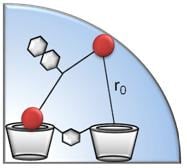
Enhanced Chiral Recognition by Cyclodextrin Dimers
Abstract
:1. Introduction
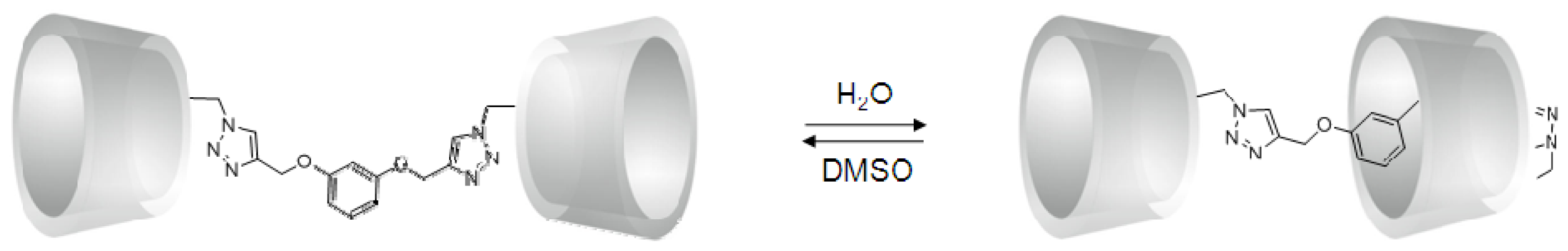
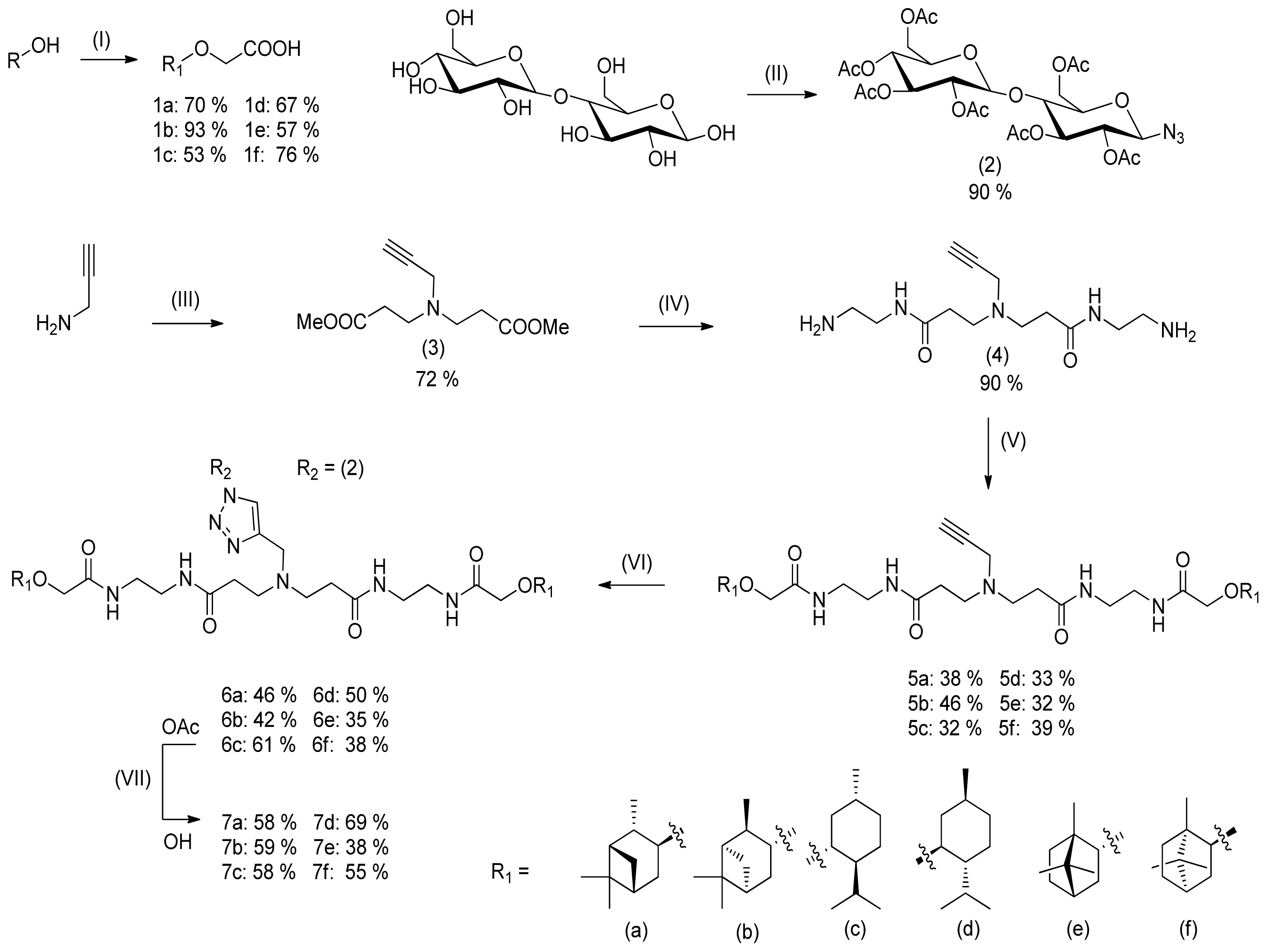
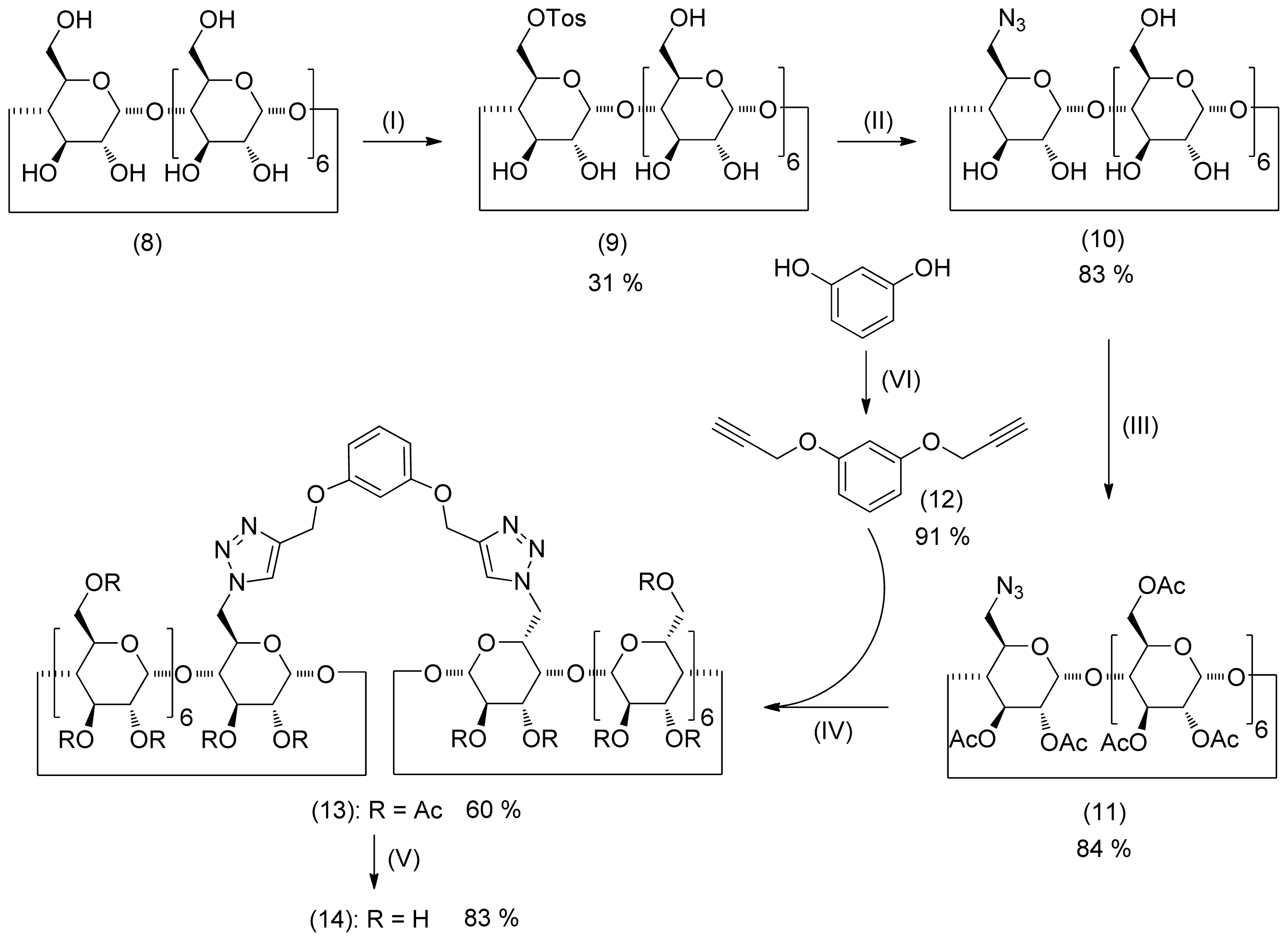
2. Results and Discussion
3. Conclusions
Supplementary Materials
ijms-12-04637-s001.pdfAcknowledgments
References
- Bressolle, F; Autran, M; Pham, TN; Vallon, JJ. Cyclodextrins and enantioselective separations of drugs by liquid chromatography and capillary electrophoresis: Basic principles and new developments. J. Chromatogr. B 1996, 687, 303–336. [Google Scholar]
- Rekharsky, M; Inoue, Y. Chiral Recognition Thermodynamics of β-Cyclodextrin: The Thermodynamic Origin of Enantioselectivity and the Enthalpy-Entropy Compensation Effect. J. Am. Chem. Soc 2000, 122, 4418–4435. [Google Scholar]
- Schwarz-Barac, S; Ritter, H; Schollmeyer, D. Cyclodextrins in polymer synthesis: Enantiodiscrimination in free-radical polymerization of cyclodextrin-complexed racemic N-Methacryloyl-D, L-phenylalanine methyl ester. Macromol. Rapid Commun 2003, 24, 325–330. [Google Scholar]
