Fungus-Mediated Green Synthesis of Silver Nanoparticles Using Aspergillus terreus
Abstract
:1. Introduction
2. Results and Discussion
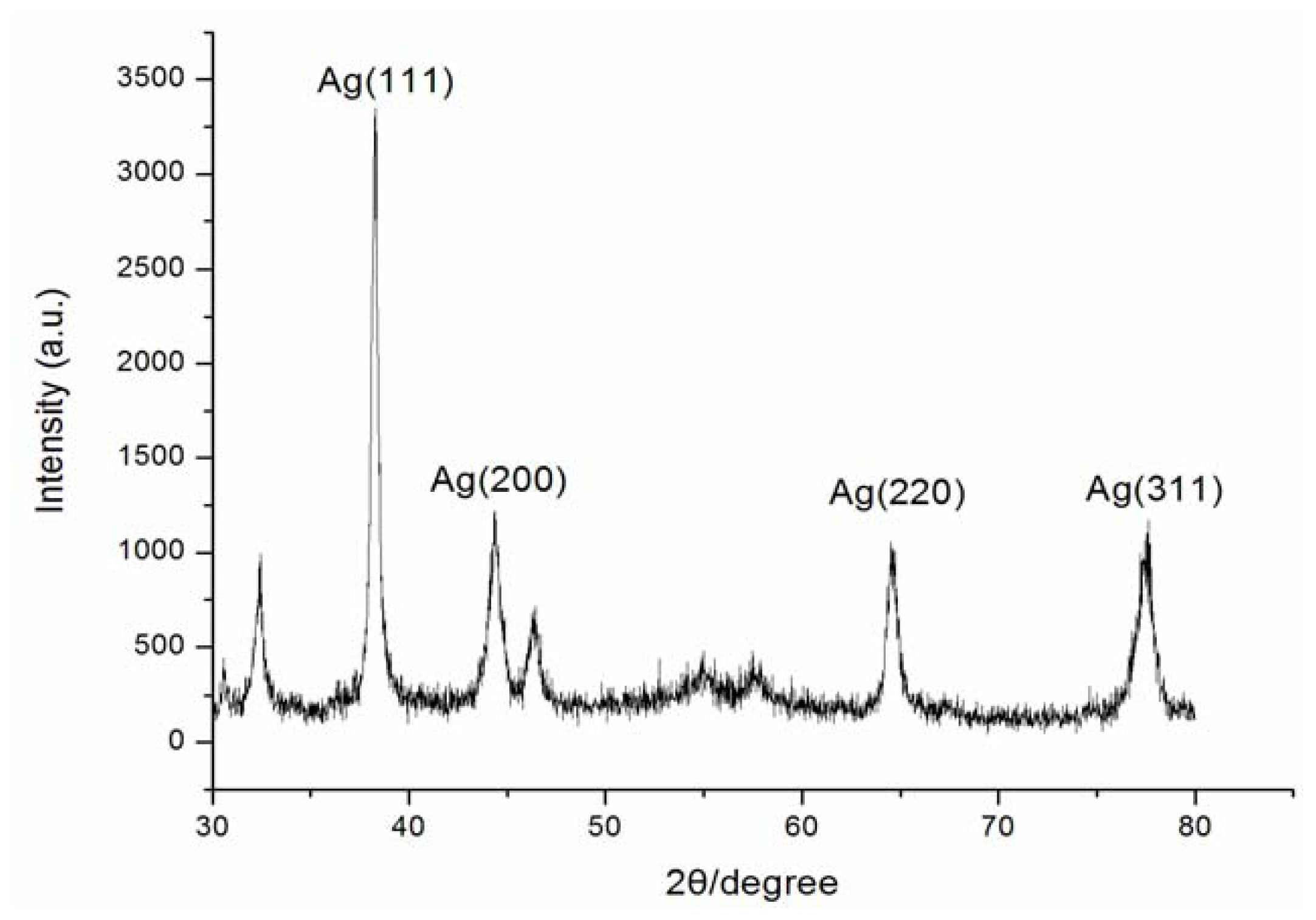
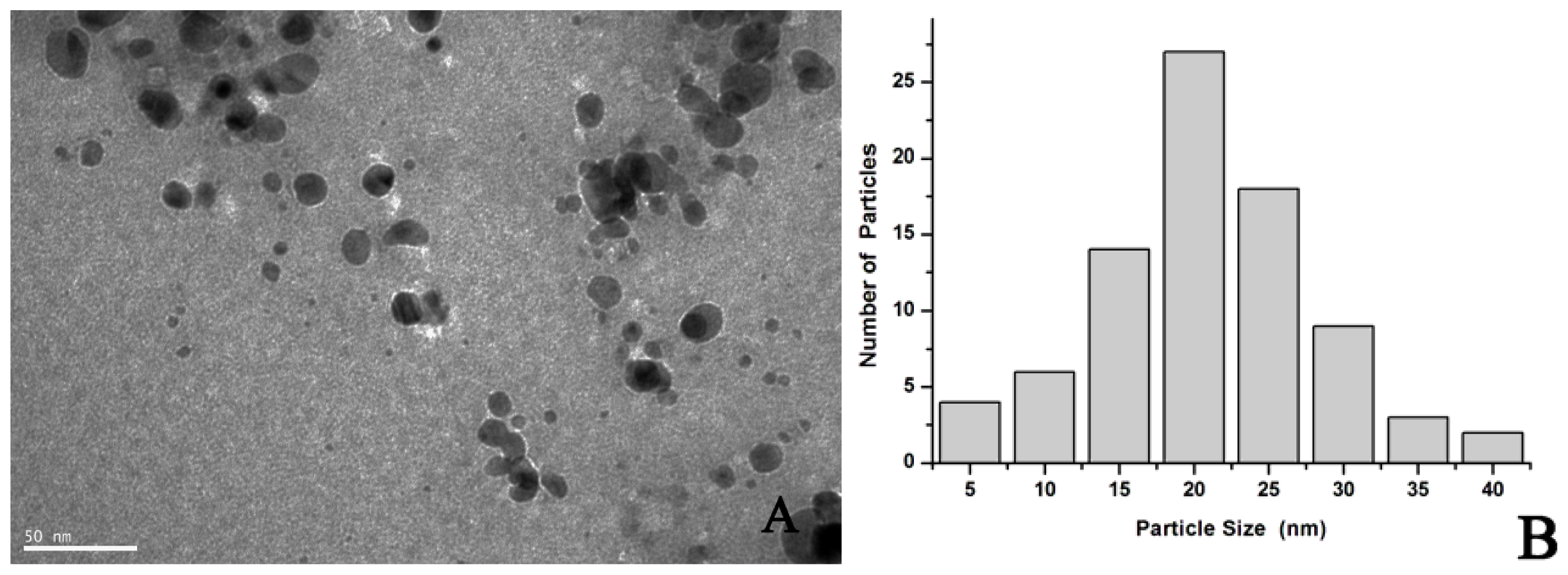
2.1. Synthesis and Characterization of AgNPs Using Aspergillus terreus
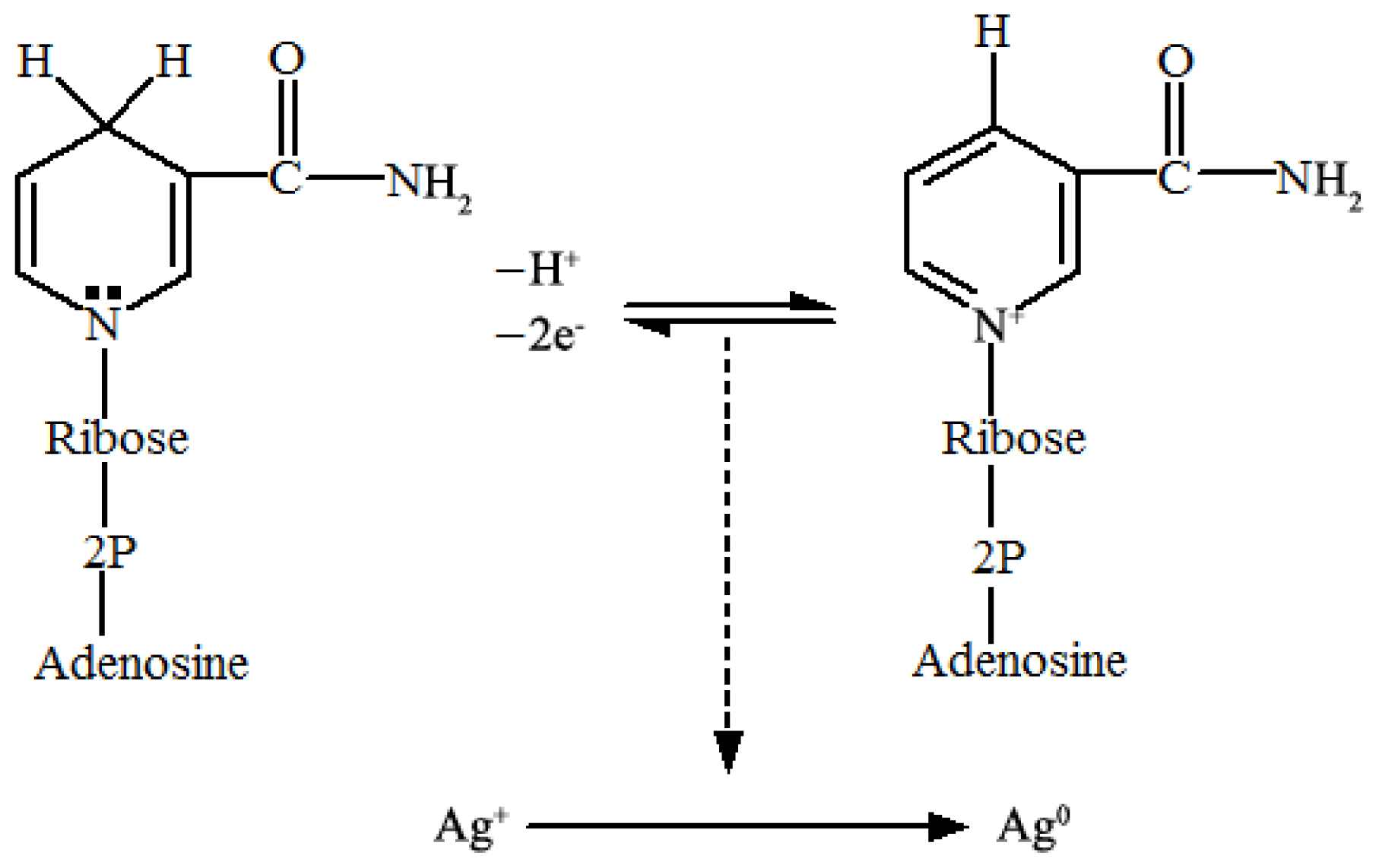
2.2. Key Factors Governing the Biosynthesis of AgNPs
2.3. Antimicrobial Activity Analysis of AgNPs
3. Experimental Section
3.1. Materials
3.2. Biomass Preparation
3.3. Biosynthesis of AgNPs
3.4. Characterization of AgNPs
3.5. Investigation of the Key Factors of Reaction
3.6. The Antimicrobial Activity Analysis of AgNPs
4. Conclusions
Acknowledgments
References
- Sun, Y.G.; Xia, Y.N. Shape-controlled synthesis of gold and silver nanoparticles. Science 2002, 298, 2176–2179. [Google Scholar]
- Gratzel, M. Photoelectrochemical cells. Nature 2001, 414, 338–344. [Google Scholar]
- Shiraishi, Y.; Toshima, N. Colloidal silver catalysts for oxidation of ethylene. J. Mol. Catal. A 1999, 141, 187–192. [Google Scholar]
- White, R.J.; Budarin, V.L.; Moir, J.W.B.; Clark, J.H. A sweet killer: Mesoporous polysaccharide confined silver nanoparticles for antibacterial applications. Int. J. Mol. Sci 2011, 12, 5782–5796. [Google Scholar]
- Groneberg, D.A.; Giersig, M.; Welte, T.; Pison, U. Nanoparticle-based diagnosis and therapy. Curr. Drug Targets 2006, 7, 643–648. [Google Scholar]
- Tao, A.; Kim, F.; Hess, C.; Goldberger, J.; He, R.R.; Sun, Y.G.; Xia, Y.N.; Yang, P.D. Langmuir-blodgett silver nanowire monolayers for molecular sensing using surface-enhanced raman spectroscopy. Nano Lett 2003, 3, 1229–1233. [Google Scholar]
