Oral Drug Delivery Systems Comprising Altered Geometric Configurations for Controlled Drug Delivery
Abstract
:1. Introduction
2. Multilayered Tablets for Controlled Drug Delivery
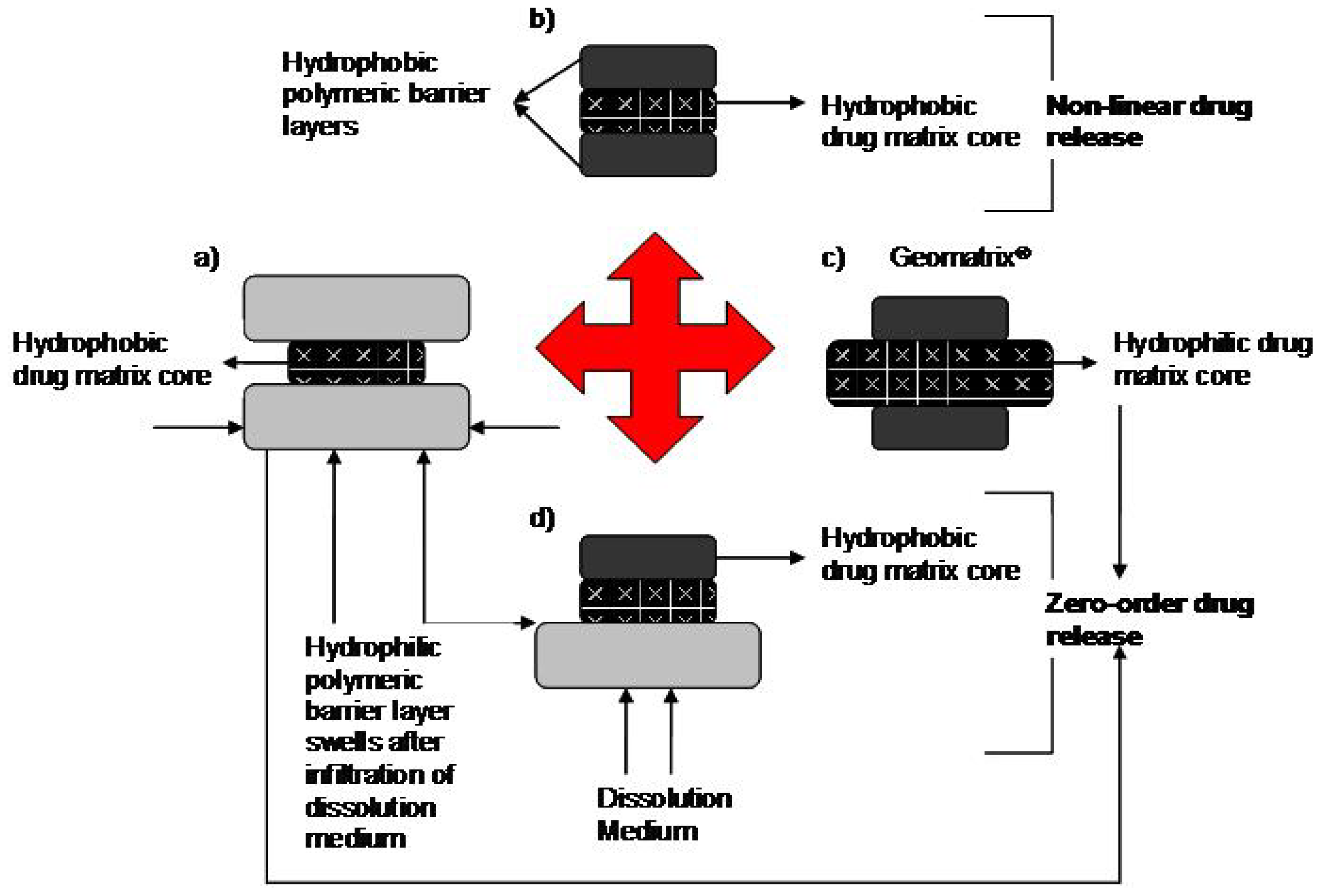
2.1. Geomatrix® Multilayer Tablet Technology
2.2. Sodas® Multilayer Tablet Technology
2.3. Factors Affecting the Rate of Drug Release from Multilayered Tablets
2.3.1. Polymers Employed in Multilayered Tablets
2.3.2. Structure of the Device
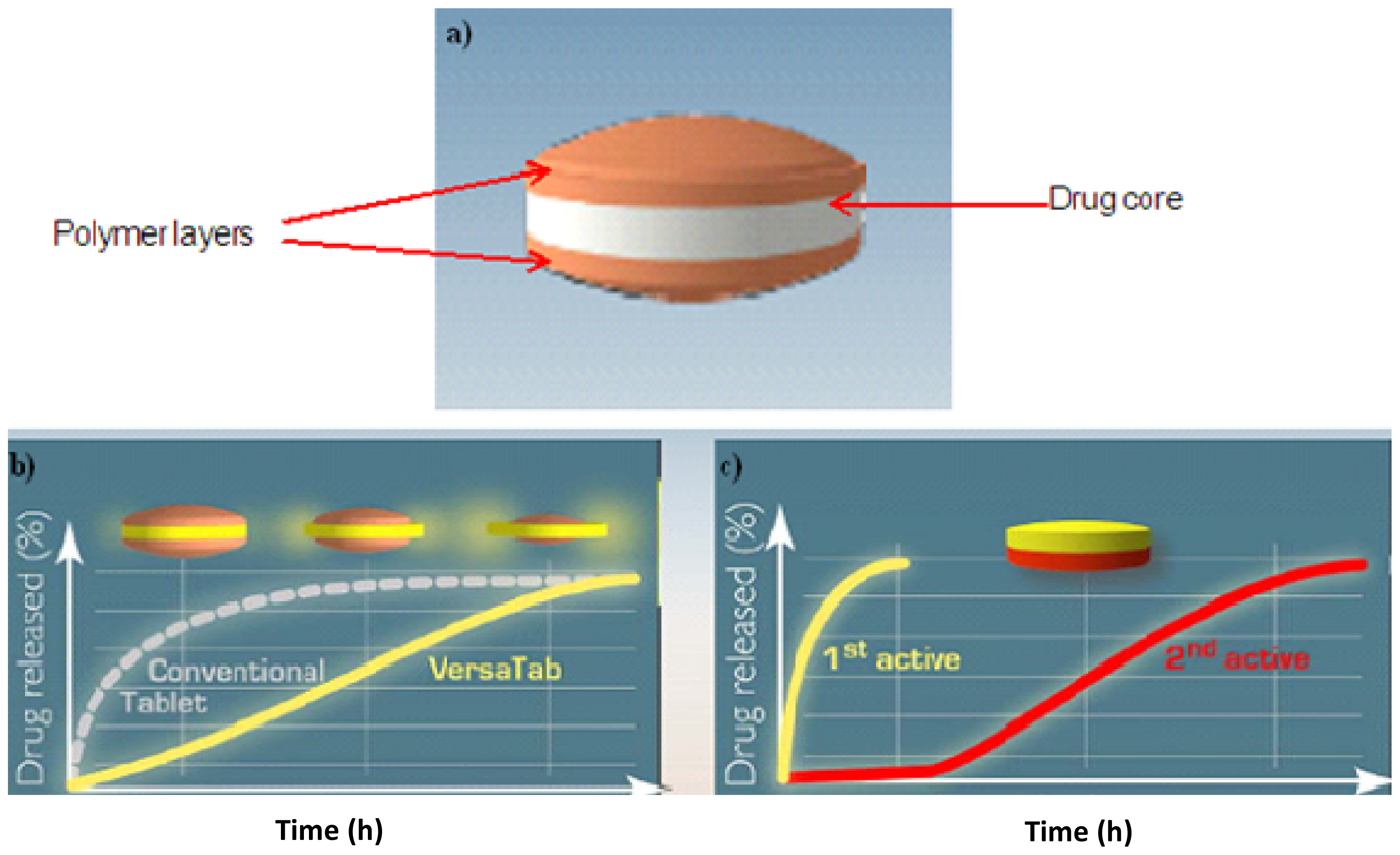
2.4. Bilayered Tablets
2.5. Triple-Layered Tablets
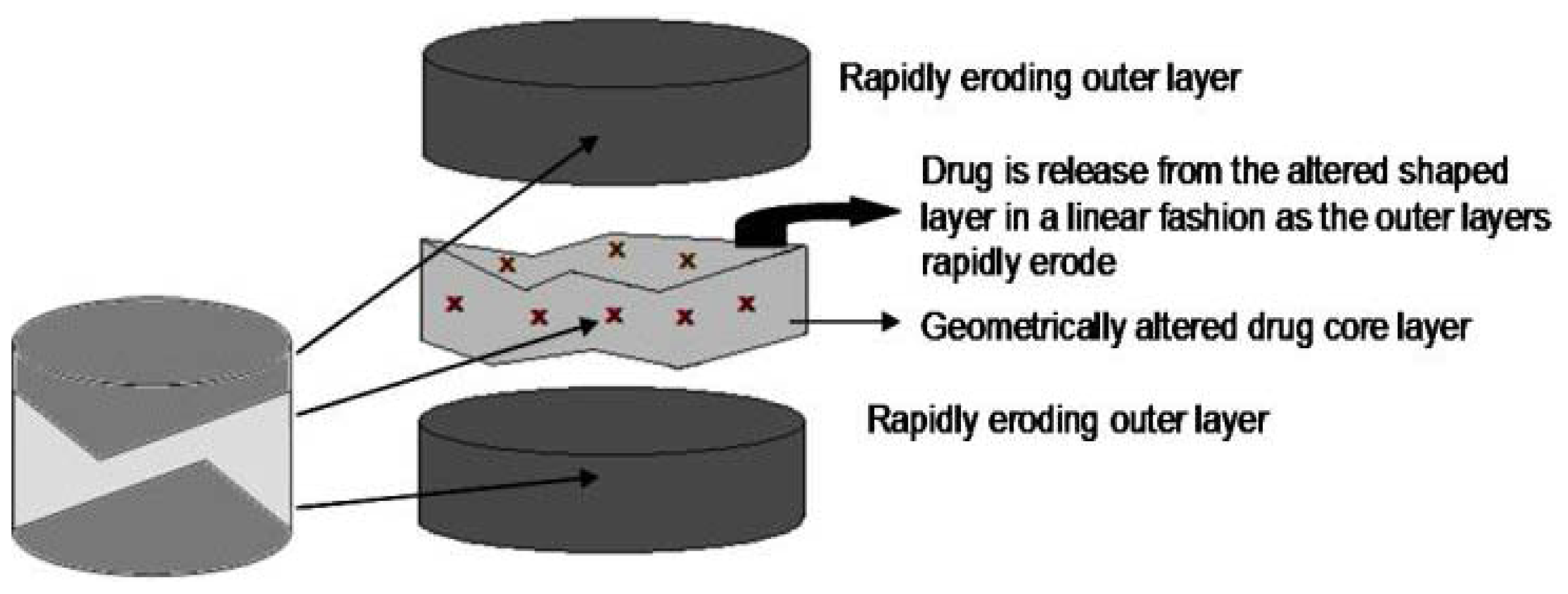
2.5.1. GeolockTM Technology
2.5.2. Various Drug Release Profiles Achievable by Triple-Layered Tablets
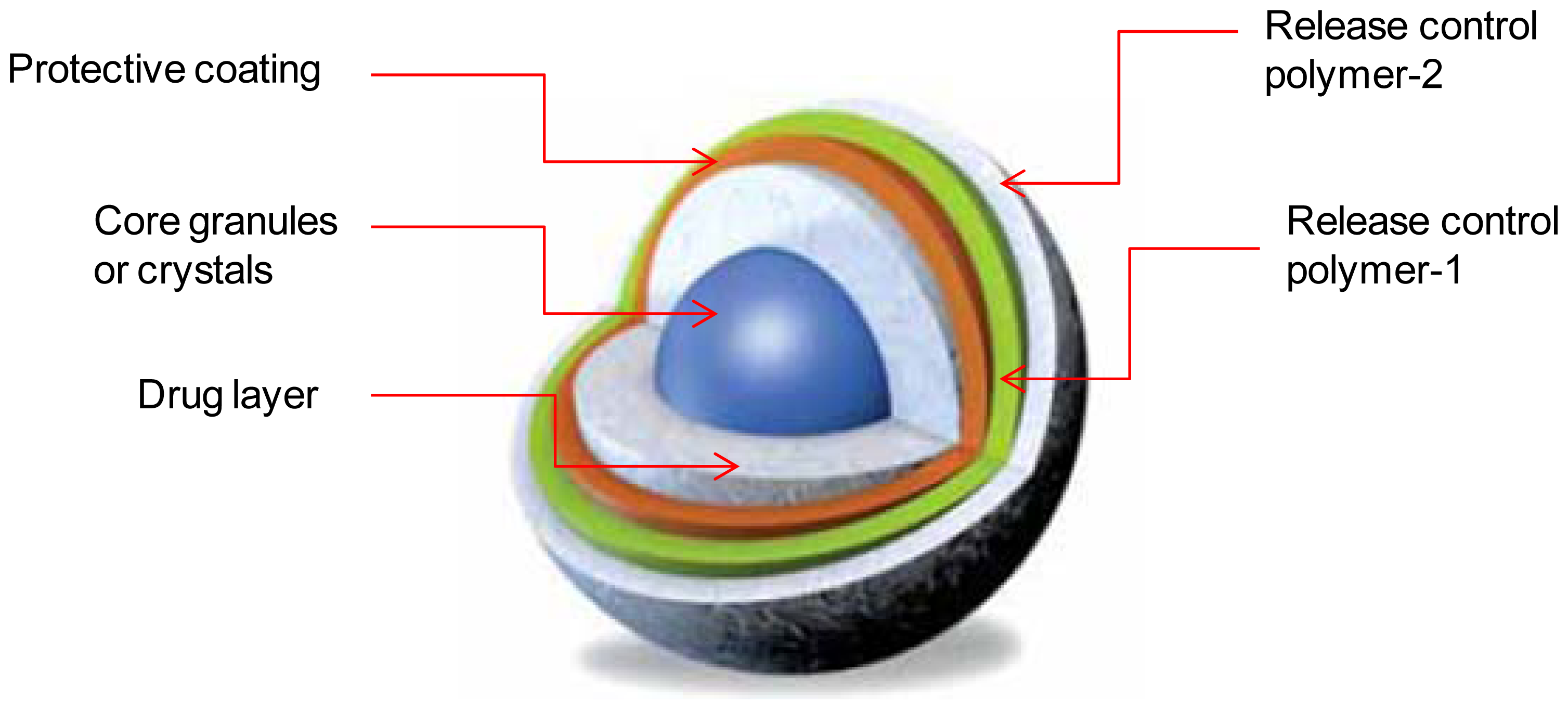
3. Multilayered Osmotic Devices
4. Multilayered Floatable Drug Delivery Systems
5. Core-in-Cup Devices
6. Procise® Technology
7. Donut-Shaped Devices for Controlled Drug Delivery
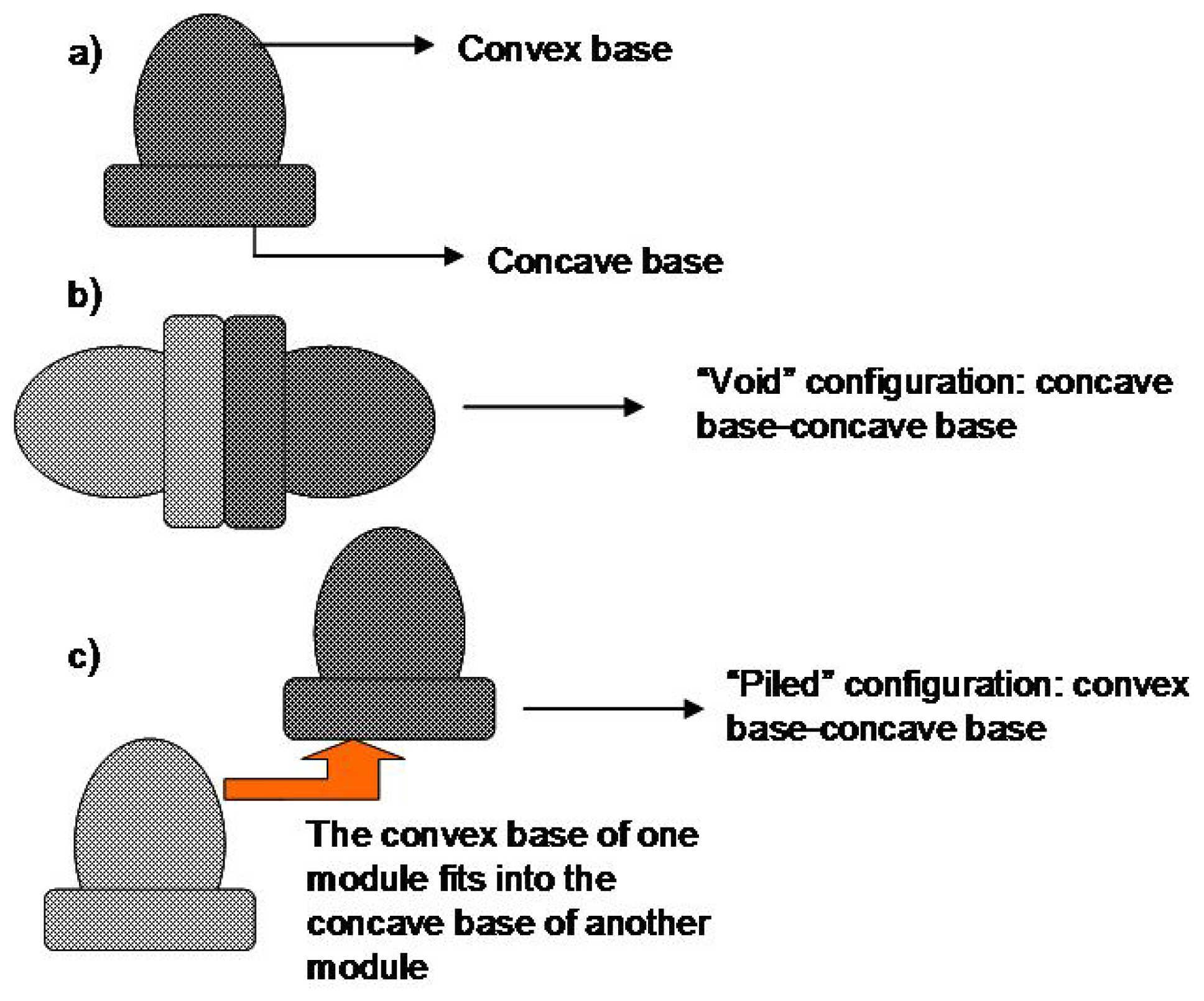
8. Dome Matrix® and “Release Modules Assemblage” Technology
9. Conclusions
References
- Deshpande, A.A.; Rhodes, C.T.; Shah, N.S.; Malick, A.W. Controlled-release drug delivery systems for prolonged gastric residence: An overview. Drug Dev. Ind. Pharm 1996, 22, 531–539. [Google Scholar]
- Green, P.G. Iontophoretic delivery of peptide drugs. J. Control. Release 1996, 41, 33–48. [Google Scholar]
- Rubinstein, A.; Friedman, M.; Baluom, M.; Tirosh, B. Controlled Release Oral Drug Delivery System. U.S. Patent 7,189,414, 13 March 2007. [Google Scholar]
- Abdul, S.; Poddar, S.S. A flexible technology for modified release of drugs: Multi layered tablets. J. Control. Release 2004, 97, 393–405. [Google Scholar]
- Yu, D.; Branford-White, C.; Ma, Z.; Zhu, L.M.; Li, X.Y.; Yang, X.L. Novel drug delivery devices for providing linear release profiles fabricated by 3DP. Int. J. Pharm 2009, 370, 160–166. [Google Scholar]