- D’Anna, F; Riela, S; Gruttadauria, M; Lo Meo, P; Noto, R. A spectrofluorimetric study of binary fluorophore-cyclodextrin complexes used as chiral selectors. Tetrahedron 2005, 61, 4577–4583. [Google Scholar]
- Chen, Y; Li, F; Liu, BW; Jiang, BP; Zhang, HY; Wang, L; Liu, Y. Thermodynamic Origin of Selective Binding of β-Cyclodextrin Derivatives with Chiral Chromophoric Substituents toward Steroids. J. Phys. Chem. B 2010, 114, 16147–16155. [Google Scholar]
- Gingter, S; Bezdushna, E; Ritter, H. Chiral Recognition of Poly(N-isopropylacrylamide-co-(D or L)-N-tryptophan-acrylamide) with Methylated β-Cyclodextrin. Macromolecules 2010, 43, 3128–3131. [Google Scholar]
- Rekharsky, M; Inoue, Y. Complexation Thermodynamics of Cyclodextrins. Chem. Rev 1998, 98, 1875–1917. [Google Scholar]
- Liu, Y; Zhang, Q; Chen, Y. Spectrophotometric and Calorimetric Titration Studies on Molecular Recognition of Camphor and Borneol by Nucleobase-Modified β-Cyclodextrins. J. Phys. Chem. B 2007, 111, 12211–12218. [Google Scholar]