- Waterhouse, G.I.N.; Bowmaker, G.A.; Metson, J.B. Oxygen chemisorption on an electrolytic silver catalyst: A combined TPD and raman spectroscopic study. Appl. Surf. Sci 2003, 214, 36–51. [Google Scholar]
- Elliott, C. The effects of silver dressings on chronic and burns wound healing. Br. J. Nurs 2010, 19, S32–S36. [Google Scholar]
- Darroudi, M.; Ahmad, M.B.; Zak, A.K.; Zamiri, R.; Hakimi, M. Fabrication and characterization of gelatin stabilized silver nanoparticles under UV-light. Int. J. Mol. Sci 2011, 12, 6346–6356. [Google Scholar]
- Kilin, D.S.; Prezhdo, O.V.; Xia, Y.N. Shape-controlled synthesis of silver nanoparticles: Ab initio study of preferential surface coordination with citric acid. Chem. Phys. Lett 2008, 458, 113–116. [Google Scholar]
- Bansal, V.; Ramanathan, R.; Bhargava, S.K. Fungus-mediated biological approaches towards “green” synthesis of oxide nanomaterials. Aust. J. Chem 2011, 64, 279–293. [Google Scholar]
- Shankar, S.S.; Rai, A.; Ankamwar, B.; Singh, A.; Ahmad, A.; Sastry, M. Biological synthesis of triangular gold nanoprisms. Nat. Mater 2004, 3, 482–488. [Google Scholar]
- Mohanpuria, P.; Rana, N.K.; Yadav, S.K. Biosynthesis of nanoparticles: Technological concepts and future applications. J. Nanopart. Res 2008, 10, 507–517. [Google Scholar]
- Klaus, T.; Joerger, R.; Olsson, E.; Granqvist, C.G. Silver-based crystalline nanoparticles, microbially fabricated. Proc. Natl. Acad. Sci. USA 1999, 96, 13611–13614. [Google Scholar]
- Ramanathan, R.; O’Mullane, A.P.; Parikh, R.Y.; Smooker, P.M.; Bhargava, S.K.; Bansal, V. Bacterial kinetics-controlled shape-directed biosynthesis of silver nanoplates using morganella psychrotolerans. Langmuir 2011, 27, 714–719. [Google Scholar]
