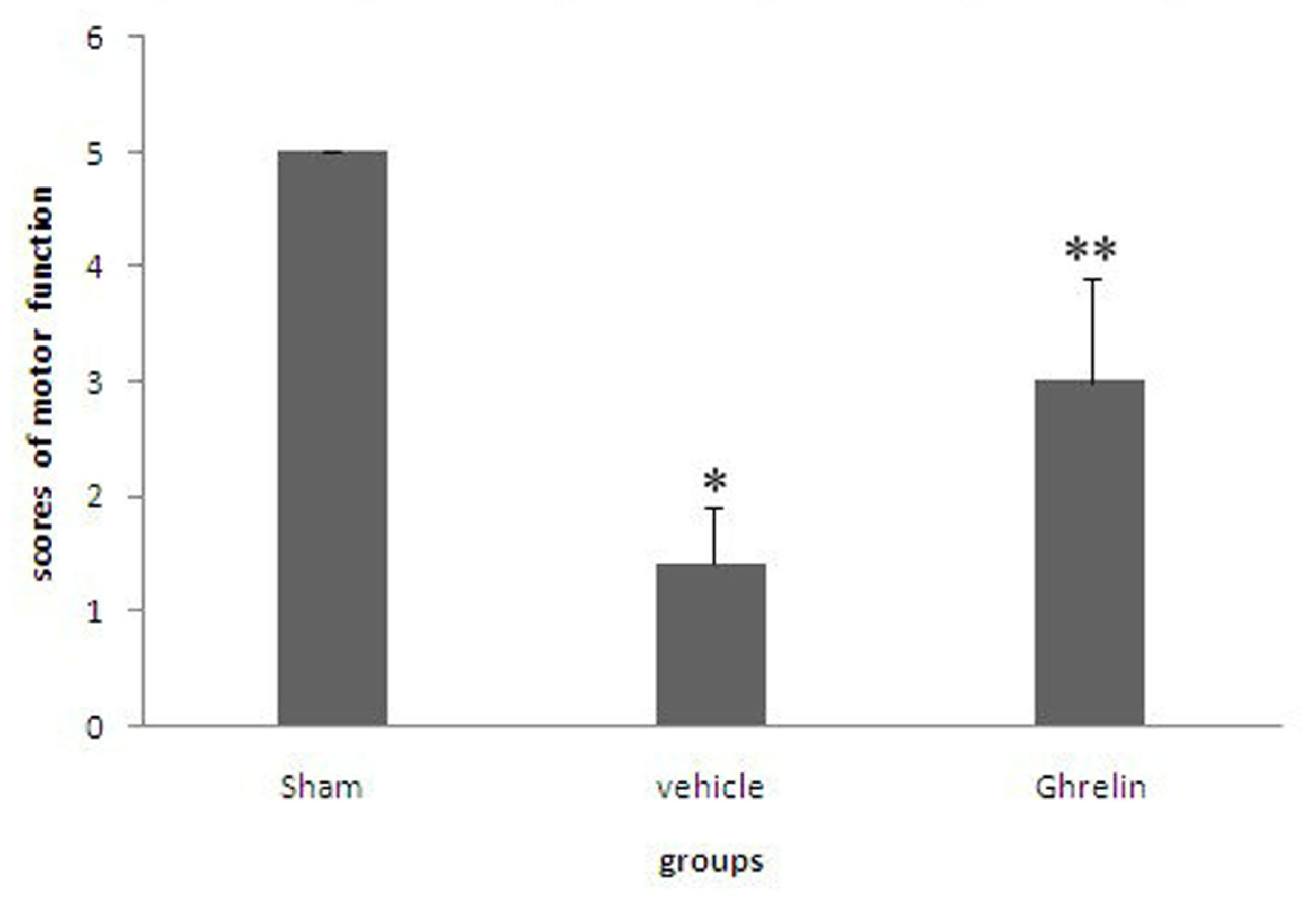
3.1. Effects of Ghrelin on Motor Function of Hind Limbs
3.2. Effects of Ghrelin Administration on TNF-α Release and MPO Activity
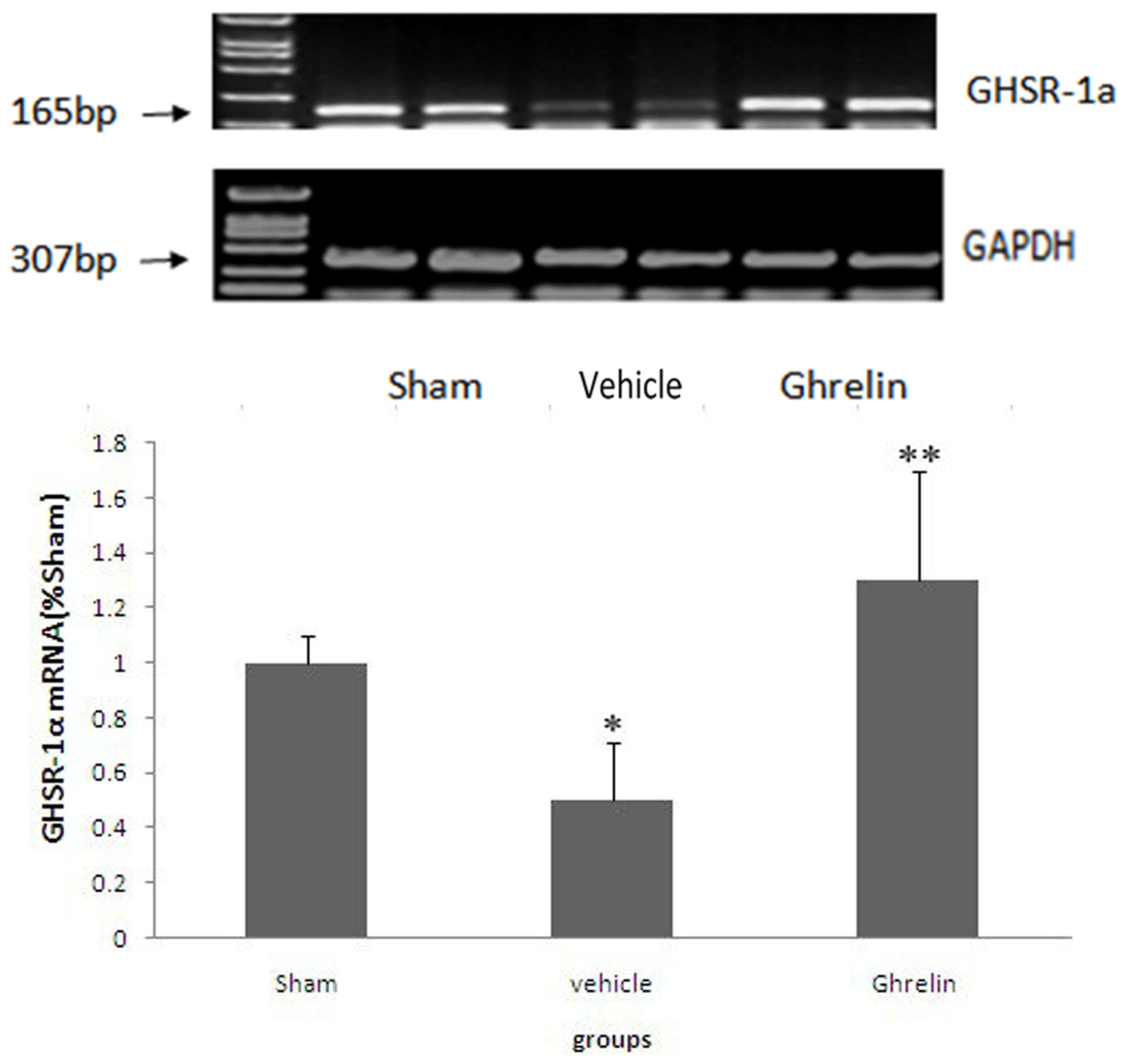
3.3. Changes of Ghrelin Receptor in Spinal Cord
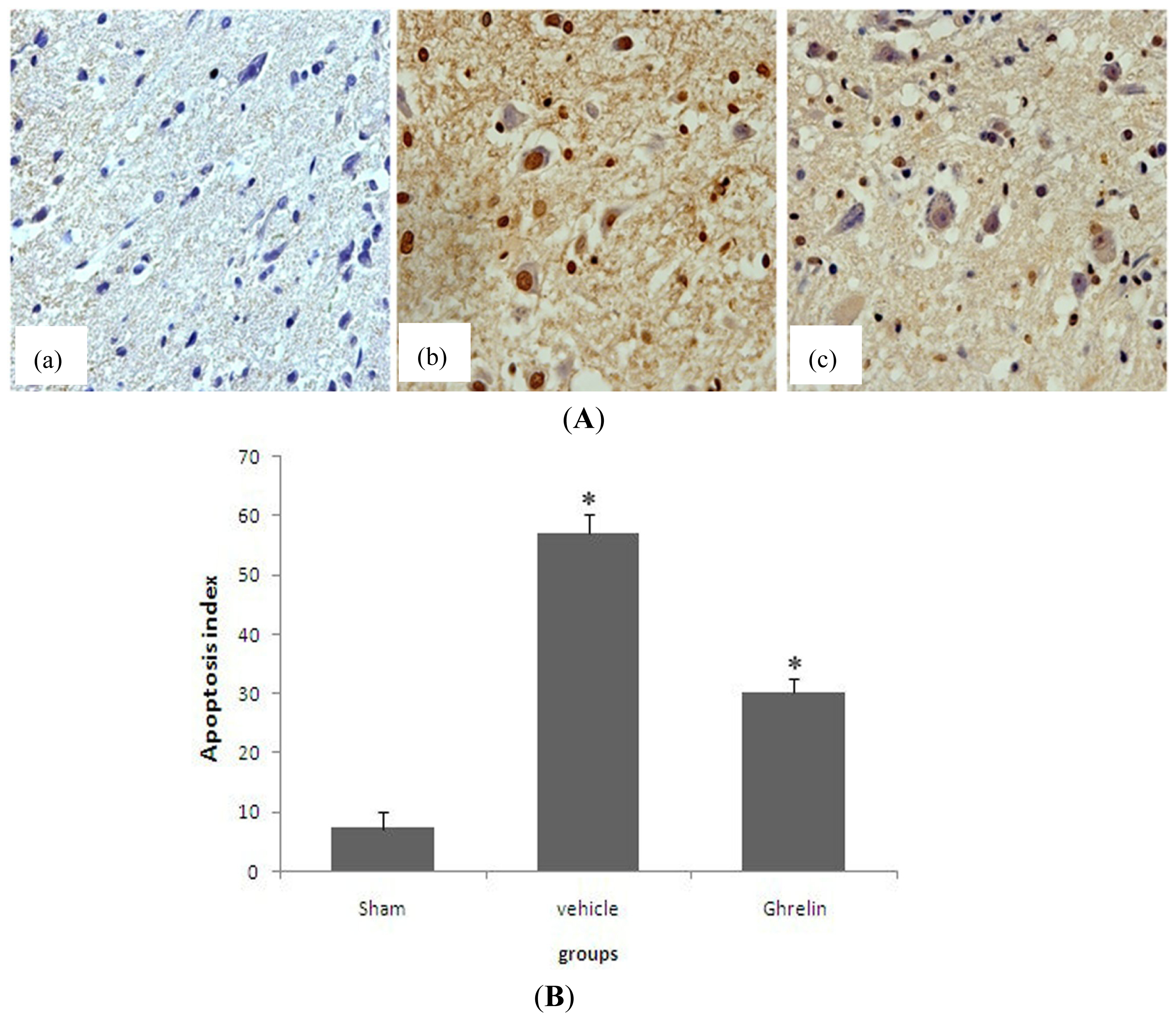
3.4. Effects of Ghrelin on TUNEL Staining
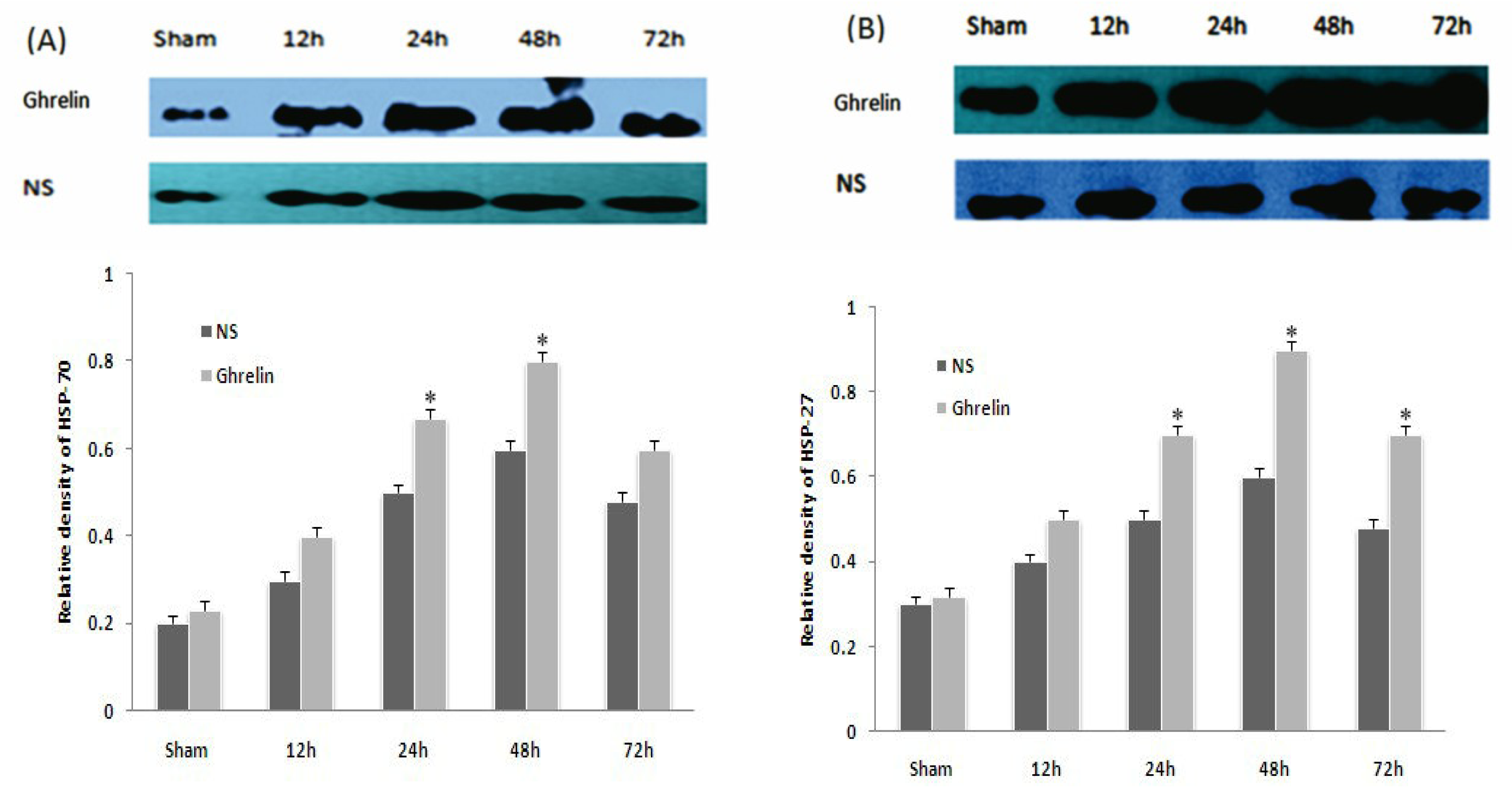
3.5. Effects of Ghrelin on Expression of HSP70, HSP27, Bax and Bcl-2
4. Discussion
Acknowledgment
List of Abbreviations
| EDTA | ethylenediaminetetraacetic acid |
| IL-β | interleukin-1β |
| TNF-α | tumor necrosis factor-α |
| I/R | ischemia/reperfusion |
| MPO | Myeloperoxidase |
| TUNEL | terminal deoxynucleotidyl transferase-mediated UTP end labeling |
| HSP | heat-shock protein |
| PBS | phosphate buffered saline |
References
- Zhou, Y.; Zhao, Y.N.; Yang, E.B.; Ling, E.A.; Wang, Y.; Hassouna, M.M.; Mack, P. Induction of neuronal and inducible nitric oxide synthase in the motoneurons of spinal cord following transient abdominal aorta occlusion in rats. J. Surg. Res 1999, 87, 185–193. [Google Scholar]
- Gilling-Smith, G.L.; Worswick, L.; Knight, P.F.; Wolfe, J.H.; Mansfield, A.O. Surgical repair of thoracoabdominal aortic aneurysm: 10 years’ experience. Br. J. Surg 1995, 82, 624–629. [Google Scholar]
- Marsala, M.; Sorkin, L.S.; Yaksh, T.L. Transient spinal ischemia in rat: Characterization of spinal cord blood flow, extracellular amino acid release, and concurrent histopathological damage. J. Cereb. Blood Flow Metab 1994, 14, 604–614. [Google Scholar]