- Mammen, M; Choi, SK; Whitesides, GM. Polyvalent interactions in biological systems: Implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Ed 1998, 37, 2754–2794. [Google Scholar]
- Kim, Y; Hechler, B; Klutz, AM; Gachet, C; Jacobson, KA. Toward Multivalent Signaling across G Protein-Coupled Receptors from Poly(amidoamine) Dendrimers. Bioconjugate Chem 2008, 19, 406–411. [Google Scholar]
- Liu, Z; Deshazer, H; Rice, AJ; Chen, K; Zhou, C; Kallenbach, NR. Multivalent Antimicrobial Peptides from a Reactive Polymer Scaffold. J. Med. Chem 2006, 49, 3436–3439. [Google Scholar]
- Bowman, MC; Ballard, TE; Ackerson, CJ; Feldheim, DL; Margolis, DM; Melander, C. Inhibition of HIV Fusion with Multivalent Gold Nanoparticles. J. Am. Chem. Soc 2008, 130, 6896–6897. [Google Scholar]
- DeFrees, SA; Phillips, L; Guo, L; Zalipsky, S. Sialyl Lewis x Liposomes as a multivalent Ligand and Inhibitor of E-Selectin Mediated Cellular Adhesion. J. Am. Chem. Soc 1996, 118, 6101–6104. [Google Scholar]
- Voskuhl, J; Stuart, MCA; Ravoo, BJ. Sugar-Decorated Sugar Vesicles: Lectin-Carbohydrate Recognition at the Surface of Cyclodextrin Vesicles. Chem. Eur. J 2010, 16, 2790–2796. [Google Scholar]
