In Vitro Phytotoxicity and Antioxidant Activity of Selected Flavonoids
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phytotoxic Activity
2.2. Antioxidant Activity
3. Experimental Section
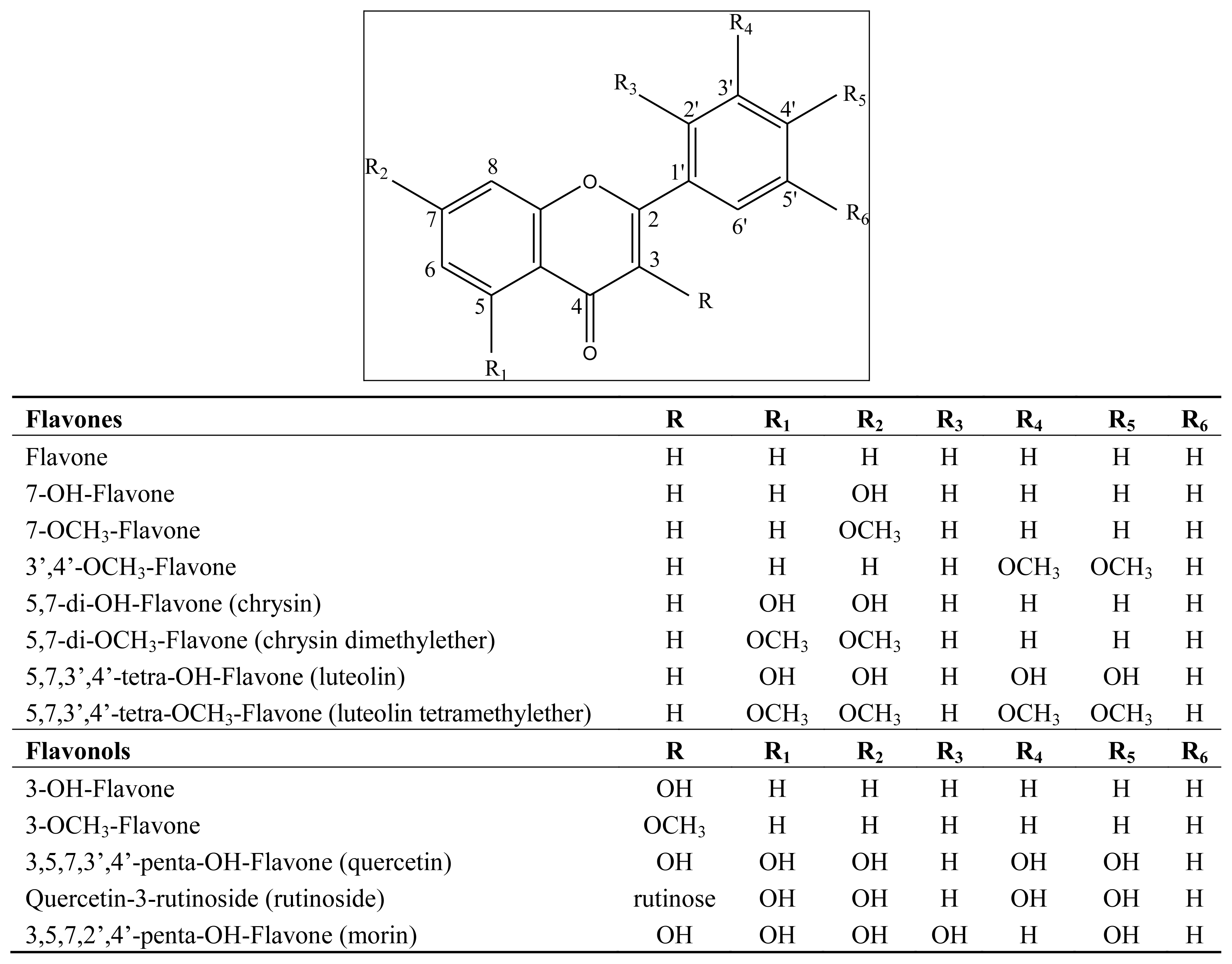
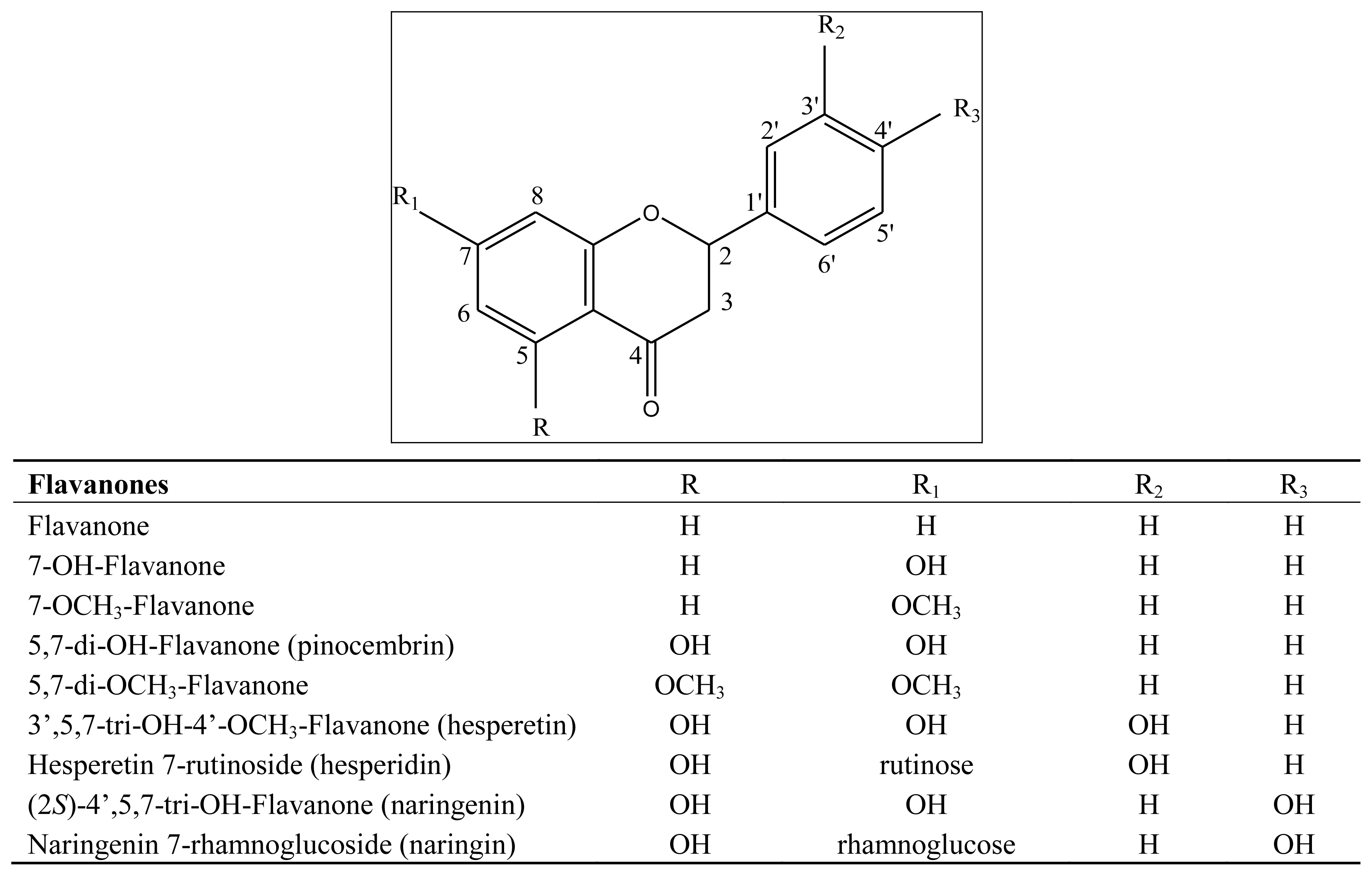
3.1. Flavonoids
3.2. Flavonoid Solutions
3.3. Phytotoxic Assay
3.4. Bleaching of the Free-Radical 1,1-Diphenyl-2-picrylhydrazyl (DPPH Test)
3.5. Statistical Analysis
4. Conclusions
References
- Dixon, R.A.; Achine, L.; Kota, P.; Liu, C.J.; Srinivasa-Reddy, M.S.; Wang, L. The phenylpropanoids pathway and plant defence—A genomic perspective. Mol. Plant Pathol 2002, 6, 371–390. [Google Scholar]
- Kinoshita, T.; Lepp, Z.; Kawai, Y.; Terao, J.; Chuman, H. An integrated database of flavonoids. Biofactors 2006, 26, 179–188. [Google Scholar]
- Mol, J.; Grotewold, E.; Koes, R. How genes paint the flowers and seeds. Trends Plant Sci 1998, 3, 212–218. [Google Scholar]
- Bais, H.P.; Vepadechedu, R.; Gilroy, S.; Callaway, R.M.; Vivanco, J.M. Allelopathy and exotic plants: From genes to invasion. Science 2003, 301, 1377–1380. [Google Scholar]
- Buer, C.S.; Imin, N.; Djordjevic, M.A. Flavonoids: New roles for old molecules. J. Integr. Plant Biol 2010, 52, 98–111. [Google Scholar]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem 2002, 13, 572–584. [Google Scholar]
- Beninger, C.W.; Abou-Zaid, M.M.; Kistner, A.L.E.; Hallett, R.H.; Iqbal, M.J.; Grodizinsky, B.; Hall, J.C. A flavanone and two phenolic acids from Crysanthemum morifolium with phytotoxic and insect growth regulating activity. J. Chem. Ecol 2004, 30, 589–606. [Google Scholar]
- An, M. Mathematical modeling of dose-response relationship (hormesis) in allelopathy and its application. Nonlinearity Biol. Toxicol. Med 2005, 3, 153–172. [Google Scholar]
- Taiz, L.; Zeiger, E. Plant Physiology, 5th ed; Sinauer Associates: Sunderland, UK, 2010. [Google Scholar]
- Cesco, S.; Neumann, G.; Tomasi, N.; Pinton, R.; Weisskopf, L. Release of plant-borne flavonoids into the rhizosphere and their role in plant nutrition. Plant Soil 2010, 329, 1–25. [Google Scholar]
