Modeling Natural Anti-Inflammatory Compounds by Molecular Topology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Analyzed Compounds
2.2. Molecular Descriptors
2.3. Modeling Techniques
- TP (True positive): Active compounds correctly classified or predicted.
- FP (False positive): Inactive compounds classified as anti-inflammatory.
- TN (True negative): Inactive compounds correctly classified.
- FN (False negative): Anti-inflammatory compounds classified as inactive.
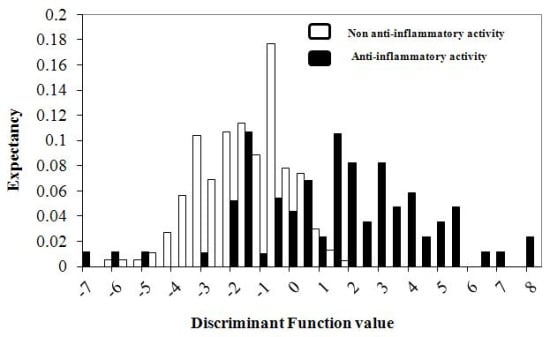
2.4. Pharmacological-Activity Distribution Diagrams
2.5. Topological Virtual Screening
3. Results and Discussion
3.1. Similarity Study
3.2. Mathematical Modeling
3.3. Topological Virtual Screening
4. Conclusions
Supplementary Material
ijms-12-09481-s001.pdfAcknowledgments
References
- Gautam, R.; Jachak, S.M. Recent developments in anti-inflammatory natural products. Med. Res. Rev 2009, 29, 767–820. [Google Scholar]
- Butler, M.S. Natural products to drugs: Natural product-derived compounds in clinical trials. Nat. Prod. Rep 2008, 25, 475–516. [Google Scholar]
- Chin, Y.W.; Balunas, M.J.; Chai, H.B.; Kinghorn, A.D. Drug discovery from natural sources. AAPS J 2006, 8, 239–253. [Google Scholar]
- Paterson, I.; Anderson, E.A. The renaissance of natural products as drug candidates. Science 2005, 310, 451–453. [Google Scholar]
- Lam, K.S. New aspects of natural products in drug discovery. Trends Microbiol 2007, 15, 279–289. [Google Scholar]
- Wilson, R.M.; Danishefsky, S.J. Small molecule natural products in the discovery of therapeutic agents: The synthesis connection. J. Org. Chem 2006, 71, 8329–8351. [Google Scholar]
- Bohlin, L.; Göransson, U.; Alsmark, C.; Wedén, C.; Backlund, A. Natural products in modern life science. Phytochem. Rev 2010, 9, 279–301. [Google Scholar]
- Galvez, J.; Villar, M.; Galvez-Llompart, M.; Amigo, M. Chemistry explained by topology: An alternative approach. Comb. Chem. High Throughput Screen 2011, 14, 279–283. [Google Scholar]
- García-Domenech, R.; Gálvez, J.; de Julián-Ortiz, J.V.; Pogliani, L. Some new trends in chemical graph theory. Chem. Rev 2008, 108, 1127–1169. [Google Scholar]
- Horrobin, D.F. Innovation in the pharmaceutical industry. J. R. Soc. Med 2000, 93, 341–345. [Google Scholar]
- Galvez-Llompart, M.; Giner, M.; Recio, C.; Candeletti, S.; Garcia-Domenech, R. Application of molecular topology to the search of novel NSAIDs: Experimental validation of activity. Lett. Drug Des. Discov 2010, 7, 438–445. [Google Scholar]
- Pla-Franco, J.; Gálvez-Llompart, M.; Gálvez, J.; García-Domenech, R. Application of molecular topology for the prediction of reaction yields and anti-inflammatory activity of heterocyclic amidine derivatives. Int. J. Mol. Sci 2011, 12, 1281–1292. [Google Scholar]
- Marrero-Ponce, Y.; Siverio-Mota, D.; Gálvez-Llompart, M.; Recio, M.C.; Giner, R.M.; García-Domènech, R.; Torrens, F.; Arán, V.J.; Cordero-Maldonado, M.L.; Esguera, C.V.; et al. Discovery of novel anti-inflammatory drug-like compounds by aligning in silico and in vivo screening: The nitroindazolinone chemotype. Eur. J. Med. Chem 2011, 46, 5736–5753. [Google Scholar]
- Kontogiorgis, C.; Bompou, E.M.; Ntella, M.; Berghe, W.V. Natural products from mediterranean diet: From anti-inflammatory agents to dietary epigenetic modulators. Anti Inflamm. Anti Allergy Agents Med. Chem 2010, 9, 101–124. [Google Scholar]
- MicroSource Discovery Systems. Available online: http://www.msdiscovery.com/home.html accessed on 14 December 2011.
- ChemDraw, version Ultra 10.0; CambridgeSoft: Cambridge, MA, USA, 2006.
- Dragon Software, version 5.4-2006; Talete, SRL: Milan, Italy, 2006.
- Statistica (data analysis software system), version 9.0; StatSoft, I: Tulsa, OK, USA, 2009.
- Matthews, B.W. Comparison of the predicted and observed secondary structure of T4 phage lysozyme. Biochim. Biophys. Acta 1975, 405, 442–451. [Google Scholar]
- Gálvez, J.; García-Domenech, R.; de Gregorio Alapont, C.; de Julián-Ortiz, J.; Popa, L. Pharmacological distribution diagrams: A tool for de novo drug design. J. Mol. Graph 1996, 5, 272–276. [Google Scholar]
- Mohar, B.; Babic, D.; Trinajstic, N. A novel definition of the Wiener index for trees. J. Chem. Inf. Comput. Sci 1993, 33, 153–154. [Google Scholar]
- Broto, P.; Devillers, J. Autocorrelation of Properties Distributed on Molecular Graphs. In Practical Application of Quantitative Structure-Activity Relationship (QSAR) in Environmental Chemistry and Toxicology; Karcher, W., Devillers, J., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1990; pp. 105–127. [Google Scholar]
- Moreau, G.; Broto, P. Autocorrelation of molecular structures, application to SAR studies. Nouv. J. Chim 1980, 4, 757–764. [Google Scholar]
- Won, S.J.; Liu, C.T.; Tsao, L.T.; Weng, J.R.; Ko, H.H.; Wang, J.P.; Lin, C.N. Synthetic chalcones as potential anti-inflammatory and cancer chemopreventive agents. Eur. J. Med. Chem 2005, 40, 103–112. [Google Scholar]
- Tewtrakul, S.; Subhadhirasakul, S.; Karalai, C.; Ponglimanont, C.; Cheenpracha, S. Anti-inflammatory effects of compounds from Kaempferia parviflora and Boesenbergia pandurata. Food Chem 2009, 115, 534–538. [Google Scholar]
- Witaicenis, A.; Seito, L.N.; di Stasi, L.C. Intestinal anti-inflammatory activity of esculetin and 4-methylesculetin in the trinitrobenzenesulphonic acid model of rat colitis. Chem. Biol. Interact 2010, 186, 211–218. [Google Scholar]
- Ruan, Q.; Zhang, W.; Hufnagl, P.; Kaun, C.; Binder, B.R.; Wojta, J. Anisodamine counteracts lipopolysaccharide-induced tissue factor and plasminogen activator inhibitor-1 expression in human endothelial cells: Contribution of the NF-kB pathway. J. Vasc. Res 2001, 38, 13–19. [Google Scholar]
