Decidual Macrophages Are Significantly Increased in Spontaneous Miscarriages and Over-Express FasL: A Potential Role for Macrophages in Trophoblast Apoptosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Tissue Samples
2.2. Immunohistochemistry
2.3. TUNEL Assay
2.4. Double Immunofluorescence
- FasL/CD68. Frozen sections were thawed, fixed in acetone for 5 min at room temperature and rehydrated in PBS. After blocking for 15min with blocking solution (Ultra V Block, Lab Vision, Fremont, CA, USA), the sections were incubated with rabbit-Anti-Fas-L (Q20 Santa Cruz Biotechnologies, Santa Cruz, CA, USA) 1:100 overnight at 4 °C and washed with PBS; incubation at room temperature with goat-anti-rabbit Cy-3 (Jackson Dianova, Hamburg, DE) diluted 1:500 for 30 min followed. After washing with PBS, the slides were incubated at room temperature with mouse-anti-CD68 (clone EBM 11, Dako, Hamburg, Germany) diluted 1:100 for 1 h. Incubation with the goat-anti-mouse Cy-2 secondary antibody (Jackson Dianova, Hamburg, Germany) diluted 1:100 for 30 min at room temperature, followed.
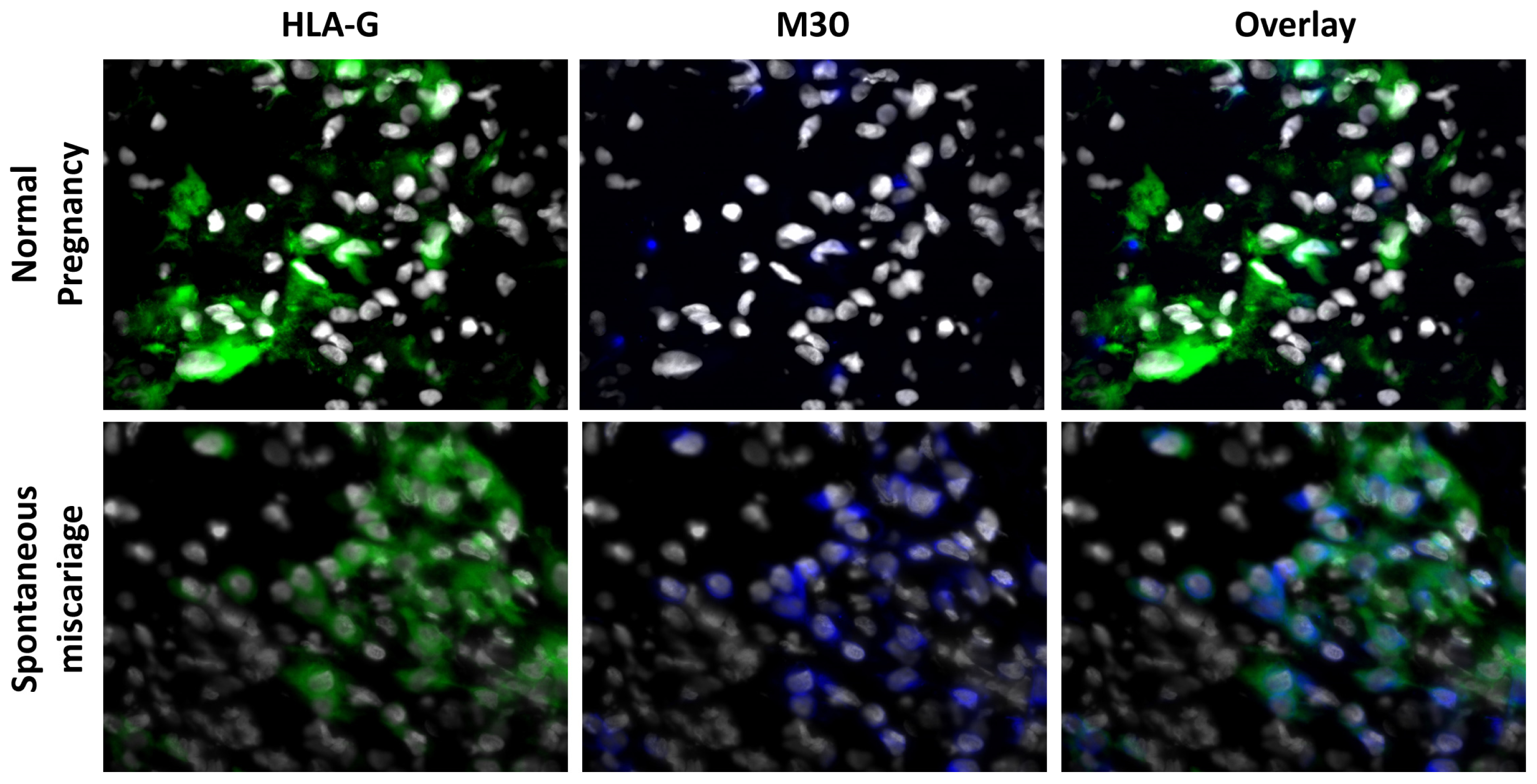
- HLA-G/M30. Thawing, fixation, rehydration and blocking were done as described above. The slides were then incubated with the biotinylated mouse-Anti-M30 (CytoDeath, clone M30, Enzo Life Sciences, Lörrach, DE, Germany) diluted 1:100 overnight at 4 °C, succeeded by the incubation at room temperature with streptavidin Cy-5 (Jackson Dianova, Hamburg, DE, Germany) diluted 1:400 for 30 min. Finally the slides were incubated with the FITC conjugated mouse-Anti-HLA-G (clone MEM G/9, Serotec, Oxford, UK) diluted 1:50, overnight at 4 °C.
2.5. Statistical Analysis
3. Results
3.1. Apoptosis Is Increased in Spontaneous Abortions Compared to Normal Pregnancies
3.2. Macrophage Population and Distribution Are Significantly Altered in Spontaneous Miscarriages
3.3. Macrophages Significantly Over-Express Fas ligand (FasL) in Spontaneous Miscarriages
4. Discussion
Acknowledgement
- Conflict of InterestsThe authors declare no conflict of interest.
References
- Macklon, N.S.; Geraedts, J.P.; Fauser, B.C. Conception to ongoing pregnancy: The “black box” of early pregnancy loss. Hum. Reprod. Update 2002, 8, 333–343. [Google Scholar]
- Barnea, E. Epidemiology, Etiology of Early Pregnancy Disorders. In The First Twelve Weeks of Gestation; Barnea, E., Hustin, J., Jaunieaux, E., Eds.; Springer-Verlag: Berlin, Germany, 1992; pp. 263–269. [Google Scholar]
- Warning, J.C.; McCracken, S.A.; Morris, J.M. A balancing act: Mechanisms by which the fetus avoids rejection by the maternal immune system. Reproduction 2011, 141, 715–724. [Google Scholar]