- Lewen, A.; Matz, P.; Chan, P.H. Free radical pathways in cns injury. J. Neurotrauma 2000, 17, 871–890. [Google Scholar]
- Barone, F.C.; Feuerstein, G.Z. Inflammatory mediators and stroke: New opportunities for novel therapeutics. J. Cereb. Blood Flow Metab 1999, 19, 819–834. [Google Scholar]
- Matsushita, K.; Wu, Y.; Qiu, J.; Lang-Lazdunski, L.; Hirt, L.; Waeber, C.; Hyman, B.T.; Yuan, J.; Moskowitz, M.A. Fas receptor and neuronal cell death after spinal cord ischemia. J. Neurosci 2000, 20, 6879–6887. [Google Scholar]
- De Haan, P.; Kalkman, C.J.; Jacobs, M.J. Pharmacologic neuroprotection in experimental spinal cord ischemia: A systematic review. J. Neurosurg. Anesthesiol 2001, 13, 3–12. [Google Scholar]
- Andrews, Z.B. Central mechanisms involved in the orexigenic actions of ghrelin. Peptides 2011, 32, 2248–2255. [Google Scholar]
- Andrews, Z.B. The extra-hypothalamic actions of ghrelin on neuronal function. Trends Neurosci 2011, 34, 31–40. [Google Scholar]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar]
- Nakazato, M.; Murakami, N.; Date, Y.; Kojima, M.; Matsuo, H.; Kangawa, K.; Matsukura, S. A role for ghrelin in the central regulation of feeding. Nature 2001, 409, 194–198. [Google Scholar]
- Shimizu, Y.; Nagaya, N.; Teranishi, Y.; Imazu, M.; Yamamoto, H.; Shokawa, T.; Kangawa, K.; Kohno, N.; Yoshizumi, M. Ghrelin improves endothelial dysfunction through growth hormone-independent mechanisms in rats. Biochem. Biophys. Res. Commun 2003, 310, 830–835. [Google Scholar]