- Nunomura, R.C.S.; Oliveira, V.G.; Silvab, S.; Nunomuraa, S.M. Characterization of bergenin in Endopleura uchi bark and its anti-inflammatory activity. J. Braz. Chem. Soc 2009, 20, 1060–1064. [Google Scholar]
- Nazir, N.; Koul, S.; Qurishi, M.A.; Taneja, S.C.; Ahmad, S.F.; Bani, S.; Qazi, G.N. Immunomodulatory effect of bergenin and norbergenin against adjuvant-induced arthritis—A flow cytometric study. J. Ethnopharmacol 2007, 112, 401–405. [Google Scholar]
- Lourens, A.; Reddy, D.; Baser, K.; Viljoen, A.; van Vuuren, S. In vitro biological activity and essential oil composition of four indigenous South African Helichrysum species. J. Ethnopharmacol 2004, 95, 253–258. [Google Scholar]
- Hsu, D.Z.; Liu, M.Y. Bicuculline methiodide attenuates hepatic injury and decreases mortality in septic rats: Role of cytokines. Shock 2004, 22, 347–350. [Google Scholar]
- Dai, S.J.; Ren, Y.; Shen, L.; Zhang, D.W. New alkaloids from Forsythia suspensa and their anti-inflammatory activities. Planta Med 2009, 75, 375–377. [Google Scholar]
- Konrath, E.L.; Santin, K.; Nassif, M.; Latini, A.; Henriques, A.; Salbego, C. Antioxidant and pro-oxidant properties of boldine on hippocampal slices exposed to oxygen-glucose deprivation in vitro. Neurotoxicology 2008, 29, 1136–1140. [Google Scholar]
- O’Brien, P.; Carrasco-Pozo, C.; Speisky, H. Boldine and its antioxidant or health-promoting properties. Chem. Biol. Interact 2006, 159, 1–17. [Google Scholar]
- Sasaki, Y.; Hosokawa, T.; Nagai, M.; Nagumo, S. In vitro study for inhibition of NO production about constituents of Sappan Lignum. Biol. Pharm. Bull 2007, 30, 193–196. [Google Scholar]
- Shen, J.; Zhang, H.; Lin, H.; Su, H.; Xing, D.; Du, L. Brazilein protects the brain against focal cerebral ischemia reperfusion injury correlating to inflammatory response suppression. Eur. J. Pharmacol 2007, 558, 88–95. [Google Scholar]
- Cashman, J.; Burt, H.; Springate, C.; Gleave, J.; Jackson, J. Camptothecin-loaded films for the prevention of postsurgical adhesions. Inflamm. Res 2004, 53, 355–362. [Google Scholar]
- Wall, M.E.; Wani, M.; Cook, C.; Palmer, K.; McPhail, A.; Sim, G. The isolation and structure of camptothecin, a novel alkaloidal leukemia and tumor inhibitor from Camptotheca acuminata. J. Am. Chem. Soc 1966, 88, 3888–3890. [Google Scholar]
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev 2010, 29, 405–434. [Google Scholar]
- Luo, X.J.; Peng, J.; Li, Y.J. Recent advances in the study on capsaicinoids and capsinoids. Eur. J. Pharmacol 2010, 650, 1–7. [Google Scholar]
- Mengoni, E.S.; Vichera, G.; Rigano, L.A.; Rodriguez-Puebla, M.L.; Galliano, S.R.; Cafferata, E.E.; Pivetta, O.H.; Moreno, S.; Vojnov, A.A. Suppression of COX-2, IL-1[beta] and TNF-[alpha] expression and leukocyte infiltration in inflamed skin by bioactive compounds from Rosmarinus officinalis L. Fitoterapia 2011, 82, 414–421. [Google Scholar]
- Trinh, H.T.; Chae, S.J.; Joh, E.H.; Son, K.H.; Jeon, S.J.; Kim, D.H. Tanshinones isolated from the rhizome of Salvia miltiorrhiza inhibit passive cutaneous anaphylaxis reaction in mice. J. Ethnopharmacol 2010, 132, 344–348. [Google Scholar]
- Wang, M.; Dai, H.; Li, X.; Li, Y.; Wang, L.; Xue, M. Structural elucidation of metabolites of tanshinone I and its analogue dihydrotanshinone I in rats by HPLC-ESI-MSn. J. Chromatogr. B 2010, 878, 915–924. [Google Scholar]
- Singh, K.; Jaggi, A.S.; Singh, N. Exploring the ameliorative potential of Punica granatum in dextran sulfate sodium induced ulcerative colitis in mice. Phytother. Res 2009, 23, 1565–1574. [Google Scholar]
- Ogawa, Y.; Kanatsu, K.; Iino, T.; Kato, S.; Jeong, Y.; Shibata, N.; Takada, K.; Takeuchi, K. Protection against dextran sulfate sodium-induced colitis by microspheres of ellagic acid in rats. Life Sci 2002, 71, 827–839. [Google Scholar]
- Min, K.R.A.K.; Wang, B.Y.; Lim, H.S.; Kang, B.S.; Oh, G.J.; Lee, J.; Kang, S.H.; Lee, K.S.; Ro, J.S.; Kim, Y. (−)-Epiafzelechin: Cyclooxygenase-1 inhibitor and anti-inflammatory agent from aerial parts of Celastrus orbiculatus. Planta Med. 1999, 65, 460–462. [Google Scholar]
- Yasukawa, K.; Akihisa, T.; Yoshida, Z.; Takido, M. Inhibitory effect of euphol, a triterpene alcohol from the roots of Euphorbia kansui, on tumour Promotion by 12-Otetradecanoylphorbol- 13-acetate in two-stage carcinogenesis in mouse skin. J. Pharm. Pharmacol 2000, 52, 119–124. [Google Scholar]
- Kroes, B.; Van den Berg, A.; Van Ufford, H.C.Q.; van Dijk, H.; Labadie, R. Anti-inflammatory activity of gallic acid. Planta Med 1992, 58, 499–499. [Google Scholar]
- Németh, Z.H.; Deitch, E.A.; Szabó, C.; Mabley, J.G.; Pacher, P.; Fekete, Z.; Hauser, C.J.; Haskó, G. Na/H exchanger blockade inhibits enterocyte inflammatory response and protects against colitis. Am. J. Physiol. Gastrointest. Liver Physiol 2002, 283, G122–G132. [Google Scholar]
- Choi, J.; Jeong, T.; Kim, D.; Kim, Y.; Na, H.; Nam, K.; Lee, S.; Kim, H.; Oh, S.R.; Choi, Y.; et al. Hematein inhibits atherosclerosis by inhibition of reactive oxygen generation and NF-κB-dependent inflammatory mediators in hyperlipidemic mice. J. Cardiovasc. Pharmacol 2003, 42, 287–295. [Google Scholar]
- Chandrasekaran, C.; Deepak, H.; Thiyagarajan, P.; Kathiresan, S.; Sangli, G.K.; Deepak, M.; Agarwal, A. Dual Inhibitory Effect of Glycyrrhiza Glabra (GutGard (TM)) on COX and LOX Products; Urban & Fischer Verlag: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Geraets, L.; Moonen, H.J.J.; Brauers, K.; Wouters, E.F.M.; Bast, A.; Hageman, G.J. Dietary flavones and flavonoles are inhibitors of poly (ADP-ribose) polymerase-1 in pulmonary epithelial cells. J. Nutr 2007, 137, 2190–2195. [Google Scholar]