- Unten, L.; Koketsu, M.; Kim, M. Antidiscoloring activity of green tea polyphenols on β-carotene. J. Agric. Food Chem 1997, 45, 2009–2019. [Google Scholar]
- Star, A.E. Frond exudate flavonoids as allelopathic agents in Pityrogramma. Bull. Torrey Bot. Club 1980, 107, 146–153. [Google Scholar]
- Moroz, P.A.; Kommissarenko, N.F. Allelopathic activity of several phenolic compounds. Allelopatii 1983, 118–122. [Google Scholar]
- Shalaby, A.M. Influence of flavonoid compounds on VA mycorrhiza Glomus mosseae and alfalfa plants. Egypt. J. Microbiol 2001, 35, 225–238. [Google Scholar]
- Dakshini, K.M.M. Allelopathic potential of the phenolics from the roots of Pluchea lanceolata. Physiol. Plant 1994, 92, 571–576. [Google Scholar]
- Tsanuo, M.K.; Hassanali, A.; Hooper, A.M.; Khan, Z.; Kaberia, F.; Pickett, J.A.; Wadhams, L.J. Isoflavones from the allelopathic aqueous root exudate of Desmodium uncinatum. Phytochemistry 2003, 64, 263–273. [Google Scholar]
- Yang, D.H.; Webster, J.; Adam, Z.; Lindhal, M.; Andersson, B. Induction of acclimative proteolysis of the light-harvesting chlorophyll a/b protein of photosystem II in response to elevated light intensities. Plant Physiol 1998, 118, 827–834. [Google Scholar]
- Basile, A.; Sorbo, S.; Giordano, S.; Ricciardi, L.; Ferrara, S.; Montesano, D.; Castaldo Cobianchi, R.; Vuotto, M.L.; Ferrara, L. Antibacterial and allelopathic activity of extract from Castanea sativa leaves. Fitoterapia 2000, 71, S110–S116. [Google Scholar]
- Parvez, M.M.; Tomita-Yokotani, K.; Fujii, Y.; Konishi, T.; Iwashina, T. Effects of quercetin and its seven derivatives on the growth of Arabidopsis thaliana and Neurospora crassa. Biochem. Syst. Ecol 2004, 32, 631–635. [Google Scholar]
- Einhellig, F.A. Mode of Allelochemical Action of Phenolic Compounds. In Allelopathy; Macias, F.A., Ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 217–238. [Google Scholar]
- Moreland, D.E.; Novitski, W.P. Interference by flavone and flavonols with cloroplasts mediated electrons transport and phosphorylation. Phytochemistry 1988, 27, 3359–3365. [Google Scholar]
- Stenlid, G. Flavonoids as inhibitors of the formation of adenosine tri-phosphate in plant mitochondria. Phytochemistry 1970, 9, 2251–2256. [Google Scholar]