- Parikh, R.Y.; Ramanathan, R.; Coloe, P.J.; Bhargava, S.K.; Patole, M.S.; Shouche, Y.S.; Bansal, V. Genus-wide physicochemical evidence of extracellular crystalline silver nanoparticles biosynthesis by Morganella spp. PLoS One 2011, 6, e21401. [Google Scholar]
- Narayanan, K.B.; Sakthivel, N. Biological synthesis of metal nanoparticles by microbes. Adv. Colloid Interface Sci 2010, 156, 1–13. [Google Scholar]
- Ahmad, A.; Mukherjee, P.; Senapati, S.; Mandal, D.; Khan, M.I.; Kumar, R.; Sastry, M. Extracellular biosynthesis of silver nanoparticles using the fungus Fusarium oxysporum. Colloid Surface B 2003, 28, 313–318. [Google Scholar]
- Ingle, A.; Gade, A.; Pierrat, S.; Sonnichsen, C.; Rai, M. Mycosynthesis of silver nanoparticles using the fungus Fusarium acuminatum and its activity against some human pathogenic bacteria. Curr. Nanosci 2008, 4, 141–144. [Google Scholar]
- Kathiresan, K.; Manivannan, S.; Nabeel, M.A.; Dhivya, B. Studies on silver nanoparticles synthesized by a marine fungus, Penicillium fellutanum isolated from coastal mangrove sediment. Colloid Surface B 2009, 71, 133–137. [Google Scholar]
- Bharde, A.; Rautaray, D.; Bansal, V.; Ahmad, A.; Sarkar, I.; Yusuf, S.M.; Sanyal, M.; Sastry, M. Extracellular biosynthesis of magnetite using fungi. Small 2006, 2, 135–141. [Google Scholar]
- Wiley, B.J.; Im, S.H.; Li, Z.Y.; McLellan, J.; Siekkinen, A.; Xia, Y. Maneuvering the surface plasmon resonance of silver nanostructures through shape-controlled synthesis. J. Phys. Chem. B 2006, 110, 15666–15675. [Google Scholar]
- Verma, V.C.; Kharwar, R.N.; Gange, A.C. Biosynthesis of antimicrobial silver nanoparticles by the endophytic fungus Aspergillus clavatus. Nanomedicine (Lond) 2010, 5, 33–40. [Google Scholar]