- Ahn, K.S.; Noh, E.J.; Cha, K.H.; Kim, Y.S.; Lim, S.S.; Shin, K.H.; Jung, S.H. Inhibitory effects of Irigenin from the rhizomes of Belamcanda chinensis on nitric oxide and prostaglandin E2 production in murine macrophage RAW 264.7 cells. Life Sci 2006, 78, 2336–2342. [Google Scholar]
- Lee, S.; Kim, J.; Seo, G.; Kim, Y.C.; Sohn, D. Isoliquiritigenin, from Dalbergia odorifera, up-regulates anti-inflammatory heme oxygenase-1 expression in RAW264.7 macrophages. Inflamm. Res 2009, 58, 257–262. [Google Scholar]
- Chan, S.C.; Chang, Y.S.; Wang, J.P.; Chen, S.C.; Kuo, S.C. Three new flavonoids and antiallergic anti-inflammatory constituents from the heartwood of Dalbergia odorifera. Planta Med 1998, 64, 153–158. [Google Scholar]
- Maes, M.; Mihaylova, I.; Ruyter, M.D.; Kubera, M.; Bosmans, E. The immune effects of TRYCATs (tryptophan catabolites along the IDO pathway): Relevance for depression—And other conditions characterized by tryptophan depletion induced by inflammation. Neuroendocrinol. Lett 2007, 28, 826–831. [Google Scholar]
- Alia, B.; Bashir, A.; Tanira, M. Anti-inflammatory, antipyretic, and analgesic effects of Lawsonia inermis L. (henna) in rats. Pharmacology 1995, 51, 356–363. [Google Scholar]
- Yadav, V.R.; Prasad, S.; Sung, B.; Kannappan, R.; Aggarwal, B.B. Targeting inflammatory pathways by triterpenoids for prevention and treatment of cancer. Toxins 2010, 2, 2428–2466. [Google Scholar]
- Du, H.; Matsushima, T.; Spyvee, M.; Goto, M.; Shirota, H.; Gusovsky, F.; Chiba, K.; Kotake, M.; Yoneda, N.; Eguchi, Y. Discovery of a potent, metabolically stabilized resorcylic lactone as an anti-inflammatory lead. Bioorg. Med. Chem. Lett 2009, 19, 6196–6199. [Google Scholar]
- Shen, Y.; Du, H.; Kotake, M.; Matsushima, T.; Goto, M.; Shirota, H.; Gusovsky, F.; Li, X.; Jiang, Y.; Schiller, S. Discovery of an in vitro and in vivo potent resorcylic lactone analog of LL-Z1640-2 as anti-inflammatory lead, II. Bioorg. Med. Chem. Lett 2010, 20, 3047–3049. [Google Scholar]
- Kubo, K. In studies on N-cyclohexylanthranilic acid analogs. I. synthesis and analgesic activity. Chem. Abstr 1978, 88, 570. [Google Scholar]
- Shah, V.O.; Ferguson, J.; Hunsaker, L.A.; Deck, L.M.; Vander Jagt, D.L. Cardiac glycosides inhibit LPS-induced activation of pro-inflammatory cytokines in whole blood through an NF-κB-dependent mechanism. Int. J. Appl. Res. Nat. Prod 2011, 4, 11–19. [Google Scholar]
- Phitak, T.; Choocheep, K.; Pothacharoen, P.; Pompimon, W.; Premanode, B.; Kongtawelert, P. The effects of p-hydroxycinnamaldehyde from Alpinia galanga extracts on human chondrocytes. Phytochemistry 2009, 70, 237–243. [Google Scholar]
- Morikawa, T.; Ando, S.; Matsuda, H.; Kataoka, S.; Muraoka, O.; Yoshikawa, M. Inhibitors of nitric oxide production from the rhizomes of Alpinia galanga: Structures of new 8–9′ linked neolignans and sesquineolignan. Chem. Pharm. Bull 2005, 53, 625–630. [Google Scholar]
- Madhukar, M.; Sawraj, S.; Sharma, P.D. Design, synthesis and evaluation of mutual prodrug of 4-biphenylacetic acid and quercetin tetramethyl ether (BPA-QTME) as gastrosparing NSAID. Eur. J. Med. Chem 2010, 45, 2591–2596. [Google Scholar]
- Al-Harbi, M.M.; Qureshi, S.; Ahmed, M.M.; Raza, M.; Miana, G.A.; Shah, A.H. Studies on the antiinflammatory, antipyretic and analgesic activities of santonin. Jpn. J. Pharmacol 1994, 64, 135–139. [Google Scholar]
- Wagoner, J.; Morishima, C.; Graf, T.N.; Oberlies, N.H.; Teissier, E.; Pécheur, E.I.; Tavis, J.E.; Polyak, S.J. Differential in vitro effects of intravenous versus oral formulations of silibinin on the HCV life cycle and inflammation. PLoS One 2011, 6. [Google Scholar] [CrossRef]
- Bannwart, C.F.; Peracoli, J.C.; Nakaira-Takahagi, E.; Peracoli, M.T. Inhibitory effect of silibinin on tumour necrosis factor-alpha and hydrogen peroxide production by human monocytes. Nat. Prod. Res 2010, 24, 1747–1757. [Google Scholar]
- Hou, Y.C.; Liou, K.T.; Chern, C.M.; Wang, Y.H.; Liao, J.F.; Chang, S.; Chou, Y.H.; Shen, Y.C. Preventive effect of silymarin in cerebral ischemia-reperfusion-induced brain injury in rats possibly through impairing NF-[kappa] B and STAT-1 activation. Phytomedicine 2010, 17, 963–973. [Google Scholar]
- Pan, C.H.; Kim, E.S.; Jung, S.H.; Nho, C.W.; Lee, J.K. Tectorigenin inhibits IFN-γ/LPS-induced inflammatory responses in murine macrophage RAW 264.7 cells. Arch. Pharm. Res 2008, 31, 1447–1456. [Google Scholar]
- Kim, Y.P.; Yamada, M.; Lim, S.S.; Lee, S.H.; Ryu, N.; Shin, K.H.; Ohuchi, K. Inhibition by tectorigenin and tectoridin of prostaglandin E2 production and cyclooxygenase-2 induction in rat peritoneal macrophages. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1999, 1438, 399–407. [Google Scholar]
- Marquez-Martin, A.; Puerta, R.D.L.; Fernandez-Arche, A.; Ruiz-Gutierrez, V.; Yaqoob, P. Modulation of cytokine secretion by pentacyclic triterpenes from olive pomace oil in human mononuclear cells. Cytokine 2006, 36, 211–217. [Google Scholar]
- Öztürk, A.; Özbek, H. The anti-inflammatory activity of Eugenia caryophyllata essential oil: An animal model of anti-inflammatory activity. Eur. J. Gen. Med 2005, 2, 159–163. [Google Scholar]
- Zou, J.; Jin, D.; Chen, W.; Wang, J.; Liu, Q.; Zhu, X.; Zhao, W. Selective cyclooxygenase-2 inhibitors from Calophyllum membranaceum. J. Nat. Prod 2005, 68, 1514–1518. [Google Scholar]
- PubChem Bioassay. Available online: http://pubchem.ncbi.nlm.nih.gov/assay/assay.cgi?cid=13650 accessed on 27 October 2011.