- Peer, W.A.; Murphy, A.S. Flavonoids and auxin transport: Modulators or regulators? Trends Plant Sci 2007, 12, 556–563. [Google Scholar]
- Macias, F.A.; Molinillo, J.M.G.; Torres, A.; Varela, R.M.; Castellano, D. Bioactive flavonoids from Helianthus annuus cultivars. Phytochemistry 1997, 45, 683–687. [Google Scholar]
- Beninger, C.W.; Hall, J.C. Allelopathic activity of luteolin 7-O-β-glucuronide isolated from Chrysanthemum morifolium L. Biochem. Syst. Ecol 2005, 33, 103–111. [Google Scholar]
- Bais, H.P.; Weir, T.L.; Perry, L.G.; Gilroy, S.; Vivanco, J.M. The role of root exudates in rhizosphere interactions with plants and other organisms. Annu. Rev. Plant Biol 2006, 57, 233–266. [Google Scholar]
- Rolim del Almeida, L.F.; Delachiave, M.E.; Sannomiya, M.; Vilegas, W.; Santos, L.C.; Mancini, E.; de Feo, V. In vitro potential allelopathic of Leonurus sibiricus leaves. J. Plant Interact 2008, 3, 39–48. [Google Scholar]
- Rolim del Almeida, L.F.; Sannomiya, M.; Rodrigues, C.M.; Delachiave, M.E.A.; Santos, L.C.; Vilegas, W.; de Feo, V. In vitro allelopathic effects of extracts and amenthoflavone from Byrsonima crassa (Malpighiaceae). J. Plant Interact 2007, 2, 121–124. [Google Scholar]
- Martínez-Flórez, S.J.; Gonzalez-Galego, J.; Culbras, J.M.; Tuñon, J. Los flavonoides: Propiedades y acciones antioxidantes. Nutrición Hospitalaria 2002, 6, 271–278. [Google Scholar]
- Nicollier, G.F.; Thompson, A.C. Phytotoxic compounds from Melilotus alba (white sweet clover) and isolation and identification of two new flavonoids. J. Agric. Food Chem 1982, 30, 760–766. [Google Scholar]
- Phillips, I.D.J. Some interactions of gibberellic acid with naringenin (4′,5,7-trihydroxyflavanone) in the control of dormancy and growth in plants. J. Exp. Bot 1962, 13, 213–226. [Google Scholar]
- Popravko, S.A.; Kononenko, G.P.; Vul’fson, N.S. Growth inhibitors in buds of Betula verrucosa. Russ. Chem. Bull 1974, 10. [Google Scholar] [CrossRef]
- Seelinger, G.; Merfort, I.; Schempp, C.M. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta Med 2008, 74, 1667–1677. [Google Scholar]
