Salivary Proteins Associated with Periodontitis in Patients with Type 2 Diabetes Mellitus
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Collection and Pre-Treatment of the Saliva Samples
3.2. First Dimensional Separation—Isoelectric Focusing
3.3. Second Dimensional Separation—Polyacrylamide Gel Electrophoresis
3.4. Scanning and Analysis of Gel Image
3.5. Identification of Saliva Proteins
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Wild, S.H.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 2569–2569. [Google Scholar]
- Loo, J.; Yan, W.; Ramachandran, P.; Wong, D. Comparative human salivary and plasma proteomes. J. Dent. Res 2010, 89, 1016–1023. [Google Scholar]
- The International Diabetes Federation, Diabetes Atlas; The International Diabetes Federation: Brussels, Belgium, 2011; Available online: http://www.idf.org/diabetesatlas/5e/regional-overviews accessed on 20 December 2011.
- Ministry of Health, 3rd National Health and Morbidity Survey; Ministry of Health: Putrajaya, Malaysia, 2006.
- Letchuman, G.; Wan Nazaimoon, W.; Wan Mohamad, W.; Chandran, L.; Tee, G.; Jamaiyah, H.; Isa, M.; Zanariah, H.; Fatanah, I.; Ahmad Faudzi, Y. Prevalence of diabetes in the malaysian national health morbidity survey III 2006. Med. J. Malays 2010, 65, 173–179. [Google Scholar]
- Löe, H. Periodontal disease. The sixth complication of diabetes mellitus. Diabetes Care 1993, 16, 329–334. [Google Scholar]
- Grossi, S.G. Treatment of periodontal disease and control of diabetes: An assessment of the evidence and need for future research. Ann. Periodontol 2001, 6, 138–145. [Google Scholar]
- Oliver, R.C.; Tervonen, T. Diabetes—A risk factor for periodontitis in adults? J. Periodontol 1994, 65, 530–538. [Google Scholar]
- Tsai, C.; Hayes, C.; Taylor, G.W. Glycemic control of type 2 diabetes and severe periodontal disease in the US adult population. Community Dent. Oral Epidemiol 2002, 30, 182–192. [Google Scholar]
- Kingman, A.; Albandar, J.M. Methodological aspects of epidemiological studies of periodontal diseases. Periodontology 2000 2002, 29, 11–30. [Google Scholar]
- Kinane, D.; Bouchard, P. Group E of European Workshop on Periodontology. Periodontal diseases and health: Consensus report of the sixth European workshop on periodontology. J. Clin. Periodontol 2008, 35, 333–337. [Google Scholar]
- Grossi, S.G.; Genco, R.J. Periodontal disease and diabetes mellitus: A two-way relationship. Ann. Periodontol 1998, 3, 51–61. [Google Scholar]
- Oral Health Division, Ministry of Health, Malaysia, National Oral Health Survey of Adults; Ministry of Health: Putrajaya, Malaysia, 2000.
- Kaufman, E.; Lamster, I.B. The diagnostic applications of saliva—A review. Crit. Rev. Oral Biol. Med 2002, 13, 197–212. [Google Scholar]
- Mandel, I.D. The functions of saliva. J. Dent. Res 1987, 66, 623–627. [Google Scholar]
- Hofman, L.F. Human saliva as a diagnostic specimen. J. Nutr 2001, 131, 1621S–1625S. [Google Scholar]
- Streckfus, C.; Bigler, L. Saliva as a diagnostic fluid. Oral Dis 2002, 8, 69–76. [Google Scholar]
- Wong, D.T. Salivary diagnostics powered by nanotechnologies, proteomics and genomics. J. Am. Dent. Assoc 2006, 137, 313–321. [Google Scholar]
