1. Introduction
Lichens are symbiotic organisms built from fungi and a photosynthetic partner, which is either alga or Cyanobacterium [
1–
3]. They usually grow on rocks, non-fertile ground, and as epiphytes on trees and leaves [
4]. For hundreds of years, lichens have been used in many Europeans country as a cure for stomach diseases, diabetes, coughs, pulmonary tuberculosis, wound curing, and dermatological diseases [
5]. Besides, many sorts are used for human nutrition, animal nutrition, the production of colors, perfumes, alcohols and in the medical industry [
6].
Lichens synthesize, mostly by a fungal metabolism, various bioactive secondary components, that sometimes constitute even more than 30% of the dry mass of talus [
7]. They are found as crystal deposits on the surface of hiphes. They are poorly soluble in water and can usually be isolated from a lichen by means of organic diluents [
8]. More than one hundred secondary metabolites, mainly monoaromatics, depsides, depsidones, pulvinates, dibenzofurans, anthraquinones and xanthones, characteristic of lichens have been detected and isolated [
9]. Chemicals structures of these classes of compounds are similar and identification is often very difficult.
Lichen metabolites exert a large variety of biological actions including antibiotic, antimycotic, antiviral, anti-inflammatory, analgesic, antipyretic, antiproliferative and cytotoxic effects [
10,
11]. Here, we report the chemical composition of
Toninia candida and
Usnea barbata extracts using HPLC-UV analysis and
in vitro antioxidant, antimicrobial and anticancer activities of these lichens and their norstictic acid and usnic acid constituents.
2. Results and Discussion
This paper deals with the phytochemical analysis of acetone extracts from the species
T. candida and
U. barbata, lichens growing in Serbia, and the isolation of their major metabolites.
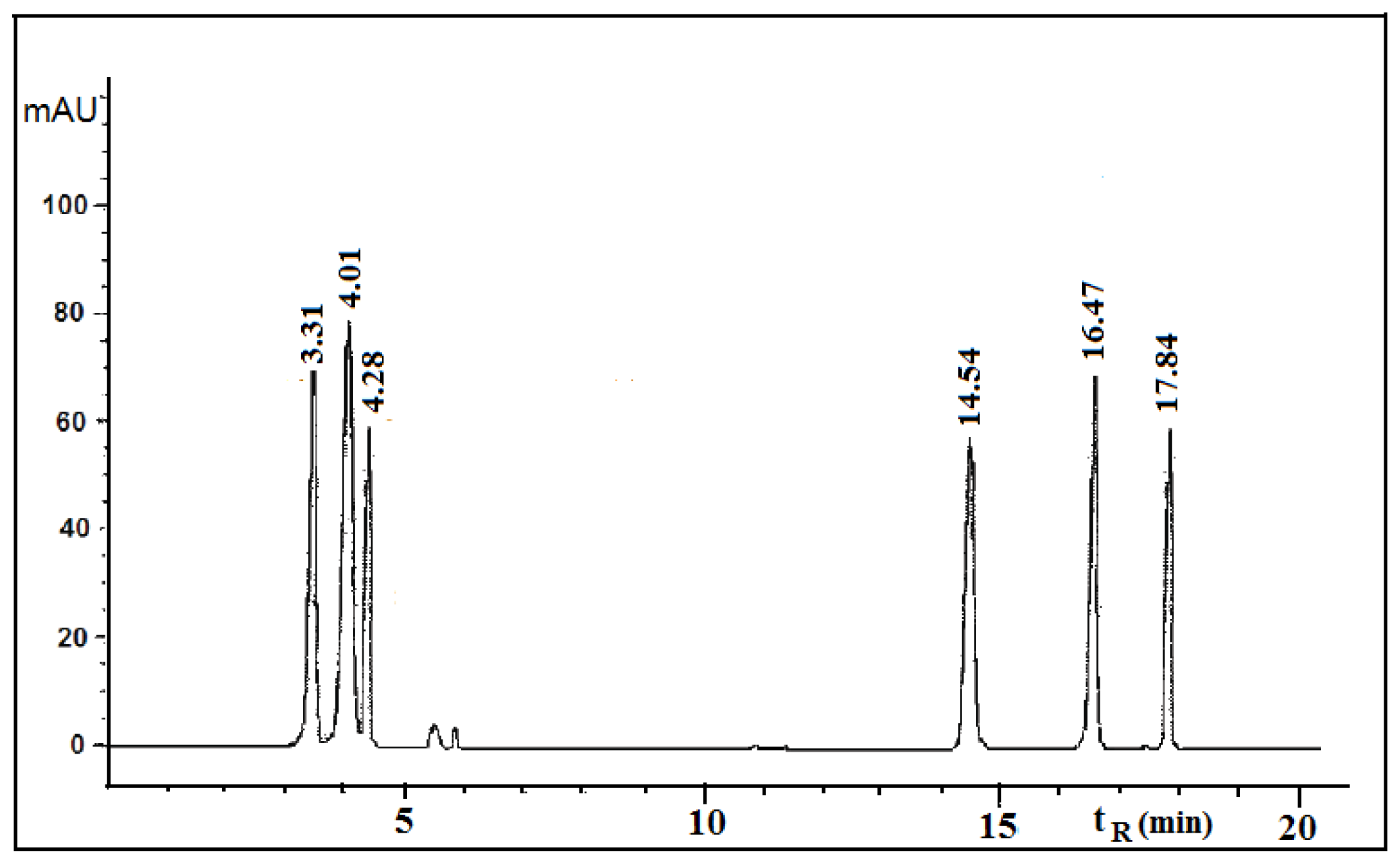
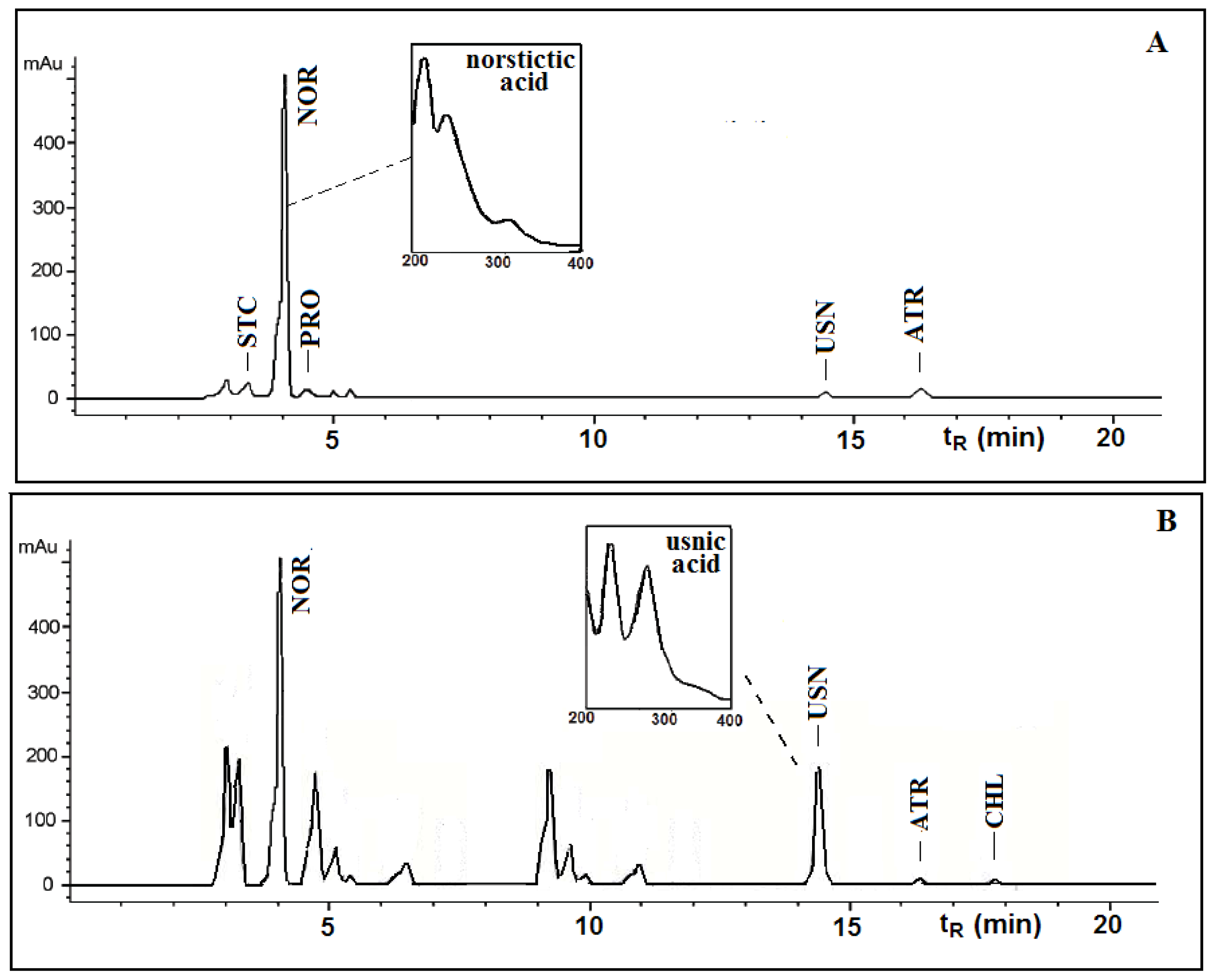
Figures 1 and
2 show the HPLC chromatograms of the standards and the acetone extracts from the species
T. candida and
U. barbata. In addition to the main chemical compound (norstictic acid), stictic acid, protocetraric acid, usnic acid and atranorin were also identified in the acetone extract of
T. candida. On the other hand, the acetone extract of