- Furusawa, M.; Tanaka, T.; Ito, T.; Nishikawa, A.; Yamazaki, N.; Nakaya, K.-I.; Matsuura, N.; Tsuchiya, H.; Nagayama, M.; Iinuma, M. Antioxidant activity of hydroflavonoids. J. Health Sci 2005, 51, 376–378. [Google Scholar]
- Furusawa, M.; Tsuchiya, H.; Nagayama, M.; Tanaka, T.; Nakaya, K.-I.; Iinuma, M. Anti-platelet and membrane-rigidifying flavonoids in brownish scale of onion. J. Health Sci 2003, 49, 475–480. [Google Scholar]
- Mendoza-Wilson, A.M.; Santacruz-Ortega, H.; Balandrán-Quintana, R.R. Relationship between structure, properties, and the radical scavenging activity of morin. J. Mol. Struct 2011, 995, 134–141. [Google Scholar]
- Choi, J.H.; Kim, H.S.; Jung, M.J.; Choi, J.S. (+)-Catechin an antioxidant principle from the leaves of Pinus densiflora that acts on 1, 1-diphenyl-2-picrylhydrazyl radical. Nat. Prod. Sci 2001, 7, 1–4. [Google Scholar]
- Ben Sghaier, M.; Skandrani, I.; Nasr, N.; Franca, M.-G.; Chekir-Ghedira, L.; Ghedira, K. Unité Flavonoids and sesquiterpenes from Teucrium ramosissimum promote antiproliferation of human cancer cells and enhance antioxidant activity: A structure–activity relationship study. Environ. Toxicol. Pharmacol 2011, 32, 336–348. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med 1996, 20, 933–956. [Google Scholar]
- Dziedzic, S.Z.; Hudson, B.J.F. Polyhydroxy chalcones and flavanones as antioxidants for edible oils. Food Chem 1983, 12, 205–212. [Google Scholar]
- Dugas, A.J., Jr; Castaneda-Acosta, J.; Bonin, G.C.; Price, K.L.; Fischer, N.H.; Winston, G.W. Evaluation of the total peroxyl radical scavenging capacity of flavonoids: Structure-activity relationships. J. Nat. Prod 2000, 63, 327–331. [Google Scholar]
- Bors, W.; Heller, W.; Michel, C.; Saran, M. Flavonoids as antioxidants: Determination of radical scavenging efficiencies. Meth. Enzymol 1990, 186, 343–355. [Google Scholar]
- Lien, E.J.; Ren, S.; Bui, H.H.; Wang, R. Quantitative structure-activity relationship analysis of phenolic antioxidants. Free Radic. Biol. Med 1999, 26, 285–294. [Google Scholar]
- Bewley, J.D.; Black, M. Seeds: Physiology of Development and Germination, 2nd ed; Plenum Press: New York, NY, USA, 1985. [Google Scholar]
- Mencherini, T.; Picerno, P.; Scesa, C.; Aquino, R. Triterpene, antioxidant, and antimicrobial compounds from Melissa officinalis. J. Nat. Prod 2007, 70, 1889–1894. [Google Scholar]