- Helmerhorst, E.; Oppenheim, F. Saliva: A dynamic proteome. J. Dent. Res 2007, 86, 680–693. [Google Scholar]
- Jessie, K.; Hashim, O.H.; Rahim, Z.H.A. Protein precipitation method for salivary proteins and rehydration buffer for two-dimensional electrophoresis. Biotechnology 2008, 7, 686–693. [Google Scholar]
- Gonçalves, L.D.R.; Soares, M.R.; Nogueira, F.; Garcia, C.; Camisasca, D.R.; Domont, G.; Feitosa, A.C.R.; Pereira, D.A.; Zingali, R.B.; Alves, G. Comparative proteomic analysis of whole saliva from chronic periodontitis patients. J. Proteomics 2010, 73, 1334–1341. [Google Scholar]
- Huang, C.M. Comparative proteomic analysis of human whole saliva. Arch. Oral Biol 2004, 49, 951–962. [Google Scholar]
- Jessie, K.; Pang, W.W.; Rahim, Z.H.A.; Hashim, O.H. Proteomic analysis of whole human saliva detects enhanced expression of interleukin-1 receptor antagonist, thioredoxin and lipocalin-1 in cigarette smokers compared to non-smokers. Int. J. Mol. Sci 2010, 11, 4488–4505. [Google Scholar]
- Rao, P.V.; Reddy, A.P.; Lu, X.; Dasari, S.; Krishnaprasad, A.; Biggs, E.; Roberts, C.T.; Nagalla, S.R. Proteomic identification of salivary biomarkers of type-2 diabetes. J. Proteome Res 2009, 8, 239–245. [Google Scholar]
- Muscari, A.; Antonelli, S.; Bianchi, G.; Cavrini, G.; Dapporto, S.; Ligabue, A.; Ludovico, C.; Magalotti, D.; Poggiopollini, G.; Zoli, M. Serum C3 is a stronger inflammatory marker of insulin resistance than C-reactive protein, leukocyte count, and erythrocyte sedimentation rate. Diabetes Care 2007, 30, 2362–2368. [Google Scholar]
- Johansen, F.; Braathen, R.; Brandtzaeg, P. Role of J chain in secretory immunoglobulin formation. Scand. J. Immunol 2000, 52, 240–248. [Google Scholar]
- Max, E.E.; Korsmeyer, S.J. Human J chain gene. Structure and expression in B lymphoid cells. J. Exp. Med 1985, 161, 832–849. [Google Scholar]
- Zikan, J.; Novotny, J.; Trapane, T.L.; Koshland, M.E.; Urry, D.W.; Bennett, J.C.; Mestecky, J. Secondary structure of the immunoglobulin J chain. Proc. Natl. Acad. Sci. USA 1985, 82, 5905–5909. [Google Scholar]
- Asano, M.; Komiyama, K. Polymeric immunoglobulin receptor. J. Oral Sci 2011, 53, 147–156. [Google Scholar]
- Bostanci, N.; Heywood, W.; Mills, K.; Parkar, M.; Nibali, L.; Donos, N. Application of label-free absolute quantitative proteomics in human gingival crevicular fluid by LC/MSE (gingival exudatome). J. Proteome Res 2010, 9, 2191–2199. [Google Scholar]
- UniProtKB Plastin-2 P13796 (PLSL_HUMAN). Available online: http://www.uniprot.org/uniprot/P13796 accessed on 11 July 2011.
- Jones, S.L.; Wang, J.; Turck, C.W.; Brown, E.J. A role for the actin-bundling protein L-plastin in the regulation of leukocyte integrin function. Proc. Natl. Acad. Sci. USA 1998, 95, 9331–9336. [Google Scholar]
- Grant, M.M.; Creese, A.J.; Barr, G.; Ling, M.R.; Scott, A.E.; Matthews, J.B.; Griffiths, H.R.; Cooper, H.J.; Chapple, I.L.C. Proteomic analysis of a noninvasive human model of acute inflammation and its resolution: The twenty-one day gingivitis model. J. Proteome Res 2010, 9, 4732–4744. [Google Scholar]
- UniProtKB Leukocyte elastase inhibitor P30740 (ILEU_HUMAN). Available online: http://www.uniprot.org/uniprot/P30740 accessed on 11 July 2011.