U. barbata showed the presence of norstictic acid, usnic acid, atranorin and chloroatranorin. Identification of these compounds was achieved by comparison of their
tR values with the standard substances previously isolated from lichens. Norstictic acid has specific UV spectra different from other depsidones with absorption maxima at 212, 238 and 310 nm. Compared with many other lichen substances, the UV spectrum of usnic acid is very specific and this acid can easily be identified by its UV spectrum (absorbance maxima at 234 and 284 nm) and its retention time by HPLC. After HPLC analysis of the tested lichen extracts, their major lichen metabolites, norstictic and usnic acids, were isolated from the acetone extracts of
T. candida and
U. barbata, respectively.
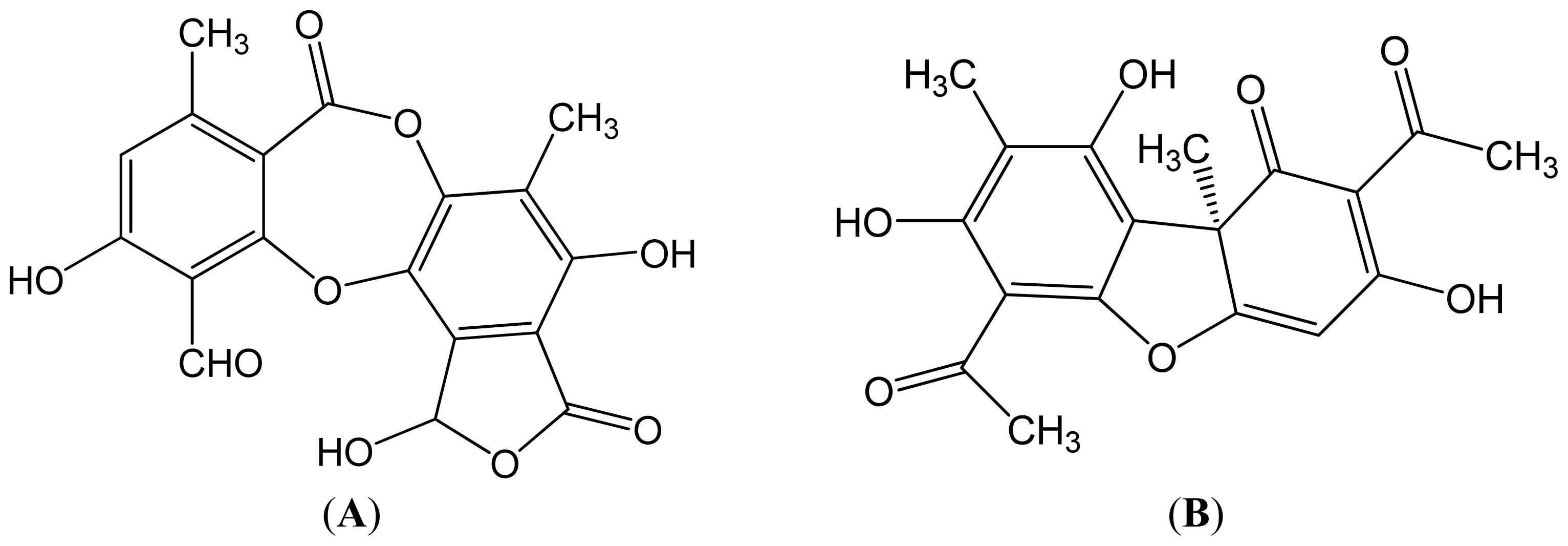
Table 1 shows the retention time of the detected lichen substances and their absorbance maxima (nm). Norstictic acid belongs to the class of depsidones, while usnic acid is a well-known lichen metabolite with a dibenzofurane structure. The structures of the detected compounds are shown in
Figure 3. These pure compounds were further used for antioxidant, antimicrobial and anticancer investigations.