- Mencherini, T.; Cau, A.; Bianco, G.; Loggia, R.D.; Aquino, R. An extract of Apium graveolens var. dulce leaves: Structure of the major constituent, apiin, and its anti-inflammatory properties. J. Pharm. Pharmacol 2007, 59, 891–897. [Google Scholar]
- Ocana, A.; Gomez-Asensio, C.; Arranz-Gutierrez, E.; Torres, C.; Senorans, F.J.; Reglero, G. In vitro study of the effect of diesterified alkoxyglycerols with conjugated linoleic acid on adipocyte inflammatory mediators. Lipids Health. Dis 2010, 9. [Google Scholar] [CrossRef]
- Matsui, J.; Ikemoto, T.; Yoshida, M.; Kakishima, H. Anti-inflammatory skin-lightening cosmetics containing (iso)bergaptol. JP Patent 2001114663, 24 April 2001. [Google Scholar]
- Reddy, M.K.; Alexander-Lindo, R.L.; Nair, M.G. Relative inhibition of lipid peroxidation, cyclooxygenase enzymes, and human tumor cell proliferation by natural food colors. J. Agric. Food Chem 2005, 53, 9268–9273. [Google Scholar]
- Washiyama, M.; Sasaki, Y.; Hosokawa, T.; Nagumo, S. Anti-inflammatory constituents of Sappan lignum. Biol. Pharm. Bull 2009, 32, 941–944. [Google Scholar]
- Horie, S.; Okuda, C.; Yamashita, T.; Watanabe, K.; Kuramochi, K.; Hosokawa, M.; Takeuchi, T.; Kakuda, M.; Miyashita, K.; Sugawara, F. Purified canola lutein selectively inhibits specific isoforms of mammalian DNA polymerases and reduces inflammatory response. Lipids 2010, 45, 713–721. [Google Scholar]
- Fernandes, E.S.; Passos, G.F.; Medeiros, R.; da Cunha, F.M.; Ferreira, J.; Campos, M.M.; Pianowski, L.F.; Calixto, J. Anti-inflammatory effects of compounds alpha-humulene and (−)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur. J. Pharmacol 2007, 569, 228–236. [Google Scholar]
- Mazura, M.; Susanti, D.; Rasadah, M. Anti-inflammatory action of components from Melastoma malabathricum. Pharm. Biol 2007, 45, 372–375. [Google Scholar]
- Lima, J.; Oliveira, A.; Miranda, A.L.P.; Rezende, C.; Pinto, A. Anti-inflammatory and antinociceptive activities of an acid fraction of the seeds of Carpotroche brasiliensis (Raddi)(Flacourtiaceae). Braz. J. Med. Biol. Res 2005, 38, 1095–1103. [Google Scholar]
- Li, X.; Yang, Y.B.; Yang, Q.; Sun, L.N.; Chen, W.S. Anti-Inflammatory and analgesic activities of Chaenomeles speciosa fractions in laboratory animals. J. Med. Food 2009, 12, 1016–1022. [Google Scholar]
- Bastos, V.P.D.; Gomes, A.S.; Lima, F.J.B.; Brito, T.S.; Soares, P.M.G.; Pinho, J.P.M.; Silva, C.S.; Santos, A.A.; Souza, M.H.L.P.; Magalhães, P.J.C. Inhaled 1,8-cineole reduces inflammatory parameters in airways of ovalbumin-challenged Guinea pigs. Basic Clin. Pharmacol. Toxicol 2010, 108, 34–39. [Google Scholar]
- Park, K.Y.; Lee, S.; Min, B.; Lee, K.S.; Choi, J.; Chung, S.R.; Min, K.R.; Kim, Y. Inhibitory effect of luteolin 4′-O-glucoside from Kummerowia striata and other flavonoids on interleukin-5 bioactivity. Planta Med 1999, 65, 457–459. [Google Scholar]
- Rehani, K.; Scott, D.A.; Renaud, D.; Hamza, H.; Williams, L.R.; Wang, H.; Martin, M. Cotinine-induced convergence of the cholinergic and PI3 kinase-dependent anti-inflammatory pathways in innate immune cells. Biochim. Biophys. Acta Mol. Cell Res 2008, 1783, 375–382. [Google Scholar]
- Kalayarasan, S.; Sriram, N.; Sudhandiran, G. Diallyl sulfide attenuates bleomycin-induced pulmonary fibrosis: Critical role of iNOS, NF-[kappa] B, TNF-[alpha] and IL-1[beta]. Life Sci 2008, 82, 1142–1153. [Google Scholar]
- Marinier, B. Unsymmetrical derivatives of djenkolic acid. U.S. Patent 3448142(A), 19 April 1969. [Google Scholar]
- Del-Vechio-Vieira, G.; Sousa, O.V.; Miranda, M.A.; Senna-Valle, L.; Kaplan, M.A.C. Analgesic and anti-inflammatory properties of essential oil from Ageratum fastigiatum. Braz. Arch. Biol. Techn 2009, 52, 1115–1121. [Google Scholar]
- Rai, S.K.; Sharma, M.; Tiwari, M. Inhibitory effect of novel diallyldisulfide analogs on HMG-CoA reductase expression in hypercholesterolemic rats: CREB as a potential upstream target. Life Sci 2009, 85, 211–219. [Google Scholar]
- Koehler, A.N. Methods for modulating NF-κB using gibberellins. U.S. Patent WO 2010062372, 3 June 2010. [Google Scholar]
- Nakamichi, K.; Nakano, T.; Yasuura, H.; Izumi, S.; Kawashima, Y. Stabilization of sodium guaiazulene sulfonate in granules for tableting prepared using a twin-screw extruder. Eur. J. Pharm. Biopharm 2003, 56, 347–354. [Google Scholar]
- Hamsa, T.; Kuttan, G. Harmine inhibits tumour specific neo vessel formation by regulating VEGF, MMP, TIMP and pro inflammatory mediators both in vivo and in vitro. Eur. J. Pharmacol 2010, 15, 64–73. [Google Scholar]
- Yamazaki, Y.; Kawano, Y. Inhibitory effects of herbal alkaloids on the tumor necrosis factor-α and nitric oxide production in lipopolysaccharide-stimulated RAW264 macrophages. Chem. Pharm. Bull 2011, 59, 388–391. [Google Scholar]
