Optimization of the Extraction of Anthocyanins from the Fruit Skin of Rhodomyrtus tomentosa (Ait.) Hassk and Identification of Anthocyanins in the Extract Using High-Performance Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (HPLC-ESI-MS)
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization of the Anthocyanin Extraction
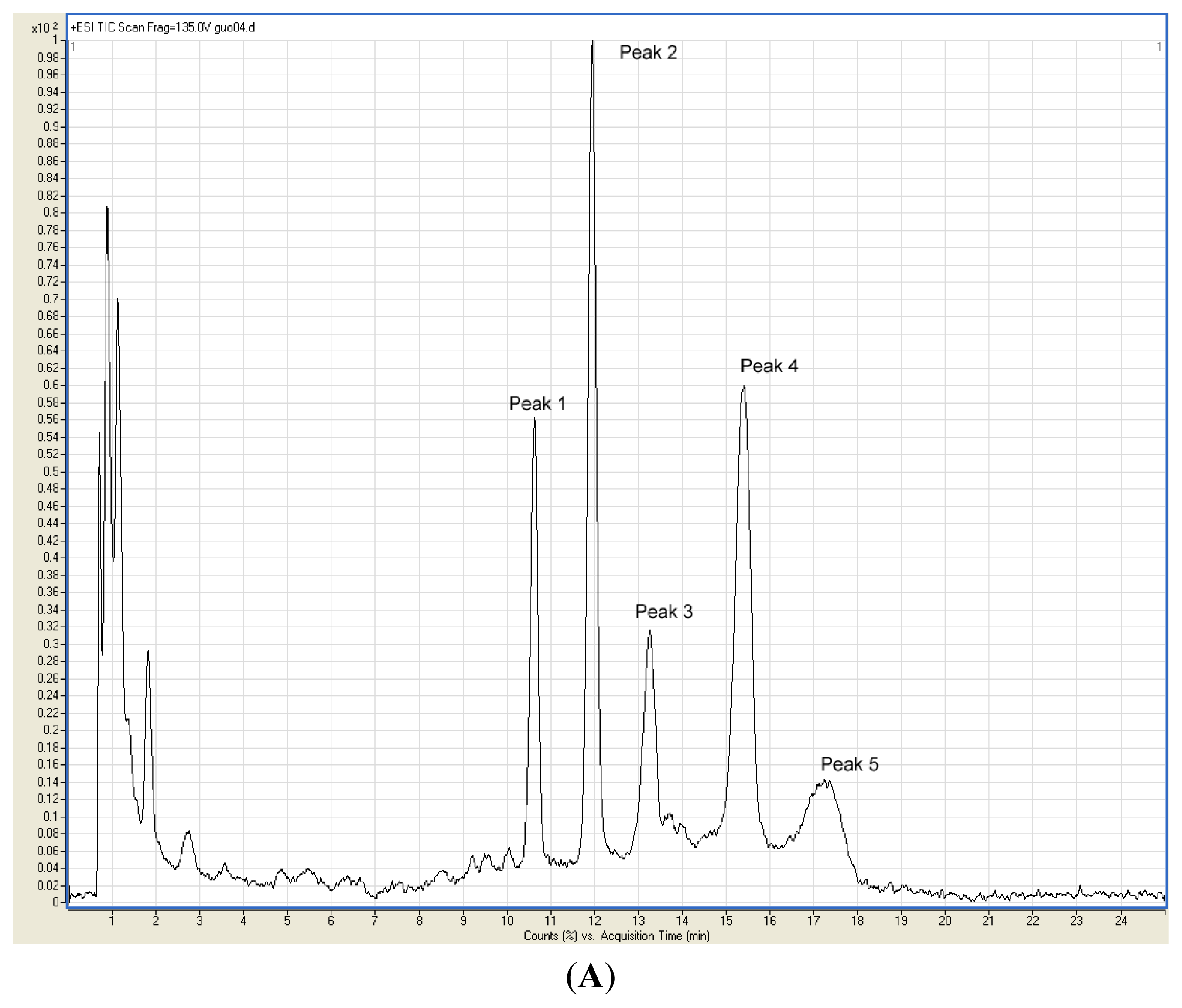
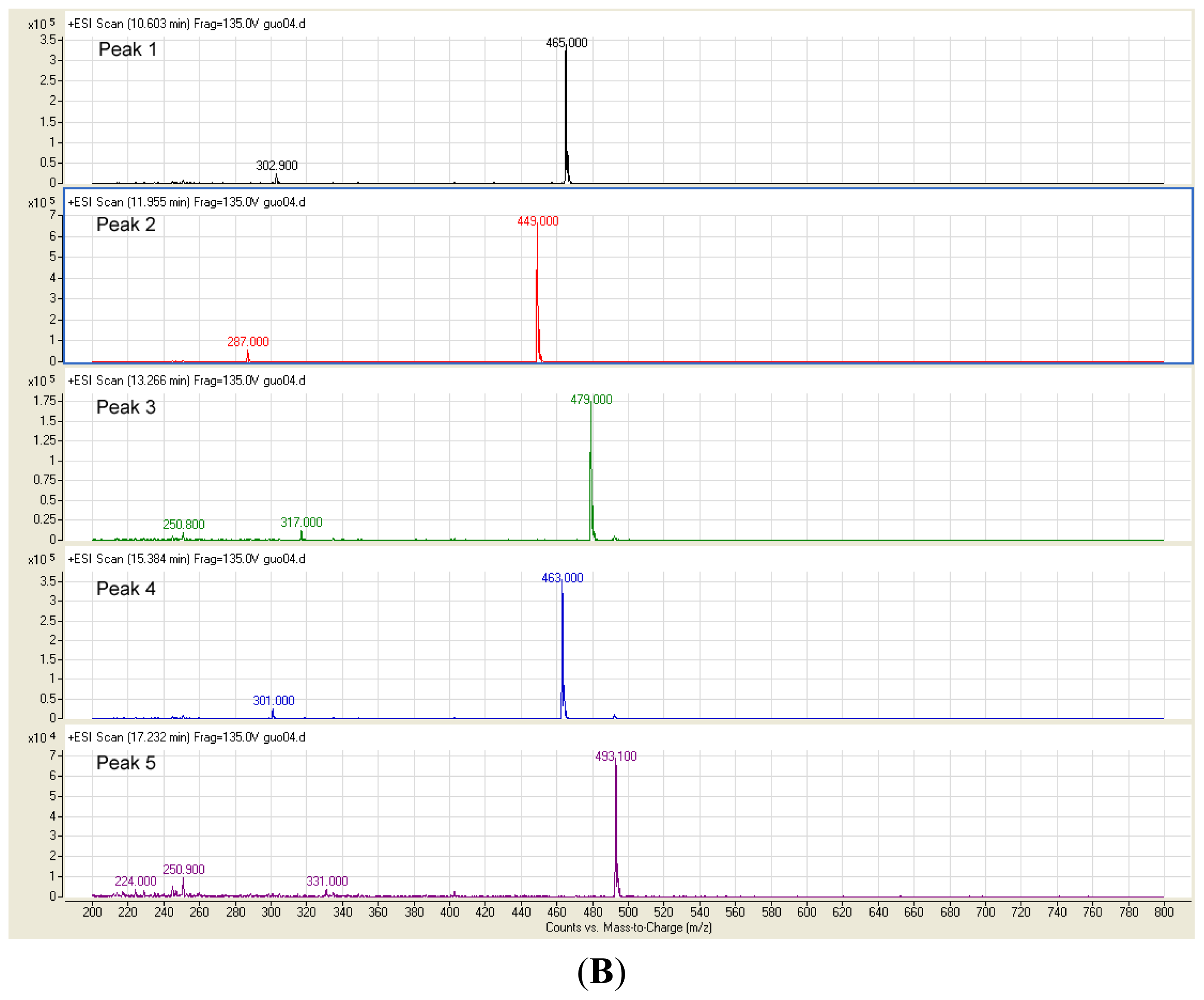
2.2. Identification of Anthocyanins in the Extract
3. Experimental Section
3.1. Sample Preparation and Chemicals
3.2. Extraction of Anthocyanins
3.3. Total Anthocyanin Content Measurement
3.4. Experimental Design for Optimization
3.5. Identification of Anthocyanins in Fruit Skin Extract by HPLC-ESI-MS
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Nabae, K.; Hayashi, S.M.; Kawabe, M.; Ichihara, T.; Hagiwara, A.; Tamano, S.; Tsushima, Y.; Uchida, K.; Koda, T.; Nakamura, M.; et al. A 90-day oral toxicity study of purple corn color, a natural food colorant, in F344 rats. Food Chem. Toxicol 2008, 46, 774–780. [Google Scholar]
- He, J.; Giusti, M.M. Anthocyanins: Natural colorants with health-promoting properties. Annu. Rev. Food Sci. Technol 2010, 1, 163–187. [Google Scholar]