- Piccinni, M.P. T cell tolerance towards the fetal allograft. J. Reprod. Immunol 2010, 85, 71–75. [Google Scholar]
- Yagel, S. The developmental role of natural killer cells at the fetal-maternal interface. Am. J. Obstet. Gynecol 2009, 201, 344–350. [Google Scholar]
- Houser, B.L. Decidual macrophages and their roles at the maternal-fetal interface. Yale J. Biol. Med 2012, 85, 105–118. [Google Scholar]
- Nagamatsu, T.; Schust, D.J. The contribution of macrophages to normal and pathological pregnancies. Am. J. Reprod. Immunol 2010, 63, 460–471. [Google Scholar]
- Nagata, S. Fas and Fas ligand: A death factor and its receptor. Adv. Immunol 1994, 57, 129–144. [Google Scholar]
- Makrigiannakis, A.; Zoumakis, E.; Kalantaridou, S.; Coutifaris, C.; Margioris, A.N.; Coukos, G.; Rice, K.C.; Gravanis, A.; Chrousos, G.P. Corticotropin-releasing hormone promotes blastocyst implantation and early maternal tolerance. Nat. Immunol 2001, 2, 1018–1024. [Google Scholar]
- Minas, V.; Jeschke, U.; Kalantaridou, S.N.; Richter, D.U.; Reimer, T.; Mylonas, I.; Friese, K.; Makrigiannakis, A. Abortion is associated with increased expression of FasL in decidual leukocytes and apoptosis of extravillous trophoblasts: A role for CRH and urocortin. Mol. Hum. Reprod 2007, 13, 663–673. [Google Scholar]
- Wongweragiat, S.; Searle, R.F.; Bulmer, J.N. Expression of Fas/Fas ligand by decidual leukocytes in hydatidiform mole. Biol. Reprod 2001, 64, 784–789. [Google Scholar]
- Mizuno, M.; Aoki, K.; Kimbara, T. Functions of macrophages in human decidual tissue in early pregnancy. Am. J. Reprod. Immunol 1994, 31, 180–188. [Google Scholar]
- Parhar, R.S.; Yagel, S.; Lala, P.K. PGE2-mediated immunosuppression by first trimester human decidual cells blocks activation of maternal leukocytes in the decidua with potential anti-trophoblast activity. Cell Immunol 1989, 120, 61–74. [Google Scholar]
- Heikkinen, J.; Mottonen, M.; Komi, J.; Alanen, A.; Lassila, O. Phenotypic characterization of human decidual macrophages. Clin. Exp. Immunol 2003, 131, 498–505. [Google Scholar]
- Miwa, N.; Hayakawa, S.; Miyazaki, S.; Myojo, S.; Sasaki, Y.; Sakai, M.; Takikawa, O.; Saito, S. IDO expression on decidual and peripheral blood dendritic cells and monocytes/macrophages after treatment with CTLA-4 or interferon-gamma increase in normal pregnancy but decrease in spontaneous abortion. Mol. Hum. Reprod 2005, 11, 865–870. [Google Scholar]
