Acute Oral Toxicity Evaluations of Some Zinc(II) Complexes Derived from 1-(2-Salicylaldiminoethyl)piperazine Schiff Bases in Rats
Abstract
:1. Introduction
2. Result and Discussion
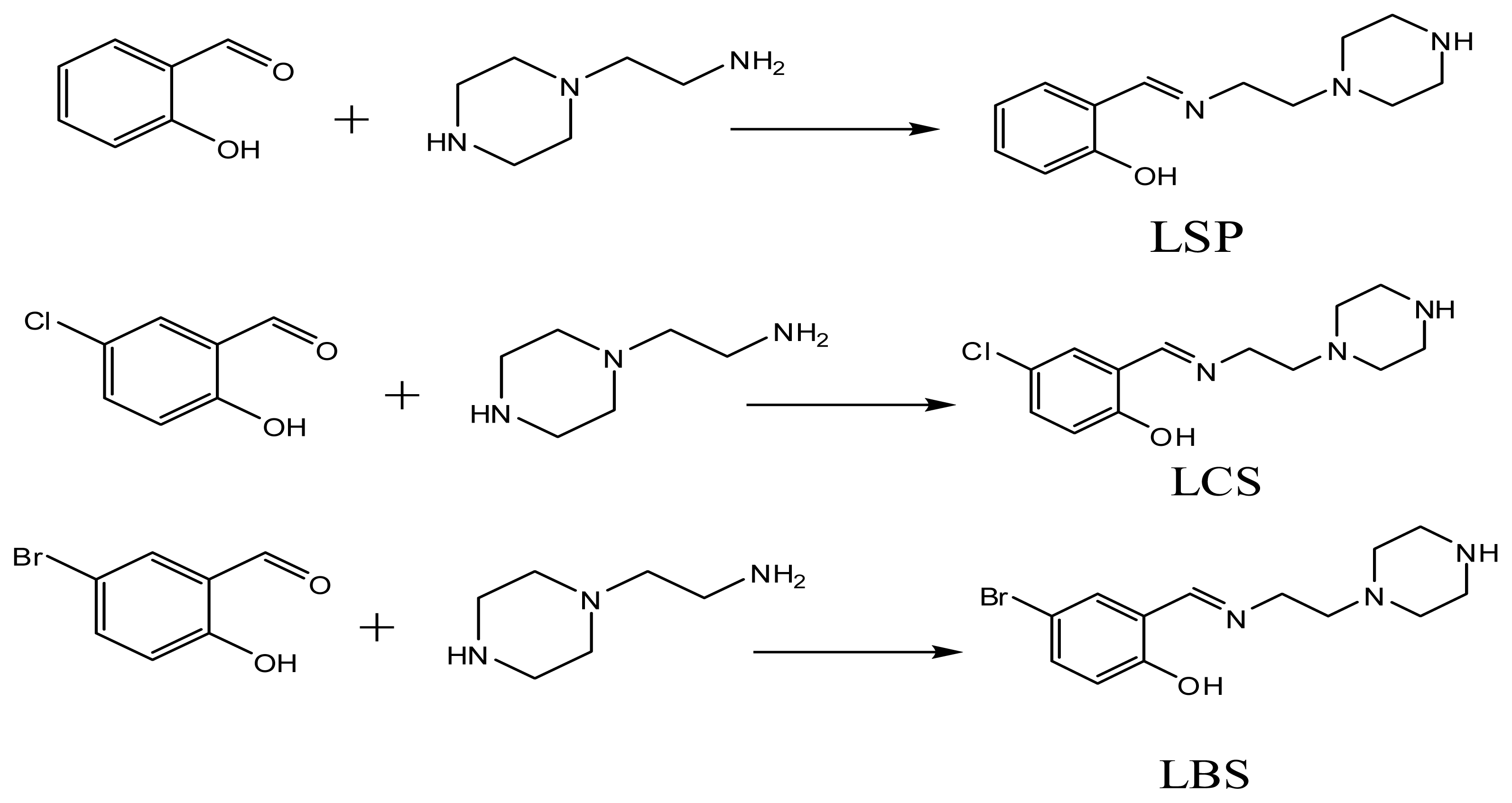
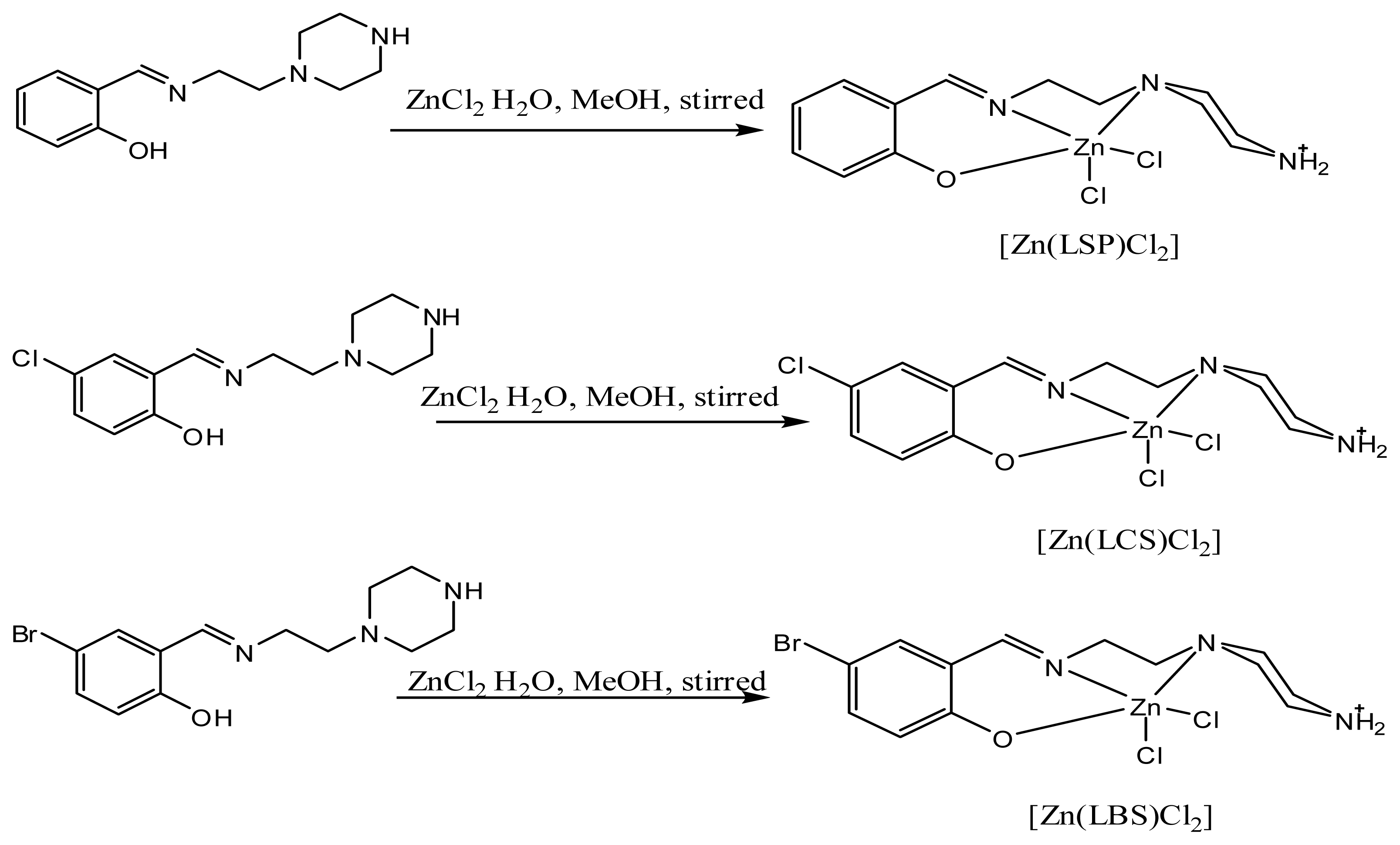
2.1. Chemistry
2.2. Acute Toxicity Study
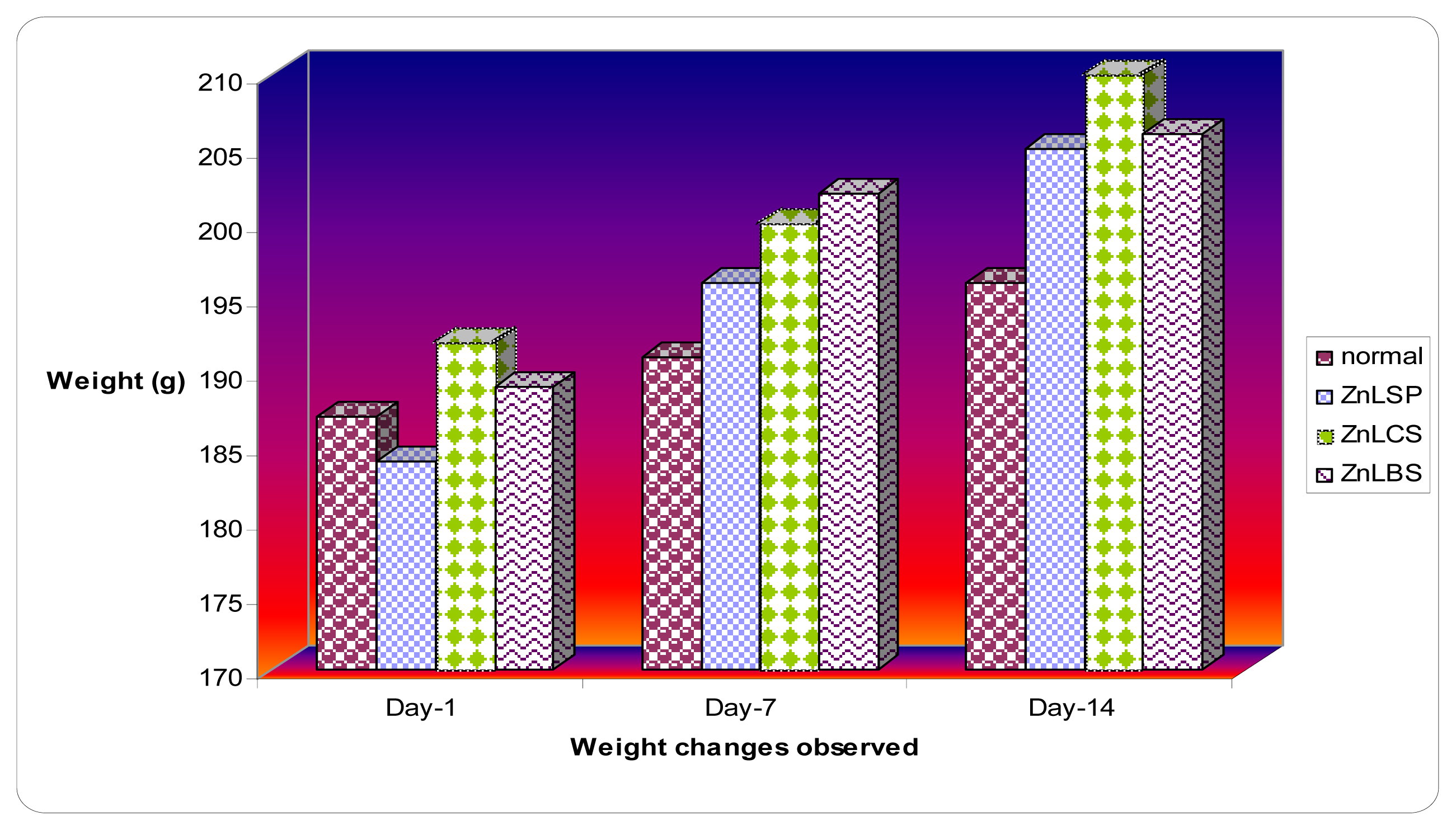
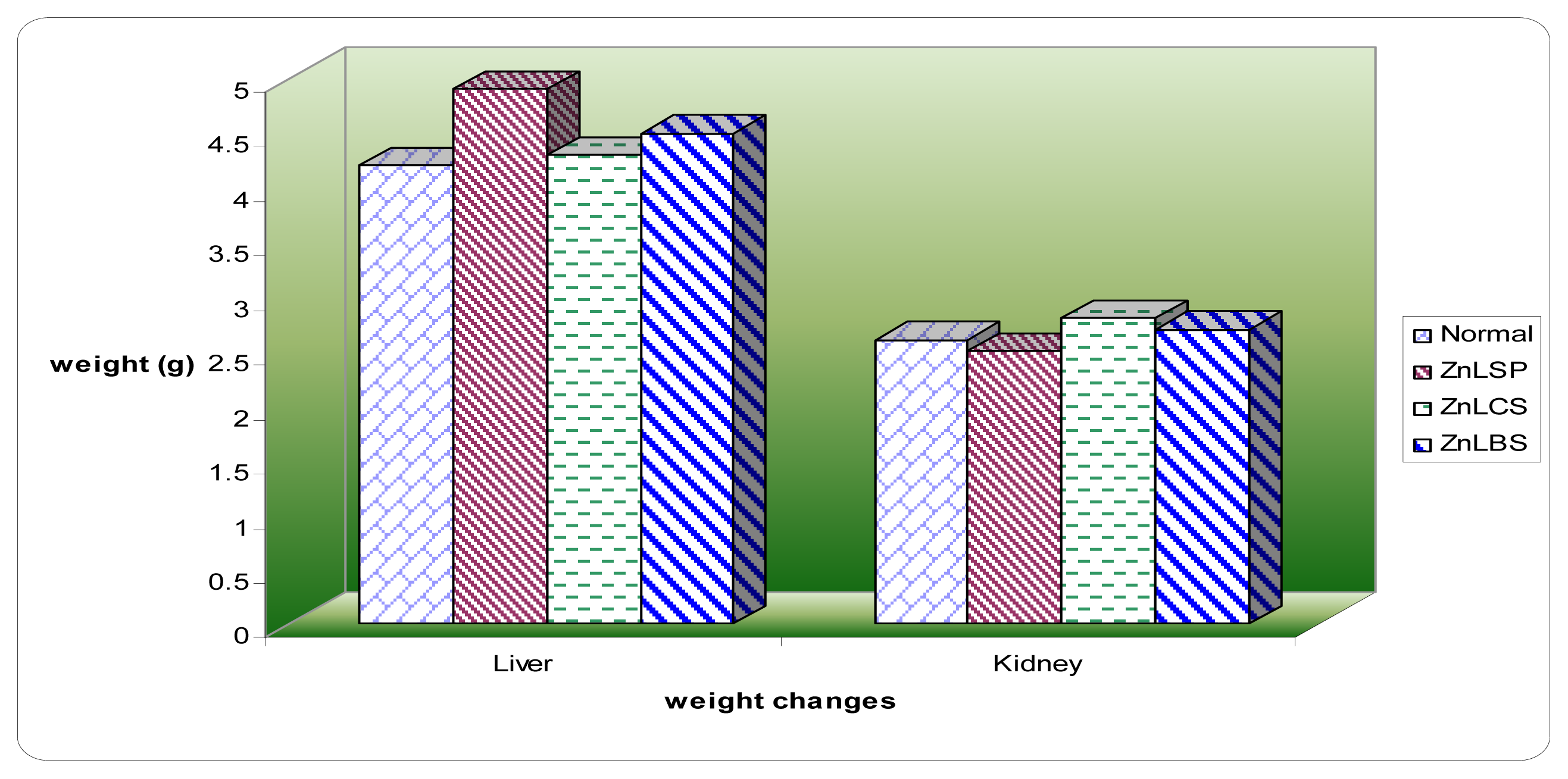
2.3. Body and Organ Weight Changes
2.4. Effects of Zinc Complexes on the Biochemical Indices
3. Experimental
3.1 Chemistry
3.2. Schiff Bases
3.3. Complexes
3.3.1. 2-((2-(piperazin-1-yl)ethylimino)methyl)phenol-dichlorido-Zn-(II): [Zn(LSP)Cl2]
3.3.2. 4-chloro-2-((2-(piperazin-1-yl)ethylimino)methyl)phenol-dichlorido-Zn(II): [Zn(LCS)Cl2]
3.3.3. 4-bromo-2-((2-(piperazin-1-yl)ethylimino)methyl)phenol-dichlorido-Zn-(II): [Zn(LBS)Cl2]
3.4. Animals
3.5. Acute Toxicity Test
3.6. Statistical Analysis
4. Conclusion
Acknowledgement
Reference
- Wintergerst, E.S.; Maggini, S.; Hornig, D.H. Immune-enhancing role of vitamin C and zinc and effect on clinical conditions. Ann. Nutr. Metab 2006, 50, 85–94. [Google Scholar]
- McCall, K.A.; Huang, C.; Fierke, C.A. Function and mechanism of zinc metalloenzymes. J. Nutr. 2000, 130, 1437S–1446S. [Google Scholar]
- O’Neil-Cutting, M.A.; Bomford, A.; Munro, H. Effect of excess dietary zinc on tissue storage of iron in rats. J. Nutr 1981, 111, 1969–1979. [Google Scholar]
- Barry, C.; Starcher, J.G.G.; Madaras, J.G. Zinc Absorption and Its Relationship to Intestinal Metallothionein. J. Nutr 1980, 110, 1391–1397. [Google Scholar]
- Cruickshank, P.; Sheehan, J.C. Gas chromatographic analysis of amino acids as N-trifluoroacetylamino acid methyl esters. Anal. Chem 1964, 36, 1191–1197. [Google Scholar]
- Jacobus, W.E.; Lehninger, A.L. Creatine kinase of rat heart mitochondria. J. Biol. Chem 1973, 248, 4803–4810. [Google Scholar]