- Tallarida, R.J.; Murray, R.B. Manual of Pharmacological Calculations; Springer-Verlag: New York, NY, USA, 1984. [Google Scholar]
- Sokal, R.R.; Rohlf, F.J. Biometry, 2nd ed; W.H. Freeman and Company: New York, NY, USA, 1981. [Google Scholar]
- Taylor, L.P.; Grotewold, E. Flavonoids as developmental regulators. Curr. Opin. Plant Biol 2005, 8, 317–323. [Google Scholar]
| Raphanus sativus L. | Lepidium sativum L. | |||||
|---|---|---|---|---|---|---|
| Germinated seeds ± SD | Germinated seeds ± SD | |||||
| 10−6 M | 10−5 M | 10−4 M | 10−6 M | 10−5 M | 10−4 M | |
| Control | 9.3 ± 0.8 | 9.3 ± 0.8 | 9.3 ± 0.8 | 9.5 ± 0.8 | 9.5 ± 0.8 | 9.5 ± 0.8 |
| Flavone | 9.7 ± 0.6 | 8.0 ± 1.0 | 9.3 ± 0.6 | 9.8 ± 0.8 | 9.4 ± 0.8 | 9.4 ± 0.8 |
| 7-OH-Flavone | 9.3 ± 1.2 | 9.7 ± 0.6 | 8.7 ± 1.2 | 10.0 ± 0.0 | 10.0 ± 0.0 | 10.0 ± 0.0 |
| 7-OCH3-Flavone | 8.7 ± 0.7 | 9.0 ± 1.0 | 9.3 ± 1.2 | 8.8 ± 0.8 | 9.4 ± 0.8 | 8.8 ± 0.8 |
| 5,7-di-OH-Flavone (chrysin) | 9.3 ± 0.8 | 8.3 ± 1.5 | 8.7 ± 0.8 | 9.5 ± 0.8 | 9.1 ± 0.8 | 8.8 ± 1.3 |
| 5,7-di-OCH3-Flavone (chrysin dimethylether) | 9.0 ± 0.0 | 9.3 ± 1.5 | 9.7 ± 0.8 | 8.5 ± 1.5 | 9.5 ± 0.8 | 8.8 ± 1.3 |
| 3′,4′-di-OCH3-Flavone | 9.6 ± 0.9 | 9.6 ± 0.5 | 10.0 ± 0.6 | 10.0 ± 0.8 * | 10.3 ± 0.0 | 10.0 ± 0.8* |
| 5,7,3′,4′-tetra-OH-Flavone (luteolin) | 8.7 ± 0.8 | 8.3 ± 2.0 | 9.0 ± 1.3 | 8.5 ± 2.8 | 9.1 ± 1.5 | 9.5 ± 0.8 |
| 5,7,3′,4′-tetra-OCH3-Flavone (luteolin tetramethylether) | 9.3 ± 1.5 | 9.0 ± 1.3 | 9.0 ± 1.3 | 9.1 ± 0.8 | 9.8 ± 1.3 | 6.9 ± 4.8 |
| 3-OH-Flavone | 9.0 ± 1.7 | 9.0 ± 1.0 | 9.3 ± 0.6 | 10.0 ± 0.0 | 10.0 ± 0.0 | 8.8 ± 2.0 |
| 3-OCH3-Flavone | 9.7 ± 0.6 | 8.7 ± 1.5 | 8.0 ± 1.0 | 7.6 ± 0.9 * | 8.4 ± 1.5 | 9.8 ± 0.8 |
| 2′,3,4′,5,7-penta-OH-Flavone (morin) | 8.7 ± 1.2 | 8.4 ± 1.0 | 9.0 ± 0.6 | 7.9 ± 1.5 | 8.9 ± 1.2 | 8.9 ± 0.6 |
| 3,5,7,3′,4′-penta-OH-Flavone (quercetin) | 9.0 ± 0.6 | 8.7 ± 0.6 | 9.0 ± 0.6 | 8.2 ± 0.6* | 8.9 ± 0.6 | 9.5 ± 0.0 |
| Quercetin-3-rutinoside (rutin) | 9.0 ± 1.3 | 9.3 ± 1.5 | 9.3 ± 1.5 | 8.9 ± 0.8 | 9.2 ± 0.0 | 8.9 ± 0.8 |
| β-Naphtoflavone | 9.0 ± 0.8 | 7.4 ± 4.3 | 8.6 ± 1.3 | 10.0 ± 0.0 | 10.0 ± 0.0 | 9.9 ± 0.8 |