- Munn, D.H.; Zhou, M.; Attwood, J.T.; Bondarev, I.; Conway, S.J.; Marshall, B.; Brown, C.; Mellor, A.L. Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 1998, 281, 1191–1193. [Google Scholar]
- Renaud, S.J.; Graham, C.H. The role of macrophages in utero-placental interactions during normal and pathological pregnancy. Immunol. Invest 2008, 37, 535–564. [Google Scholar]
- Saito, S.; Nakashima, A.; Shima, T.; Ito, M. Th1/Th2/Th17 and regulatory T-cell paradigm in pregnancy. Am. J. Reprod. Immunol 2010, 63, 601–610. [Google Scholar]
- Gordon, S. Alternative activation of macrophages. Nat. Rev. Immunol 2003, 3, 23–35. [Google Scholar]
- Mills, C.D.; Kincaid, K.; Alt, J.M.; Heilman, M.J.; Hill, A.M. M-1/M-2 macrophages and the Th1/Th2 paradigm. J. Immunol 2000, 164, 6166–6173. [Google Scholar]
- Devaraj, S.; Jialal, I. C-reactive protein polarizes human macrophages to an M1 phenotype and inhibits transformation to the M2 phenotype. Arterioscler. Thromb. Vasc. Biol 2011, 31, 1397–1402. [Google Scholar]
- Chieppa, M.; Bianchi, G.; Doni, A.; Del Prete, A.; Sironi, M.; Laskarin, G.; Monti, P.; Piemonti, L.; Biondi, A.; Mantovani, A.; et al. Cross-linking of the mannose receptor on monocyte-derived dendritic cells activates an anti-inflammatory immunosuppressive program. J. Immunol 2003, 171, 4552–4560. [Google Scholar]
- Gustafsson, C.; Mjosberg, J.; Matussek, A.; Geffers, R.; Matthiesen, L.; Berg, G.; Sharma, S.; Buer, J.; Ernerudh, J. Gene expression profiling of human decidual macrophages: Evidence for immunosuppressive phenotype. PLoS One 2008, 3, e2078. [Google Scholar]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Invest 2012, 122, 787–795. [Google Scholar]
- Vassiliadou, N.; Bulmer, J.N. Immunohistochemical evidence for increased numbers of ‘classic’ CD57+ natural killer cells in the endometrium of women suffering spontaneous early pregnancy loss. Hum. Reprod 1996, 11, 1569–1574. [Google Scholar]
- Quack, K.C.; Vassiliadou, N.; Pudney, J.; Anderson, D.J.; Hill, J.A. Leukocyte activation in the decidua of chromosomally normal and abnormal fetuses from women with recurrent abortion. Hum. Reprod 2001, 16, 949–955. [Google Scholar]
- Dockrell, D.H.; Badley, A.D.; Villacian, J.S.; Heppelmann, C.J.; Algeciras, A.; Ziesmer, S.; Yagita, H.; Lynch, D.H.; Roche, P.C.; Leibson, P.J.; et al. The expression of Fas Ligand by macrophages and its upregulation by human immunodeficiency virus infection. J. Clin. Invest 1998, 101, 2394–2405. [Google Scholar]
- Borges, V.M.; Falcao, H.; Leite-Junior, J.H.; Alvim, L.; Teixeira, G.P.; Russo, M.; Nobrega, A.F.; Lopes, M.F.; Rocco, P.M.; Davidson, W.F.; et al. Fas ligand triggers pulmonary silicosis. J. Exp. Med 2001, 194, 155–164. [Google Scholar]