- Lehninger, A.L.; Vercesi, A.; Bababunmi, E.A. Regulation of Ca2+ release from mitochondria by the oxidation-reduction state of pyridine nucleotides. Proc. Nat. Acad. Sci. USA 1978, 75, 1690–1694. [Google Scholar]
- Vazquez, M.A.; Munoz, F.; Donoso, J.; Blanco, F.G. Spectroscopic study of the Schiff bases of dodecylamine with pyridoxal 5′-phosphate and 5′-deoxypyridoxal. A model for the Schiff bases of pyridoxal 5′-phosphate in biological systems. Biochem. J 1991, 279, 759–767. [Google Scholar]
- Salata, C.A.; Youinou, M.T.; Burrows, C.J. Preparation and structural characterization of dicopper (II) and dinickel (II) imidazolate-bridged macrocyclic Schiff base complexes. Inorg. Chem 1991, 30, 3454–3461. [Google Scholar]
- Dowling, C.; Murphy, V.J.; Parkin, G. Bis (pyrazolylethyl) ether ligation to zinc and cobalt: Meridional vs facial coordination and the suitability of such ligands in providing a NNO donor set for modeling bioinorganic aspects of zinc chemistry. Inorg. Chem 1996, 35, 2415–2420. [Google Scholar]
- Cohen, S.M. New approaches for medicinal applications of bioinorganic chemistry. Curr. Opin. Chem. Biol 2007, 11, 115–120. [Google Scholar]
- Rafique, S.; Idrees, M.; Nasim, A.; Akbar, H.; Athar, A. Transition metal complexes as potential therapeutic agents. Biotech. Mol. Biol. Rev 2010, 5, 38–45. [Google Scholar]
- Nishat, N.; Haq, M.M.; Ahamad, T.; Kumar, V. Synthesis, spectral and antimicrobial studies of a novel macrocyclic ligand containing a piperazine moiety and its binuclear metal complexes. J. Coord. Chem 2007, 60, 85–96. [Google Scholar]
- Keypour, H.; Rezaeivala, M.; Valencia, L.; Perez-Lourido, P. Synthesis and crystal structure of Mn (II) complexes with novel macrocyclic Schiff-base ligands containing piperazine moiety. Polyhedron 2008, 27, 3172–3176. [Google Scholar]
- Islam, M.K.; Miyoshi, T.; Yamada, M.; Alim, M.A.; Huang, X.; Motobu, M.; Tsuji, N. Effect of piperazine (diethylenediamine) on the moulting, proteome expression and pyrophosphatase activity of Ascaris suum lung-stage larvae. Acta. Trop 2006, 99, 208–217. [Google Scholar]