The scavenging DPPH radicals of the studied samples are shown in
Table 2. The IC
50 values of all extracts and compounds ranged from 102.65 to 979.30 μg/mL. There was a statistically significant difference between samples and the control (
p < 0.05). Among the tested extracts, the acetone extract from
T. candida showed the largest DPPH radical scavenging activity (IC
50 = 115.77 μg/mL). The lichen components isolated demonstrated very strong DPPH radical scavenging activity, larger than those from extracts. Usnic acid showed a smaller DPPH radical scavenging activity than norstictic acid. The IC
50 values for norstictic acid and usnic acid were 102.65 and 130.73 μg/mL respectively.
The results of the reducing power assay of the tested extracts and compounds are summarized in
Table 3. Higher absorbance indicates higher reducing power. Among the lichen extracts,
T. candida showed the highest reducing power. Norstictic acid and usnic acid showed a very high reducing power, which was higher than those from extracts.
The scavenging of superoxide anion radicals by the tested lichen extracts and compounds is shown in
Table 2. There was a statistically significant difference between samples and the control (
p < 0.05). The lichen components isolated showed the highest superoxide anion radical scavenging activity (the IC
50 was 133.46 μg/mL for norstictic and 197.28 μg/mL for usnic acid). The scavenging activities of tested extracts were somewhat lower.
The total phenolic content of the tested lichen extracts are given in
Table 4. The total amount of phenolic compounds was determined as the pyrocatechol equivalent using an equation obtained from a standard pyrocatechol graph. The total phenolic contents of the acetone extracts of
T. candida and
U. barbata were 49.81 and 31.25 μg PE/mg respectively.
Various antioxidant activities were compared to ascorbic acid. The results showed that the standard antioxidant had a stronger activity than the tested samples.
The tested lichen extracts have a strong antioxidant activity against various oxidative systems
in vitro. The strong antioxidant activity of the tested lichen extracts is correlated with a high content of total phenols. In fact, it was observed that the examined lichen extracts with the higher content of phenols exert a stronger radical scavenging effect, suggesting that phenolics are the main agents for their antioxidant activity. These results mostly correspond to the literature, where we have found a number of reports for the antioxidant activity of extracts with a high content of phenolic compounds [
12]. In most lichens, phenols, including depsides, depsidones, and dibenzofurans, are important antioxidants because of their ability to scavenge free radicals such as singlet oxygen, superoxide, and hydroxyl radicals [
10]. As well, in our experiment the isolated components norstictic acid and usnic acid which are classified as phenols, also exhibited a significantly stronger antioxidant activity than the extracts, which indicates the important role of phenols in antioxidant activity. However, some authors believe that the antioxidant activity of extracts may not necessarily be correlated with the content of polyphenolics, suggesting that the antioxidant activity of different lichens may also depend on other, non-phenol components [
13].
The antioxidant effect of some other lichens was also studied by other researchers. For example, Luo
et al.[
14] determined the antioxidant activity of methanol extracts from the lichen
Thamnolia vermicularis. Praveen Kumar
et al.[
15] determined the antioxidant activity of the extracts of the lichen
Ramalina hossei and
Ramalina conduplicans. Manojlović
et al.[
16] explored the antioxidant properties of
Laurera benguelensis.
The antimicrobial activities of the lichen extracts and lichen components against the test microorganisms are shown in
Table 5. The extract from
U. barbata showed a moderate antibacterial and antifungal activity. It inhibited the microorganisms tested at concentrations from 0.125 to 12.5 mg/mL. The extract from
T. candida inhibited all the tested microorganisms, but at higher concentrations. The lichen components isolated demonstrated very strong antimicrobial activity. The MIC for different components relative to the tested microorganisms ranged from 0.0008 to 1 mg/mL. The strongest antimicrobial activity was found in usnic acid, which in extremely low amounts (
Table 5) inhibited all species of bacteria and fungi.
The antimicrobial activity was compared with the standard antibiotics, streptomycin (for bacteria) and ketoconazole (for fungi). The results showed that standard antibiotics had a similar or stronger activity than the tested samples as shown in
Table 5. In a negative control, DMSO had no inhibitory effect on the tested organisms.