- Varum, F.J.O.; Merchant, H.A.; Basit, A.W. Oral modified-release formulations in motion: The relationship between gastrointestinal transit and drug absorption. Int. J. Pharm 2010, 395, 1–2. [Google Scholar]
- Chien, T.W. Fundamentals of Controlled-Release and Drug Administration. In Novel Drug Delivery System; Swarbrick, J., Ed.; Marcel Dekker Inc: New York, NY, USA, 1982; pp. 465–574. [Google Scholar]
- Wilding, I.R.; Coupe, A.J.; Davis, S.S. The role of gamma scintigraphy in oral drug delivery. Adv. Drug Deliv. Rev 1991, 7, 87–117. [Google Scholar]
- Fassihi, R.; Yang, L. Controlled Release Drug Delivery System. U.S. Patent 5,783,212, 21 July 1998. [Google Scholar]
- Singh, B.N.; Kim, K.H. Floating drug delivery systems: An approach to oral controlled drug delivery via gastric retention. J. Control. Release 2000, 63, 235–259. [Google Scholar]
- Lingam, M.; Ashok, T.; Venkateswarlu, V.; Rao, Y.M. Design and evaluation of a novel matrix type multiple units as biphasic gastroretentive drug delivery systems. AAPS PharmSci 2006, 9, 1253–1261. [Google Scholar]
- Shah, A.C. Design of Oral Sustained and Release Drug Delivery Systems: In Vitro/in Vivo Considerations. In Oral Sustained Release Formulation Design and Evaluation; Yacobi, A., Halperin-Walega, E., Eds.; Pergamon: New York, NY, USA, 1988; pp. 35–56. [Google Scholar]
- Hardy, I.J.; Windberg-Baarup, A.; Neri, C.; Byway, P.V.; Booth, S.W.; Fitzpatrick, S. Modulation of drug release kinetics from hydroxypropyl methyl cellulose matrix tablets using polyvinyl pyrrolidone. Int. J. Pharm 2007, 337, 246–253. [Google Scholar]
- Landgraf, W.; Li, N.H.; Benson, J.R. New polymer enables near zero order release of drugs. Drug Deliv. Technol 2005, 5, 48–55. [Google Scholar]
- Shahiwala, A.; Misra, A. Pulmonary absorption of liposomal levonorgestrel. AAPS PharmSci 2004, 5. [Google Scholar] [CrossRef]