- Lanhers, M.C.; Fleurentin, J.; Mortier, F.; Vinche, A.; Younos, C. Anti-inflammatory and analgesic effects of an aqueous extract of Harpagophytum procumbens. Planta Med 1992, 58, 117–123. [Google Scholar]
- Rao, K. Screening of anti-inflammatory and hepatoprotective activities of alantolactone, isolated from the roots of Inula racemosa. Indian Drugs 1997, 34, 571–575. [Google Scholar]
- Kang, S.R.; Park, K.I.; Park, H.S.; Lee, D.H.; Kim, J.A.; Nagappan, A.; Kim, E.H.; Lee, W.S.; Shin, S.C.; Park, M.K. Anti-inflammatory effect of flavonoids isolated from Korea Citrus Aurantium L. on lipopolysaccharide-induced mouse macrophage RAW 264.7 cells by blocking of nuclear factor-kappa B (NF-[kappa]B) and mitogen-activated protein kinase (MAPK) signaling pathways. Food Chem 2011, 129, 1721–1728. [Google Scholar]
- Byeon, S.E.; Lee, Y.G.; Kim, J.; Han, J.G.; Lee, H.Y.; Cho, J.Y. Hinokitiol, a natural tropolone derivative, inhibits TNF-alpha production in LPS-activated macrophages via suppression of NF-kappa B. Planta Med 2008, 74, 828–833. [Google Scholar]
- Kim, E.J.; Park, H.; Kim, J.; Park, J.H.Y. 3,3′-diindolylmethane suppresses 12-O-tetradecanoylphorbol-13-acetate-induced inflammation and tumor promotion in mouse skin via the downregulation of inflammatory mediators. Mol. Carcinog 2010, 49, 672–683. [Google Scholar]
- Claxson, A.; Morris, C.; Blake, D.; Siren, M.; Halliwell, B.; Gustafsson, T.; Löfkvist, B.; Bergelin, I. The anti-inflammatory effects of D-myo-inositol-1,2,6-trisphosphate (PP56) on animal models of inflammation. Inflamm. Res 1990, 29, 68–70. [Google Scholar]
- Maruo, S.; Kuriyama, I.; Kuramochi, K.; Tsubaki, K.; Yoshida, H.; Mizushina, Y. Inhibitory effect of novel 5-O-acyl juglones on mammalian DNA polymerase activity, cancer cell growth and inflammatory response. Bioorg. Med. Chem 2011, 19, 5803–5812. [Google Scholar]
- Keiss, H.P.; Dirsch, V.M.; Hartung, T.; Haffner, T.; Trueman, L.; Auger, J.; Kahane, R.; Vollmar, A.M. Garlic (Allium sativum L.) modulates cytokine expression in lipopolysaccharide-activated human blood thereby inhibiting NF-kappaB activity. J. Nutr 2003, 133, 2171–2175. [Google Scholar]
- Orhan, I.; Kupeli, E.; Sener, B.; Yesilada, E. Appraisal of anti-inflammatory potential of the clubmoss, Lycopodium clavatum L. J. Ethnopharmacol 2007, 109, 146–150. [Google Scholar]
- Kawamura, F.; Nakanishi, M.; Hirashima, N. Effects of menadione, a reactive oxygen generator, on leukotriene secretion from RBL-2H3 cells. Biol. Pharm. Bull 2010, 33, 881–885. [Google Scholar]
- Juergens, U.R.; Stober, M.; Vetter, H. The anti-inflammatory activity of L-menthol compared to mint oil in human monocytes in vitro: A novel perspective for its therapeutic use in inflammatory diseases. Eur. J. Med. Res 1998, 3, 539–545. [Google Scholar]
- Kawata, J.; Kameda, M.; Miyazawa, M. Cyclooxygenase-2 inhibitory effects of monoterpenoids with a p-methane skeleton. Int. J. Essent. Oil Ther 2008, 2, 145–148. [Google Scholar]
- Frydas, S.; Papaioannou, N.; Papazahariadou, M.; Hatzistilianou, M.; Karagouni, E.; Trakatelli, M.; Brellou, G.; Petrarca, C.; Castellani, M.L.; Conti, P.; et al. Inhibition of MCP-1 and MIP-2 chemokines in murine trichinellosis: Effect of the anti-inflammatory compound L-mimosine. Int. J. Immunopathol. Pharmacol 2005, 18, 85–94. [Google Scholar]
- Luft, P.; Oostingh, G.J.; Gruijthuijsen, Y.; Horejs-Hoeck, J.; Lehmann, I.; Duschl, A. Patulin influences the expression of Th1/Th2 cytokines by activated peripheral blood mononuclear cells and T cells through depletion of intracellular glutathione. Environ. Toxicol 2008, 23, 84–95. [Google Scholar]
- Lim, H.; Son, K.H.; Chang, H.W.; Bae, K.H.; Kang, S.S.; Kim, H.P. Anti-inflammatory activity of Pectolinarigenin and Pectolinarin isolated from Cirsium chanroenicum. Biol. Pharm. Bull 2008, 31, 2063–2067. [Google Scholar]
- Gemmell, D.K.; Cottney, J.; Lewis, A.J. Comparative effects of drugs on four paw oedema models in the rat. Inflamm. Res 1979, 9, 107–116. [Google Scholar]
- Bae, G.S.; Kim, M.S.; Jung, W.S.; Seo, S.W.; Yun, S.W.; Kim, S.G.; Park, R.K.; Kim, E.C.; Song, H.J.; Park, S.J. Inhibition of lipopolysaccharide-induced inflammatory responses by piperine. Eur. J. Pharmacol 2010, 642, 154–162. [Google Scholar]
- Luo, P.; Wong, Y.F.; Ge, L.; Zhang, Z.F.; Liu, Y.; Liu, L.; Zhou, H. Anti-inflammatory and analgesic effect of plumbagin through inhibition of nuclear factor-κB activation. J. Pharmacol. Exp. Ther 2010, 335, 735–742. [Google Scholar]
- Kim, J.-Y.; Lee, W.-K.; Yu, Y.G.; Kim, J.-H. Blockade of LTB4-induced chemotaxis by bioactive molecules interfering with the BLT2-Gαi interaction. Biochem. Pharmacol 2010, 79, 1506–1515. [Google Scholar]