- Huang, J.L.; Li, Q.B.; Sun, D.H.; Lu, Y.H.; Su, Y.B.; Yang, X.; Wang, H.X.; Wang, Y.P.; Shao, W.Y.; He, N.; et al. Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Bansal, V.; Li, V.; O’Mullane, A.P.; Bhargava, S.K. Shape dependent electrocatalytic behaviour of silver nanoparticles. CrystEngComm 2010, 12, 4280–4286. [Google Scholar]
- Dudev, T.; Lim, C. Factors controlling the mechanism of NAD+ non-redox reactions. J. Am. Chem. Soc 2010, 132, 16533–16543. [Google Scholar]
- Lin, H. Nicotinamide adenine dinucleotide: Beyond a redox coenzyme. Org. Biomol. Chem 2007, 5, 2541–2554. [Google Scholar]
- Ahmad, A.; Mukherjee, P.; Mandal, D.; Senapati, S.; Khan, M.I.; Kumar, R.; Sastry, M. Enzyme mediated extracellular synthesis of CdS nanoparticles by the fungus, Fusarium oxysporum. J. Am. Chem. Soc 2002, 124, 12108–12109. [Google Scholar]
- Govender, Y.; Riddin, T.L.; Gericke, M.; Whiteley, C.G. On the enzymatic formation of platinum nanoparticles. J. Nanopart. Res 2010, 12, 261–271. [Google Scholar]
- Khan, S.S.; Mukherjee, A.; Chandrasekaran, N. Studies on interaction of colloidal silver nanoparticles (SNPs) with five different bacterial species. Colloid Surface B 2011, 87, 129–138. [Google Scholar]
- Espinel-Ingroff, A.; Warnock, D.W.; Vazquez, J.A.; Arthington-Skaggs, B.A. In vitro antifungal susceptibility methods and clinical implications of antifungal resistance. Med. Mycol 2000, 38, 293–304. [Google Scholar]
- Wang, L.; Yokoyama, K.; Miyaji, M.; Nishimura, K. The identification and phylogenetic relationship of pathogenic species of Aspergillus based on the mitochondrial cytochrome b gene. Med. Mycol 1998, 36, 153–164. [Google Scholar]