- Reed, R.C.; Dutta, S.; Liu, W. Once-daily dosing is appropriate for extended-release divalproex over a wide dose range, but not for enteric-coated, delayed-release divalproex: Evidence via computer simulations and implications for epilepsy therapy. Epilepsy Res 2009, 87, 260–267. [Google Scholar]
- Kim, C.J. Coated Tablet With Long Term Parabolic and Zero-Order Release Kinetics. U.S. Patent 6,110,500, 29 August 2000. [Google Scholar]
- Sershen, S.; West, J. Implantable, polymeric systems for modulated drug delivery. Adv. Drug Deliv. Rev 2002, 54, 1225–1235. [Google Scholar]
- Vandamme, T.F.; Ellis, K.J. Issues and challenges in developing ruminal drug delivery systems. Adv. Drug Deliv. Rev 2004, 56, 1415–1436. [Google Scholar]
- Ayres, J.W. Coated, Platform-Generating Tablet. U.S. Patent 6,733,784, 11 May 2004. [Google Scholar]
- Shivaraj, A.; Selvam, R.P.; Mani, T.T.; Sivakumar, T. Design and evaluation of transdermal drug delivery of ketotifen fumarate. Int. J. Pharm. Biomed. Res 2010, 1, 42–47. [Google Scholar]
- Siegel, S.; Winey, K. Long-Term Delivery Formulations and Methods of Use Thereof. U.S. Pat. Appl. 20080305140, 12 November 2008. [Google Scholar]
- Cheng, P.; Chen, M.; Udipi, K. Dry Diazeniumdiolation Methods for Producing Nitric Oxide Releasing Medical Devices. U.S. Pat. Appl. 20100159119, 24 June 2010. [Google Scholar]
- Varelas, C.G.; Dixon, D.G.; Steiner, C.A. Zero-order release from biphasic polymer hydrogels. J. Control. Release 1995, 34, 185–192. [Google Scholar]
- Qiu, Y.; Chidambaram, N.; Flood, K. Design and evaluation of layered diffusional matrices for zero-order sustained-release. J. Control. Release 1998, 51, 123–130. [Google Scholar]
- Pillay, V.; Fassihi, R. Electrolyte-induced compositional heterogeneity: A novel approach for rate-controlled oral drug delivery. J. Pharm. Sci 1999, 88, 1140–1148. [Google Scholar]
