Synthesis and Insecticidal Activities of New Ester-Derivatives of Celangulin-V
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis of Celangulin-V Derivatives
2.2. Insecticidal Activities
3. Experimental
3.1. General Experimental Procedures
3.2. Synthesis of the Derivatives of Celangulin-V
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-acetyloxy-4α-hydroxy-β-dihydroagarofuran (1.1):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-propyloxy-4α-hydroxy-β-dihydroagarofuran (1.2):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-butanoyloxy-4α-hydroxy-β-dihydroagarofuran (1.3):
1β,2β-diacetyloxy-6α,8α,12-triisobutanoyloxy-9β-benzoyloxy-4α-hydroxy-β-dihydro-agarofuran (1.7):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-pentanoyloxy-4α-hydroxy-β-dihydroagarofuran (1.4):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-hexanoyloxy-4α-hydroxy-β-dihydroagarofuran (1.5):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-heptanoyloxy-4α-hydroxy-β-dihydroagarofuran (1.6):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-(2-ethylbutanoyloxy)-4α-hydroxy-β-dihydro-agarofuran (1.8):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-9β-benzoyloxy-6α-(3-phenylpropanoyloxy)-4α-hydroxy- β-dihydro-agarofuran (1.9):
1β,2β-diacetyloxy-8α,12-diisobutanoyloxy-6α,9β-dibenzoyloxy-4α-hydroxy-β-dihydroagarofuran (1.10):
3.3. Bioassay for Insecticidal Activities
4. Conclusions
Acknowledgements
References
- Gao, J.M.; Wu, W.J.; Zhang, J.W.; Konishi, Y. The dihydro-β-agarofuran sesquiterpenoids. Nat. Prod. Rep 2007, 24, 1153–1189. [Google Scholar]
- Spivey, A.C.; Weston, M.; Woodhead, S. Celastraceae sesquiterpenoids: Biological activity and synthesis. Chem. Soc. Rev 2002, 31, 43–59. [Google Scholar]