- Masuda, Y.; Tanaka, T.; Inomata, N.; Ohnuma, N.; Tanaka, S.; Itoh, Z.; Hosoda, H.; Kojima, M.; Kangawa, K. Ghrelin stimulates gastric acid secretion and motility in rats. Biochem. Biophys. Res. Commun 2000, 276, 905–908. [Google Scholar]
- Xia, Q.; Pang, W.; Pan, H.; Zheng, Y.; Kang, J.S.; Zhu, S.G. Effects of ghrelin on the proliferation and secretion of splenic t lymphocytes in mice. Regul. Pept 2004, 122, 173–178. [Google Scholar]
- Wu, R.; Dong, W.; Cui, X.; Zhou, M.; Simms, H.H.; Ravikumar, T.S.; Wang, P. Ghrelin down-regulates proinflammatory cytokines in sepsis through activation of the vagus nerve. Ann. Surg 2007, 245, 480–486. [Google Scholar]
- Park, J.M.; Kakimoto, T.; Kuroki, T.; Shiraishi, R.; Fujise, T.; Iwakiri, R.; Fujimoto, K. Suppression of intestinal mucosal apoptosis by ghrelin in fasting rats. Exp. Biol. Med (Maywood) 2008, 233, 48–56. [Google Scholar]
- Miao, Y.; Xia, Q.; Hou, Z.; Zheng, Y.; Pan, H.; Zhu, S. Ghrelin protects cortical neuron against focal ischemia/reperfusion in rats. Biochem. Biophys. Res. Commun 2007, 359, 795–800. [Google Scholar]
- Chung, H.; Kim, E.; Lee, D.H.; Seo, S.; Ju, S.; Lee, D.; Kim, H.; Park, S. Ghrelin inhibits apoptosis in hypothalamic neuronal cells during oxygen-glucose deprivation. Endocrinology 2007, 148, 148–159. [Google Scholar]