- Yang, L.; Fassihi, R. Zero-order release kinetics from a self-correcting floatable asymmetric configuration drug delivery system. J. Pharm. Sci 2000, 85, 170–173. [Google Scholar]
- Gallardo, A.; Rodríguez, G.; Aguilar, M.R.; Fernández, M.; San Román, J. A kinetic model to explain the zero-order release of drugs from ionic polymeric drug conjugates: Application to AMPS-Triflusal-Derived polymeric drugs. Macromolecules 2003, 36, 8876–8880. [Google Scholar]
- Andjelić, S.; Yuan, J.; Jamiolkowski, D.D.; Diluccio, R.; Bezwada, R.; Zhang, H.; Mijovic, J. Hydrophilic absorbable copolyester exhibiting zero-order drug release. Pharm. Res 2006, 23, 821–834. [Google Scholar]
- Pryce Lewis, W.E.; Rowe, C.W.; Cima, M.J.; Materna, P.A. System for Manufacturing Controlled Release Dosage Forms, Such as Zero-Order Release Profile Dosage Form Manufactured by Three-Dimensional Printing. U.S. Patent 7,820,201, 26 October 2010. [Google Scholar]
- Wang, S.X.; Bao, A.; Phillips, W.T.; Goins, B.; Herrera, S.J.; Santoyo, C.; Miller, F.R.; Otto, R.A. Intraoperative therapy with liposomal drug delivery: Retention and distribution in human head and neck squamous cell carcinoma xenograft model. Int. J. Pharm 2009, 373, 156–164. [Google Scholar]
- Brooke, D.; Walshkuhn, R.J. Zero-order drug delivery system: Theory and preliminary testing. J. Pharm. Sci 1977, 66, 159–162. [Google Scholar]
- Ford, J.L.; Rubenstein, M.H.; McCaul, F.; Hogan, J.E.; Edgar, P.J. Importance of drug type, tablet shape and added diluents on drug release kinetics from hydroxypropylmethylcellulose matrix tablets. Int. J. Pharm 1987, 40, 233–234. [Google Scholar]
- Sangalli, M.E.; Conte, U.; Gazzaniga, A.; La Manna, A. Washington, DC, USA, 25–30 July 1993; Academic Press Inc: New York NY, USA, 1980; 20, pp. 316–317.
