The Effect of the Acetone Extract of Arctotis arctotoides (Asteraceae) on the Growth and Ultrastructure of Some Opportunistic Fungi Associated with HIV/AIDS
Abstract
:1. Introduction
2. Results
2.1. Inhibition of Fungal Growth
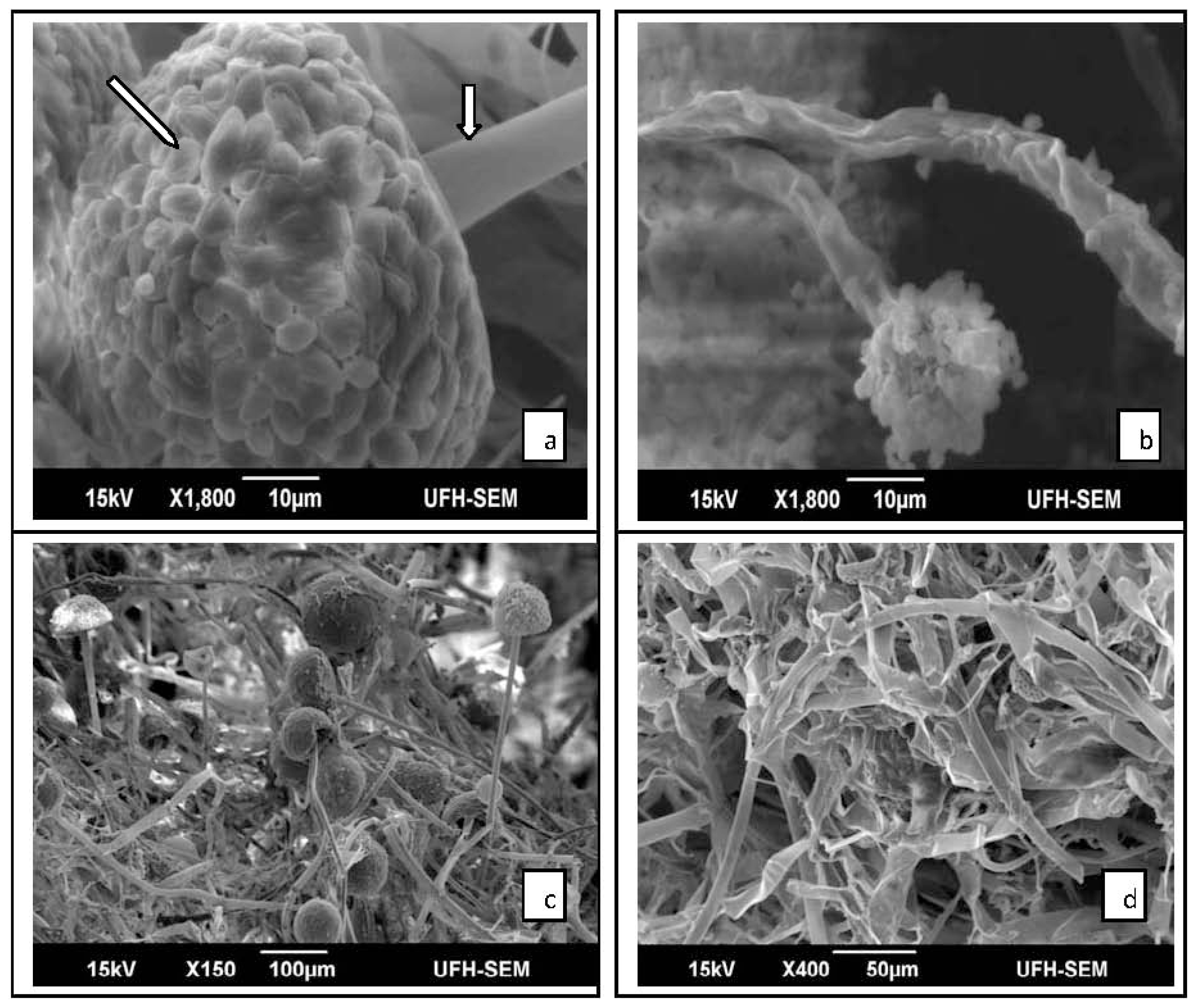
2.2. Results of SEM
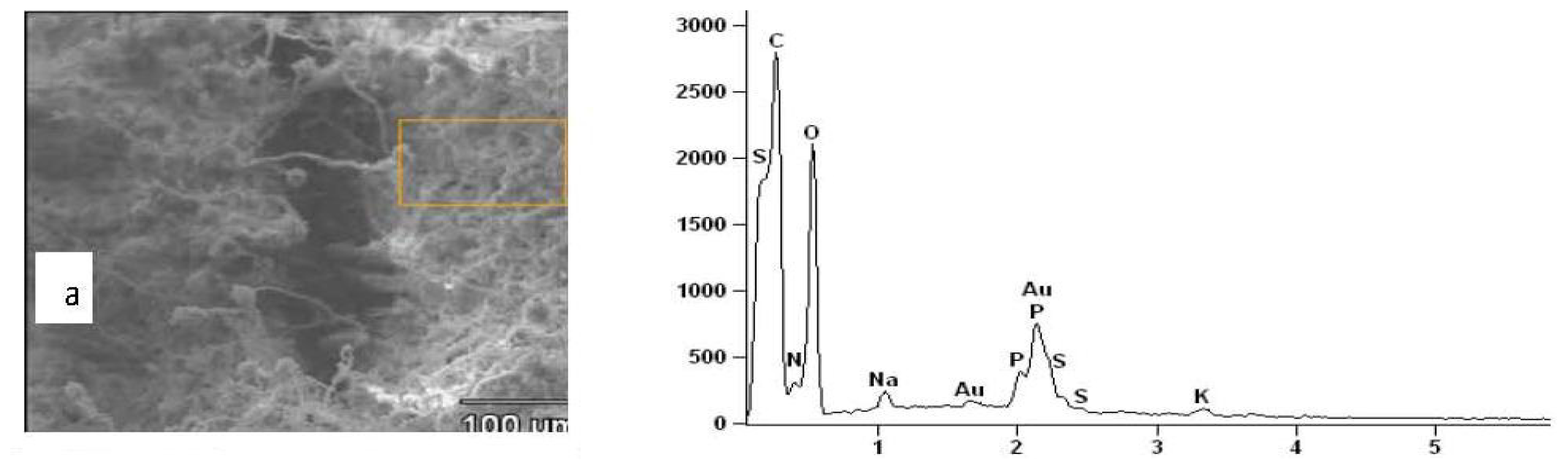
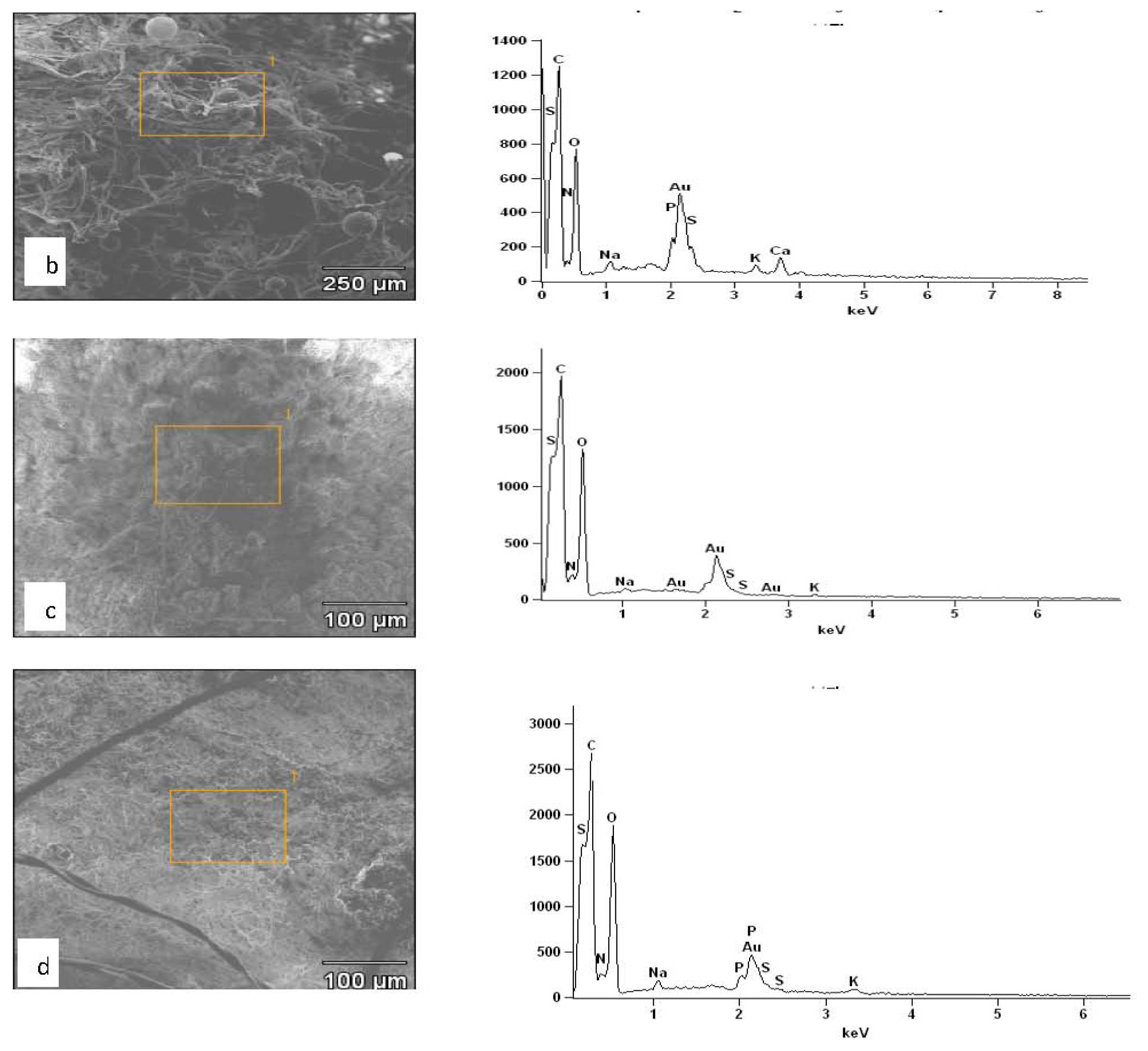
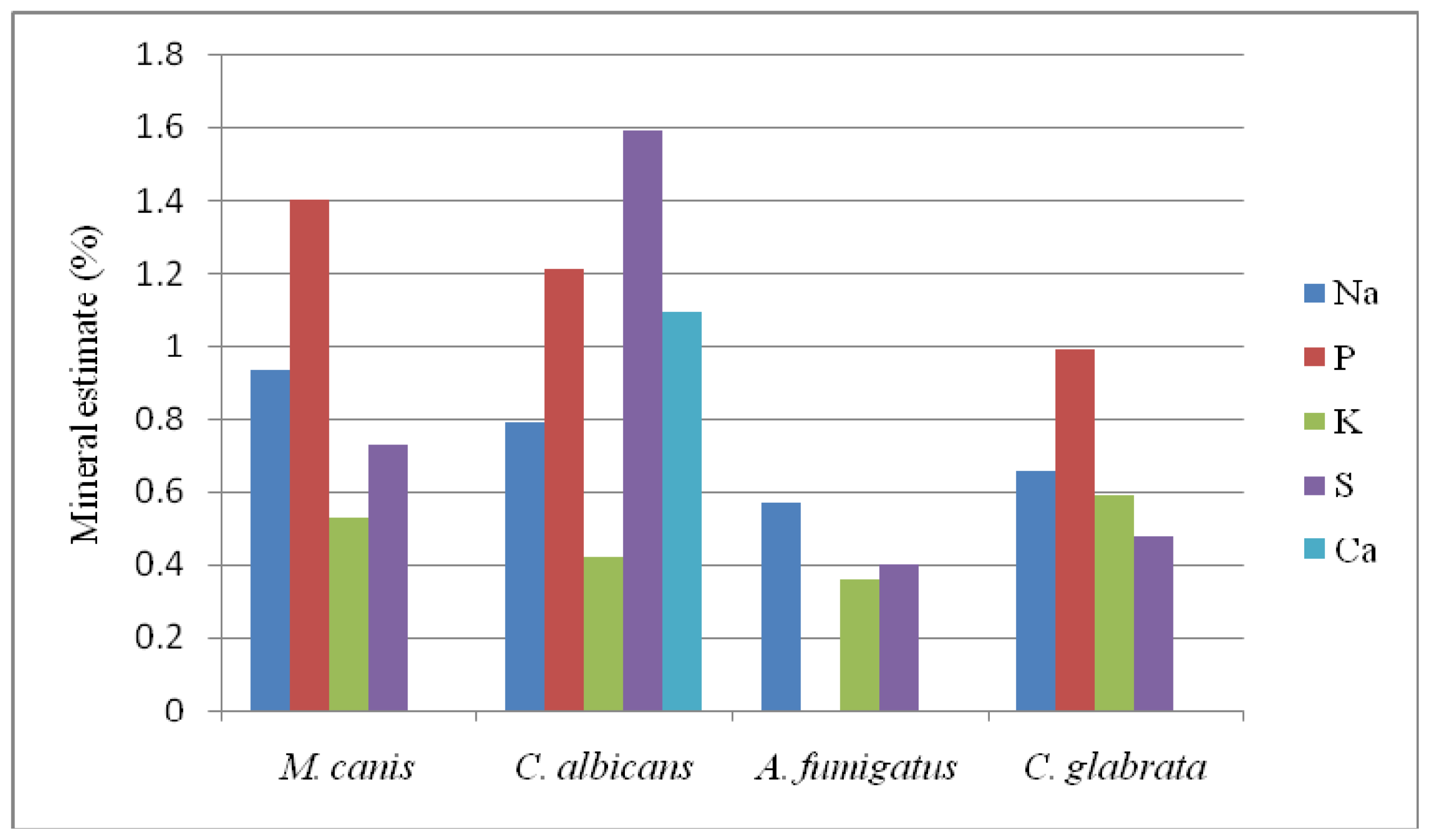
2.3. Results of EDXS
3. Discussion
4. Experimental Section
4.1. Plant Material and Culture Conditions
4.2. Inhibition of Fungal Growth
4.3. SEM and Energy Dispersive X-ray Spectroscopy (SEM-EDXS)
5. Conclusion
Acknowledgements
References
- Walsh, T.J.; Groll, A.; Hiemenz, J.; Fleming, R.; Roilides, E.; Anaissie, E. Infections due to emerging and uncommon medically important fungal pathogens. Clin. Microbiol. Infect 2004, 10, 48–66. [Google Scholar]
- Bharathi, M.; Rani, A.U. Pathogenic fungal isolates in sputum of HIV positive patients. J. AIDS HIV Res 2011, 107–113. [Google Scholar]
- Miceli, H.M.; Diaz, J.A; Lee, D.S. Emerging opportunistic yeast infections. Lancet Infect. Dis. 2011, 11, 142–151. [Google Scholar]
- Sun, J.N.; Solis, N.V.; Phan, Q.T.; Bajwa, J.S.; Kashleva, H.; Thompson, A.; Liu, Y.; Dongari-Bagtzoglou, A.; Edgerton, M.; Filler, S.G. Host cell invasion and virulence mediated by Candida albicans. PLoS Pathog 2010, 6, e1001181. [Google Scholar]
