Fabrication of Porous Scaffolds with a Controllable Microstructure and Mechanical Properties by Porogen Fusion Technique
Abstract
:1. Introduction
2. Results and Discussion
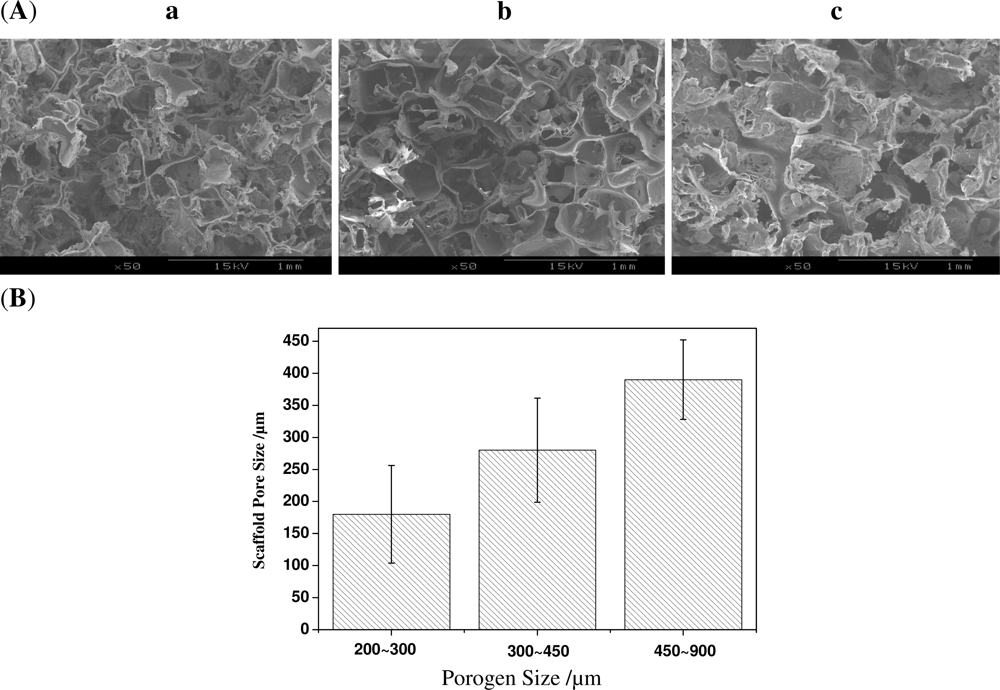
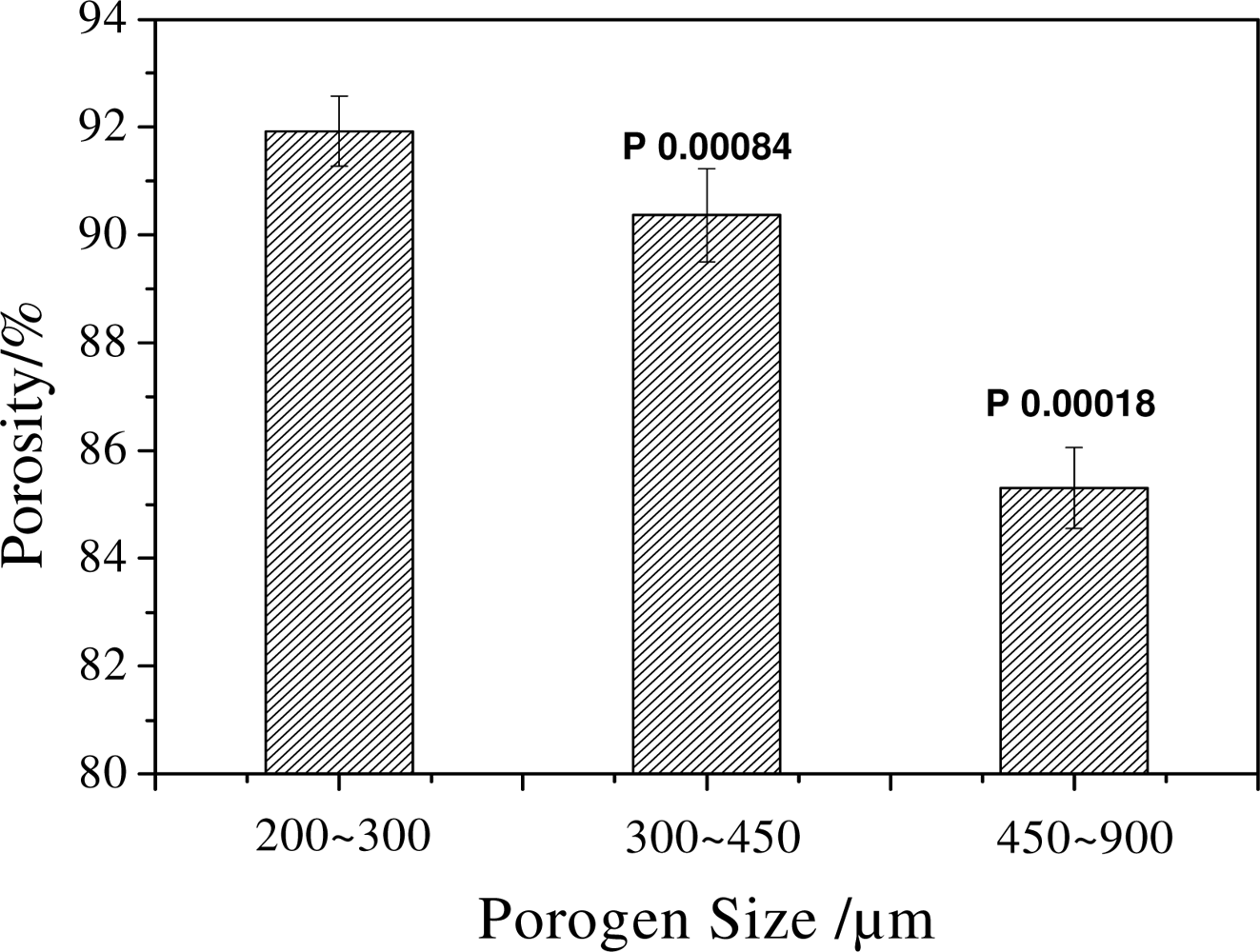
2.1. Effect of Porogen Sizes on the Structure and Mechanical Properties of Scaffolds
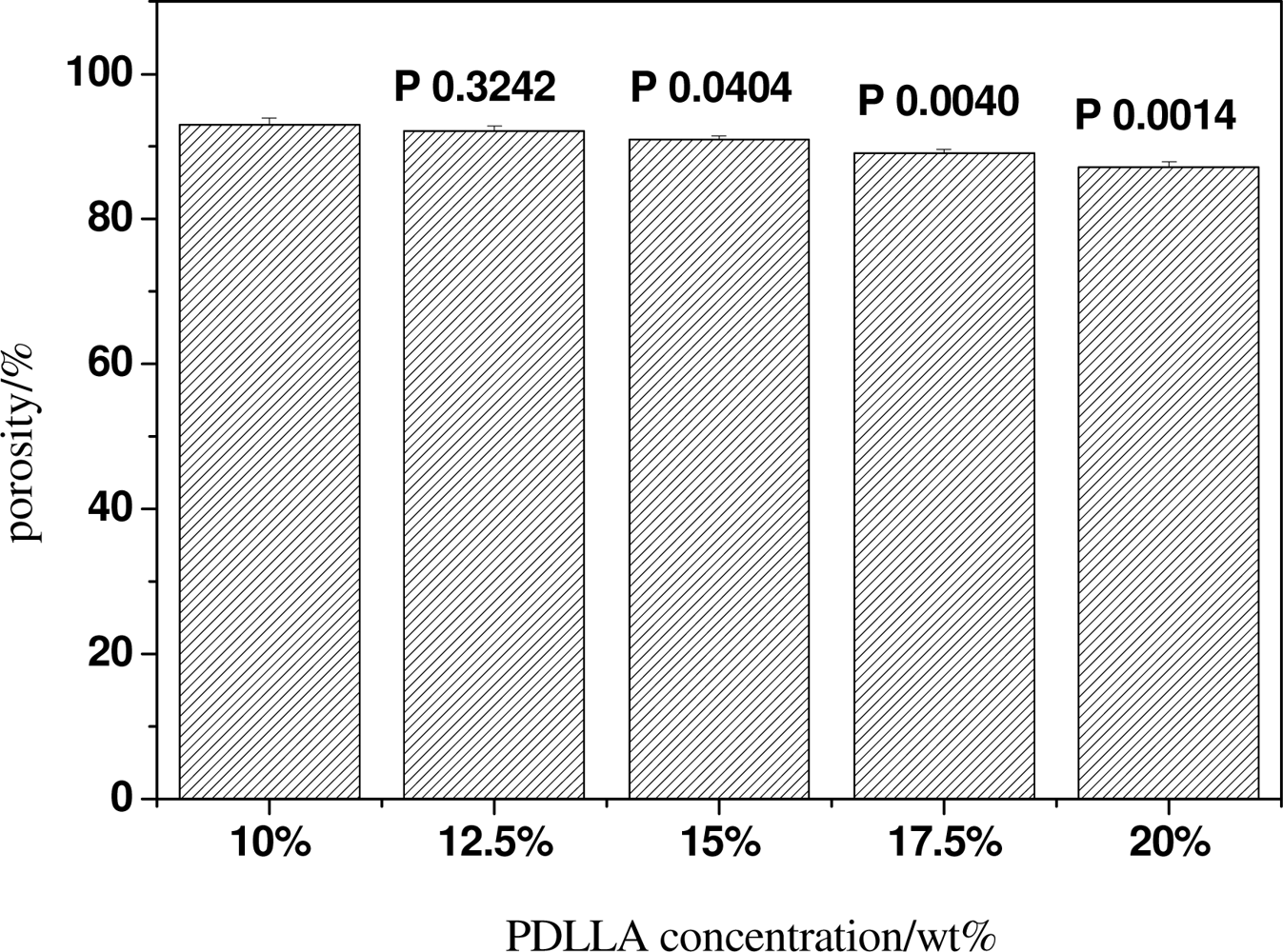
2.2. Effect of PDLLA Concentration on the Structure and Mechanical Properties of Scaffolds
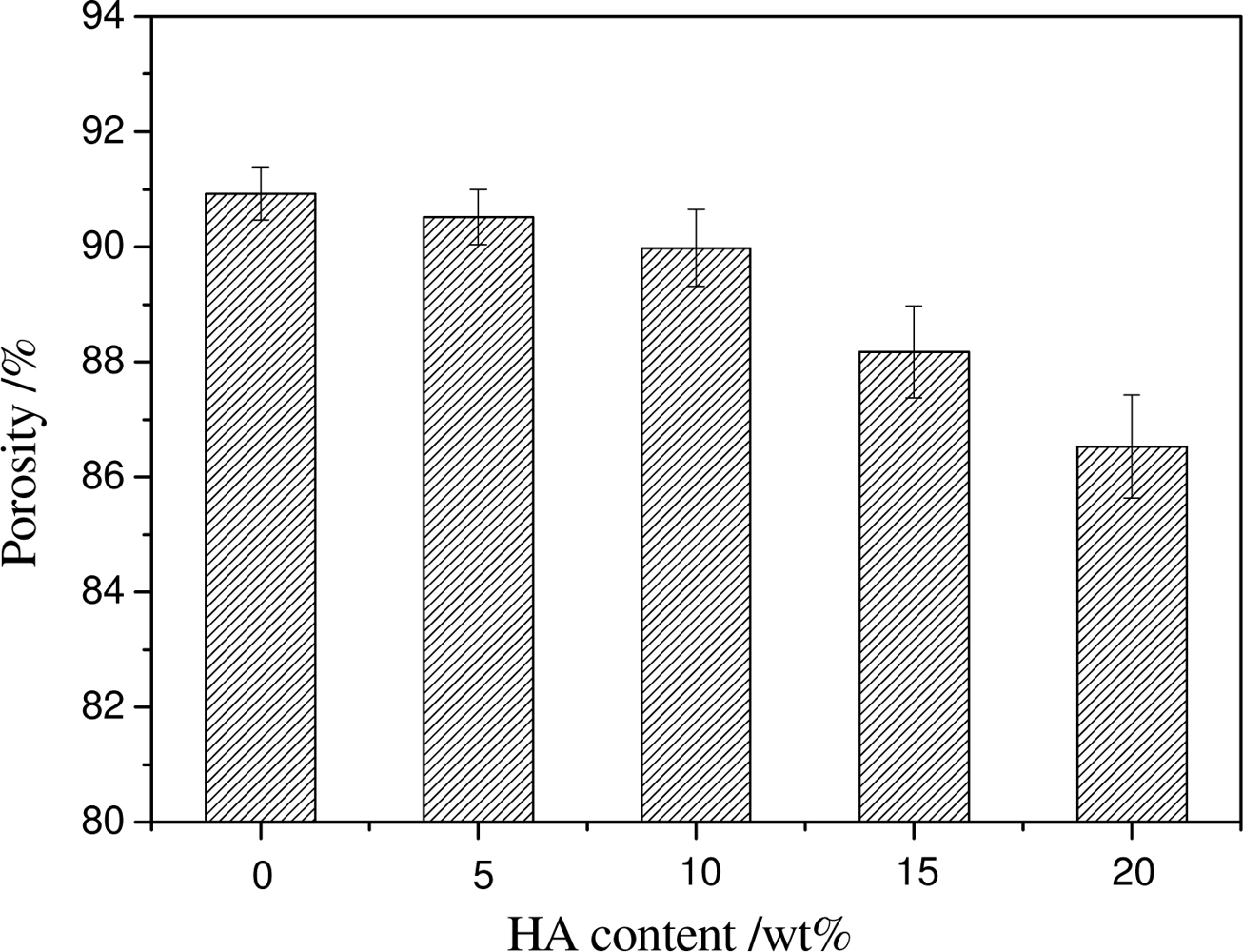
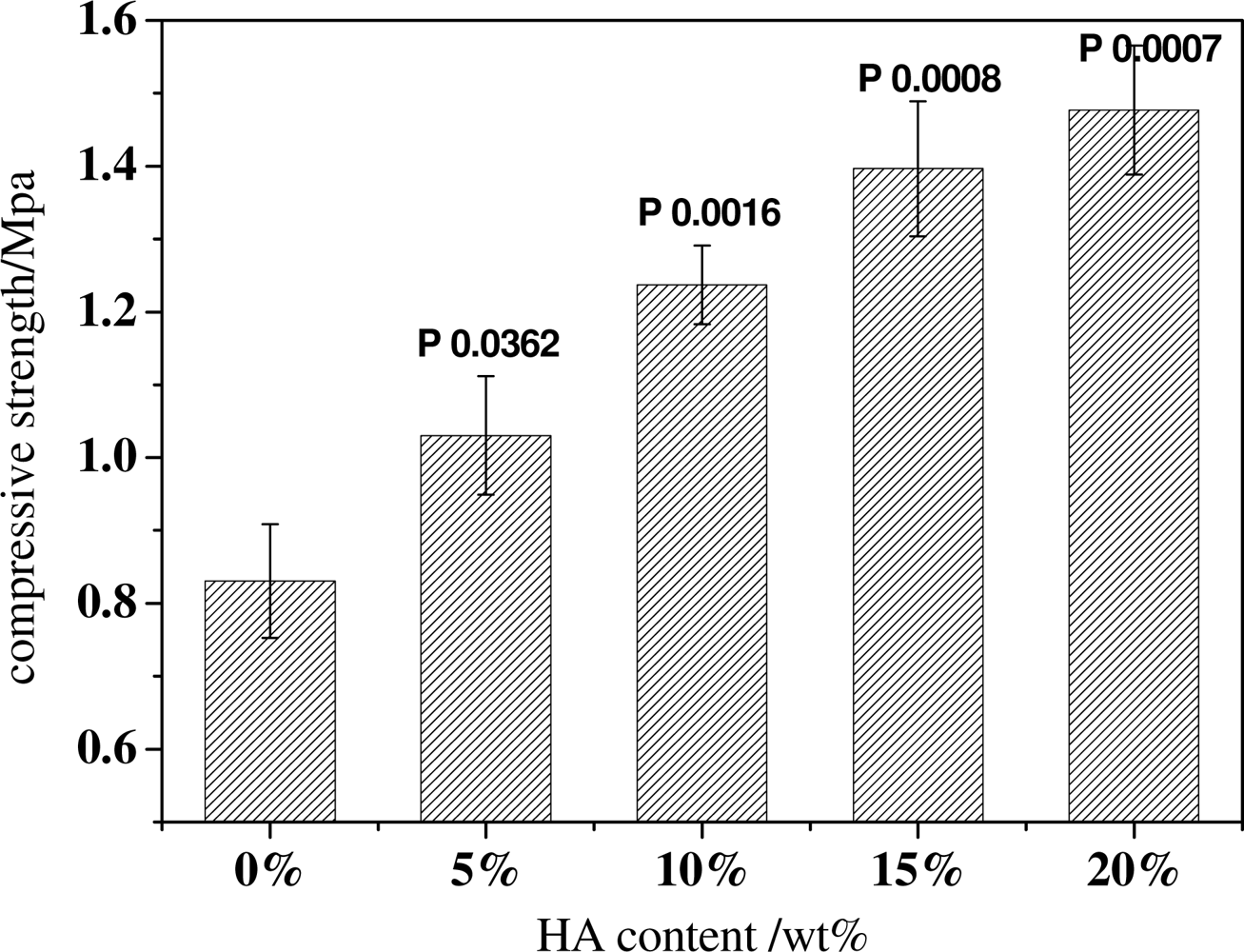
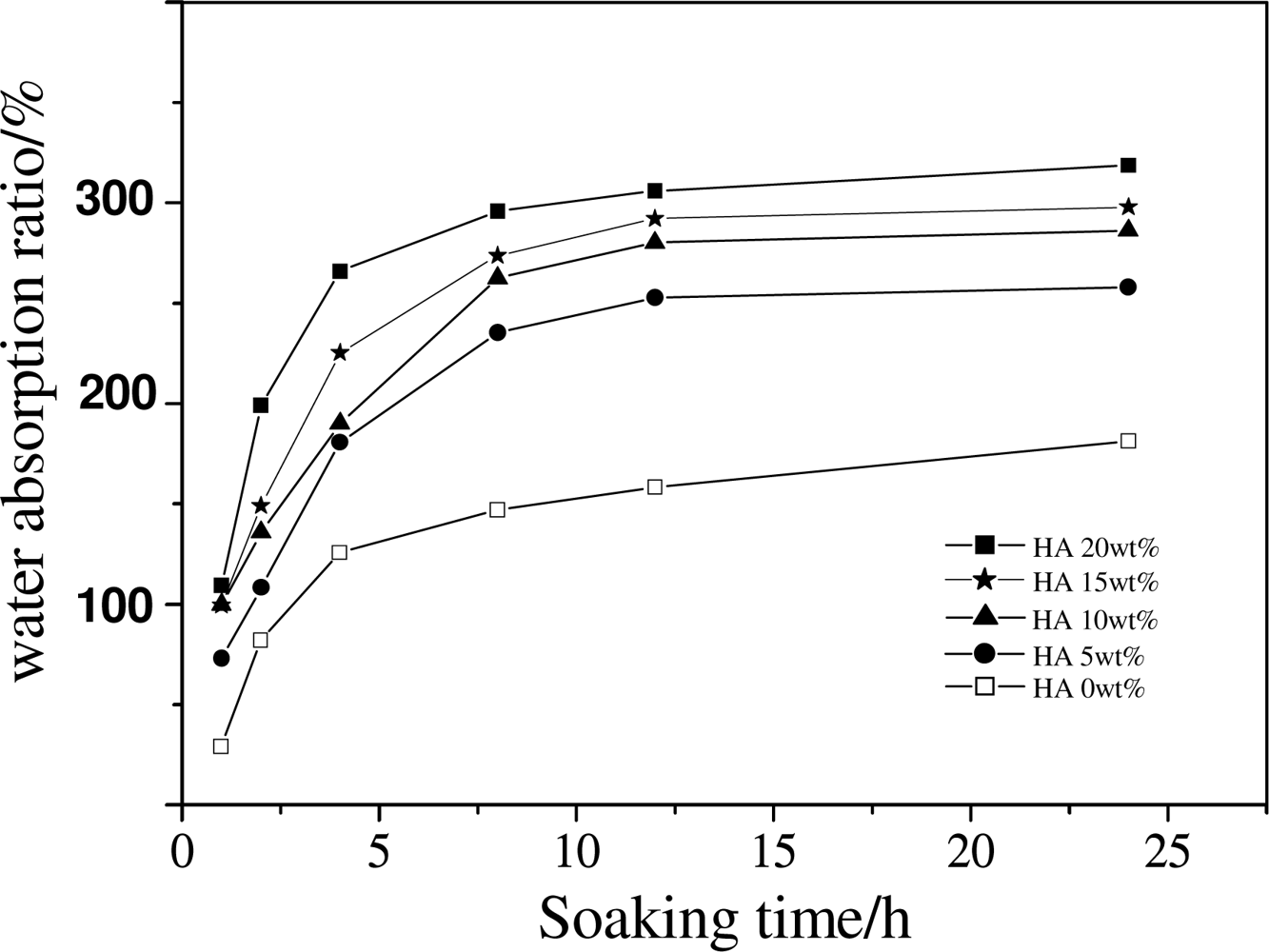
2.3. Effect of HA Content on the Structure and Mechanical Properties of Scaffold
3. Experimental Section
3.1. Materials
3.2. Scaffold Preparation
3.3. Characterization
3.3.1. Morphology
3.3.2. Porosity
3.3.3. Compressive Strength
3.3.4. Water Absorption Ability
4. Conclusions
Acknowledgments
References
- Hutmacher, DW. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar]