- Siepmann, J.; Streubel, A.; Peppas, N.A. Understanding and predicting drug delivery from hydrophilic matrix tablets using the “Sequential Layer”. Model. Pharm. Res 2002, 19, 306–314. [Google Scholar]
- Chopra, S.K. Drug Delivery and Systems Based on Geometric Configuration. In Modified-Release Drug Delivery Technology; Rathbone, M.J., Hadgraft, J., Roberts, M.S., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 35–48. [Google Scholar] [CrossRef]
- Cobby, J.; Mayersohn, M.; Walker, G.C. Influence of shape factors on kinetics of drug release from matrix tablets I: Theoretical. J. Pharm. Sci 1974, 63, 725–732. [Google Scholar]
- Sakamoto, J.H.; van de Ven, A.L.; Godin, B.; Blanco, E.; Serda, R.E.; Grattoni, A.; Ziemys, A.; Bouamrani, A.; Hu, T.; Ranganathan, S.I.; et al. Enabling individualized therapy through nanotechnology. Pharmacol. Res 2010, 62, 57–89. [Google Scholar]
- Cao, X.; Lai, S.; Lee, L.J. Design of a self-regulated drug delivery device. Biomed. Microdevices 2001, 3, 109–118. [Google Scholar]
- Efentakis, M.; Politis, S. Comparative evaluation of various structures in polymer controlled drug delivery systems and the effect of their morphology and characteristics on drug release. Eur. Polym. J 2006, 42, 1183–1195. [Google Scholar]
- Martin del Valle, E.M.; Galan, M.A.; Carbonell, R.G. Drug delivery technologies: The way forward in the new decade. Ind. Eng. Chem. Res 2009, 48, 2475–2486. [Google Scholar]
- Sundy, E.; Danckwerts, M.P. A novel compression-coated doughnut-shaped tablet design for zero-order sustained release. Eur. J. Pharm. Sci 2004, 22, 477–485. [Google Scholar]
- Dash, S.; Murthy, P.N.; Nath, L.; Chowdhury, P. Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol. Pharm 2010, 67, 217–223. [Google Scholar]
- Conte, U.; Maggi, L.; Colombo, P.; Manna, L. Multilayered hydrophilic matrices as constant release devices. J. Control. Release 1993, 26, 39–47. [Google Scholar]
- Kim, C.J. Release kinetics of coated, donut-shaped tablets for water soluble drugs. Eur. J. Pharm. Sci 1999, 12, 237–242. [Google Scholar]
- Survase, S.; Kumar, N. Pulsatile drug delivery: Current scenario. Curr. Res. Info. Pharm. Sci 2007, 8, 27–33. [Google Scholar]
- Losi, E.; Bettini, R.; Santi, P.; Sonvico, F.; Colombo, G.; Lofthus, K.; Colombo, P.; Peppas, N.A. Assemblage of novel release modules for the development of adaptable drug delivery systems. J. Control. Release 2006, 111, 212–218. [Google Scholar]
- Gohel, M.C.; Nagori, S.A. A novel colonic drug delivery system of ibuprofen. Asian J. Pharm 2009, 3, 233–239. [Google Scholar]
- Colombo, P.; La Manna, A.; Conte, U. System for the Controlled-Rate Release of Active Substances. U.S. Patent 4,839,177, 13 June 1989. [Google Scholar]
- Song, F.; Zhang, L.; Yang, C.; Yan, L. Genipin-crosslinked casein hydrogels for controlled drug delivery. Int. J. Pharm 2009, 373, 41–47. [Google Scholar]
- Herrlich, S.; Ziolek, S.; Hoefemann, H.; Zengerle, R.; Haeberle, S. Adjustable Diffusion Barrier for Controlled Drug Release in Spastic and Pain Therapy. Proceedings of the 4th European Conference of the International Federation of Medical and Biological Engineering, Antwerp, Belgium, 23–27 November 2008; Springer: New York, NY, USA, 2009; 22, pp. 2368–2371. [Google Scholar]
- Mueller, K.F.; Heiber, S.J. Crosslinked, Porous Polymers for Controlled Drug Delivery. U.S. Patent 4,548,990, 22 October 1985. [Google Scholar]
- Crotts, G.; Park, T.G. Preparation of porous and nonporous biodegradable polymeric hollow microspheres. J. Control. Release 1995, 35, 91–105. [Google Scholar]
- Learoyd, T.P.; Burrows, J.L.; French, E.; Seville, P.C. Sustained delivery by leucine-modified chitosan spray-dried respirable powders. Int. J. Pharm 2009, 372, 97–104. [Google Scholar]
- Naveen, R. Biodegradable polymers in controlled drug delivery. Pharmainfo.net. 2009. Available online: http://www.pharmainfo.net/raghanaveen/biodegradable-polymers-controlled-drug-delivery (accessed on 3 May 2010).