- Hong, E.S.; Ahn, G.U.; Cho, B.G. Cosmetics containing rhapontin. KR Patent 2011013813, 10 February 2011. [Google Scholar]
- Takahashi, A.; Nakata, K. Anti-inflammatory effects of peppermint. Aromatopia 1995, 13, 42–45. [Google Scholar]
- Arjumand, W.; Seth, A.; Sultana, S. Rutin attenuates cisplatin induced renal inflammation and apoptosis by reducing NFkappaB, TNF-alpha and caspase-3 expression in wistar rats. Food Chem. Toxicol 2011, 49, 2013–2021. [Google Scholar]
- Yan, W.; Guo, Y.; Xu, Z.; Huang, R. Vacuum distillation preparation and pharmaceutical application of Asarum heterotropoides volatile oil. CN Patent 101530442, 16 September 2009. [Google Scholar]
- Jin, M.H.; Bae, K.; Chang, H.W.; Son, J.K. Anti-inflammatory compounds from the leaves of Ailanthus altissima. Biomol. Ther.(Seoul. ) 2009, 32, 86–91. [Google Scholar]
- Xing, J.; Sun, J.; Dong, Y.; Qu, Q.; Yuan, Z.; Hu, S.; Shi, R. Application of shikimic acid to prepare medicines for treating ulcerative colitis. CN Patent 101352427, 28 January 2009. [Google Scholar]
- Xing, J.-F.; Sun, J.-N.; Hou, J.-Y.; Guo, Y.-J.; Wang, X.-K.; Zheng, X.-H.; Guo, R.-T. Antiinflammatory effects of 3,4-oxo-isopropylidene-shikimic acid. Zhongguo Yaoxue Zazhi 2006, 41, 1861–1863. [Google Scholar]
- El Gazzar, M.; El Mezayen, R.; Marecki, J.C.; Nicolls, M.R.; Canastar, A.; Dreskin, S.C. Anti-inflammatory effect of thymoquinone in a mouse model of allergic lung inflammation. Int. Immunopharmacol 2006, 6, 1135–1142. [Google Scholar]
- Ganguly, K.; Sharma, A.V.; Reiter, R.J.; Swarnakar, S. Melatonin promotes angiogenesis during protection and healing of indomethacin-induced gastric ulcer: Role of matrix metalloproteinase-2. J. Pineal Res 2010, 49, 130–140. [Google Scholar]



| Symbol | Name | Ref. |
|---|---|---|
| TI1 | first Mohar index | [21] |
| ATS7m | 2D Broto-Moreau autocorrelation of a topological structure-lag7/weighted by atomic masses | [22] |
| ATS4v | 2D Broto-Moreau autocorrelation of a topological structure-lag4/weighted by Van der Waal volumes | [23] |
| ATS7v | 2D Broto-Moreau autocorrelation of a topological structure-lag7/weighted by Van der Waal volumes | [23] |
| ATS1p | 2D Broto-Moreau autocorrelation of a topological structure-lag4/weighted by Atomic polarizabilities | [23] |
| Percent-Correct | Compounds Classified as Active | Compounds Classified as Inactive | |
|---|---|---|---|
| Training Set | |||
| Active Group | 50 | 61 | 62 |
| Inactive Group | 98 | 5 | 284 |
| Test Set | |||
| Active Group | 59 | 24 | 17 |
| Inactive Group | 86 | 6 | 37 |
| Compound | TI1 | ATS7m | ATS4v | ATS7v | ATS1p | DF | Class. | Ref. |
|---|---|---|---|---|---|---|---|---|
| Value | Pred. | |||||||
| Active Group | ||||||||
| 2′,β-DIHYDROXYCHALCONE | 25.6 | 2.79 | 2.81 | 2.57 | 2.91 | 0.82 | A | [24] |
| 3,7-DIMETHOXYFLAVONE | 49 | 3 | 3.27 | 2.73 | 3.01 | −2.19 | I | [25] |
| 4-METHYLESCULETIN | 11.21 | 1.02 | 2.51 | 0.23 | 2.59 | 6.44 | A | [26] |
| ANISODAMINE | 66.85 | 3.2 | 3.02 | 2.93 | 3.05 | 1.22 | A | [27] |
| BERGENIN | 51.11 | 3.32 | 3.37 | 2.52 | 2.97 | −0.82 | I | [28,29] |
| β-CARYOPHYLLENE ALCOHOL | 23.75 | 0 | 3.12 | 0 | 2.92 | 5.13 | A | [30] |
| BICUCULLINE (+) | 158.89 | 3.69 | 3.66 | 3.24 | 3.25 | −0.95 | I | [31,32] |
| BOLDINE | 73.83 | 3.38 | 3.7 | 2.83 | 3.16 | −1.75 | I | [33,34] |
| BRAZILEIN | 58.35 | 2.98 | 3.35 | 2.24 | 3.08 | 3.31 | A | [35,36] |
| CAMPTOTHECIN | 113.6 | 3.45 | 3.64 | 3.14 | 3.27 | −0.33 | I | [37,38] |
| CAPSAICIN | 0 | 3.01 | 2.95 | 2.71 | 3 | 1.8 | A | [39,40] |
| CARNOSIC ACID | 54.11 | 2.93 | 3.81 | 2.68 | 3.21 | −2.66 | I | [41] |
| CARVACROL | 0 | 0 | 2.08 | 0 | 2.44 | 3.68 | A | [14] |
| DIHYDROTANSHINONE I | 57.02 | 2.66 | 3.51 | 2.4 | 3.13 | −0.49 | I | [42,43] |
| ELLAGIC ACID | 56.29 | 2.61 | 3.43 | 1.72 | 3.02 | 3.35 | A | [44,45] |
| EPIAFZELECHIN (2R,3R)(−) | 44.79 | 3.02 | 3.06 | 2.53 | 2.98 | 1.45 | A | [46] |
| EUPHOL | 146.22 | 3.66 | 4.11 | 3.61 | 3.54 | −1.62 | I | [47] |
| GALLIC ACID | 0 | 0 | 1.93 | 0 | 2.33 | 2.36 | A | [48] |
| GENISTEIN | 44.57 | 2.88 | 3.09 | 2.45 | 2.98 | 1.15 | A | [14] |
| HARMALINE | 26.93 | 1.98 | 2.93 | 1.48 | 2.8 | 1.62 | A | [49] |
| HEMATEIN | 63.65 | 3.11 | 3.4 | 2.34 | 3.1 | 3.01 | A | [50,51] |
| HIERACIN | 53.56 | 3.3 | 3.17 | 2.72 | 3.03 | 1.38 | A | [52] |
| IRIGENIN TRIMETHYL ETHER | 90.88 | 3.73 | 3.75 | 3.27 | 3.17 | −4.15 | I | [53] |
| ISOLIQUIRITIGENIN | 29.12 | 2.87 | 2.78 | 2.61 | 2.94 | 2 | A | [54] |
| KOPARIN 2′-METHYL ETHER | 59.21 | 3.2 | 3.37 | 2.81 | 3.05 | −1.78 | I | [55] |
| KYNURENINE | 0 | 2.45 | 2.51 | 1.7 | 2.61 | 1.89 | A | [56] |
| LAWSONE | 9.48 | 0 | 2.43 | 0 | 2.59 | 4 | A | [57] |
| LUTEOLIN GLUCOSIDE | 171.97 | 4.01 | 3.57 | 3.45 | 3.35 | 2.44 | A | [14] |
| MADECASSIC ACID | 189.69 | 4.04 | 4.43 | 3.75 | 3.63 | −2.1 | I | [58] |