- Baddley, J.W. Clinical risk factors for invasive aspergillosis. Med. Mycol 2011, 49, 7–12. [Google Scholar]
- Thompson, G.R.; Patterson, T.F. Pulmonary aspergillosis. Semin. Respir Crit. Care Med 2008, 28, 103–110. [Google Scholar]
- Rodwell, G.E.; Bayles, C.L.; Towersey, L.; Aly, R. The prevalence of dermatophyte infection in patients infected with human immunodeficiency virus. Int. J. Dermatol 2008, 47, 339–343. [Google Scholar]
- Alvarez-Castellanos, P.P.; Bishop, C.D.; Pascual-Villalobos, M.J. Antifungal activity of the essential oil of flowerheads of garland chrysanthemum (Chrysanthemum coronarium) against agricultural pathogens. Phytochemistry 2001, 5, 99–102. [Google Scholar]
- Otang, W.M.; Grierson, D.S.; Ndip, R.N. Antifungal activity of Arctotis arctotoides (L.f.) O. Hoffm. and Gasteria bicolor Haw. against opportunistic fungi associated with HIV/AIDS. Pharmacog. Mag 2011, in press. [Google Scholar]
- Afolayan, A.J.; Jimoh, F.O.; Sofidiya, M.O.; Koduru, S.; Lewu, F.B. Medicinal potential of the root of Arctotis arctotoides. Pharm. Biol 2007, 45, 486–493. [Google Scholar]