- Heller, J.; Pangburn, S.H.; Penhale, D.W.H. Use of Bioerodible Polymers in Self-Regulated Drug Delivery Systems. In Controlled-Release Technology, Pharmaceutical Applications; Lee, P.I., Good, W.R., Eds.; ACS Symposium Series: Washington, DC, USA, 1987; pp. 172–187. [Google Scholar]
- Harland, R.S.; Gazzaniga, A.; Sangalli, M.E.; Colombo, P.; Peppas, N.A. Drug/polymer matrix swelling and dissolution. Pharm. Res 1998, 5, 488–494. [Google Scholar]
- Lee, P.I. Kinetics of drug release from the hydrogel matrices. J. Control. Release 1985, 2, 277–288. [Google Scholar]
- Nelson, K.G.; Smith, S.J.; Bennet, R.M. Constant-Release Diffusion Systems: Rate Control by Means of Geometric Configuration. In Controlled-Release Technology, Pharmaceutical Applications; Lee, P.I., Good, W.R., Eds.; ACS Symposium Series: Washington, DC, USA, 1987; pp. 324–340. [Google Scholar]
- Peppas, N.A.; Sahlin, J.J. A simple equation for the description of solute release: III. Coupling of diffusion and relaxation. Int. J. Pharm 1989, 57, 162–172. [Google Scholar]
- Narasimhan, B.; Langer, R. Zero-order release of micro and macromolecules from polymeric devices: The role of the burst effect. J. Control. Release 1997, 47, 13–20. [Google Scholar]
- Conte, U.; Maggi, L. A flexible technology for the linear, pulsatile and delayed release of drugs allowing for easy accommodation of difficult in vitro targets. J. Control. Release 2000, 64, 263–268. [Google Scholar]
- Zerbe, H.G.; Szabo, P. Oral Dosage Formulation. U.S. Pat. Appl. 20060127478, 15 June 2006. [Google Scholar]
- Namdeo, B. Barrier layers in multilayered tablets. Express Pharma. 2008. Available online: www.expresspharmaonline.com/20080731/research03.shtml accessed on 24 October 2010.
- Chidambaram, N.; Porter, W.; Flood, K.; Qui, Y. Formulation and characterisation of new layered diffusional matrices for zero-order sustained release. J. Control. Release 1998, 52, 149–158. [Google Scholar]
- Kim, C.J. Controlled release from triple layer, donut-shaped tablets with enteric polymers. AAPS PharmSci 2005, 6, 429–436. [Google Scholar]
- Shionogi Pharma Inc. Once a day Sular® (Nisoldipine) with Geomatrix® delivery system for the treatment of hypertension. 2008. Available online: http://www.sular.com/html/geomatrix.html (accessed on 24 August 2010).
- Patel, V.M.; Prajapati, B.G.; Patel, M.M. Formulation, evaluation, and comparison of bilayered and multilayered mucoadhesive buccal devices of propranolol hydrochloride. AAPS PharmSci 2007, 8, E147–E154. [Google Scholar]
- Streubel, A.; Siepmann, J.; Peppas, N.A.; Bodmeier, R. Bimodal drug release achieved with multi-layer matrix tablets: Transport mechanisms and device design. J. Control. Release 2000, 69, 455–468. [Google Scholar]
- Efentakis, M.; Peponaki, C. Formulation study and evaluation of matrix and three-layer tablet sustained drug delivery systems based on carbopols with isosorbide mononitrate. AAPS PharmSci 2008, 9, 917–923. [Google Scholar]
- Elan drug technologies. Spheroidal drug absorption system (SODAS®). 2010. Available online: http://www.elandrugtechnologies.com/oral_controlled_release/sodas (accessed on 26 August 2010).
- Ye, I.M.; Karasulu, H.; Ertan, G. Different geometric shaped hydrogel theophylline tablets: Statistical approach for estimating drug release. Farmaco 2003, 57, 939–945. [Google Scholar]
- Hongtao, L.; Xiaochen, G.U. Correlation between drug dissolution and polymer hydration: A study using texture analysis. Int. J. Pharm 2007, 342, 18–25. [Google Scholar]
- Conti, S.; Maggi, L.; Segale, L.; Ochoa Machiste, E.; Conte, U.; Grenier, P.; Vergnault, G. Matrices containing NaCMC and HPMC: 2. Swelling and release mechanism study. Int. J. Pharm 2007, 333, 143–151. [Google Scholar]
- Li, H.; Hardy, R.J.; Gu, X. Effect of drug solubility on polymer hydration and drug dissolution from polyethylene oxide (PEO) matrix tablets. AAPS PharmSci 2008, 9, 437–443. [Google Scholar]
- Barakat, N.S.; Elbagory, I.M.; Almurshedi, A.S. Controlled-release carbamazepine matrix granules and tablets comprising lipophilic and hydrophilic components. Drug Deliv 2009, 16, 57–65. [Google Scholar]
- Yang, L.; Johnson, B.; Fassihi, R. Determination of continuous changes in the gel layer thickness of poly(ethylene oxide) and HPMC tablets undergoing hydration: A textural analysis study. Pharm. Res 1998, 15, 1902–1906. [Google Scholar]
- Zerbe, H.G.; Krumme, M. Design Characteristics and Release Properties of a Novel Erosion-Controlled Oral Delivery System. In Modified-Release Drug Delivery Technology; Rathbone, M.J., Hadgraft, J., Roberts, M.S., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 59–76. [Google Scholar] [CrossRef]
- Porter, S.C. Novel drug delivery: Review of current trends with oral solid dosage forms. Am. Pharm. Rev 2009, 85, 28–35. [Google Scholar]
- Doshi, M.M.; Joshi, M.D.; Mehta, B.P. Pharmaceutical Composition for Controlled Drug Delivery System. U.S. Patent 7,157,100, 2 January 2007. [Google Scholar]
- Patra, C.N.; Kumar, A.B.; Pandit, H.K.; Singh, S.P.; Devi, M.V. Design and evaluation of sustained release bilayer tablets of propranolol hydrochloride. Acta Pharm 2007, 57, 479–489. [Google Scholar]
- Nirmal, J.; Saisivam, S.; Peddanna, C.; Muralidharan, S.; Godwinkumar, S.; Nagarajan, M. Bilayer tablets of Atorvastatin calcium and Nicotinic acid: Formulation and evaluation. Chem. Pharm. Bull 2008, 56, 1455–1458. [Google Scholar]
- Kim, S.W.; Jun, S.S.; Jo, Y.G.; Koo, J.S.; Jun, Y.S. Combination Formulation with Controlled Release Comprising Metformin and Glimepiride. WIPO Pat. Appl. WO/2008/050987, 2 May 2008. [Google Scholar]
- IntelGelx Corp. Innovative Drug Delivery Solutions. 2009. Available online: http://www.intelgenx.com/_assets/pdf/Rodman-Renshaw-Sept-2009.pdf (accessed on 26 August 2010).
- SkyePharma. A chronotherapy focused-real time oral drug delivery. 2010. Available online: http://www.skyepharma.com/ (accessed on 26 August 2010).