- Wei, S.P.; Wang, M.A.; Zhang, J.W.; Qian, Y.; Ji, Z.Q.; Wu, W.J. Three novel sesquiterpene esters from Celastrus angulatus. Nat. Prod. Commun 2009, 4, 461–466. [Google Scholar]
- Wu, W.J.; Tu, Y.Q.; Liu, H.X.; Zhu, J.B. Celangulin II, III and IV: New insecticidal sesquiterpenoids from Celastrus angulatus. J. Nat. Prod 1992, 55, 1294–1298. [Google Scholar]
- Wang, M.T.; Qin, H.L.; Kong, M.; Li, Y.Z. Insecticidal sesquiterpene polyol ester from Celastrus angulatus. Phytochemistry 1991, 30, 3931–3933. [Google Scholar]
- Wu, W.J.; Li, S.B.; Zhu, J.B.; Liu, H.X. New Sesquiterpenoid celangulin V: Isolation and determination. J. Northwest A&F Univ 1994, 22, 116–117. [Google Scholar]
- Qi, Z.J.; Xue, X.P.; Wu, W.J.; Zhang, J.W.; Yang, R.Y. Preparation of monoclonal antibody against celangulin V and immunolocalization of receptor in the oriental armyworm, Mythimna separata walker (Lepidoptera: Noctuidae). J. Agric. Food Chem 2006, 54, 7600–7605. [Google Scholar]
- Zhang, J.W.; Hu, Z.N.; Yang, H.; Wu, W.J. Synthesis and insecticidal activities of new ether-derivatives of celangulin-V. Nat. Prod. Commun 2010, 5, 845–848. [Google Scholar]
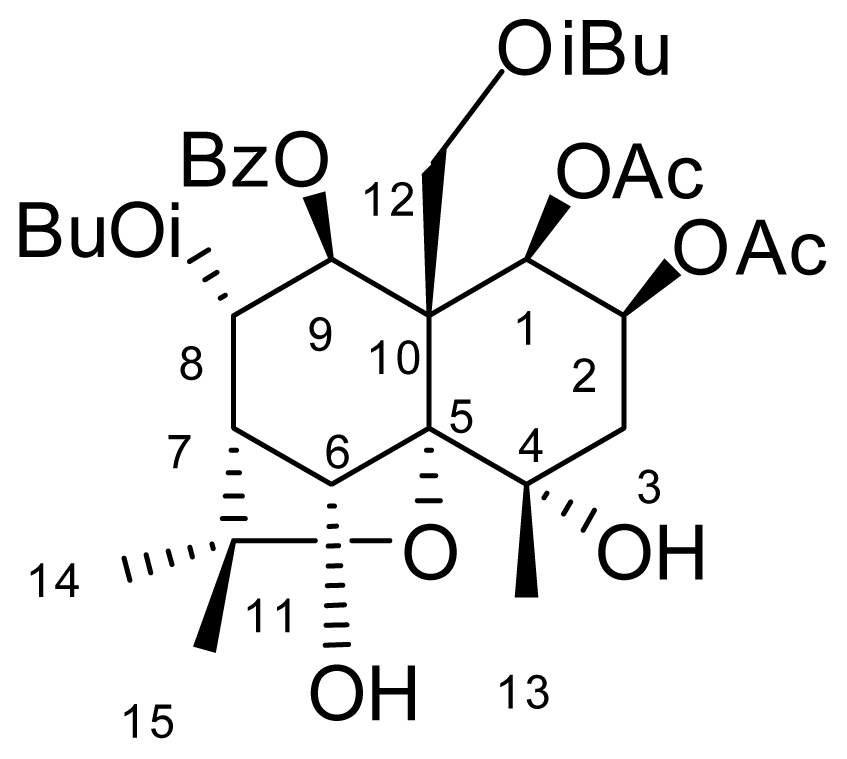
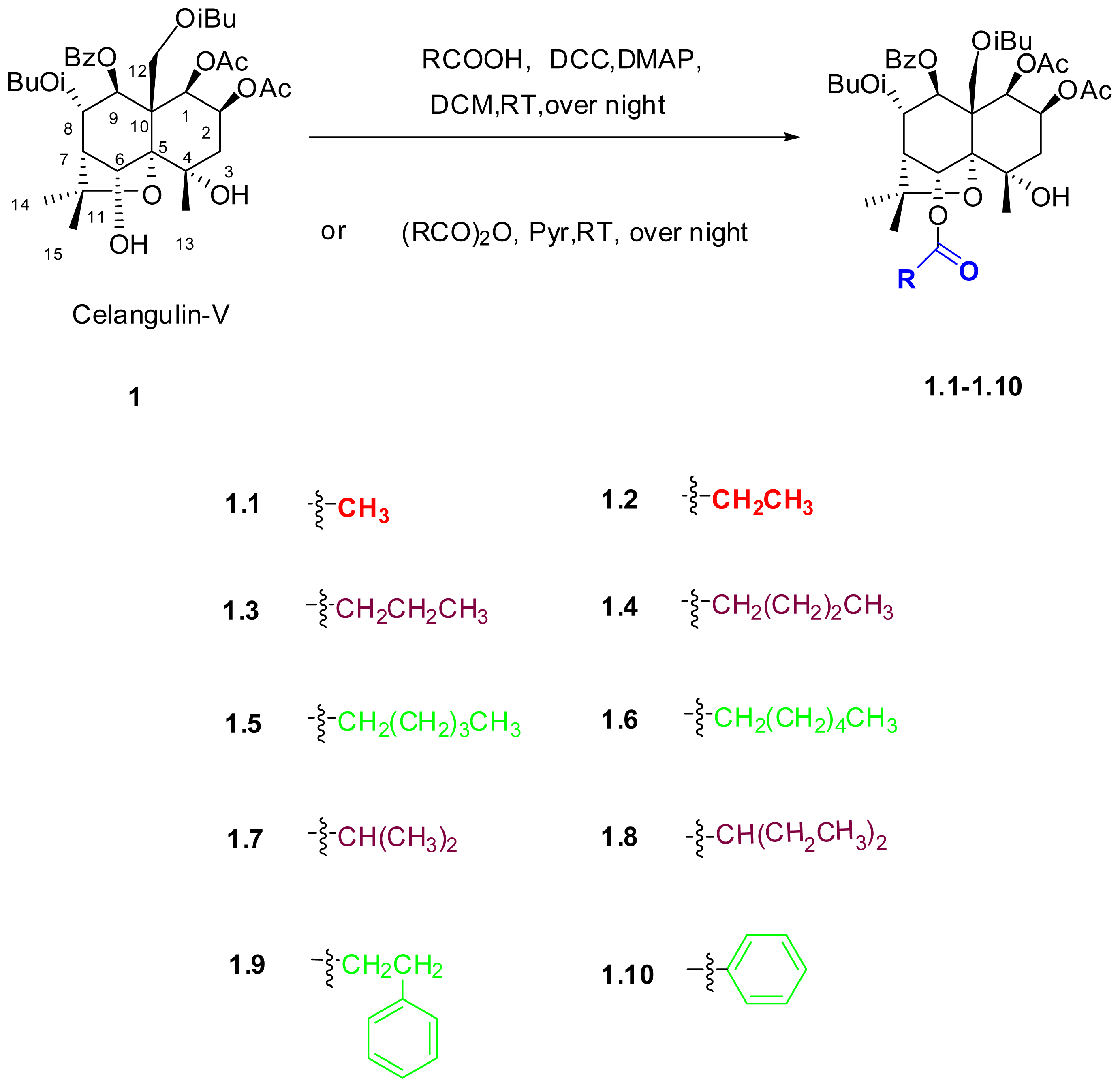
| Compounds | Molecular Formula | M.P °C | ESI-MS (M + Na) | Optical Rotation [α]°, (MeOH, c = mg.100 mL−1) |
|---|---|---|---|---|
| Celangulin-V | C34H46O13 | 198–200 | 685 | −12.2, (10.8) |
| 1.1 | C36H48O14 | 214–216 | 727 | −10.6, (16.0) |
| 1.2 | C37H50O14 | 94–96 | 741 | −10.1, (15.0) |
| 1.3 | C38H52O14 | 114–116 | 755 | −10.3, (18.5) |
| 1.4 | C39H54O14 | 102–104 | 769 | −33.0, (18.0) |
| 1.5 | C40H56O14 | 64–66 | 783 | −15.2, (15.0) |