- Chaudhary, P.; Kumar, R.; Verma, A.K.; Singh, D.; Yadav, V.; Chhillar, A.K.; Sharma, G.L.; Chandra, R. Synthesis and antimicrobial activity of N-alkyl and N-aryl piperazine derivatives. Bioorgan. Med. Chem 2006, 14, 1819–1826. [Google Scholar]
- Narendra, S.C.J.N.; Sadashiva, C.T.; Kavitha, C.V.; Rangappa, K.S. Synthesis and in vitro antimicrobial studies of medicinally important novel N-alkyl and N-sulfonyl derivatives of 1-[bis (4-fluorophenyl)-methyl] piperazine. Bioorg. Med. Chem 2006, 14, 6621–6627. [Google Scholar]
- Sadashiva, C.T.; Narendra, S.C.J.N.; Ponnappa, K.C.; Veerabasappa, G.T.; Rangappa, K.S. Synthesis and efficacy of 1-[bis (4-fluorophenyl)-methyl] piperazine derivatives for acetylcholinesterase inhibition, as a stimulant of central cholinergic neurotransmission in Alzheimer’s disease. Bioorgan. Med. Chem. Lett. 2006, 16, 3932–3936. [Google Scholar]
- Nozawa, D.; Okubo, T.; Ishii, T.; Takamori, K.; Chaki, S.; Okuyama, S.; Nakazato, A. Novel piperazines: Potent melanocortin-4 receptor antagonists with anxiolytic-like activity. Bioorg. Med. Chem 2007, 15, 2375–2385. [Google Scholar]
- Chen, C.; Jiang, W.; Tucci, F.; Tran, J.A.; Fleck, B.A.; Hoare, S.R.; Joppa, M.; Markison, S.; Wen, J.; Chen, C.W.; et al. Discovery of 1-{2-[(1S)-(3-Dimethylamino-propionyl)amino-2-methylpropyl]-4-methyl-phenyl}-4-[(2R)-methyl-3-(4-chlorophenyl)-propionyl] piperazine as an Orally Active Antagonist of the Melanocortin-4 Receptor for the Potential Treatment of Cachexia. J. Med. Chem 2007, 50, 5249–5252. [Google Scholar]
- Staack, R.F.; Paul, L.D.; Springer, D.; Kraemer, T.; Maurer, H.H. Cytochrome P450 dependent metabolism of the new designer drug 1-(3-trifluoromethylphenyl) piperazine (TFMPP): In vivo studies in Wistar and Dark Agouti rats as well as in vitro studies in human liver microsomes. Biochem. Pharmacol 2004, 67, 235–244. [Google Scholar]