- Temenoff, JS; Mikos, AG. Tissue engineering for regeneration of articular cartilage. Biomaterials 2000, 21, 431–440. [Google Scholar]
- Zhang, Y; Zhang, M. Synthesis and characterization of macroporous chitosan/calcium phosphate composite scaffolds for tissue engineering. J. Biomed. Mater. Res 2001, 55, 304–312. [Google Scholar]
- Rizzi, SC; Heath, DJ; Coombes, AGA; Bock, N; Textor, M; Downes, S. Biodegradable polymer/hydroxyapatite composites: Surface analysis and initial attachment of human osteoblasts. J. Biomed. Mater. Res 2001, 55, 475–486. [Google Scholar]
- Wang, M. Developing bioactive composite materials for tissue replacement. Biomaterials 2003, 24, 2133–2151. [Google Scholar]
- Karageorgiou, V; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar]
- Widmer, MS; Puneet, K; Gupta, PK; Lu, L; Meszlenyi, RK; Evans, GRD; Brandt, K; Savel, T; Gurlek, A; Charles, W; Patrick, CW; Mikos, AG. Manufacture of porous biodegradable polymer conduits by an extrusion process for guided tissue regeneration. Biomaterials 1998, 19, 1945–1955. [Google Scholar]
- Mikos, AG; Lyman, MD; Freed, LE; Langer, R. Wetting of poly(l-lactic acid) and poly(d,l-lactic-co-glycolic acid) foams for tissue culture. Biomaterials 1994, 15, 55–58. [Google Scholar]
- Kim, SS; Park, MS; Jeon, O; Choi, CY; Kim, BS. Poly(lactide-co-glycolide)/hydroxyapatite composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 1399–1409. [Google Scholar]
- Chen, G; Ushida, T; Tateishi, T. Preparation of poly(l-lactic acid) and poly(dl-lactic-co-glycolic acid) foams by use of ice microparticulates. Biomaterials 2001, 22, 2563–2567. [Google Scholar]
- Mooney, DJ; Baldwin, DF; Suh, NP; Vacanti, JP; Langer, R. Novel approach to fabricate porous sponges of poly (dl-lactic-co-glycolic acid) without the use of organic solvents. Biomaterials 1996, 17, 1417–1422. [Google Scholar]
- Wang, K; Thomas, CH; Healy, KE; Nuber, G. A novel method to fabricate bioabsorbable scaffolds. Polymer 1995, 36, 837–842. [Google Scholar]