| 4′,5,7-tri-OH-Isoflavone | 9.0 ± 1.0 | 8.7 ± 0.6 | 9.0 ± 1.0 | 8.9 ± 0.8 | 9.2 ± 1.3 | 8.0 ± 0.8 * |
| Flavanone | 9.3 ± 0.6 | 9.6 ± 0.0 | 8.2 ± 0.8 | 9.9 ± 0.8 | 10.0 ± 0.8 | 10.0 ± 0.8 |
| 7-OH-Flavanone | 9.3 ± 0.3 | 10.0 ± 0.3 | 9.3 ± 0.3 | 10.0 ± 0.8 | 10.0 ± 0.8 | 10.0 ± 0.8 * |
| 7-OCH3-Flavanone | 9.6 ± 0.9 | 9.6 ± 0.5 | 10.0 ± 0.3 | 10.0 ± 0.8 | 9.9 ± 2.0 | 10.0 ± 0.8 |
| 5,7-di-OH-Flavanone (pinocembrin) | 8.3 ± 1.5 | 8.0 ± 0.0 * | 9.7 ± 0.8 | 9.2 ± 1.3 | 8.9 ± 0.8 | 8.9 ± 0.8 |
| 5,7-di-OCH3-Flavanone | 9.3 ± 1.5 | 9.0 ± 1.3 | 9.0 ± 0.0 | 9.1 ± 0.8 | 9.8 ± 0.0 | 8.5 ± 0.8 |
| (2S)-4′,5,7-tri-OH-Flavanone (naringenin) | 9.0 ± 1.3 | 9.3 ± 0.8 | 9.3 ± 1.5 | 8.6 ± 1.5 | 8.3 ± 0.0 | 8.6 ± 0.8 |
| Naringin (naringenin 7-rhamnoglucoside) | 8.7 ± 2.0 | 8.3 ± 0.8 | 9.3 ± 0.8 | 8.3 ± 1.3 | 8.9 ± 0.8 | 8.3 ± 1.3 |
| 3′,5,7-tri-OH-4′-OCH3-Flavanone (hesperetin) | 8.6 ± 1.3 | 9.0 ± 1.5 | 8.6 ± 1.3 | 10.0 ± 0.0 | 10.0 ± 0.0 | 9.9 ± 0.8 |
| Hesperidin (hesperetin 7-rutinoside) | 9.0 ± 0.8 | 8.3 ± 2.0 | 8.3 ± 0.8 | 9.7 ± 0.9 | 8.7 ± 2.8 | 10.0 ± 1.9 |
| (+)-Catechin | 9.3 ± 0.8 | 9.3 ± 0.8 | 9.3 ± 0.8 | 9.9 ± 0.8 | 9.9 ± 0.8 | 8.2 ± 1.3 |
| Catechol | 9.6 ± 0.0 | 9.6 ± 0.0 | 9.6 ± 0.0 | 9.2 ± 2.3 | 9.5 ± 1.5 | 8.2 ± 2.3 |
| Phloridzin | 9.0 ± 0.6 | 9.0 ± 0.6 | 8.7 ± 0.6 | 8.9 ± 0.6 | 9.2 ± 0.6 | 9.2 ± 0.6 |
| Raphanus sativus L. | Lepidium sativum L. | |||||
|---|---|---|---|---|---|---|
| Radical elongation ± SD | Radical elongation ± SD | |||||
| 10−6 M | 10−5 M | 10−4 M | 10−6 M | 10−5 M | 10−4 M | |
| Control | 5.2 ± 2.8 | 5.2 ± 2.8 | 5.2 ± 2.8 | 3.6 ± 2.3 | 3.6 ± 2.3 | 3.6 ± 2.3 |
| Flavone | 7.0 ± 4.7 | 4.5 ± 3.2 | 5.0 ± 2.4 | 3.7 ± 1.9 | 4.2 ± 2.3 | 2.3 ± 1.4** |
| 7-OH-Flavone | 7.1 ± 2.8* | 3.7 ± 2.1** | 5.8 ± 3.1 | 3.7 ± 2.6 | 3.2 ± 2.4 | 3.7 ± 2.3 |
| 7-OCH3-Flavone | 4.3 ± 3.5 | 4.1 ± 3.2 | 2.9 ± 1.9*** | 2.8 ± 2.0 | 2.6 ± 2.0 | 2.4 ± 1.6** |
| 5,7-di-OH-Flavone (chrysin) | 7.1 ± 2.4* | 4.9 ± 2.5 | 2.7 ± 1.5*** | 4.7 ± 2.2 | 4.9 ± 2.6 | 4.8 ± 2.7 |
| 5,7-di-OCH3-Flavone (chrysin dimethylether) | 3.9 ± 2.5 | 1.9 ± 1.7*** | 0.4 ± 0.2*** | 4.9 ± 2.8 | 2.1 ± 1.1** | 0.2 ± 0.1*** |