- Johansson, I.; Destefanis, S.; Aberg, N.D.; Aberg, M.A.; Blomgren, K.; Zhu, C.; Ghe, C.; Granata, R.; Ghigo, E.; Muccioli, G.; et al. Proliferative and protective effects of growth hormone secretagogues on adult rat hippocampal progenitor cells. Endocrinology 2008, 149, 2191–2199. [Google Scholar]
- Jiang, X.; Ai, C.; Shi, E.; Nakajima, Y.; Ma, H. Neuroprotection against spinal cord ischemia-reperfusion injury induced by different ischemic postconditioning methods: Roles of phosphatidylinositol 3-kinase-akt and extracellular signal-regulated kinase. Anesthesiology 2009, 111, 1197–1205. [Google Scholar]
- Tarlov, I.M. Acute spinal cord compression paralysis. J. Neurosurg 1972, 36, 10–20. [Google Scholar]
- Ferens, D.M.; Yin, L.; Bron, R.; Hunne, B.; Ohashi-Doi, K.; Kitchener, P.D.; Sanger, G.J.; Witherington, J.; Shimizu, Y.; Furness, J.B. Functional and in situ hybridization evidence that preganglionic sympathetic vasoconstrictor neurons express ghrelin receptors. Neuroscience 2010, 166, 671–679. [Google Scholar]
- Kushimoto, S.; Okajima, K.; Uchiba, M.; Murakami, K.; Harada, N.; Okabe, H.; Takatsuki, K. Role of granulocyte elastase in ischemia/reperfusion injury of rat liver. Crit. Care Med 1996, 24, 1908–1912. [Google Scholar]
- Kodama, T.; Ashitani, J.; Matsumoto, N.; Kangawa, K.; Nakazato, M. Ghrelin treatment suppresses neutrophil-dominant inflammation in airways of patients with chronic respiratory infection. Pulm. Pharmacol. Ther 2008, 21, 774–779. [Google Scholar]
