Green Extraction of Natural Products: Concept and Principles
Abstract
:1. Introduction
2. Definition of Green Extraction
3. The Six Principles of Green Extraction
- Principle 1: Innovation by selection of varieties and use of renewable plant resources.
- Principle 2: Use of alternative solvents and principally water or agro-solvents.
- Principle 3: Reduce energy consumption by energy recovery and using innovative technologies.
- Principle 4: Production of co-products instead of waste to include the bio-and agro-refining industry.
- Principle 5: Reduce unit operations and favour safe, robust and controlled processes.
- Principle 6: Aim for a non denatured and biodegradable extract without contaminants.
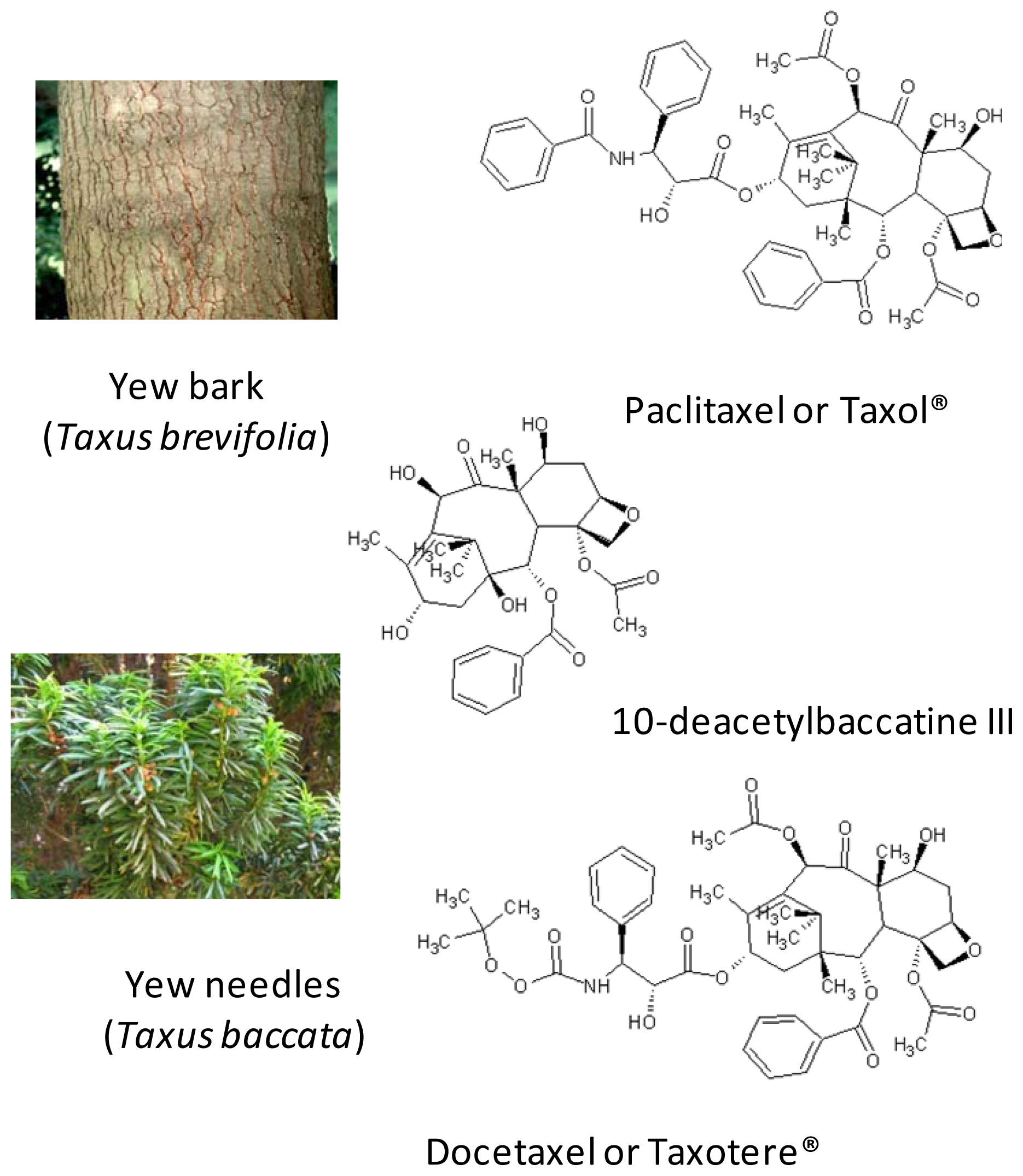
3.1. Principle 1: Innovation by the Selection of Varieties and the Use of Renewable Plant Resources
3.2. Principle 2: Use of Alternative Solvents and Principally Water or Agro-Solvents Issued from Agricultural Resources
3.3. Principle 3: Reduce Energy Consumption by Energy Recovery and Using Innovative Technologies
3.4. Principle 4: Production of Co-Products Instead of Waste to Include the Bio and Agro Refining Industry
- Waste is any material that industry has the only choice to eliminate by waste disposal centre, incineration, or landfill.
- By-product is a residual product that appears during the extraction process. It is unintentional and unpredictable. It can be used directly or be an ingredient in another production process to manufacture another finished product. By-products have economic value, for example, oil cakes (rape, sunflower, flax), cereal grain (wheat, barley), beet pulp, potato fibre and proteins, etc.