- Serradji, N.; Bensaid, O.; Martin, M.; Sallem, W.; Dereuddre-Bosquet, N.; Benmehdi, H.; Redeuilh, C.; Lamouri, A.; Dive, G.; Clayette, P.; et al. Structure-activity relationships in platelet-activating factor. Part 13: Synthesis and biological evaluation of piperazine derivatives with dual anti-PAF and anti-HIV-1 or pure antiretroviral activity. Bioorg. Med. Chem 2006, 14, 8109–8125. [Google Scholar]
- Benmehdi, H.; Lamouri, A.; Serradji, N.; Pallois, F.; Heymans, F. Synthesis of new trisubstituted 4-aminopiperidines as PAF-receptor antagonists. Eur. J. Org. Chem 2008, 2008, 299–307. [Google Scholar]
- Sallem, W.; Serradji, N.; Dereuddre-Bosquet, N.; Dive, G.; Clayette, P.; Heymans, F. Structure-activity relationships in platelet-activating factor. Part 14: Synthesis and biological evaluation of piperazine derivatives with dual anti-PAF and anti-HIV-1 activity. Bioorg. Med. Chem 2006, 14, 7999–8013. [Google Scholar]
- Salga, M.S.; Ali, H.M.; Abdullah, M.A.; Abdelwahab, S.I.; Hussain, P.D.; Hadi, A.H.A. Mechanistic studies of the anti-ulcerogenic activity and acute toxicity evaluation of dichlorido-copper (II)-4-(2-5-Bromo-benzylideneamino) ethyl) piperazin-1-ium phenolate complex against ethanol-induced gastric injury in rats. Molecules 2011, 16, 8654–8669. [Google Scholar]
- Yu, M.; Lizarzaburu, M.; Beckmann, H.; Connors, R.; Dai, K.; Haller, K.; Li, C.; Liang, L.; Lindstrom, M.; Ma, J.; et al. Identification of piperazine-bisamide GHSR antagonists for the treatment of obesity. Bioorg. Med. Chem. Lett 2010, 20, 1758–1762. [Google Scholar]
- Salga, S.M.; Ali, H.M.; Abdullah, M.A.; Abdelwahab, S.I.; Wai, L.K.; Buckle, M.J.C.; Sukumaran, S.D.; Hadi, A.H.A. Synthesis, characterization, acetylcholinesterase inhibition, molecular modeling and antioxidant activities of some novel schiff bases derived from 1-(2-ketoiminoethyl) piperazines. Molecules 2011, 16, 9316–9330. [Google Scholar]