- Remold-O’Donnell, E.; Chin, J.; Alberts, M. Sequence and molecular characterization of human monocyte/neutrophil elastase inhibitor. Proc. Natl. Acad. Sci. USA 1992, 89, 5635–5639. [Google Scholar]
- Kivelä, J.; Parkkila, S.; Parkkila, A.K.; Leinonen, J.; Rajaniemi, H. Salivary carbonic anhydrase isoenzyme VI. J. Physiol 1999, 520, 315–320. [Google Scholar]
- Jiang, W.; Gupta, D. Structure of the carbonic anhydrase VI (CA6) gene: Evidence for two distinct groups within the alpha-CA gene family. Biochem. J 1999, 344, 385–390. [Google Scholar]
- Karhumaa, P.; Leinonen, J.; Parkkila, S.; Kaunisto, K.; Tapanainen, J.; Rajaniemi, H. The identification of secreted carbonic anhydrase VI as a constitutive glycoprotein of human and rat milk. Proc. Natl. Acad. Sci. USA 2001, 98, 11604–11608. [Google Scholar]
- Kivelä, J.; Laine, M.; Parkkila, S.; Rajaniemi, H. Salivary carbonic anhydrase VI and its relation to salivary flow rate and buffer capacity in pregnant and non-pregnant women. Arch. Oral Biol 2003, 48, 547–551. [Google Scholar]
- UniProtKB Actin-related protein 3 P61158 (ARP3_HUMAN). Available online: http://www.uniprot.org/uniprot/P61158 accessed on 10 July 2011.
- Dayel, M.J.; Holleran, E.A.; Mullins, R.D. Arp2/3 complex requires hydrolyzable ATP for nucleation of new actin filaments. Proc. Natl. Acad. Sci. USA 2001, 98, 14871–14876. [Google Scholar]
- Dayel, M.J.; Mullins, R.D. Activation of Arp2/3 complex: Addition of the first subunit of the new filament by a WASP protein triggers rapid ATP hydrolysis on Arp2. PLoS Biol 2004, 2. [Google Scholar] [CrossRef] [Green Version]
- Aksentijevich, I.; Masters, S.L.; Ferguson, P.J.; Dancey, P.; Frenkel, J.; van Royen-Kerkhoff, A.; Laxer, R.; Tedgård, U.; Cowen, E.W.; Pham, T.H. An autoinflammatory disease with deficiency of the interleukin-1–receptor antagonist. N. Engl. J. Med 2009, 360, 2426–2437. [Google Scholar]
- Arend, W.P.; Guthridge, C.J. Biological role of interleukin 1 receptor antagonist isoforms. Ann. Rheum. Dis 2000, 59, i60–i64. [Google Scholar]
- Gabay, C.; Smith, M.; Eidlen, D.; Arend, W.P. Interleukin 1 receptor antagonist (IL-1Ra) is an acute-phase protein. J. Clin. Investig 1997, 99, 2930–2940. [Google Scholar]