- Maggi, L.; Shepard, T.; Rochdi, M.; Grenier, P.; Halbeisen, S.; Zimmer, R.; Conte, U. A Simulation Approach for Efficient Development of a Naproxen Geomatrix Quick/Slow Formulation. Proceedings of the 24th International Symposium on Controlled Release of Bioactive Materials; The controlled release society: Stockholm, Sweden, 1997; pp. 327–328. [Google Scholar]
- Desai, S. Multilayer Controlled Release Tablets Containing both Naproxen and Naproxen Sodium Salt. Eur. Patent EP 0656776, 20 March 1996. [Google Scholar]
- Iyer, K.; Jha, R.J.; Saoji, D.G. Cardiovascular Therapeutic Combinations. WIPO Pat. Appl. WO/2006/085128, 17 August 2006. [Google Scholar]
- Lopes, C.M.; Lobo, J.M.S.; Pinto, J.F.; Costa, P. Compressed mini-tablet as a biphasic delivery system. Int. J. Pharm 2006, 303, 93–100. [Google Scholar]
- Fukui, J.; Uemura, K.; Kobayashi, M. Studies on applicability of press-coated tablets using hydroxypropylcellulose (HPC) in the outer shell for timed-release preparations. J. Control. Release 2000, 68, 215–223. [Google Scholar]
- Siahi, M.R.; Barzegar-Jalali, M.; Monajjemzadeh, F.; Ghaffar, I.F.; Azarmi, S. Design and evaluation of 1- and 3-layer matrices of verapamil hydrochloride for sustaining its release. AAPS PharmSci 2005, 6, 626–632. [Google Scholar]
- Krishnaiah, Y.S.R.; Karthikeyan, R.S.; Gouri Sankar, V.; Satyanarayana, V. Three-layer guar gum matrix tablet formulations for oral controlled delivery of highly soluble trimetazidine dihydrochloride. J. Control Release 2002, 81, 45–56. [Google Scholar]
- Longxiao, L.; Binjie, C. Preparation of monolithic osmotic pump system by coating the indented core tablet. Eur. J. Pharm. Biopharm 2006, 64, 180–184. [Google Scholar]
- Longxiao, L.; Xiangning, X. Preparation of bilayer-core osmotic pump tablet by coating the indented core tablet. Int. J. Pharm 2008, 352, 225–239. [Google Scholar]
- Faour, J.; Mayorga, J. Multi-Layered Osmotic Device. U.S. Reissued Patent RE39069, 18 April 2006. [Google Scholar]
- Flanner, H.H.; Mcknight, L.C.; Burnside, B.A. System for Osmotic Delivery of Pharmaceutically Active Agents. U.S. Patent 6,838,093, 4 January 2005. [Google Scholar]
- Liu, Q.; Fassihi, R. Zero-order delivery of a highly soluble, low dose drug alfuzosin hydrochloride via gastro-retentive system. Int. J. Pharm 2008, 348, 27–34. [Google Scholar]
- Yang, L.; Eshragh, J.; Fassihi, R. A new intragastric delivery system for the treatment of Helicobacter pylori associated gastric ulcer: In vitro evaluation. J. Control. Release 1999, 57, 215–222. [Google Scholar]
- Danckwerts, M.P. Development of a zero-order release oral compressed tablet with potential for commercial tabletting production. Int. J. Pharm 1994, 112, 37–45. [Google Scholar]
- Guimarães, G.G.; Katsuki, G.I.; Zanardo, N.D.; Ribeiro, D.A.; Cavalcanti, O.A. Evaluation of pectin-HPMC as compression coating. I–A study of the swelling properties of coated tablet. Rev. Bras. Cienc. Farm 2008, 44, 133–141. [Google Scholar]
- Cleave, J.P. Some geometrical considerations concerning the design of tablets. J. Pharm. Pharmacol 1965, 17, 698–702. [Google Scholar]
- Rhine, W.D.; Hsieh, D.S.T.; Langer, R. Polymers for sustained macromolecule release: Procedures to fabricate reproducible delivery systems and control release kinetics. J. Pharm. Sci 1980, 69, 265–270. [Google Scholar]
- Hsieh, D.S.T.; Rhine, W.D.; Langer, R. Zero-order controlled release polymer matrices for micro and macromolecules. J. Pharm. Sci 1983, 72, 17–22. [Google Scholar]
- Kim, C.J. Compressed donut-shaped tablets with zero-order release kinetics. Pharm. Res 1995, 12, 1045–1048. [Google Scholar]
- Cheng, K.; Zhu, J.; Song, X.; Sun, L.; Junhong, Z. Studies of hydroxypropylmethylcellulose donut-shaped tablets. Drug Dev. Ind. Pharm 1999, 25, 1067–1071. [Google Scholar]


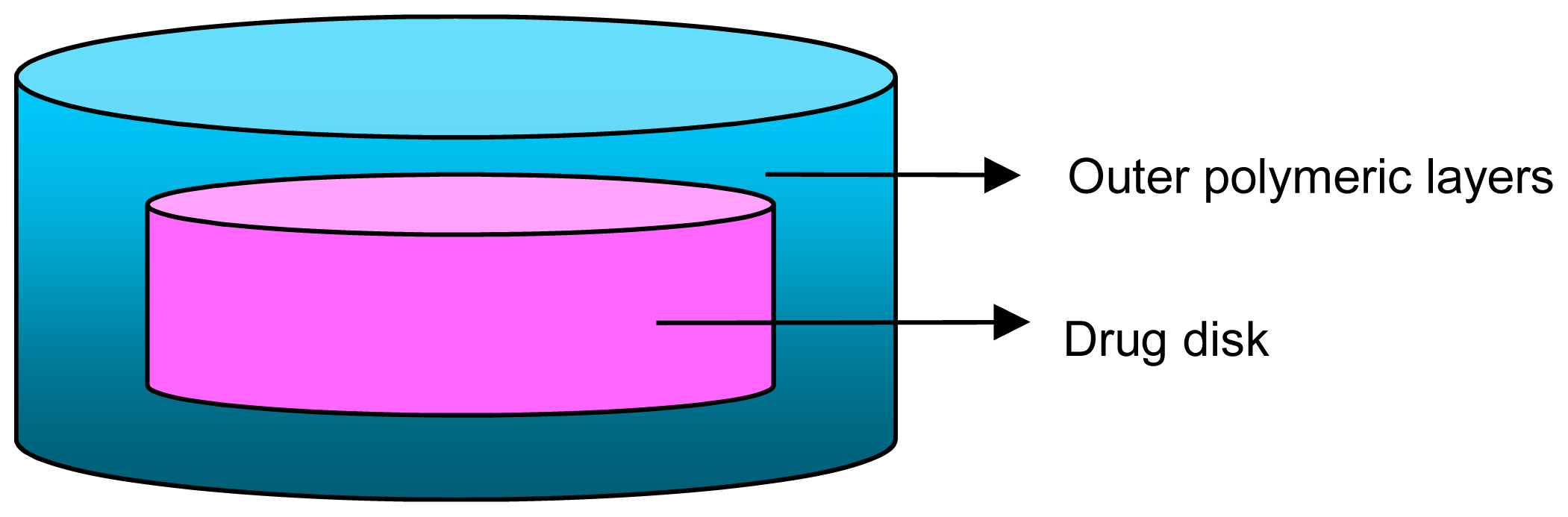



| Conventional Tablet | Multi-Layered Matrix Tablets |
|---|---|
 |  |
| Drug is released in only one kinetic model | May be used to incorporate more than one drug and separate them if any chemical incompatibilities exist. |