- Crespo-Biel, O; Lim, CW; Ravoo, BJ; Reinhoudt, DN; Huskens, J. Expression of a supramolecular complex at a multivalent interface. J. Am. Chem. Soc 2006, 128, 17024–17032. [Google Scholar]
- Harada, A; Furue, M; Nozakura, SI. Cooperative Binding by Cyclodextrin Dimers. Polym. J 1980, 12, 29–33. [Google Scholar]
- Zhang, B; Breslow, R. Enthalpic domination of the chelate effect in cyclodextrin dimers. J. Am. Chem. Soc 1993, 115, 9353–9354. [Google Scholar]
- Jiang, T; Sukumaran, DK; Soni, SD; Lawrence, DS. The Synthesis and Characterization of a Pyridine-Linked Cyclodextrin Dimer. J. Org. Chem 1994, 59, 5149–5155. [Google Scholar]
- Breslow, R; Zhang, B. Cholesterol Recognition and Binding by Cyclodextrin Dimers. J. Am. Chem. Soc 1996, 118, 8495–8496. [Google Scholar]
- Haskard, CA; Easton, CJ; May, BL; Lincoln, SF. Cooperative Binding of 6-(p-Toluidinyl) naphthalene-2-sulfonate by β-Cyclodextrin Dimers. J. Phys. Chem. B 1996, 100, 14457–14461. [Google Scholar]
- Zhang, B; Breslow, R. Ester Hydrolysis by a Catalytic Cyclodextrin Dimer Enzyme Mimic with a Metallobipyridyl Linking Group. J. Am. Chem. Soc 1997, 119, 1676–1681. [Google Scholar]