- Afolayan, A.J. Extracts from the shoots of Arctotis arctotoides inhibit the growth of bacteria and fungi. Pharm. Biol 2003, 41, 22–25. [Google Scholar]
- Salama, H.M.H.; Marraiki, N. Antimicrobial activity and phytochemical analysis of Polygonum Aviculare L. (Polygonaceae), naturally growing in Egypt. Aust. J. Basic Appl. Sci 2009, 3, 2008–2015. [Google Scholar]
- Sharma, N.; Tripathi, A. Effect of Citrus sinensis (L.) Osbeck epicarp essential oil on growth and morphogenesis of Aspergillus niger (L.) Van Tieghem. Microbiol. Res 2008, 163, 337–344. [Google Scholar]
- Gandomi, H.; Misaghi, A.; Basti, A.A.; Hamedi, H.; Shirvani, Z.R. Effect of Zataria multiflora Boiss. Essential oil on growth and aflatoxin formation by Aspergillus flavus in culture media and cheese. Food Chem. Toxicol 2009, 47, 2397–2400. [Google Scholar]
- Kaminskyj, S.G.; Dahms, T.S. High spatial resolution surface imaging and analysis of fungal cells using SEM and AFM. Micron 2008, 39, 349–361. [Google Scholar]
- Nogueira, J.H.C.; Goncalez, E.; Galleti, S.R.; Facanali, R.; Marques, M.O.M.; Felício, J.D. Ageratum conyzoides essential oil as aflatoxin suppressor of Aspergillus flavus. Int. J. Food Microbiol 2010, 137, 55–60. [Google Scholar]