- Granowitz, E.V.; Clark, B.; Mancilla, J.; Dinarello, C. Interleukin-1 receptor antagonist competitively inhibits the binding of interleukin-1 to the type II interleukin-1 receptor. J. Biol. Chem 1991, 266, 14147–14150. [Google Scholar]
- Juge-Aubry, C.E.; Somm, E.; Giusti, V.; Pernin, A.; Chicheportiche, R.; Verdumo, C.; Rohner-Jeanrenaud, F.; Burger, D.; Dayer, J.M.; Meier, C.A. Adipose tissue is a major source of interleukin-1 receptor antagonist. Diabetes 2003, 52, 1104–1110. [Google Scholar]
- Larsen, C.M.; Faulenbach, M.; Vaag, A.; Ehses, J.A.; Donath, M.Y.; Mandrup-Poulsen, T. Sustained effects of interleukin-1 receptor antagonist treatment in type 2 diabetes. Diabetes Care 2009, 32, 1663–1668. [Google Scholar]
- Larsen, C.M.; Faulenbach, M.; Vaag, A.; Vølund, A.; Ehses, J.A.; Seifert, B.; Mandrup-Poulsen, T.; Donath, M.Y. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med 2007, 356, 1517–1526. [Google Scholar]
- Heukeshoven, J.; Dernick, R. Simplified method for silver staining of proteins in polyacrylamide gels and the mechanism of silver staining. Electrophoresis 1985, 6, 103–112. [Google Scholar]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar]
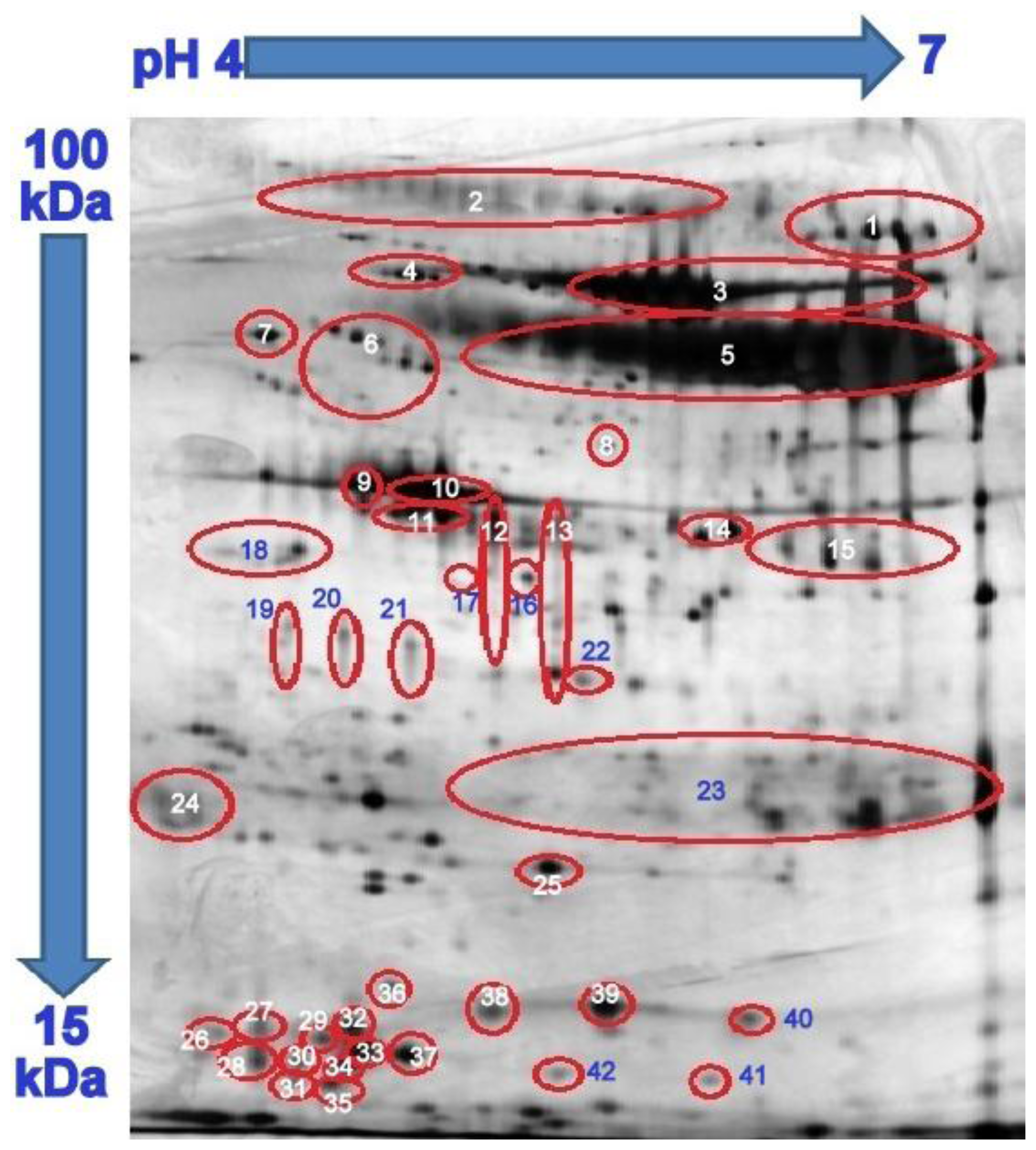
| Proteins | Protein Label | Diabetes with Periodontitis % Volume a (±SEM) | RP b/15 | Diabetes with Healthy Periodontium % Volume a (±SEM) | RP b//10 |
|---|---|---|---|---|---|
| Serotransferrin | 1 | 2.602 (±1.274) | 15 | 3.430 (±1.254) | 10 |
| pIgR | 2 | 5.093 (±1.591) | 15 | 6.848 (±1.273) | 10 |
| Plastin 2 | 4 | 0.434 (±0.161) | 15 | 0.182 (±0.039) | 8 |
| Alpha-1-antitrypsin | 6 | 1.858 (±0.641) | 15 | 1.818 (±0.836) | 10 |
| Protein disulfide isomerase | 7 | 0.230 (±0.090) | 14 | 0.199 (±0.093) | 8 |
| Actin-related protein 3 | 8 | 0.023 (±0.004) | 14 | 0.135 (±0.088) | 9 |
| Zinc-alpha-2-glycoprotein | 9 | 3.816 (±1.236) | 15 | 3.735 (±0.671) | 10 |
| 11 | |||||
| 12 | |||||
| 13 | |||||
| Actin beta | 10 | 1.567 (±0.584) | 15 | 1.913 (±0.883) | 10 |
| 16 | |||||
| 17 | |||||
| Leukocyte elastase inhibitor | 14 | 0.386 (±0.162) | 14 | 0.201 (±0.126) | 9 |
| Carbonic anhydrase VI | 15 | 1.665 (±0.595) | 14 | 2.273 (±0.549) | 10 |