- French, RR; Holzer, P; Leuenberger, MG; Woggon, WDA. Supramolecular Enzyme Mimic That Catalyzes the 15,15′ Double Bond Scission of β,β-Carotene. Angew. Chem. Int. Ed 2000, 39, 1267–1269. [Google Scholar]
- De Jong, MR; Engbersen, JFJ; Huskens, J; Reinhoudt, DN. Cyclodextrin dimers as receptor molecules for steroid sensors. Chem. Eur. J 2000, 6, 4034–4040. [Google Scholar]
- Edwards, WB; Reichert, DE; d’Avignon, DA; Welch, MJ. β-Cyclodextrin dimers as potential tumor pretargeting agents. Chem Commun 2001, 1312–1313. [Google Scholar] [CrossRef]
- Mulder, A; Auletta, T; Sartori, A; Del Ciotto, S; Casnati, A; Ungaro, R; Huskens, J; Reinhoudt, DN. Divalent Binding of a Bis(adamantyl)-Functionalized Calix[4]arene to β-cyclodextrin-based Hosts: An Experimental and Theoretical Study on Multivalent Binding in Solution and at Self-Assembled Monolayers. J. Am. Chem. Soc 2004, 126, 6627–6636. [Google Scholar]
- Liu, Y; Ding, F. Molecular Recognition Thermodynamics of Bile Salts by β-Cyclodextrin Dimers: Factors Governing the Cooperative Binding of Cyclodextrin Dimers. J. Phys. Chem. B 2005, 109, 4129–4134. [Google Scholar]