- Nath, M.; Saini, P.K.; Kumar, A. New di-and triorganotin (IV) complexes of tripodal Schiff base ligand containing three imidazole arms: Synthesis, structural characterization, anti-inflammatory activity and thermal studies. J. Organomet. Chem 2010, 695, 1353–1362. [Google Scholar]
- Yuan, R.; Chai, Y.; Liu, D.; Gao, D.; Li, J.; Yu, R. Schiff base complexes of cobalt (II) as neutral carriers for highly selective iodide electrodes. Anal. Chem 1993, 65, 2572–2575. [Google Scholar]
- Holm, R.H. Studies on Ni (II) complexes. I. Spectra of tricyclic schiff base complexes of Ni (II) and Cu (II). J. Am. Chem. Soc 1960, 82, 5632–5636. [Google Scholar]
- Cimerman, Z.; Galesic, N.; Bosner, B. Structure and spectroscopic characteristics of Schiff bases of salicylaldehyde with 2,3-diaminopyridine. J. Mol. Struct 1992, 274, 131–144. [Google Scholar]
- Vlcek, A. Mechanistic roles of metal-to-ligand charge-transfer excited states in organometallic photochemistry. Coord. Chemi. Rev 1998, 177, 219–256. [Google Scholar]
- Koester, V.J. Interligand transmetallic charge-transfer transitions in mixed-ligand chelates. Chem. Phys. Lett 1975, 32, 575–580. [Google Scholar]
- Zalis, S.; Farrell, I.R.; Vlcek, A., Jr. The involvement of metal-to-CO charge transfer and ligand-field excited states in the spectroscopy and photochemistry of mixed-ligand metal carbonyls. A theoretical and spectroscopic study of [W (CO) 4(1,2-ethylenediamine)] and [W (CO) 4 (N,N′-bis-alkyl-1,4-diazabutadiene)]. J. Am. Chem. Soc. 2003, 125, 4580–4592. [Google Scholar]
- Bagus, P.S.; Hermann, K.; Bauschlicher, C.W., Jr. A new analysis of charge transfer and polarization for ligand–metal bonding: Model studies of AlCO and AlNH. J. Chem. Phys 1984, 80. [Google Scholar] [CrossRef]