- Felgines, C.; Talavera, S.; Texier, O.; Besson, C.; Fogliano, V.; Lamaison, J.L.; la Fauci, L.; Galvano, G.; Remesy, C.; Galvano, F. Absorption and metabolism of red orange juice anthocyanins in rats. Br. J. Nutr 2006, 95, 898–904. [Google Scholar]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem 2006, 54, 4069–4075. [Google Scholar]
- Ghosh, D.; Konishi, T. Anthocyanins and anthocyanin-rich extracts: Role in diabetes and eye function. Asia Pac. J. Clin. Nutr 2007, 16, 200–208. [Google Scholar]
- Saising, J.; Ongsakul, M.; Voravuthikunchai, S.P. Rhodomyrtus tomentosa (Aiton) Hassk. ethanol extract and rhodomyrtone: A potential strategy for the treatment of biofilm-forming staphylococci. J. Med. Microbiol 2011, 60, 1793–1800. [Google Scholar]
- Bagchi, D.; Roy, S.; Patel, V.; He, G.; Khanna, S.; Ojha, N.; Phillips, C.; Ghosh, S.; Bagchi, M.; Sen, C.K. Safety and whole-body antioxidant potential of a novel anthocyanin-rich formulation of edible berries. Mol. Cell. Biochem 2006, 281, 197–209. [Google Scholar]
- Bechtold, T.; Mahmud-Ali, A.; Mussak, R. Anthocyanin dyes extracted from grape pomace for the purpose of textile dyeing. J. Sci. Food Agric 2007, 87, 2589–2595. [Google Scholar]
- Mundra, P.; Desai, K.; Lele, S.S. Application of response surface methodology to cell immobilization for the production of palatinose. Bioresour. Technol 2007, 98, 2892–2896. [Google Scholar]