- Dixit, V.D.; Schaffer, E.M.; Pyle, R.S.; Collins, G.D.; Sakthivel, S.K.; Palaniappan, R.; Lillard, J.W., Jr; Taub, D.D. Ghrelin inhibits leptin- and activation-induced proinflammatory cytokine expression by human monocytes and t cells. J. Clin. Invest. 2004, 114, 57–66. [Google Scholar]
- Hattori, N. Expression, regulation and biological actions of growth hormone (gh) and ghrelin in the immune system. Growth Horm. IGF Res 2009, 19, 187–197. [Google Scholar]
- Konturek, P.C.; Brzozowski, T.; Engel, M.; Burnat, G.; Gaca, P.; Kwiecien, S.; Pajdo, R.; Konturek, S.J. Ghrelin ameliorates colonic inflammation. Role of nitric oxide and sensory nerves. J. Physiol. Pharmacol 2009, 60, 41–47. [Google Scholar]
- Kiyoshima, T.; Fukuda, S.; Matsumoto, M.; Iida, Y.; Oka, S.; Nakakimura, K.; Sakabe, T. Lack of evidence for apoptosis as a cause of delayed onset paraplegia after spinal cord ischemia in rabbits. Anesth. Analg 2003, 96, 839–846. [Google Scholar]
- Li, M.; Ona, V.O.; Chen, M.; Kaul, M.; Tenneti, L.; Zhang, X.; Stieg, P.E.; Lipton, S.A.; Friedlander, R.M. Functional role and therapeutic implications of neuronal caspase-1 and -3 in a mouse model of traumatic spinal cord injury. Neuroscience 2000, 99, 333–342. [Google Scholar]
- Okutan, O.; Solaroglu, I.; Beskonakli, E.; Taskin, Y. Recombinant human erythropoietin decreases myeloperoxidase and caspase-3 activity and improves early functional results after spinal cord injury in rats. J. Clin. Neurosci 2007, 14, 364–368. [Google Scholar]
- Savas, S.; Delibas, N.; Savas, C.; Sutcu, R.; Cindas, A. Pentoxifylline reduces biochemical markers of ischemia-reperfusion induced spinal cord injury in rabbits. Spinal Cord 2002, 40, 224–229. [Google Scholar]