- Bea, I; Kollman, P. Chelate Effect in Cyclodextrin Dimers: A Computational (MD, MM/PBSA, and MM/GBSA) Study. J. Org. Chem 2006, 71, 2056–2063. [Google Scholar]
- Ohga, K; Harada, A. Preparation of Supramolecular Polymers from a Cyclodextrin Dimer and Ditopic Guest Molecules: Control of Structure by Linker Flexibility. Macromolecules 2005, 38, 5897–5904. [Google Scholar]
- Hasegawa, Y; Harada, A. Supramolecular Polymers Formed from β-Cyclodextrins Dimer Linked by Poly (ethylene glycol) and Guest Dimers. Macromolecules 2005, 38, 3724–3730. [Google Scholar]
- Leggio, C; Tato, J. Study on the Structure of Host-Guest Supramolecular Polymers. Macromolecules 2007, 40, 5899–5906. [Google Scholar]
- Mulder, A; Jukovic, A; Lucas, LN; van Esch, J; Feringa, BL; Huskens, J; Reinhoudt, DN. A dithienylethene-tethered beta-cyclodextrin dimer as a photoswitchable host. Chem Commun 2002, 2734–2735. [Google Scholar] [CrossRef]
- Kuad, P; Harada, A. External Stimulus-Responsive Supramolecular Structures Formed by a Stilbene Cyclodextrin Dimer. J. Am. Chem. Soc 2007, 129, 12630–12631. [Google Scholar]