- Ku, C.S.; Mun, S.P. Optimization of the extraction of anthocyanin from Bokbunja (Rubus coreanus Miq.) marc produced during traditional wine processing and characterization of the extracts. Bioresour. Technol 2008, 99, 8325–8330. [Google Scholar]
- Zou, T.B.; Wang, M.; Gan, R.Y.; Ling, W.H. Optimization of ultrasound-assisted extraction of anthocyanins from mulberry, using response surface methodology. Int. J. Mol. Sci 2011, 12, 3006–3017. [Google Scholar]
- Yang, Z.; Zhai, W. Optimization of microwave-assisted extraction of anthocyanins from purple corn (Zea mays L.) cob and identification with HPLC-MS. Innov. Food Sci. Emerg 2010, 11, 470–476. [Google Scholar]
- Le Man, H.; Behera, S.; Park, H. Optimization of operational parameters for ethanol production from Korean food waste leachate. Int. J. Environ. Sci. Technol 2010, 7, 157–164. [Google Scholar]
- Chauhan, B.; Gupta, R. Application of statistical experimental design for optimization of alkaline protease production from Bacillus sp. RGR-14. Process. Biochem 2004, 39, 2115–2122. [Google Scholar]
- Koponen, J.M.; Happonen, A.M.; Mattila, P.H.; Torronen, A.R. Contents of anthocyanins and ellagitannins in selected foods consumed in Finland. J. Agric. Food Chem 2007, 55, 1612–1619. [Google Scholar]