- Xu, J.; Wang, S.; Lin, Y.; Cao, L.; Wang, R.; Chi, Z. Ghrelin protects against cell death of hippocampal neurons in pilocarpine-induced seizures in rats. Neurosci. Lett 2009, 453, 58–61. [Google Scholar]
- Hu, X.L.; Olsson, T.; Johansson, I.M.; Brannstrom, T.; Wester, P. Dynamic changes of the anti- and pro-apoptotic proteins Bcl-w, Bcl-2, and Bax with smac/diablo mitochondrial release after photothrombotic ring stroke in rats. Eur. J. Neurosci 2004, 20, 1177–1188. [Google Scholar]
- Lee, J.E.; Yenari, M.A.; Sun, G.H.; Xu, L.; Emond, M.R.; Cheng, D.; Steinberg, G.K.; Giffard, R.G. Differential neuroprotection from human heat shock protein 70 overexpression in in vitro and in vivo models of ischemia and ischemia-like conditions. Exp. Neurol 2001, 170, 129–139. [Google Scholar]
- Seok, Y.M.; Kim, J.; Choi, K.C.; Yoon, C.H.; Boo, Y.C.; Park, Y.; Park, K.M. Wen-pi-tang-hab-wu-ling-san attenuates kidney ischemia/reperfusion injury in mice. A role for antioxidant enzymes and heat-shock proteins. J. Ethnopharmacol 2007, 112, 333–340. [Google Scholar]
- Chen, Y.; Voegeli, T.S.; Liu, P.P.; Noble, E.G.; Currie, R.W. Heat shock paradox and a new role of heat shock proteins and their receptors as anti-inflammation targets. Inflamm. Allergy Drug Targets 2007, 6, 91–100. [Google Scholar]
- Robinson, M.B.; Tidwell, J.L.; Gould, T.; Taylor, A.R.; Newbern, J.M.; Graves, J.; Tytell, M.; Milligan, C.E. Extracellular heat shock protein 70: A critical component for motoneuron survival. J. Neurosci 2005, 25, 9735–9745. [Google Scholar]