- Mourer, M; Hapiot, F; Monflier, E; Menuel, S. Click chemistry as an efficient tool to access β-cyclodextrin dimers. Tetrahedron 2008, 64, 7159–7163. [Google Scholar]
- Menuel, S; Azaroual, N; Landy, D; Six, N; Hapiot, F; Monflier, E. Unusual Inversion Phenomenon of β-Cyclodextrin Dimers in Water. Chem. Eur. J 2011, 14, 3949–3955. [Google Scholar]
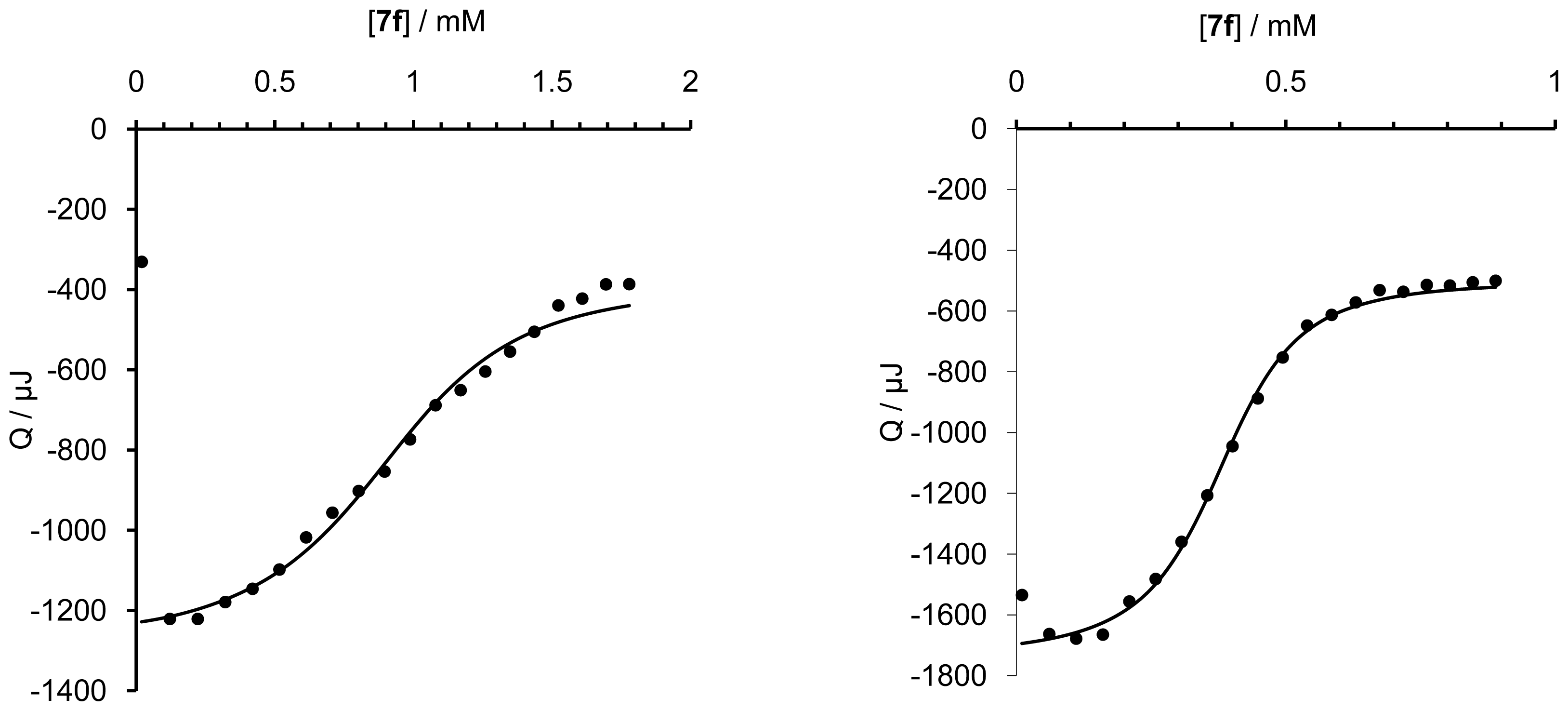
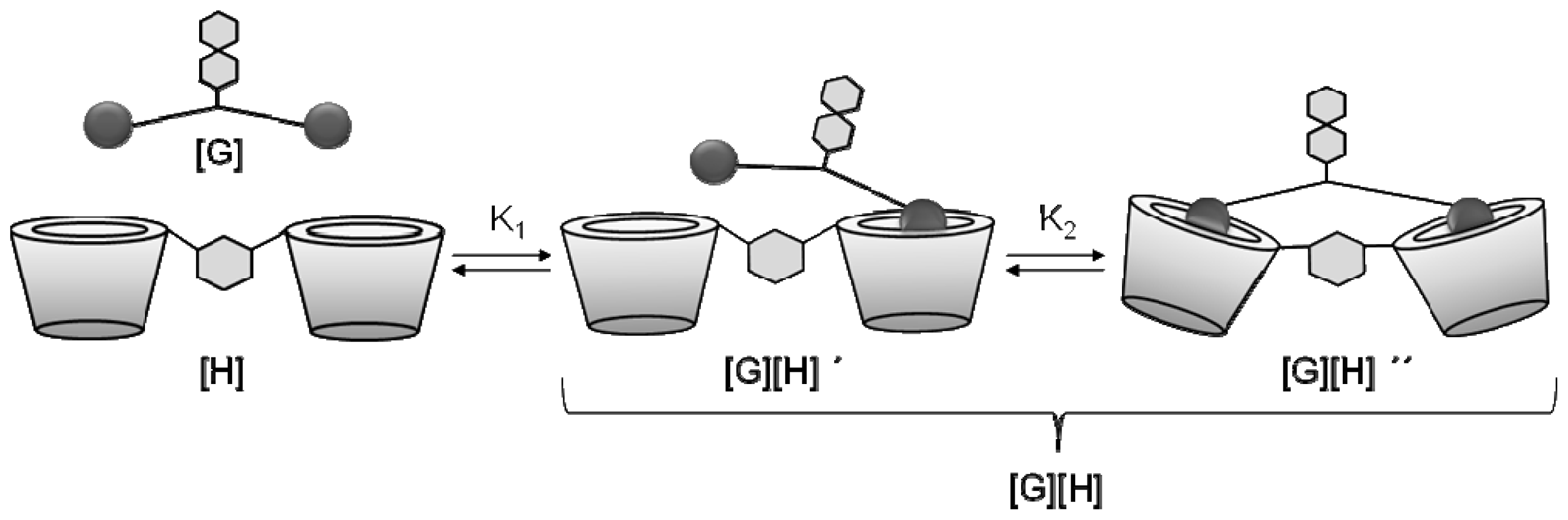
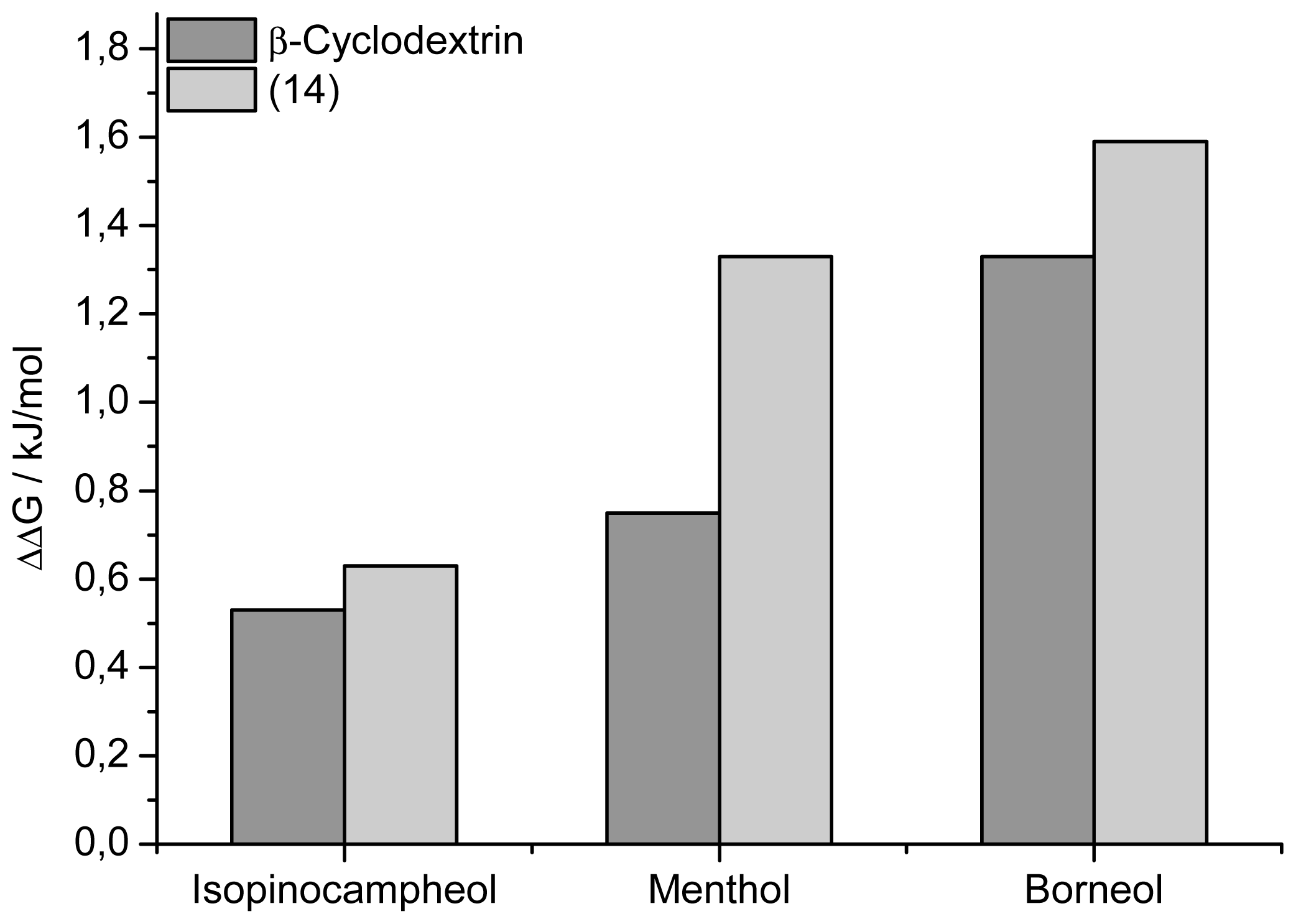
| Nr. | Guest (G) | Host (H) | ΔH (kJ mol−1) | ΔG (kJ mol−1) | ΔS (JK−1mol−1) | Ka (M−1) ×104 | n (H/G) | Ceff (mM) | |
|---|---|---|---|---|---|---|---|---|---|
| A | 7a | (+)Iso2 | β − | −13.16 | −24.01 | 36.70 | 1.68 | 1.06 | |
| B | 7a | (+)Iso2 | 14 | −45.56 | −27.69 | −27.69 | 7.14 | 0.77 | 0.126 |
| C | 7b | (−)Iso2 | β − | −14.90 | −23.48 | 28.81 | 1.31 | 0.97 | |
| D | 7b | (−)Iso2 | 14 | −34.50 | −28.32 | −28.32 | 9.22 | 0.80 | 0.268 |
| E | 7c | (−)Men2 | β − | −14.22 | −22.31 | 27.24 | 0.83 | 0.92 | |