- de Billerbeck, V.G.; Roques, C.G.; Bessiere, J.M.; Fonvielle, J.L.; Dargent, R. Effect of Cymbopogon nardus (L.) W. Watson essential oil on the growth and morphogenesis of Aspergillus niger. Can. J. Microbiol 2001, 47, 9–17. [Google Scholar]
- Figueras, M.J.; Guarro, J. X-ray microanalysis of black piedra. Antonie van Leeuwenhoek 1997, 72, 275–281. [Google Scholar]
- Sutherland, I. Microbial polysaccharides—A comparison with eukaryotic polymers. Symp. Soc. Exp. Biol 1989, 43, 389–402. [Google Scholar]
- Tolouee, M.; Alinezhad, S.; Saberi, R.; Eslamifar, A.; Zad, S.J.; Jaimand, K.; Taeb, J.; Rezaee, M.B.; Kawachi, M.; Shams-Ghahfarokhi, M.; et al. Effect of Matricaria chamomilla L. flower essential oil on the growth and ultrastructure of Aspergillus niger van Tieghem. Int. J. Food Microbiol 2010, 139, 127–133. [Google Scholar]
- Shams-Ghahfarokhi, M.; Goodarzi, M.; Razzaghi-Abyaneh, M.; Al-Tiraihi, T.; Seyedipour, G. Morphological evidences for onion-induced growth inhibition of Trichophyton rubrum and Trichophyton mentagrophytes. Fitoterapia 2004, 75, 645–655. [Google Scholar]
- Das, K.; Tiwari, R.K.S.; Shrivastava, D.K. Techniques for evaluation of medicinal plant products as antimicrobial agent: Current methods and future trends. J. Med. Plant Res 2010, 4, 104–111. [Google Scholar]