| 3′,4′-di-OCH3-Flavone | 3.1 ± 2.2** | 5.4 ± 2.5 | 3.2 ± 1.9** | 2.7 ± 1.5 | 2.3 ± 1.5* | 2.3 ± 1.5* |
| 5,7,3′,4′-tetra-OH-Flavone (luteolin) | 5.1 ± 2.0 | 6.8 ± 2.2* | 4.2 ± 2.7 | 4.0 ± 1.8 | 6.2 ± 2.3*** | 4.6 ± 2.7 |
| 5,7,3′,4′-tetra-OCH3-Flavone (luteolin tetramethylether) | 3.1 ± 1.8** | 4.7 ± 2.1 | 3.9 ± 2.4 | 4.4 ± 2.0 | 5.3 ± 2.6* | 3.0 ± 1.7 |
| 3-OH-Flavone | 4.3 ± 3.5 | 3.4 ± 2.6 | 4.6 ± 2.6 | 4.1 ± 1.7 | 3.5 ± 2.1 | 3.0 ± 1.5 |
| 3-OCH3-Flavone | 5.5 ± 2.3 | 3.6 ± 2.0 | 4.8 ± 2.4 | 2.2 ± 1.0* | 2.0 ± 1.3*** | 2.8 ± 1.3 |
| 2′,3,4′,5,7-penta-OHFlavone (morin) | 3.9 ± 2.1* | 4.7 ± 2.6 | 4.1 ± 2.2 | 3.5 ± 1.9 | 4.2 ± 3.0 | 2.5 ± 2.0* |
| 3,5,7,3′,4′-penta-OHFlavone (quercetin) | 5.0 ± 2.9 | 4.6 ± 2.4 | 5.8 ± 3.1 | 5.5 ± 2.9** | 4.6 ± 3.0 | 3.5 ± 2.1 |
| Quercetin-3-rutinoside (rutin) | 5.1 ± 2.3 | 4.8 ± 3.0 | 5.3 ± 3.3 | 4.4 ± 2.5 | 4.3 ± 2.4 | 3.8 ± 2.6 |
| β-Naphtoflavone | 6.0 ± 2.8 | 3.6 ± 2.5 | 5.1 ± 2.4 | 4.2 ± 2.2 | 5.4 ± 3.2** | 3.7 ± 2.5 |
| 4′,5,7-tri-OH-Isoflavone | 4.6 ± 2.2 | 5.8 ± 2.5 | 4.3 ± 2.5 | 4.1 ± 3.5 | 4.3 ± 2.4 | 4.2 ± 3.3 |
| Flavanone | 5.2 ± 2.2 | 5.3 ± 2.3 | 4.7 ± 1.9 | 4.2 ± 2.0 | 3.2 ± 1.6 | 3.4 ± 1.4 |
| 7-OH-Flavanone | 4.9 ± 2.4 | 5.0 ± 2.3 | 5.1 ± 2.1 | 4.0 ± 1.9 | 4.2 ± 2.0 | 4.0 ± 2.4 |
| 7-OCH3-Flavanone | 3.7 ± 2.3* | 5.3 ± 2.6 | 5.0 ± 2.1 | 4.4 ± 2.1 | 3.5 ± 1.7 | 2.0 ± 1.2** |
| 5,7-di-OH-Flavanone (pinocembrin) | 4.5 ± 2.6 | 3.4 ± 2.7* | 5.1 ± 2.5 | 4.7 ± 3.3 | 4.1 ± 3.5 | 4.9 ± 3.7 |
| 5,7-di-OCH3-Flavanone | 5.2 ± 3.1 | 3.1 ± 1.5** | 1.0 ± 1.2*** | 5.0 ± 2.4* | 3.2 ± 1.4 | 1.1 ± 0.5*** |
| (2S)-4′,5,7-tri-OH-Flavanone (naringenin) | 4.5 ± 3.9 | 4.9 ± 2.7 | 3.8 ± 2.8 | 4.3 ± 2.7 | 5.4 ± 3.1 | 5.0 ± 2.5 |
| Naringin (naringenin 7-rhamnoglucoside) | 4.1 ± 3.1 | 5.5 ± 2.6 | 3.6 ± 3.0 | 4.0 ± 2.6 | 5.4 ± 4.4* | 3.2 ± 2.8 |
| 3′,5,7-tri-OH-4′-OCH3-Flavanone (hesperetin) | 6.1 ± 2.4 | 5.1 ± 2.6 | 6.3 ± 2.5 | 5.3 ± 3.1** | 5.1 ± 2.6** | 6.0 ± 3.6*** |
| Hesperidin (hesperetin 7-rutinoside) | 7.5 ± 3.2* | 4.6 ± 1.9 | 5.8 ± 2.8 | 5.0 ± 3.3* | 4.9 ± 3.6 | 4.1 ± 3.7 |
| (+)-Catechin | 4.8 ± 2.9 | 7.0 ± 3.8 | 6.9 ± 3.8 | 6.7 ± 3.4*** | 4.8 ± 3.5 | 4.0 ± 2.1 |
| Catechol | 5.7 ± 3.1 | 6.4 ± 3.7 | 6.3 ± 3.4 | 5.2 ± 3.3* | 4.2 ± 3.8 | 4.7 ± 3.5 |