| Complement C3 | 18 | 0.582 (±0.278) | 15 | 0.626 (±0.268) | 10 |
| SPLUNC | 19 | 2.193 (±0.962) | 12 | 1.117 (±0.776) | 9 |
| 20 | |||||
| 21 | |||||
| Annexin A3 | 22 | 0.199 (±0.098) | 15 | 0.133 (±0.091) | 8 |
| Ig Kappa chain c region | 23 | 23.031 (±3.968) | 15 | 17.870 (±8.369) | 10 |
| Immunoglobulin J chain | 24 | 2.402 (±0.338) | 15 | 1.378 (±1.706) | 10 |
| Glutathione S Transferase | 25 | 0.369 (±0.252) | 15 | 0.270 (±0.112) | 10 |
| Prolactin inducible protein | 26 | 6.362 (±2.176) | 15 | 4.648 (±1.388) | 10 |
| 27 | |||||
| 28 | |||||
| 32 | |||||
| 33 | |||||
| 34 | |||||
| Lipocalin -1 | 29 | 1.948 (±0.926) | 15 | 1.602 (±0.455) | 10 |
| 30 | |||||
| 31 | |||||
| 35 | |||||
| 37 | |||||
| Interleukin-1 receptor antagonist protein | 36 | 0.041 (±0.016) | 15 | 0.171 (±0.060) | 9 |
| Haptoglobin | 38 | 0.397 (±0.246) | 15 | 0.578 (±0.323) | 10 |
| 39 | |||||
| 40 | |||||
| Transthyretin | 41 | 0.213 (±0.125) | 13 | 0.215 (±0.145) | 5 |
| 42 |
| Proteins | Fold Change a | p-Value |
|---|---|---|
| PIGR | −1.344 | 0.008 |
| Plastin 2 | +2.381 | 0 |
| Actin-related protein 3 | −5.802 | 0.001 |
| Leukocyte elastase inhibitor | +1.919 | 0.004 |
| Carbonic anhydrase VI | −1.365 | 0.012 |
| Immunoglobulin J chain | +1.743 | 0 |
| Interleukin-1 receptor antagonist | −4.132 | 0 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chan, H.H.; Rahim, Z.H.A.; Jessie, K.; Hashim, O.H.; Taiyeb-Ali, T.B. Salivary Proteins Associated with Periodontitis in Patients with Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2012, 13, 4642-4654. https://doi.org/10.3390/ijms13044642
Chan HH, Rahim ZHA, Jessie K, Hashim OH, Taiyeb-Ali TB. Salivary Proteins Associated with Periodontitis in Patients with Type 2 Diabetes Mellitus. International Journal of Molecular Sciences. 2012; 13(4):4642-4654. https://doi.org/10.3390/ijms13044642
Chicago/Turabian StyleChan, Hang Haw, Zubaidah H. A. Rahim, Kala Jessie, Onn H. Hashim, and Tara B. Taiyeb-Ali. 2012. "Salivary Proteins Associated with Periodontitis in Patients with Type 2 Diabetes Mellitus" International Journal of Molecular Sciences 13, no. 4: 4642-4654. https://doi.org/10.3390/ijms13044642