| 1.6 | C41H58O14 | colorless oils | 797 | −26.4, (19.0) |
| 1.7 | C38H52O14 | 98–100 | 755 | −39.6, (17.0) |
| 1.8 | C40H56O14 | 112–114 | 783 | −95.8, (11.5) |
| 1.9 | C43H54O14 | 124–126 | 817 | −29.7, (12.5) |
| 1.10 | C41H06O14 | 96–98 | 789 | −51.7, (13.5) |
| No. | 1 | 1.1 | 1.2 | 1.3 | 1.4 | 1.5 | 1.6 | 1.7 | 1.8 | 1.9 | 1.10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 75.17 | 75.20 | 75.22 | 75.22 | 75.23 | 75.22 | 75.23 | 75.23 | 75.25 | 75.19 | 75.20 |
| 2 | 67.44 | 67.69 | 67.71 | 67.70 | 67.72 | 67.71 | 67.72 | 67.71 | 67.72 | 67.68 | 67.74 |
| 3 | 41.25 | 42.01 | 41.98 | 41.94 | 41.94 | 41.93 | 41.94 | 41.99 | 41.79 | 41.99 | 42.51 |
| 4 | 72.23 | 69.61 | 69.64 | 69.63 | 69.63 | 69.63 | 69.65 | 69.64 | 69.64 | 69.59 | 69.81 |
| 5 | 91.56 | 92.09 | 92.11 | 92.08 | 92.08 | 92.08 | 92.08 | 92.17 | 92.04 | 92.08 | 91.94 |
| 6 | 77.04 | 75.60 | 75.58 | 75.59 | 75.60 | 75.60 | 75.60 | 75.61 | 75.69 | 75.63 | 76.43 |
| 7 | 53.69 | 52.31 | 52.36 | 52.40 | 52.39 | 52.39 | 52.39 | 52.43 | 52.62 | 52.31 | 52.31 |
| 8 | 73.86 | 75.19 | 73.57 | 73.54 | 73.55 | 73.54 | 73.55 | 73.60 | 73.48 | 73.51 | 73.39 |
| 9 | 75.45 | 75.54 | 75.49 | 75.46 | 75.49 | 75.49 | 75.50 | 75.42 | 75.69 | 75.53 | 75.70 |
| 10 | 50.71 | 51.46 | 51.47 | 51.47 | 51.48 | 51.47 | 51.48 | 51.49 | 51.54 | 51.45 | 51.57 |
| 11 | 84.61 | 84.34 | 84.31 | 84.29 | 84.29 | 84.28 | 84.29 | 84.29 | 84.21 | 84.27 | 84.44 |
| 12 | 61.82 | 61.79 | 61.80 | 61.79 | 61.80 | 61.80 | 61.81 | 61.79 | 61.79 | 61.79 | 61.87 |
| 13 | 24.32 | 24.85 | 24.93 | 24.93 | 24.96 | 24.97 | 24.98 | 24.92 | 25.13 | 24.76 | 24.57 |
| 14 | 26.45 | 25.89 | 25.90 | 25.89 | 25.90 | 25.90 | 25.91 | 25.89 | 25.87 | 25.86 | 25.89 |