| METHYL ORSELLINATE | 0 | 0.85 | 2.39 | 0.41 | 2.42 | 0.59 | I | [59,60] |
| N-METHYLANTHRANILIC ACID | 0 | 0 | 2.14 | 0 | 2.32 | −0.42 | I | [61] |
| OLEANOLIC ACID | 163.48 | 3.85 | 4.28 | 3.68 | 3.59 | −1.63 | I | [14] |
| OUABAIN | 358.36 | 4.4 | 4.26 | 3.88 | 3.66 | 0.84 | A | [62] |
| p-HYDROXYCINNAMALDEHYDE | 0 | 1.3 | 1.95 | 0.71 | 2.39 | 4.75 | A | [63,64] |
| QUERCETIN | 52.38 | 3.27 | 3.2 | 2.67 | 3.03 | 1.29 | A | [65] |
| QUERCETIN TETRAMETHYL (5,7,3′,4′) ETHER | 74.79 | 3.59 | 3.54 | 3.09 | 3.11 | −2.49 | I | [14] |
| SANTONIN | 31.04 | 1.86 | 3.2 | 1.19 | 2.94 | 4.27 | A | [66] |
| SILIBININ | 243.99 | 4.1 | 3.85 | 3.61 | 3.48 | 1.68 | A | [67–69] |
| TECTORIGENIN | 53.39 | 3.12 | 3.26 | 2.68 | 3.03 | −0.42 | I | [70,71] |
| UMBELLIFERONE | 8.36 | 0 | 2.12 | 0 | 2.47 | 4.15 | A | [14] |
| UVAOL | 153.27 | 3.74 | 4.29 | 3.61 | 3.58 | −2.13 | I | [72] |
| Inactive Group | ||||||||
| 2-METHYL GRAMINE | 13.23 | 1.1 | 2.73 | 1.1 | 2.65 | −1.78 | I | - |
| 3-DEACETYLKHIVORIN | 285.72 | 4.57 | 4.44 | 4.14 | 3.62 | −3.59 | I | - |
| 3-PINANONE OXIME | 9.02 | 0 | 2.19 | 0 | 2.56 | 5.84 | A | - |
| ASARYLALDEHYDE | 0 | 1.2 | 2.61 | 0.92 | 2.41 | −4.9 | I | - |
| BAEOMYCESIC ACID | 47.58 | 3.65 | 3.52 | 3.25 | 3.16 | −1.82 | I | - |
| BOVINOCIDIN (3-NITROPROPIONIC ACID) | −8.58 | 0 | 1.01 | 0 | 1.63 | −6.92 | I | - |
| CHOLEST-5-EN-3-ONE | 128.51 | 3.42 | 3.89 | 3.37 | 3.45 | −0.65 | I | - |
| CONESSINE | 112.88 | 3.25 | 3.84 | 3.16 | 3.36 | −1.57 | I | - |
| CRASSIN ACETATE | 37.49 | 3.91 | 3.71 | 3.52 | 3.23 | −3.06 | I | - |
| CRINAMINE | 78.42 | 2.9 | 3.48 | 2.38 | 3.09 | 0.22 | I | - |
| DEACETOXY-7-OXISOGEDUNIN | 188.23 | 3.82 | 4.25 | 3.46 | 3.5 | −2.23 | I | - |
| DEOXYANDIROBIN | 161.25 | 3.95 | 4.14 | 3.62 | 3.47 | −2.48 | I | - |
| DIPHENYLUREA | 21.73 | 2.4 | 2.5 | 2.4 | 2.77 | −0.23 | I | - |
| DUARTIN. DIMETHYL ETHER | 76.8 | 3.39 | 3.6 | 3.04 | 3.11 | −3.9 | I | - |
| EPI(13)TORULOSOL | 30.87 | 3.08 | 3.56 | 2.87 | 3.13 | −2.72 | I | - |
| EUDESMIC ACID | 0 | 1.3 | 2.7 | 0.71 | 2.45 | −2.3 | I | - |
| EVOXINE | 76.98 | 3.47 | 3.43 | 2.99 | 3.08 | −1.8 | I | - |
| GLUCITOL-4-GUCOPYANOSIDE | 0 | 3.66 | 3.06 | 2.82 | 2.83 | −1.71 | I | - |
| HEXAMETHYLQUERCETAGETIN | 88.94 | 3.82 | 3.75 | 3.33 | 3.17 | −4.27 | I | - |
| ISOOSAJIN | 153.22 | 3.79 | 3.85 | 3.61 | 3.42 | −1.22 | I | - |
| JUAREZIC ACID | 0 | 2.23 | 2.2 | 1.8 | 2.56 | 1.88 | A | - |
| KHAYASIN | 274.26 | 4.48 | 4.38 | 4.11 | 3.65 | −2.18 | I | - |
| LEOIDIN | 68.47 | 3.83 | 3.74 | 3.25 | 3.22 | −1.74 | I | - |
| LOMATIN | 31.81 | 2.35 | 3.04 | 1.99 | 2.88 | −0.07 | I | - |
| MEDICARPIN | 56.7 | 2.78 | 3.17 | 2.43 | 3.01 | 0.37 | I | - |
| MEROGEDUNIN | 72.24 | 3.21 | 3.94 | 2.75 | 3.27 | −1.63 | I | - |
| METAMECONINE | 12.34 | 0.85 | 2.5 | 0.41 | 2.5 | 1.53 | A | - |
| METHYL EVERNINATE | 0 | 1.61 | 2.56 | 0.93 | 2.46 | −0.68 | I | - |
| METHYL ROBUSTONE | 146.49 | 3.65 | 3.68 | 3.34 | 3.3 | −1.03 | I | - |
| N-METHYLISOLEUCINE | −11.17 | 0 | 2.05 | 0 | 2.1 | −5.69 | I | - |
| PLECTOCOMINE METHYL ETHER | 23.69 | 1.52 | 2.76 | 1.21 | 2.74 | 1.61 | A | - |
| PODOTOTARIN | 346.77 | 4.54 | 4.56 | 4.38 | 3.85 | −1.06 | I | - |
| PRENYLETIN | 24.04 | 2.78 | 2.76 | 2.43 | 2.82 | −0.07 | I | - |
| PTAEROXYLIN | 35.11 | 2.49 | 3.15 | 2.26 | 2.93 | −1.55 | I | - |
| RETUSOQUINONE | 0 | 1.3 | 2.35 | 0.71 | 2.52 | 3.8 | A | - |
| RHETSININE | 94.88 | 3.23 | 3.46 | 2.98 | 3.19 | −0.36 | I | - |
| RHODINYL ACETATE | −25.56 | 2.04 | 2.36 | 1.8 | 2.52 | −2.14 | I | - |
| ROBUSTIC ACID | 114.56 | 3.69 | 3.71 | 3.36 | 3.28 | −1.77 | I | - |
| SARMENTOSIDE B | 484.87 | 4.66 | 4.42 | 4.15 | 3.75 | −0.04 | I | - |
| SENECRASSIDIOL 6-ACETATE | 41.44 | 2.6 | 3.42 | 2.06 | 3.04 | 0.77 | I | - |
| THEANINE | −17.76 | 1.76 | 2.01 | 1.31 | 2.22 | −3.61 | I | - |
| TROPINE | 5.68 | 0 | 1.22 | 0 | 2.34 | 10.88 | N.C. | - |
| XANTHOXYLIN | 0 | 1.2 | 2.63 | 0.92 | 2.46 | −3.75 | I | - |
| Compound | TI1 | ATS7m | ATS4v | ATS7v | ATS1p | DF Value | Ref. |
|---|---|---|---|---|---|---|---|
| 1,5-NORCARYOPHYLLEN-3-ONE | 12.95 | 0 | 3.07 | 0 | 2.8 | 2.43 | [73] |
| 2′,4′-DIHYDROXYCHALCONE 4′-GLUCOSIDE | 119.2 | 3.71 | 3.36 | 3.29 | 3.28 | 3.08 | nr |
| 2-HYDROXYXANTHONE | 24.98 | 1.73 | 2.85 | 1.39 | 2.82 | 2.58 | [74] |
| 3-HYDROXYCOUMARIN | 8.25 | 0 | 2.15 | 0 | 2.47 | 3.79 | [75] |
| 3-NOR-3-OXOPANASINSAN-6-OL | 24.04 | 0 | 3.03 | 0 | 2.89 | 5.32 | nr |
| alpha-TOCHOPHEROL | 83.77 | 3.37 | 3.68 | 3.28 | 3.45 | 2.53 | nr |
| APIIN | 345.29 | 4.46 | 3.8 | 3.88 | 3.55 | 3.35 | [76] |
| AVOCADANE ACETATE | −70.27 | 3.07 | 2.96 | 2.76 | 3.01 | 2.16 | nr |
| BATYL ALCOHOL | −83.55 | 2.94 | 2.96 | 2.81 | 3.08 | 3.3 | [77] |
| BERGAPTOL | 22.16 | 1.3 | 2.67 | 0.71 | 2.69 | 4.85 | [78] |
| BIXIN | −103.7 | 3.31 | 3.33 | 3.18 | 3.27 | 2.98 | [79] |