- Dace, D.S.; Khan, A.A.; Stark, J.L.; Kelly, J.; Cross, A.H.; Apte, R.S. Interleukin-10 overexpression promotes Fas-ligand-dependent chronic macrophage-mediated demyelinating polyneuropathy. PLoS One 2009, 4, e7121. [Google Scholar]
- Angioni, S.; Petraglia, F.; Gallinelli, A.; Cossarizza, A.; Franceschi, C.; Muscettola, M.; Genazzani, A.D.; Surico, N.; Genazzani, A.R. Corticotropin-releasing hormone modulates cytokines release in cultured human peripheral blood mononuclear cells. Life Sci 1993, 53, 1735–1742. [Google Scholar]
- Pestka, A.; Toth, B.; Kuhn, C.; Hofmann, S.; Wiest, I.; Wypior, G.; Friese, K.; Jeschke, U. Retinoid X receptor alpha and retinoids are key regulators in apoptosis of trophoblasts of patients with recurrent miscarriages. J. Mol. Endocrinol 2011, 47, 145–156. [Google Scholar]


| Antibody (AB) | AB Incubation Conditions | Blocking Solution | Blocking Conditions |
|---|---|---|---|
| mouse-Anti-CD68 (clone EBM 11, Dako, Hamburg, Germany) | 1:100, 1, RT | Power block (Dako, Hamburg, DE, Germany) | 1:10 in distilled water, 3 min, RT |
| mouse-Anti-M30 (CytoDeath, clone M30, Enzo Life Sciences, Lörrach, Germany) | 1:400, overnight, 4 °C | Ultra V Block (Labvision, Fremont, CA, USA) | undiluted, 45 min, RT |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guenther, S.; Vrekoussis, T.; Heublein, S.; Bayer, B.; Anz, D.; Knabl, J.; Navrozoglou, I.; Dian, D.; Friese, K.; Makrigiannakis, A.; et al. Decidual Macrophages Are Significantly Increased in Spontaneous Miscarriages and Over-Express FasL: A Potential Role for Macrophages in Trophoblast Apoptosis. Int. J. Mol. Sci. 2012, 13, 9069-9080. https://doi.org/10.3390/ijms13079069
Guenther S, Vrekoussis T, Heublein S, Bayer B, Anz D, Knabl J, Navrozoglou I, Dian D, Friese K, Makrigiannakis A, et al. Decidual Macrophages Are Significantly Increased in Spontaneous Miscarriages and Over-Express FasL: A Potential Role for Macrophages in Trophoblast Apoptosis. International Journal of Molecular Sciences. 2012; 13(7):9069-9080. https://doi.org/10.3390/ijms13079069
Chicago/Turabian StyleGuenther, Sabine, Thomas Vrekoussis, Sabine Heublein, Birgit Bayer, David Anz, Julia Knabl, Iordanis Navrozoglou, Darius Dian, Klaus Friese, Antonis Makrigiannakis, and et al. 2012. "Decidual Macrophages Are Significantly Increased in Spontaneous Miscarriages and Over-Express FasL: A Potential Role for Macrophages in Trophoblast Apoptosis" International Journal of Molecular Sciences 13, no. 7: 9069-9080. https://doi.org/10.3390/ijms13079069