| F | 7c | (−)Men2 | 14 | −42.63 | −25.65 | −57.00 | 3.14 | 0.69 | 0.228 |
| G | 7d | (+)Men2 | β − | −9.18 | −23.06 | 46.57 | 1.10 | 1.03 | |
| H | 7d | (+)Men2 | 14 | −25.04 | −26.98 | 6.51 | 5.35 | 0.81 | 0.220 |
| I | 7e | (−)Bor2 | β − | −18.89 | −23.21 | 14.66 | 1.28 | 0.99 | |
| J | 7e | (−)Bor2 | 14 | −41.58 | −27.22 | −48.19 | 5.90 | 0.77 | 0.168 |
| K | 7f | (+)Bor2 | β − | −19.02 | −24.54 | 18.51 | 2.00 | 0.93 | |
| L | 7f | (+)Bor2 | 14 | −40.67 | −28.81 | −39.80 | 11.21 | 0.80 | 0.140 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Voskuhl, J.; Schaepe, K.; Ravoo, B.J. Enhanced Chiral Recognition by Cyclodextrin Dimers. Int. J. Mol. Sci. 2011, 12, 4637-4646. https://doi.org/10.3390/ijms12074637
Voskuhl J, Schaepe K, Ravoo BJ. Enhanced Chiral Recognition by Cyclodextrin Dimers. International Journal of Molecular Sciences. 2011; 12(7):4637-4646. https://doi.org/10.3390/ijms12074637
Chicago/Turabian StyleVoskuhl, Jens, Kira Schaepe, and Bart Jan Ravoo. 2011. "Enhanced Chiral Recognition by Cyclodextrin Dimers" International Journal of Molecular Sciences 12, no. 7: 4637-4646. https://doi.org/10.3390/ijms12074637