- Nicoue, E.E.; Savard, S.; Belkacemi, K. Anthocyanins in wild blueberries of Quebec: Extraction and identification. J. Agric. Food Chem 2007, 55, 5626–5635. [Google Scholar]
- Guo, H.; Ling, W.; Wang, Q.; Liu, C.; Hu, Y.; Xia, M.; Feng, X.; Xia, X. Effect of anthocyanin-rich extract from black rice (Oryza sativa L. indica) on hyperlipidemia and insulin resistance in fructose-fed rats. Plant Foods Hum. Nutr 2007, 62, 1–6. [Google Scholar]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int 2005, 88, 1269–1278. [Google Scholar]
- Zou, T.; Wang, D.; Guo, H.; Zhu, Y.; Luo, X.; Liu, F.; Ling, W. Optimization of microwave-assisted extraction of anthocyanins from mulberry and identification of anthocyanins in extract using HPLC-ESI-MS. J. Food Sci 2012, 77, C46–C50. [Google Scholar]
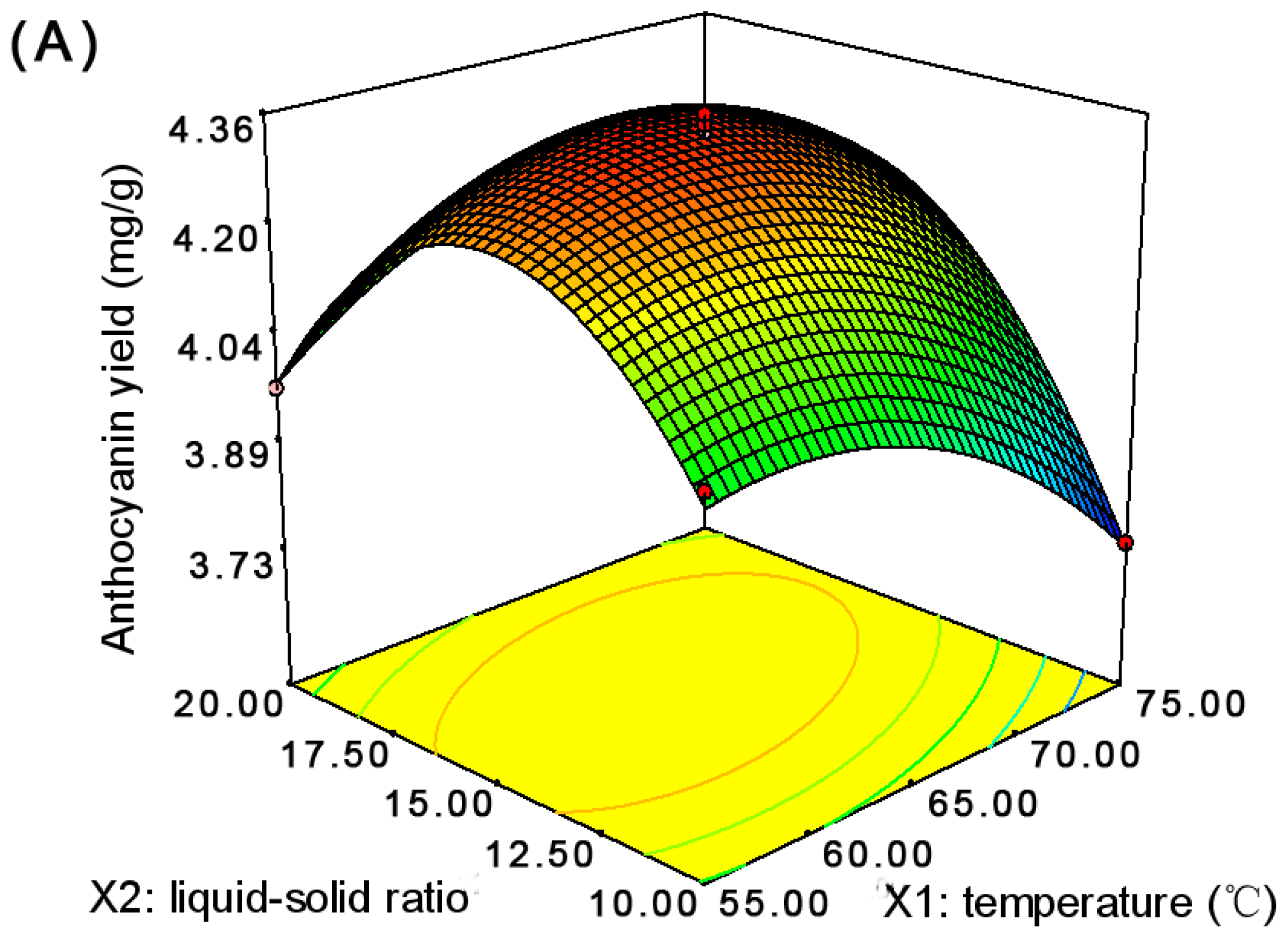
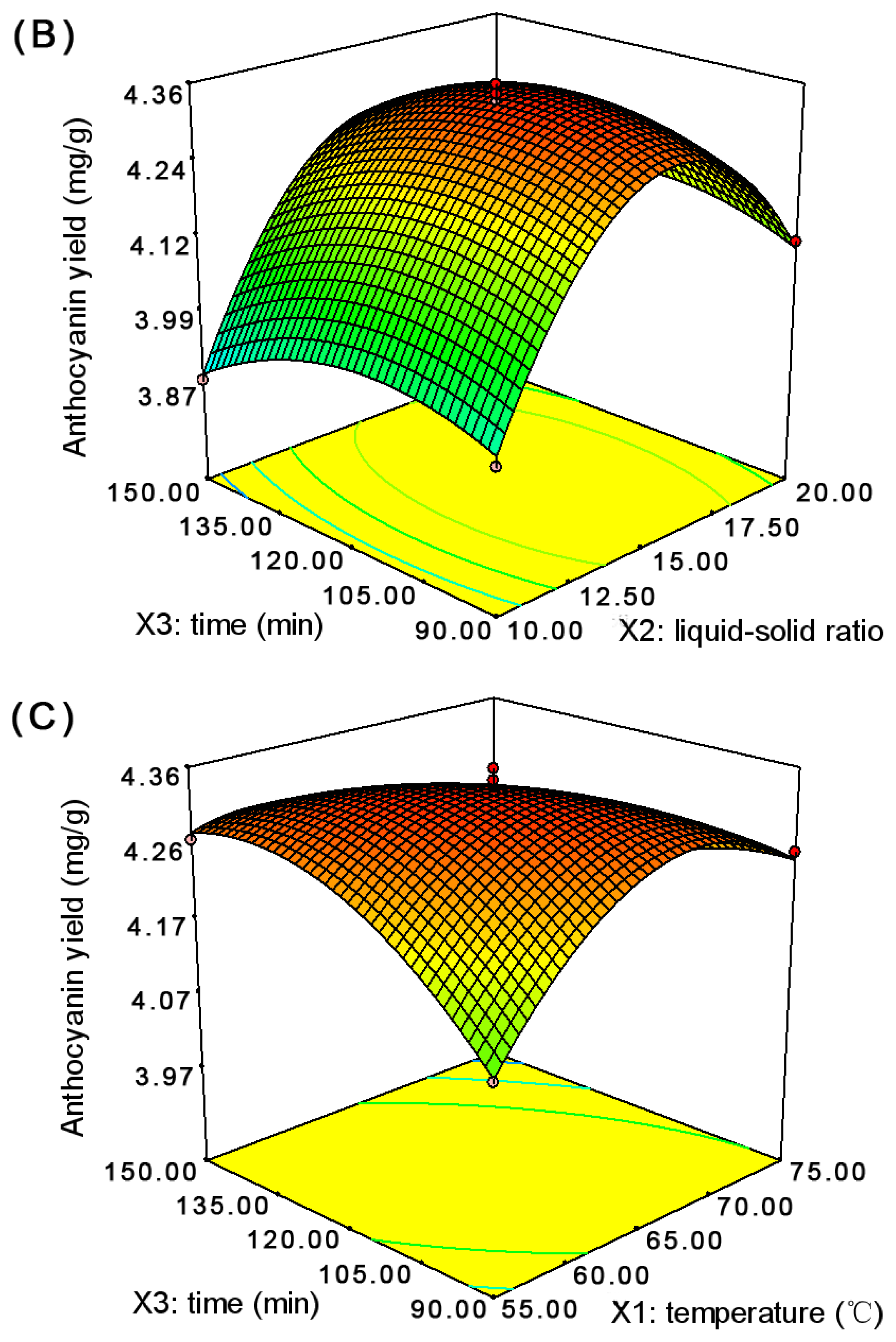
| Run | Temperature (°C) | Liquid-Solid Ratio (v/w) | Time (min) | Anthocyanin Yield a (mg/g) | |
|---|---|---|---|---|---|
| Observed | Predicted | ||||
| 1 | 65 | 20 | 150 | 4.088 | 4.071 |
| 2 | 65 | 15 | 120 | 4.358 | 4.337 |
| 3 | 55 | 10 | 120 | 4.048 | 4.027 |
| 4 | 75 | 10 | 120 | 3.737 | 3.730 |
| 5 | 55 | 15 | 150 | 4.268 | 4.278 |
| 6 | 75 | 15 | 150 | 3.983 | 3.979 |
| 7 | 55 | 20 | 120 | 3.968 | 3.975 |
| 8 | 65 | 10 | 150 | 3.877 | 3.888 |
| 9 | 65 | 15 | 120 | 4.343 | 4.337 |