In our experiments, the tested lichen extracts showed a relatively strong antimicrobial activity but the antimicrobial activity of their components was much stronger. This means that the lichen components are responsible for the antimicrobial activity of the lichens. Differences in antimicrobial activity of different species of lichens are probably a consequence of the presence of different components with varying antimicrobial activity [
17]. However, it is necessary to understand that extracts are mixtures of natural compounds, and their antimicrobial activity is not only a result of the different activities of the individual components but may also be the result of their interactions, which can cause different effects on the overall activity of the extracts.
The intensity of the antimicrobial effect depended on the species of organism tested. The extracts and compounds used in this study, had stronger antibacterial than antifungal activity. This observation is in accordance with other studies [
17,
18], focused on antimicrobial activity which have demonstrated that bacteria are more sensitive to the antimicrobial activity than the fungi due to differences in the composition and permeability of the cell wall. The cell wall of Gram-positive bacteria is composed of peptidoglucanes and teichoic acids, while the cell wall of Gram-negative bacteria is composed of peptidoglucanes, lipopolysacharides and lipoproteins [
10,
19]. The cell wall of fungi is poorly permeable and consists of polysaccharides such as hitchin and glucan [
20].
Numerous lichens were screened for antimicrobial activity in search of new antimicrobial agents. Kosanić
et al.[
10] find antimicrobial activity for the acetone extract of the lichens
Umbilicaria crustulosa,
U. cylindrica, and
U. polyphylla. Similar results were reported by Karthikai Devi
et al.[
21] for different extracts extracted from the lichen
Roccella belangeriana. Goel
et al.[
22] found that the lichen
Parmotrema reticulatum had a strong antimicrobial influence.
The cytotoxic activity of the tested lichen extracts and compounds against the tested cell lines is shown in
Table 6. The tested samples exhibited high cytotoxic activity against the target cells
in vitro. The IC
50 value for different samples relative to the tested cells ranged from 12.72 to 56.96 μg/mL. The best cytotoxic activity was exhibited for usnic acid. The IC
50 against FemX and LS174 cell lines was very low (12.72 and 15.66 μg/mL, respectively).
As shown in the table, the positive control (Cis-DDP) had a slightly better cytotoxic activity than the tested samples.
The effect of tested samples on the cell cycle was evaluated using flow cytometric analysis.
Table 7 shows a cell-cycle distribution of FemX and LS174 cells incubated in the absence or presence of compounds (IC
50) for 24 h, approximately double the time of this cell line. Treatment of FemX cells with tested extracts for 24 h led to a significant decrease of G1 cells and an increase of G2/M cells, revealing cell cycle arrest at this phase. On the other hand, treatment of the tested compounds led to an increase in the number of cells in the sub-G1 phase whiled the percentage of cells in the S-phase and G2/M phase remained unchanged compared to the controls. Interestingly, LS174 cells treated with the tested samples showed a significant increase of the sub-G1 phase and concomitant decrease in G2/M was observed, supporting a G1 phase arrest. These results suggested that the tested samples have a prominent ability to induce apoptosis in FemX and LS174 cells.
In the present study, the results clearly demonstrate that the studied lichen components induced a significant cytotoxic effect on the tested cancer cell lines, which was stronger than the lichen extracts. Some literature data have reported that lichen components are responsible for the anticancer activities of lichens [
23,
24]. However, it is difficult to determine the contribution of individual components on the overall anticancer effect. Often, the activity of the extracts may be the result of synergistic or antagonistic effects of several compounds.
The importance of lichens as anticancer agents has been confirmed in recent years, suggesting that lichens can be used as biological agents in the treatment of cancer. The mechanism of action of the tested extracts and their compounds is yet to be tested. Further research will be necessary for fractionation in order to identify the compounds responsible for the observed anti-tumor effects, and to establish the opportunities for reinforcement activities as well as to improve the selectivity.
Until now, only a few research examples have proved that lichens have anticancer activity. Bezivin
et al.[
25] reported a significant anticancer effect for
Parmelia caperata,
Cladonia convoluta,
Cladonia rangiformis,
Platismatia glauca and
Ramalina cuspidata. Manojlović,
et al.[
11] explored the anticancer properties of
Thamnolia vermicularis. Trigiani
et al.[
26] found strong anticancer activity for
Xanthoria parietina.