- Nam, YS; Park, TG. Biodegradable polymeric microcellular foams by modified thermally induced phase separation method. Biomaterials 1999, 20, 1783–1790. [Google Scholar]
- Zhang, R; Ma, PX. Poly(α-hydroxyl acids)/hydroxyapatite porous composites for bone-tissue engineering. I. Preparation and morphology. J. Biomed. Mater. Res 1999, 44, 446–455. [Google Scholar]
- Hua, FJ; Park, TG; Lee, DS. A facile preparation of highly interconnected macroporous poly (d,l-lactic acid-co-glycolic acid) (PLGA) scaffold by liquid-liquid phase separation of a PLGA-dioxane-water ternary system. Polymer 2003, 44, 1911–1920. [Google Scholar]
- Hua, FJ; Nam, JD; Lee, DS. Preparation of a macroporous poly(l-lactide) scaffold by liquid-liquid phase separation of a PLLA/1,4-dioxane/water ternary system in the presence of NaCl. Macromol. Rapid Commun 2001, 22, 1053–1057. [Google Scholar]
- Hutmacher, DW. Scaffold design and fabrication technologies for engineering tissuesstate of the art and future perspectives. J. Biomater. Sci. Polym. Ed 2001, 1, 107–124. [Google Scholar]
- Guan, L; Davies, JE. Preparation and characterization of a highly macroporous biodegradable composite tissue engineering scaffold. J. Biomed. Mater. Res 2004, 71A, 480–487. [Google Scholar]
- Murphy, WL; Dennis, RG; Kileny, JL; Mooney, DJ. Salt fusion: an approach to improve pore interconnectivity within tissue engineering scaffolds. Tiss. Eng 2002, 8, 43–52. [Google Scholar]
- Lu, LC; Garcia, CA; Mikos, AG. In vitro degradation of thin poly (dl-lactic-co-glycolic acid) films. J. Biomed. Mater. Res 1999, 46, 236–244. [Google Scholar]
- Migliaresi, C; Fambri, L; Cohn, D. A study on the in vitro degradation of poly (lactic acid). J. Biomed. Sci. Polym. Ed 1994, 6, 591–606. [Google Scholar]
- Ge, Z; Yang, F; Goh, JCH; Ramakrishna, S; Lee, EH. Biomaterial and scaffolds for ligament tissue engineering. J. Biomed. Mater. Res 2006, 77A, 639–652. [Google Scholar]