- Co-product is a material, intentional and inevitable, created during a single manufacturing process and at the same time as the main product. The main final product and co-product must always meet specifications for their characteristics, and each may be used directly for a particular application.
3.5. Principle 5: Reduce Unit Operations through Technical Innovation and Favour Safe, Robust and Controlled Processes
3.6. Principle 6: Aim for a Non Denatured and Biodegradable Extract without Contaminants with “Green” Values
4. Conclusions
Acknowledgments
References
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: Oxford, UK, 2000. [Google Scholar]
- Chemat, F. Eco-Extraction du Végétal; Dunod; Paris, France, 2011. [Google Scholar]
- Virot, M.; Tomao, V.; Ginies, C.; Chemat, F. Total lipid extraction of food using d-limonene as an alternative to n-hexane. Chromatographia 2008, 68, 311–313. [Google Scholar]
- Lapkin, A.; Plucinski, P.K.; Cutler, M. Comparative assessment of technologies for extraction of artemisinin. J. Nat. Prod 2006, 69, 1653–1664. [Google Scholar]
- Teo, C.C.; Tana, S.N.; Yong, J.W.; Hew, C.S.; Ong, E.S. Pressurized hot water extraction (PHWE). J. Chromatogr. A 2010, 1217, 2484–2494. [Google Scholar]
- Mantegna, S.; Binello, A.; Boffa, L.; Giorgis, M.; Cena, C.; Cravotto, G. A one-pot ultrasound-assisted water extraction/cyclodextrin encapsulation of resveratrol from Polygonum cuspidatum. Food Chem 2012, 130, 746–750. [Google Scholar]
- Herrero, M.; Cifuentes, A.; Ibañez, E. Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, food-by-products, algae and microalgae: A review. Food Chem 2006, 98, 136–148. [Google Scholar]
- Abert Vian, M.; Fernandez, X.; Visinoni, F.; Chemat, F. Microwave hydro-diffusion and gravity: A new device for extraction essential oils. J. Chromatogr. A 2008, 1190, 14–17. [Google Scholar]
- Sawamura, M. Citrus Essential Oil: Flavor and Fragrance; Wiley: Weinheim, Germany, 2010. [Google Scholar]
- Lebovka, N.; Vorobiev, E.; Chemat, F. Enhancing Extraction Processes in the Food Industry; CRC Press: Cambridge, UK, 2011. [Google Scholar]
- Armenta, S.; Garrigues, S.; de la Guardia, M. Green analytical chemistry. Trends Anal. Chem 2008, 27, 497–511. [Google Scholar]
- Mason, T.; Chemat, F.; Vinatoru, M. The extraction of natural products using ultrasound and microwaves. Curr. Org. Chem 2011, 15, 237–247. [Google Scholar]



| Solvent | Extraction Technique (Application) | Solvent Power | Health & Safety | Cost | Environmental Impact | ||
|---|---|---|---|---|---|---|---|
| Polar | Weakly Polar | Non-Polar | |||||
| Solvent-free | Microwave Hydrodiffusion and Gravity (antioxidants, essential oils) | +++ | + | +++ | + | +++ | |
| Pulse Electric Field (antioxidants, pigments) | +++ | + | +++ | + | +++ | ||
| Water | Steam distillation (essential oils) | ++ | + | + | ++ | + | |
| Microwave-assisted distillation (essential oils) | +++ | +++ | + | + | + | ++ | |
| Extraction by sub-critical water (Aromas) | + | ++ | + | + | + | ||
| CO2 | Supercritical fluid extraction (decaffeination of tea and coffee) | − | + | +++ | + | + | + |
| Ionic liquids | Ammonium salts (Artemisinin) | − | + | +++ | − | − | ++ |
| Agrosolvents | Ethanol (pigments and antioxidants) | + | + | − | − | ++ | + |
| Glycerol (polyphenols) | + | + | − | − | + | + | |
| Terpenes such as d-limonene (fats and oils) | − | − | ++ | − | + | + | |
| Petrochemical solvents | n-Hexane (fats and oils) | − | + | +++ | −−− | ++ | −−− |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chemat, F.; Vian, M.A.; Cravotto, G. Green Extraction of Natural Products: Concept and Principles. Int. J. Mol. Sci. 2012, 13, 8615-8627. https://doi.org/10.3390/ijms13078615
Chemat F, Vian MA, Cravotto G. Green Extraction of Natural Products: Concept and Principles. International Journal of Molecular Sciences. 2012; 13(7):8615-8627. https://doi.org/10.3390/ijms13078615
Chicago/Turabian StyleChemat, Farid, Maryline Abert Vian, and Giancarlo Cravotto. 2012. "Green Extraction of Natural Products: Concept and Principles" International Journal of Molecular Sciences 13, no. 7: 8615-8627. https://doi.org/10.3390/ijms13078615
APA StyleChemat, F., Vian, M. A., & Cravotto, G. (2012). Green Extraction of Natural Products: Concept and Principles. International Journal of Molecular Sciences, 13(7), 8615-8627. https://doi.org/10.3390/ijms13078615