| If more than one drug is incorporated, there is no way of avoiding chemical incompatibilities. | Drug release behavior is not restricted to one type, this system may offer varied drug release kinetics of the same or different drugs such as extended and immediate release. |
| Type of Polymer Used as Drug Carrier | Type of Polymer Used in Barrier Layers | Type/Dimensions of Tablet | Drug Release Achieved |
|---|---|---|---|
| Hydrophilic | Hydrophilic | Bilayered tablet | Extended drug release |
| Hydrophilic | Hydrophobic | Bilayered tablet | Drug release retarded to lesser extent |
| Hydrophobic | Hydrophilic (Methocel® K4M) | Triple-layered tablet | Zero-order drug release kinetics |
| Hydrophobic (CW) | hydrophobic (carnauba wax) | Triple-layered tablet | Non-linear drug release |
| Hydrophobic (CW) | Hydrophilic (Methocel® K15M) and Hydrophobic (CW) | Triple-layered tablet | Zero-order drug release kinetics. |
| Hydrophilic (HPMCAS&HPMC) | Hydrophobic (EC) | Triple-layered tablet | Zero-order release kinetics. |
| Technology | Design | Factors Affecting Drug Release | Type of Drug Release That May Be Achieved |
|---|---|---|---|
| Geomatrix® | Triple/bilayered tablet. | Type of polymer used, thickness of layers. | Zero-order kinetics |
| Smartrix® | Triple-layered tablet with core layer having a specific shape different to that of the outer layers. | Shape of core layer. | According to shape of core, zero-order kinetics |
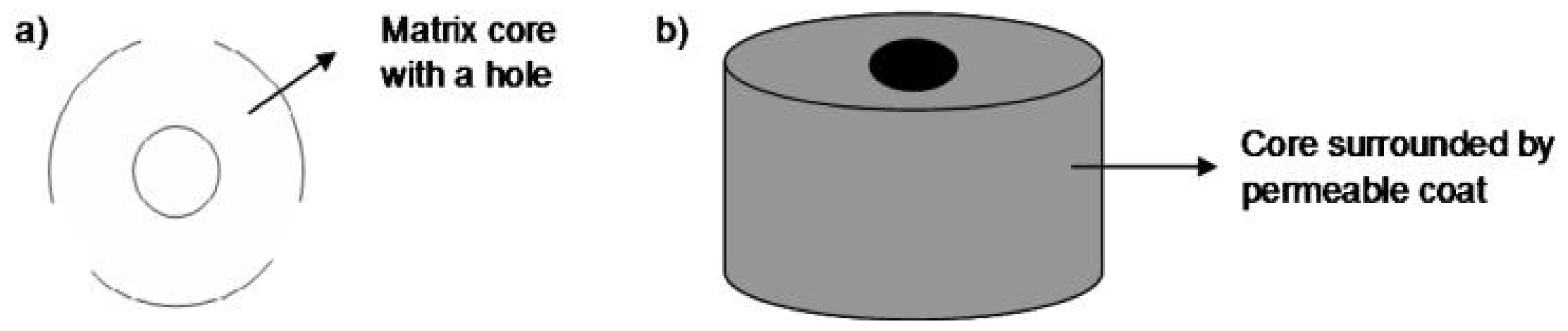
| Procise® | Uniformly dispersed drug core containing a hole. | Geometry of core | According to geometry of core, zero-order kinetics |
| Dome Matrix®/“Release modules assemblage” | Elementary module containing a concave base side and a convex base side. Various arrangements of modules to form different structures | Arrangement of modules, type of polymeric material used. | Various according to arrangement of modules e.g., Concave base attached to concave base. |
| Core-in-cup devices | Disc-shaped core compression coated on one surface and circumference to form a cup around it. | Type of polymer, dimensions of core and cup. | Zero-order kinetics |
| Doughnut-shaped tablets | Single/triple-layered tablets with a central hole/holes | Size and number of holes, type of polymer used Zero-order kinetics Sodas® Multilayer tablet Type of polymer used, thickness of layers. Shape of core layer | Pursatile drug release |
| VersaTab® | Bilayered tablet | Core drug, polymer layers | Immediate release and controlled release |
| GeolockTM | Triple layered tablet | Polymer layers, single or combination of drugs in the inner core | Immediate or modified release |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Moodley, K.; Pillay, V.; Choonara, Y.E.; Du Toit, L.C.; Ndesendo, V.M.K.; Kumar, P.; Cooppan, S.; Bawa, P. Oral Drug Delivery Systems Comprising Altered Geometric Configurations for Controlled Drug Delivery. Int. J. Mol. Sci. 2012, 13, 18-43. https://doi.org/10.3390/ijms13010018
Moodley K, Pillay V, Choonara YE, Du Toit LC, Ndesendo VMK, Kumar P, Cooppan S, Bawa P. Oral Drug Delivery Systems Comprising Altered Geometric Configurations for Controlled Drug Delivery. International Journal of Molecular Sciences. 2012; 13(1):18-43. https://doi.org/10.3390/ijms13010018
Chicago/Turabian StyleMoodley, Kovanya, Viness Pillay, Yahya E. Choonara, Lisa C. Du Toit, Valence M. K. Ndesendo, Pradeep Kumar, Shivaan Cooppan, and Priya Bawa. 2012. "Oral Drug Delivery Systems Comprising Altered Geometric Configurations for Controlled Drug Delivery" International Journal of Molecular Sciences 13, no. 1: 18-43. https://doi.org/10.3390/ijms13010018
APA StyleMoodley, K., Pillay, V., Choonara, Y. E., Du Toit, L. C., Ndesendo, V. M. K., Kumar, P., Cooppan, S., & Bawa, P. (2012). Oral Drug Delivery Systems Comprising Altered Geometric Configurations for Controlled Drug Delivery. International Journal of Molecular Sciences, 13(1), 18-43. https://doi.org/10.3390/ijms13010018