| 15 | 30.15 | 29.72 | 29.72 | 29.73 | 29.74 | 29.74 | 29.75 | 29.75 | 29.83 | 29.66 | 29.81 |
| No. | 1 | 1.1 | 1.2 | 1.3 | 1.4 | 1.5 | 1.6 | 1.7 | 1.8 | 1.9 | 1.10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 5.49 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.48 (d,1H, 3.5) | 5.49 (d,1H, 3.5) | 5.46 (d,1H, 3.5) | 5.52 (d,1H, 3.5) |
| 2 | 5.39 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.36 (m,1H) | 5.37 (m,1H) | 5.35 (m,1H) | 5.39 (m,1H) |
| 3 | 2.09 (m,1H), 2.03 (m,1H) | 2.11 (m,1H), 1.97 (m,1H) | 2.11 (m,1H), 1.96 (m,1H) | 2.12 (m,1H), 2.07 (m,1H) | 2.14 (m,1H), 2.09 (m,1H) | 2.12 (m,1H), 1.95 (m,1H) | 2.12 (m,1H), 1.95 (m,1H) | 2.13 (m,1H), 1.95 (m,1H) | 2.12 (m,1H), 1.95 (m,1H) | 2.11 (m,1H), 1.95 (m,1H) | 2.17 (m,1H), 2.02 (m,1H) |
| 6 | 5.22(s, 1H) | 6.48(s, 1H) | 6.49(s, 1H) | 6.49(s, 1H) | 6.48(s, 1H) | 6.48(s, 1H) | 6.48(s, 1H) | 6.49(s, 1H) | 6.48(s, 1H) | 6.47(s, 1H) | 6.61(s, 1H) |
| 7 | 2.58 (d,1H, 3.5) | 2.49 (d,1H, 3.5) | 2.48 (d,1H, 3.5) | 2.47 (d,1H, 3.5) | 2.47 (d,1H, 3.5) | 2.47 (d,1H, 3.5) | 2.47 (d,1H, 3.5) | 2.46 (d,1H, 3.5) | 2.43 (d,1H, 3.5) | 2.39 (d,1H, 3.5) | 2.67 (d,1H, 3.5) |
| 8 | 5.62 (dd,1H, 9.5,3.5) | 5.77 (dd,1H, 9.5,3.5) | 5.78 (dd,1H, 9.5,3.5) | 5.79 (dd,1H, 9.5,3.5) | 5.78 (dd,1H, 9.5,3.5) | 5.79 (dd,1H, 9.5,3.5) | 5.78 (dd,1H, 9.5,3.5) | 5.79 (dd,1H, 9.5,3.5) | 5.81 (dd,1H, 9.5,3.5) | 5.76 (dd,1H, 9.5,3.5) | 5.93 (dd,1H, 9.5,3.5) |
| 9 | 6.06 (d,1H, 9.5) | 6.03 (d,1H, 9.5) | 6.04 (d,1H, 9.5) | 6.04 (d,1H, 9.5) | 6.04 (d,1H, 9.5) | 6.03 (d,1H, 9.5) | 6.03 (d,1H, 9.5) | 6.04 (d,1H, 9.5) | 6.03 (d,1H, 9.5) | 6.02 (d,1H, 9.5) | 6.09 (d,1H, 9.5) |