- Barrera, DA; Zylstra, E; Lansbury, PT; Langer, R. Synthesis and RGD peptide modification of a new biodegradable copolymer: poly (lactic acid-co-lysine). J. Am. Chem. Soc 1993, 115, 11010–11011. [Google Scholar]
- Suh, H; Hwang, YS; Lee, JE; Han, CD; Park, JC. Behavior of osteoblasts on a type I atelocollagen grafted ozone oxidized poly-l-lactic acid membrane. Biomaterials 2001, 22, 219–230. [Google Scholar]
- Hench, LL. Bioceramics: From concept to clinic. J. Am. Ceram. Soc 1991, 74, 1487–1510. [Google Scholar]
- Rizzi, SC; Heath, DJ; Coombes, AGA; Bock, N; Textor, M; Downes, S. Biodegradable polymer/hydroxyapatite composites: Surface analysis and initial attachment of human osteoblasts. J. Biomed. Mater. Res 2001, 55, 475–486. [Google Scholar]
- Bleach, NC; Nazhat, SN; Tanner, KE; Kellomaki, M; Tormala, P. Effect of filler content on mechanical and dynamic mechanical properties of particulate biphasic calcium phosphate-polylactide composites. Biomaterials 2002, 7, 1579–1585. [Google Scholar]
- Montse, CH; Sergio, DV; Hentges, E; Bleuet, P; Lacroix, D; Planell, JA. Mechanical and structural characterisation of completely degradable polylactic acid/calcium phosphate glass scaffolds. Biomaterials 2007, 30, 4429–4438. [Google Scholar]





© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tan, Q.; Li, S.; Ren, J.; Chen, C. Fabrication of Porous Scaffolds with a Controllable Microstructure and Mechanical Properties by Porogen Fusion Technique. Int. J. Mol. Sci. 2011, 12, 890-904. https://doi.org/10.3390/ijms12020890
Tan Q, Li S, Ren J, Chen C. Fabrication of Porous Scaffolds with a Controllable Microstructure and Mechanical Properties by Porogen Fusion Technique. International Journal of Molecular Sciences. 2011; 12(2):890-904. https://doi.org/10.3390/ijms12020890
Chicago/Turabian StyleTan, Qinggang, Songgang Li, Jie Ren, and Chu Chen. 2011. "Fabrication of Porous Scaffolds with a Controllable Microstructure and Mechanical Properties by Porogen Fusion Technique" International Journal of Molecular Sciences 12, no. 2: 890-904. https://doi.org/10.3390/ijms12020890