| Concentration of plant extract (mg/mL) | Inhibition of mycelial growth (%) a | |||
|---|---|---|---|---|
| Af | Cg | Ca | Mc | |
| 0.32 | 0 | 14.3 | 9.4 | 15.6 |
| 0.63 | 0 | 19.5 | 20.7 | 26.3 |
| 1.25 | 0 | 14.3 | 25.3 * | 29 * |
| 2.5 | 0 | 29 * | 41.8 * | 41.4 * |
| 5 | 9.7 | 25 * | 65.3 * | 59.5 * |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Otang, W.M.; Grierson, D.S.; Ndip, R.N. The Effect of the Acetone Extract of Arctotis arctotoides (Asteraceae) on the Growth and Ultrastructure of Some Opportunistic Fungi Associated with HIV/AIDS. Int. J. Mol. Sci. 2011, 12, 9226-9235. https://doi.org/10.3390/ijms12129226
Otang WM, Grierson DS, Ndip RN. The Effect of the Acetone Extract of Arctotis arctotoides (Asteraceae) on the Growth and Ultrastructure of Some Opportunistic Fungi Associated with HIV/AIDS. International Journal of Molecular Sciences. 2011; 12(12):9226-9235. https://doi.org/10.3390/ijms12129226
Chicago/Turabian StyleOtang, Wilfred M., Donald S. Grierson, and Roland N. Ndip. 2011. "The Effect of the Acetone Extract of Arctotis arctotoides (Asteraceae) on the Growth and Ultrastructure of Some Opportunistic Fungi Associated with HIV/AIDS" International Journal of Molecular Sciences 12, no. 12: 9226-9235. https://doi.org/10.3390/ijms12129226