| BRAZILIN | 58.35 | 2.98 | 3.35 | 2.24 | 3.08 | 3.31 | [80] |
| CANTHARIDIN | 18.37 | 0 | 2.32 | 0 | 2.62 | 6.04 | nr |
| CAPSANTHIN | 187.67 | 3.83 | 3.96 | 3.77 | 3.77 | 6.06 | [81] |
| CARYOPHYLLENE [t(−)] | 11.37 | 0 | 2.89 | 0 | 2.77 | 3.75 | [82] |
| CEDROL | 23.5 | 0 | 3.14 | 0 | 2.92 | 4.87 | [83] |
| CHAULMOOGRIC ACID | 0 | 2.69 | 2.77 | 2.57 | 2.99 | 3.14 | [84] |
| CHLOROGENIC ACID | 48.22 | 3.36 | 3.09 | 2.78 | 3.07 | 3.29 | [85] |
| CINEOLE | 6.55 | 0 | 2.08 | 0 | 2.48 | 4.81 | [86] |
| COSMOSIIN | 162.44 | 3.92 | 3.54 | 3.41 | 3.33 | 2.27 | [87] |
| COTININE | 11.53 | 0.85 | 2.43 | 0.41 | 2.53 | 3.21 | [88] |
| CULMORIN | 25.72 | 0 | 3.2 | 0 | 2.94 | 4.8 | nr |
| DESOXYPEGANINE HYDROCHLORIDE | 16.37 | 0 | 2.49 | 0 | 2.65 | 4.82 | nr |
| DIALLYL SULFIDE | −7.5 | 0 | 1.39 | 0 | 2.12 | 2.63 | [89] |
| DICTAMNINE | 22.62 | 0 | 2.88 | 0 | 2.71 | 2.05 | nr |
| DIGITOXIN | 1046.2 | 4.61 | 4.4 | 4.15 | 3.93 | 2.34 | [62] |
| DIGOXIN | 1069.1 | 4.64 | 4.43 | 4.16 | 3.94 | 2.11 | [62] |
| DJENKOLIC ACID | −28.53 | 2.37 | 2.28 | 1.86 | 2.69 | 4.92 | [90] |
| EPOXY (1,11)HUMULENE | 15.16 | 0 | 3.05 | 0 | 2.83 | 3.48 | [91] |
| GARLICIN | −9.74 | 0.69 | 1.64 | 0.69 | 2.4 | 5.34 | [92] |
| GIBBERELLIC ACID | 92.61 | 2.83 | 3.8 | 2.26 | 3.27 | 2.08 | [93] |
| GUAIAZULENE | 13.61 | 0 | 3 | 0 | 2.83 | 4.22 | [94] |
| GUVACINE HYDROCHLORIDE | 0 | 0 | 1.32 | 0 | 2.1 | 2.91 | nr |
| HAEMATOXYLIN | 63.65 | 3.11 | 3.4 | 2.34 | 3.1 | 3.01 | nr |
| HARMALOL HYDROCHLORIDE | 22.47 | 1.65 | 2.81 | 0.87 | 2.77 | 5.94 | nr |
| HARMANE | 19.16 | 0 | 2.76 | 0 | 2.74 | 4.34 | [95] |
| HARMOL HYDROCHLORIDE | 22.47 | 1.65 | 2.81 | 0.87 | 2.77 | 5.94 | [96] |
| HARPAGOSIDE | 218.6 | 4.16 | 3.56 | 3.65 | 3.4 | 2.86 | [97] |
| HELENINE | 28.19 | 1.79 | 3.07 | 1.26 | 2.91 | 4.05 | [98] |
| HESPERIDIN | 421.88 | 4.48 | 3.87 | 3.88 | 3.6 | 3.8 | [99] |
| HINOKITIOL | 0 | 0 | 2.26 | 0 | 2.48 | 2.74 | [100] |
| HUMULENE (alpha) | 0 | 0 | 3 | 0 | 2.77 | 2.56 | [82] |
| INDOLE-3-CARBINOL | 7.49 | 0 | 2.09 | 0 | 2.46 | 4.19 | [101] |
| INOSITOL | 0 | 0 | 1.73 | 0 | 2.28 | 3.12 | [102] |
| ISOBERGAPTENE | 25.9 | 0.85 | 2.91 | 0.41 | 2.72 | 2.91 | nr |
| ISOKOBUSONE | 14.57 | 1.3 | 3.12 | 0.71 | 2.83 | 3.55 | nr |
| JUGLONE | 9.48 | 0 | 2.47 | 0 | 2.59 | 3.48 | [103] |
| KOBUSONE | 26.62 | 0 | 3.12 | 0 | 2.85 | 3.35 | nr |
| L(+/−)-ALLIIN | −14.57 | 1.3 | 1.97 | 0.71 | 2.36 | 3.65 | [104] |
| LYCOPODINE PERCHLORATE | 36.79 | 0 | 3.48 | 0 | 3.01 | 3.55 | [105] |
| MENADIONE | 9.48 | 0 | 2.53 | 0 | 2.63 | 3.95 | [106] |
| MENTHOL(−) | 0 | 0 | 2.2 | 0 | 2.44 | 2.26 | [107] |
| MENTHONE | 0 | 0 | 2.2 | 0 | 2.44 | 2.26 | [108] |
| MIMOSINE | 0 | 2.31 | 2.25 | 1.23 | 2.43 | 3.19 | [109] |
| MUUROLLADIE-3-ONE | 15 | 1.3 | 3.09 | 0.71 | 2.86 | 4.75 | nr |
| OCTOPAMINE HYDROCHLORIDE | 0 | 0.94 | 1.94 | 0.3 | 2.36 | 5.54 | nr |
| PATULIN | 7.06 | 0 | 1.81 | 0 | 2.28 | 2.2 | [110] |
| PECTOLINARIN | 427.48 | 4.54 | 3.93 | 3.96 | 3.61 | 3.01 | [111] |
| PHLORIDZIN | 127.51 | 4.08 | 3.47 | 3.57 | 3.31 | 2.28 | [112] |
| PICROTOXININ | 68.36 | 2.09 | 3.46 | 1.2 | 3.05 | 5.58 | nr |
| PIPERINE | 74.81 | 2.87 | 2.93 | 2.6 | 3 | 2.01 | [113] |
| PLUMBAGIN | 10.92 | 0 | 2.67 | 0 | 2.67 | 3.28 | [114] |
| PUNCTAPORONIN B | 17.9 | 1.41 | 3.3 | 0.57 | 2.91 | 5.6 | nr |
| PURPURIN | 33.76 | 2.12 | 3.22 | 1.62 | 2.96 | 2.28 | [115] |
| RHAPONTIN | 125.6 | 3.82 | 3.42 | 3.38 | 3.28 | 2.06 | [116] |
| RHOIFOLIN | 357.18 | 4.51 | 3.85 | 3.93 | 3.57 | 3.3 | [117] |
| RUTOSIDE (rutin) | 392.19 | 4.68 | 3.88 | 4.07 | 3.6 | 3.12 | [118] |
| SAFROLGLYCOL | 14.58 | 2.26 | 2.4 | 1.4 | 2.54 | 2.79 | [119] |
| SCOPOLETIN | 11.82 | 1.33 | 2.47 | 0.82 | 2.54 | 2.11 | [120] |
| SECURININE | 31.65 | 0.85 | 3.02 | 0.41 | 2.85 | 5.28 | nr |
| SHIKIMIC ACID | 0 | 0 | 1.93 | 0 | 2.33 | 2.36 | [121,122] |
| THYMOQUINONE | 0 | 0 | 2.31 | 0 | 2.48 | 2.11 | [123] |
| TRYPTAMINE | 9.75 | 0.77 | 2.38 | 0.53 | 2.56 | 3.05 | [124] |
| XANTHURENIC ACID | 13.51 | 1.3 | 2.65 | 0.71 | 2.64 | 3.73 | [56] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Galvez-Llompart, M.; Zanni, R.; García-Domenech, R. Modeling Natural Anti-Inflammatory Compounds by Molecular Topology. Int. J. Mol. Sci. 2011, 12, 9481-9503. https://doi.org/10.3390/ijms12129481
Galvez-Llompart M, Zanni R, García-Domenech R. Modeling Natural Anti-Inflammatory Compounds by Molecular Topology. International Journal of Molecular Sciences. 2011; 12(12):9481-9503. https://doi.org/10.3390/ijms12129481
Chicago/Turabian StyleGalvez-Llompart, María, Riccardo Zanni, and Ramón García-Domenech. 2011. "Modeling Natural Anti-Inflammatory Compounds by Molecular Topology" International Journal of Molecular Sciences 12, no. 12: 9481-9503. https://doi.org/10.3390/ijms12129481