- Ganter, B.; Tugendreich, S.; Pearson, C.I.; Ayanoglu, E.; Baumhueter, S.; Bostian, K.A.; Brady, L.; Browne, L.J.; Calvin, J.T.; Day, G.J.; et al. Development of a large-scale chemogenomics database to improve drug candidate selection and to understand mechanisms of chemical toxicity and action. J. Biotechnol 2005, 119, 219–244. [Google Scholar]
- Rozman, K.K.; Doull, J. Dose and time as variables of toxicity. Toxicology 2000, 144, 169–178. [Google Scholar]
- Bugrim, A.; Nikolskaya, T.; Nikolsky, Y. Early prediction of drug metabolism and toxicity: Systems biology approach and modeling. Drug Dis. Today 2004, 9, 127–135. [Google Scholar]
- Benigni, R.; Tatiana, I.N.; Benfenati, E.; Bossa, C.; Franke, R.; Helma, C.; Hulzebos, E.; Marchant, C.; Richard, A.; Woo, Y.T.; et al. The expanding role of predictive toxicology: An update on the (Q) SAR models for mutagens and carcinogens. J. Environ. Sci. Hea. C 2007, 25, 53–97. [Google Scholar]
- Cronin, M.T.D.; Jaworska, J.S.; Walker, J.D.; Comber, M.H.I.; Watts, C.D.; Worth, A.P. Use of QSARs in international decision-making frameworks to predict health effects of chemical substances. Environ. Health Persp 2003, 111, 1391–1401. [Google Scholar]
- Das, B.K.; Mukherjee, S.C. Toxicity of cypermethrin in Labeo rohita fingerlings: Biochemical, enzymatic and haematological consequences. Comp. Biochem. Phys. C 2003, 134, 109–121. [Google Scholar]
- Basketter, D.A.; Bremmer, J.N.; Buckley, P.; Kammuller, M.E.; Kawabata, T.; Kimber, I.; Loveless, S.E.; Magda, S.; Stringer, D.A.; Vohr, H.W. Pathology considerations for, and subsequent risk assessment of, chemicals identified as immunosuppressive in routine toxicology. Food Chem. Toxicol 1995, 33, 239–243. [Google Scholar]
- Mukhopadhyay, S.; Mandal, D.; Ghosh, D.; Goldberg, I.; Chaudhury, M. Equilibrium studies in solution involving nickel (II) complexes of flexidentate Schiff base ligands: Isolation and structural characterization of the planar red and octahedral green species involved in the equilibrium. Inorg. Chem 2003, 42, 8439–8445. [Google Scholar]