- Borchert, H.; Shevehenko, E.V.; Robert, A.; Mekis, I.; Kornowski, A.; Grubel, G.; Weller, H. Determination of nanocrystal sizes: A comparison of TEM, SAXS, and XRD studies of highly monodisperse CoPt3 particles. Langmuir 2005, 21, 1931–1936. [Google Scholar]
- Wang, Y.; Lu, Z.; Wu, H.; Lv, F. Study on the antibiotic activity of microcapsule curcumin against foodborne pathogens. Int. J. Food. Microbiol 2009, 136, 71–74. [Google Scholar]




| Tested Pathogenic Organisms | Mean Size of Inhibition Zone (mm)
| |
|---|---|---|
| Control | Test | |
| Candida albicans (ATCC 90028) | 9 | 16 ± 1 |
| Candida krusei (ATCC 6258) | 10 | 14 ± 2 |
| Candida parapsilosis (ATCC 22019) | 9 | 13 ± 1 |
| Candida tropicalis (JLCC 30394) | 10 | 14 ± 1 |
| Aspergillus flavus (IFM 55648) | 9 | 13 ± 2 |
| Aspergillus fumigates (IFM 40808) | 9 | 14 ± 2 |
| Staphylococcus aureus (ATCC 25923) | 9 | 16 ± 1 |
| Pseudomonas aeruginosa (ATCC 27853) | 9 | 12 ± 1 |
| Escherichia coli (ATCC 25922) | 10 | 13 ± 1 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, G.; He, D.; Qian, Y.; Guan, B.; Gao, S.; Cui, Y.; Yokoyama, K.; Wang, L. Fungus-Mediated Green Synthesis of Silver Nanoparticles Using Aspergillus terreus. Int. J. Mol. Sci. 2012, 13, 466-476. https://doi.org/10.3390/ijms13010466
Li G, He D, Qian Y, Guan B, Gao S, Cui Y, Yokoyama K, Wang L. Fungus-Mediated Green Synthesis of Silver Nanoparticles Using Aspergillus terreus. International Journal of Molecular Sciences. 2012; 13(1):466-476. https://doi.org/10.3390/ijms13010466
Chicago/Turabian StyleLi, Guangquan, Dan He, Yongqing Qian, Buyuan Guan, Song Gao, Yan Cui, Koji Yokoyama, and Li Wang. 2012. "Fungus-Mediated Green Synthesis of Silver Nanoparticles Using Aspergillus terreus" International Journal of Molecular Sciences 13, no. 1: 466-476. https://doi.org/10.3390/ijms13010466