| 12 | 4.87, 4.65 (Abq, 2H, 13.5) | 4.90, 4.65 (Abq, 2H, 13.5) | 4.90, 4.65 (Abq, 2H, 13.5) | 4.90, 4.64 (Abq, 2H, 13.5) | 4.90,4.64 (Abq,2H, 13.5) | 4.90, 4.64 (Abq, 2H, 13.5) | 4.890, 4.64 (Abq, 2H, 13.5) | 4.89, 4.65 (Abq, 2H, 13.5) | 4.91, 4.64 (Abq, 2H, 13.5) | 4.88, 4.64 (Abq, 2H, 13.5) | 4.93, 4.70 (Abq, 2H, 13.5) |
| 13 | 1.77(s, 3H) | 1.57(s, 3H) | 1.57(s, 3H) | 1.73(s, 3H) | 1.73(s, 3H) | 1.74(s, 3H) | 1.72(s, 3H) | 1.72(s, 3H) | 1.72(s, 3H) | 1.69(s, 3H) | 1.75(s, 3H) |
| 14 | 1.72(s, 3H) | 1.72(s, 3H) | 1.72(s, 3H) | 1.57(s, 3H) | 1.57(s, 3H)) | 1.56(s, 3H) | 1.56(s, 3H) | 1.58(s, 3H) | 1.58(s, 3H) | 1.47(s, 3H) | 1.59(s, 3H) |
| 15 | 1.61(s, 3H) | 1.50(s, 3H) | 1.49(s, 3H) | 1.49(s, 3H) | 1.49(s, 3H) | 1.49(s, 3H) | 1.49(s, 3H) | 1.49(s, 3H) | 1.51(s, 3H) | 1.42(s, 3H) | 1.53(s, 3H) |
| Compounds | Mortality(%) |
|---|---|
| Acetone | 0 |
| Celangulin-V | 66.7 |
| 1.1 | 75.0 |
| 1.2 | 83.3 |
| 1.3 | 16.7 |
| 1.4 | 33.3 |
| 1.5 | 0 |
| 1.6 | 0 |
| 1.7 | 8.3 |
| 1.8 | 8.3 |
| 1.9 | 0 |
| 1.10 | 0 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, J.; Hu, Z.; Li, S.; Wu, W. Synthesis and Insecticidal Activities of New Ester-Derivatives of Celangulin-V. Int. J. Mol. Sci. 2011, 12, 9596-9604. https://doi.org/10.3390/ijms12129596
Zhang J, Hu Z, Li S, Wu W. Synthesis and Insecticidal Activities of New Ester-Derivatives of Celangulin-V. International Journal of Molecular Sciences. 2011; 12(12):9596-9604. https://doi.org/10.3390/ijms12129596
Chicago/Turabian StyleZhang, Jiwen, Zhan Hu, Shenkun Li, and Wenjun Wu. 2011. "Synthesis and Insecticidal Activities of New Ester-Derivatives of Celangulin-V" International Journal of Molecular Sciences 12, no. 12: 9596-9604. https://doi.org/10.3390/ijms12129596