- Salga, M.S.; Ali, H.M.; Abdulla, M.A.; Abdelwahab, S.I. Gastroprotective activity and mechanism of novel dichlorido-zinc (II)-4-(2-5-methoxybenzylideneamino) ethyl) piperazin-1-iumphenolate complex on ethanol-induced gastric ulceration. Chem. Biol. Interact 2011, in press. [Google Scholar]
| Indices | Normal | [Zn(LSP)Cl2] | [Zn(LCS)Cl2] | [Zn(LBS)Cl2] |
|---|---|---|---|---|
| Sodium | 139.5 ± 2.6 | 139.2 ± 2.4 | 138.2 ± 2.7 | 138.5 ± 2.9 |
| Potassium | 5.05 ± 0.8 | 5.50 ± 0.6 | 4.70 ± 0.9 | 5.100 ± 0.7 |
| Chloride | 103.8 ± 1.3 | 104.6 ± 2.3 | 105.1 ± 1.2 | 102.3 ± 1.7 |
| CO2 | 23.9 ± 2.1 | 22.2 ± 2.6 | 21.3 ± 3.2 | 23.10 ± 3.3 |
| Anion gap | 17.4 ± 1.2 | 18.5 ± 0.7 | 16.5 ± 3.1 | 18.50 ± 3.5 |
| Urea | 6.10 ± 1.3 | 7.40 ± 0.6 | 7.60 ± 1.7 | 9.300 ± 2.6 |
| Creatinine | 42.5 ± 1.9 | 28.4 ± 17 | 40.8 ± 1.9 | 50.50 ± 1.9 |
| Indices | Normal | [Zn(LSP)Cl2] | [Zn(LCS)Cl2] | [Zn(LBS)Cl2] |
|---|---|---|---|---|
| Total protein | 70.5 ± 3.6 | 87.3 ± 4.9 | 75.5 ± 3.2 | 81.8 ± 2.6 |
| Albumin | 59.5 ± 2.4 | 68.6 ± 4.1 | 62.5 ± 3.2 | 69.3 ± 0.5 |
| Globulin | 59.5 ± 3.4 | 69.5 ± 4.9 | 61.9 ± 2.2 | 65.3 ± 2.4 |
| Alk. Phosphate | 59.30 ± 11.3 | 81.30 ± 10.8 | 82.3 ± 11.3 | 92.80+12.5 |
| ALT | 49.8 ± 7.2 | 61.0 ± 3.6 | 57.8 ± 3.4 | 55.3 ± 4.9 |
| AST | 259.8 ± 12.7 | 292.3 ± 10.6 | 278.5 ± 9.8 | 281.5 ± 12.6 |
| Total bilirubin | 6.25 ± 0.5 | 7.50 ± 0.7 | 6.88 ± 0.8 | 7.32 ± 0.8 |
| C.bilurubin | 3.61 ± 0.9 | 5.83 ± 1.3 | 3.85 ± 1.4 | 4.22 ± 2.1 |
| Triglyceride | 0.45 ± 0.1 | 0.60 ± 0.4 | 0.30 ± 0.05 | 0.80 ± 0.8 |
| Total cholesterol | 2.20 ± 0.3 | 3.70 ± 0.5 | 3.30 ± 0.1 | 3.6 0± 0.4 |
| HDL | 1.53 ± 0.4 | 1.47 ± 0.2 | 1.39 ± 0.4 | 1.50 ± 0.6 |
| Indices | Normal | [Zn(LSP)Cl2] | [Zn(LCS)Cl2] | [Zn(LBS)Cl2] |
|---|---|---|---|---|
| HGB | 151.3 ± 11.0 | 162.3 ± 11.8 | 158.3 ± 12.2 | 163.3 ± 13.5 |
| HCT | 0.540 ± 0.21 | 0.980 ± 0.03 | 0.830 ± 0.01 | 0.920 ± 0.04 |