| Group | TNF-α (pg/mL) | ||||
|---|---|---|---|---|---|
| 12 h | 24 h | 48 h | 72 h | ||
| Sham | 234 ± 22.3 | 257 ± 12.45 | 185 ± 33.21 | 188.5 ± 10.1 | |
| Control | 495 ± 5.67 * | 629 ± 11.98 * | 424 ± 50.2 * | 303.6 ± 18.0 * | |
| Ghrelin | 378 ± 32.16 ** | 425 ± 8.14 ** | 343 ± 42.1 ** | 232.2 ± 21.3 ** | |
| MPO (U/g wet tissue) | |||||
| 12 h | 24 h | 48 h | 72 h | ||
| Sham | 0.31 ± 0.03 | 0.35 ± 0.08 | 0.30 ± 0.06 | 0.25 ± 0.01 | |
| Control | 0.92 ± 0.18 * | 1.53 ± 0.21 * | 1.09 ± 0.27 * | 0.75 ± 0.13 * | |
| Ghrelin | 0.44 ± 0.06 ** | 0.88 ± 0.15 ** | 0.78 ± 0.08 ** | 0.32 ± 0.04 ** | |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Q.; Huang, C.; Meng, B.; Tang, T.; Shi, Q.; Yang, H. Acute Effect of Ghrelin on Ischemia/Reperfusion Injury in the Rat Spinal Cord. Int. J. Mol. Sci. 2012, 13, 9864-9876. https://doi.org/10.3390/ijms13089864
Zhang Q, Huang C, Meng B, Tang T, Shi Q, Yang H. Acute Effect of Ghrelin on Ischemia/Reperfusion Injury in the Rat Spinal Cord. International Journal of Molecular Sciences. 2012; 13(8):9864-9876. https://doi.org/10.3390/ijms13089864
Chicago/Turabian StyleZhang, Qin, Chen Huang, Bin Meng, Tiansi Tang, Qin Shi, and Huilin Yang. 2012. "Acute Effect of Ghrelin on Ischemia/Reperfusion Injury in the Rat Spinal Cord" International Journal of Molecular Sciences 13, no. 8: 9864-9876. https://doi.org/10.3390/ijms13089864