| 10 | 65 | 15 | 120 | 4.313 | 4.337 |
| 11 | 65 | 10 | 90 | 3.938 | 3.955 |
| 12 | 65 | 15 | 120 | 4.328 | 4.337 |
| 13 | 55 | 15 | 90 | 4.103 | 4.107 |
| 14 | 75 | 20 | 120 | 4.088 | 4.109 |
| 15 | 65 | 20 | 90 | 4.108 | 4.097 |
| 16 | 75 | 15 | 90 | 4.253 | 4.243 |
| 17 | 65 | 15 | 120 | 4.343 | 4.337 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 0.5600 | 9 | 0.0620 | 138.63 | <0.0001 |
| X1 | 0.0130 | 1 | 0.0130 | 29.55 | 0.0010 |
| X2 | 0.0530 | 1 | 0.0530 | 118.22 | <0.0001 |
| X3 | 0.0043 | 1 | 0.0043 | 9.62 | 0.0173 |
| X1X2 | 0.0460 | 1 | 0.0460 | 103.32 | <0.0001 |
| X1X3 | 0.0470 | 1 | 0.0470 | 105.24 | <0.0001 |
| X2X3 | 0.0004 | 1 | 0.0004 | 0.93 | 0.3658 |
| X12 | 0.0550 | 1 | 0.0550 | 121.47 | <0.0001 |
| X22 | 0.2900 | 1 | 0.2900 | 647.30 | <0.0001 |
| X32 | 0.0210 | 1 | 0.0210 | 47.72 | 0.0002 |
| Residual | 0.0031 | 7 | 0.0004 | ||
| Lack of Fit | 0.0020 | 3 | 0.0006 | 2.25 | 0.2245 |
| Pure Error | 0.0012 | 4 | 0.0002 | ||
| Cor Total | 0.5600 | 16 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, G.-L.; Guo, H.-H.; Sun, Y.-M. Optimization of the Extraction of Anthocyanins from the Fruit Skin of Rhodomyrtus tomentosa (Ait.) Hassk and Identification of Anthocyanins in the Extract Using High-Performance Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (HPLC-ESI-MS). Int. J. Mol. Sci. 2012, 13, 6292-6302. https://doi.org/10.3390/ijms13056292