| Phloridzin | 3.5 ± 2.2** | 4.7 ± 2.3 | 4.4 ± 2.6 | 5.0 ± 3.4* | 3.5 ± 2.6 | 2.6 ± 1.6 |
| Compound | DPPH test [IC50 (μg of compound/mL)] |
|---|---|
| Flavone | >100 |
| 7-OH-Flavone | >100 |
| 7-OCH3-Flavone | >100 |
| 5,7-di-OH-Flavone (chrysin) | >100 |
| 5,7-di-OCH3-Flavone (chrysin dimethylether) | >100 |
| 3′,4′-di-OCH3-Flavone | >100 |
| 5,7,3′,4′-tetra-OH-Flavone (luteolin) | 2.051 ± 0.638 a |
| 5,7,3′,4′-tetra-OCH3-Flavone (luteolin tetramethylether) | >100 |
| 3-OH-Flavone | 93.721 ± 3.201 a |
| 3-OCH3-Flavone | >100 |
| 2′,3,4′,5,7-penta-OH-Flavone (morin) | 5.803 ± 1.375 a |
| 3,5,7,3′,4′-penta-OH-Flavone (quercetin) | 2.355 ± 0.847 a |
| Quercetin-3-rutinoside (rutin) | 11.406 ± 1.302 a |
| β-Naphtoflavone | >100 |
| 4′,5,7-tri-OH-Isoflavone | >100 |
| Flavanone | >100 |
| 7-OH-Flavanone | >100 |
| 7-OCH3-Flavanone | >100 |
| 5,7-di-OH-Flavanone (pinocembrin) | >100 |
| 5,7-di-OCH3-Flavanone | >100 |
| (2S)-4′,5,7-tri-OH-Flavanone (naringenin) | >100 |
| Naringin (naringenin 7-rhamnoglucoside) | >100 |
| 3′,5,7-tri-OH-4′-OCH3-Flavanone (hesperetin) | 176.893 ± 2.428 a |
| Hesperidin (hesperetin 7-rutinoside) | >100 |
| (+)-Catechin | 15.819 ± 1.273 a |
| Catechol | 2.774 ± 0.289 a |
| Phloridzin | >100 |
| α-Tocopherol b | 10.100 ± 1.300 a |
| Vitamin C b | 5.851 ± 0.9206 a |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Martino, L.; Mencherini, T.; Mancini, E.; Aquino, R.P.; De Almeida, L.F.R.; De Feo, V. In Vitro Phytotoxicity and Antioxidant Activity of Selected Flavonoids. Int. J. Mol. Sci. 2012, 13, 5406-5419. https://doi.org/10.3390/ijms13055406
De Martino L, Mencherini T, Mancini E, Aquino RP, De Almeida LFR, De Feo V. In Vitro Phytotoxicity and Antioxidant Activity of Selected Flavonoids. International Journal of Molecular Sciences. 2012; 13(5):5406-5419. https://doi.org/10.3390/ijms13055406
Chicago/Turabian StyleDe Martino, Laura, Teresa Mencherini, Emilia Mancini, Rita Patrizia Aquino, Luiz Fernando Rolim De Almeida, and Vincenzo De Feo. 2012. "In Vitro Phytotoxicity and Antioxidant Activity of Selected Flavonoids" International Journal of Molecular Sciences 13, no. 5: 5406-5419. https://doi.org/10.3390/ijms13055406