| RBC | 7.90 ± 0.4 | 8.10 ± 0.3 | 8.60 ± 0.3 | 8.80 ± 0.7 |
| MCV | 65.2 ± 2.4 | 69.3 ± 1.5 | 67.9 ± 1.8 | 76.9 ± 1.8 |
| MCH | 17.9 ± 0.9 | 18.1 ± 0.3 | 18.9 ± 0.6 | 18.6 ± 0.5 |
| MCHC | 291.2 ± 2.3 | 260.5 ± 4.4 | 302.3 ± 5.6 | 269.3 ± 4.5 |
| RDW | 15.8 ± 1.4 | 17.5 ± 1.2 | 16.6 ± 1.5 | 17.9 ± 2.1 |
| WBC | 10.7 ± 3.1 | 11.3 ± 0.4 | 12.9 ± 4.4 | 12.4 ± 1.8 |
| Platelet | 654.3 ± 9.5 | 846.6 ± 8.5 | 689.3 ± 9.7 | 708.3 ± 9.4 |
| Indices | Normal | [Zn(LSP)Cl2] | [Zn(LCS)Cl2] | [Zn(LBS)Cl2] |
|---|---|---|---|---|
| ImmunoglobulinG | 933.4 ± 2.3 | 933.8 ± 4.2 | 933.5 ± 3.6 | 933.9 ± 4.8 |
| Immunoglobulin A | 97.5 ± 2.2 | 89.6 ± 3.7 | 98.7 ± 4.4 | 99.2 ± 4.3 |
| Immunoglobulin M | 43.9 ± 7.9 | 42.3 ± 3.5 | 52.7 ± 8.3 | 63.5 ± 9.6 |
| Complement 3 | 96.8 ± 2.3 | 96.2 ± 1.2 | 96.6 ±3.2 | 96.9 ±1.7 |
| Complement 4 | 29.8 ±2.1 | 52.9 ±3.2 | 57.2 ± 4.2 | 57.9 ± 4.2 |
| C-reactive Protein | 0.42 ± 0.3 | 0.34 ± 0.5 | 0.45 ± 0.2 | 0.53 ± 0.2 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Salga, M.S.; Ali, H.M.; Abdulla, M.A.; Abdelwahab, S.I. Acute Oral Toxicity Evaluations of Some Zinc(II) Complexes Derived from 1-(2-Salicylaldiminoethyl)piperazine Schiff Bases in Rats. Int. J. Mol. Sci. 2012, 13, 1393-1404. https://doi.org/10.3390/ijms13021393
Salga MS, Ali HM, Abdulla MA, Abdelwahab SI. Acute Oral Toxicity Evaluations of Some Zinc(II) Complexes Derived from 1-(2-Salicylaldiminoethyl)piperazine Schiff Bases in Rats. International Journal of Molecular Sciences. 2012; 13(2):1393-1404. https://doi.org/10.3390/ijms13021393
Chicago/Turabian StyleSalga, Muhammad Saleh, Hapipah Mohd Ali, Mahmood Ameen Abdulla, and Siddig Ibrahim Abdelwahab. 2012. "Acute Oral Toxicity Evaluations of Some Zinc(II) Complexes Derived from 1-(2-Salicylaldiminoethyl)piperazine Schiff Bases in Rats" International Journal of Molecular Sciences 13, no. 2: 1393-1404. https://doi.org/10.3390/ijms13021393