Liu G-L, Guo H-H, Sun Y-M. Optimization of the Extraction of Anthocyanins from the Fruit Skin of Rhodomyrtus tomentosa (Ait.) Hassk and Identification of Anthocyanins in the Extract Using High-Performance Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (HPLC-ESI-MS). International Journal of Molecular Sciences. 2012; 13(5):6292-6302. https://doi.org/10.3390/ijms13056292
Chicago/Turabian StyleLiu, Guo-Ling, Hong-Hui Guo, and Yuan-Ming Sun. 2012. "Optimization of the Extraction of Anthocyanins from the Fruit Skin of Rhodomyrtus tomentosa (Ait.) Hassk and Identification of Anthocyanins in the Extract Using High-Performance Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (HPLC-ESI-MS)" International Journal of Molecular Sciences 13, no. 5: 6292-6302. https://doi.org/10.3390/ijms13056292
APA StyleLiu, G.-L., Guo, H.-H., & Sun, Y.-M. (2012). Optimization of the Extraction of Anthocyanins from the Fruit Skin of Rhodomyrtus tomentosa (Ait.) Hassk and Identification of Anthocyanins in the Extract Using High-Performance Liquid Chromatography-Electrospray Ionization-Mass Spectrometry (HPLC-ESI-MS). International Journal of Molecular Sciences, 13(5), 6292-6302. https://doi.org/10.3390/ijms13056292





