AFM and Multiple Transmission-Reflection Infrared Spectroscopy (MTR-IR) Studies on Formation of Air-Stable Supported Lipid Bilayers
Abstract
:1. Introduction
2. Results and Discussion
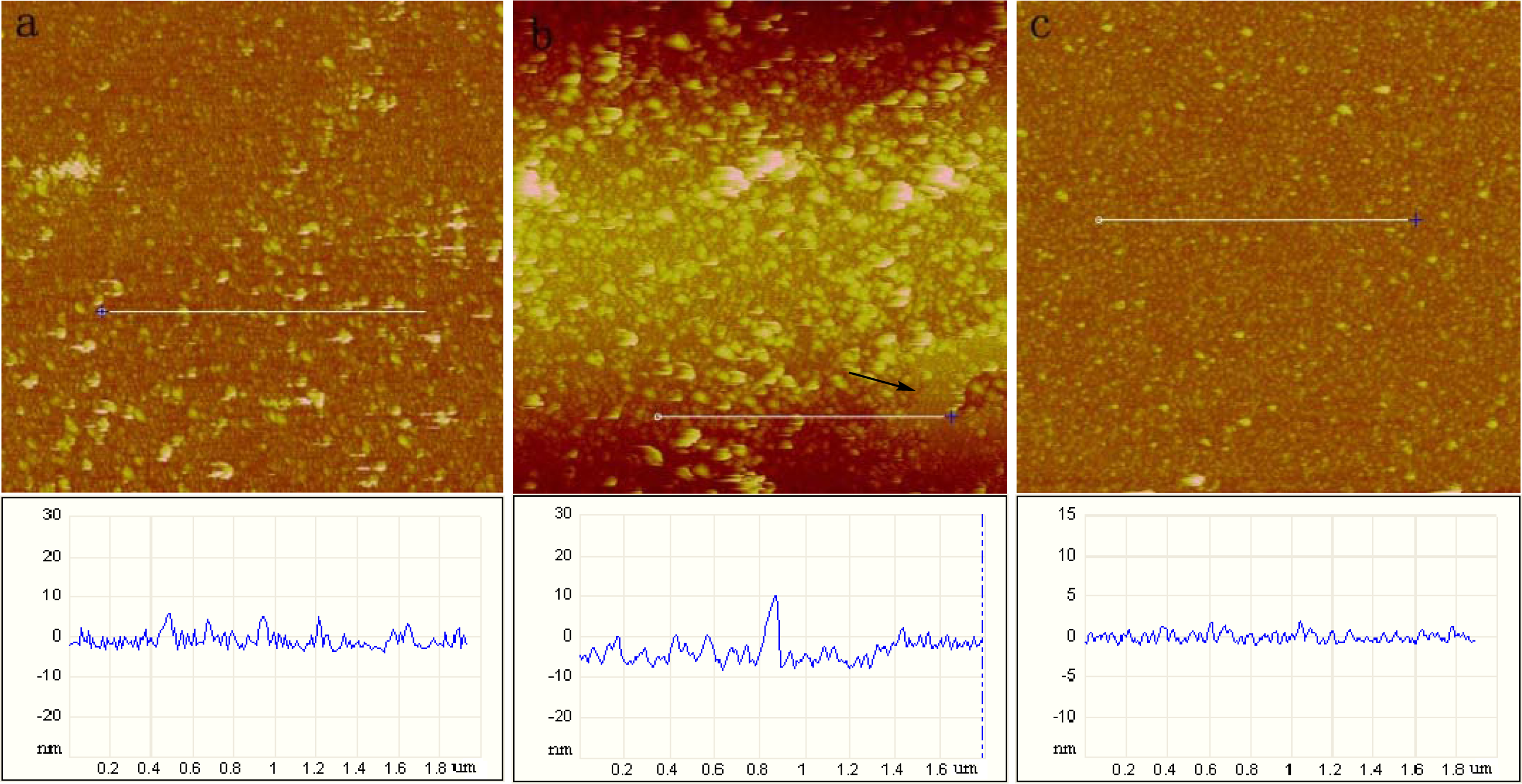
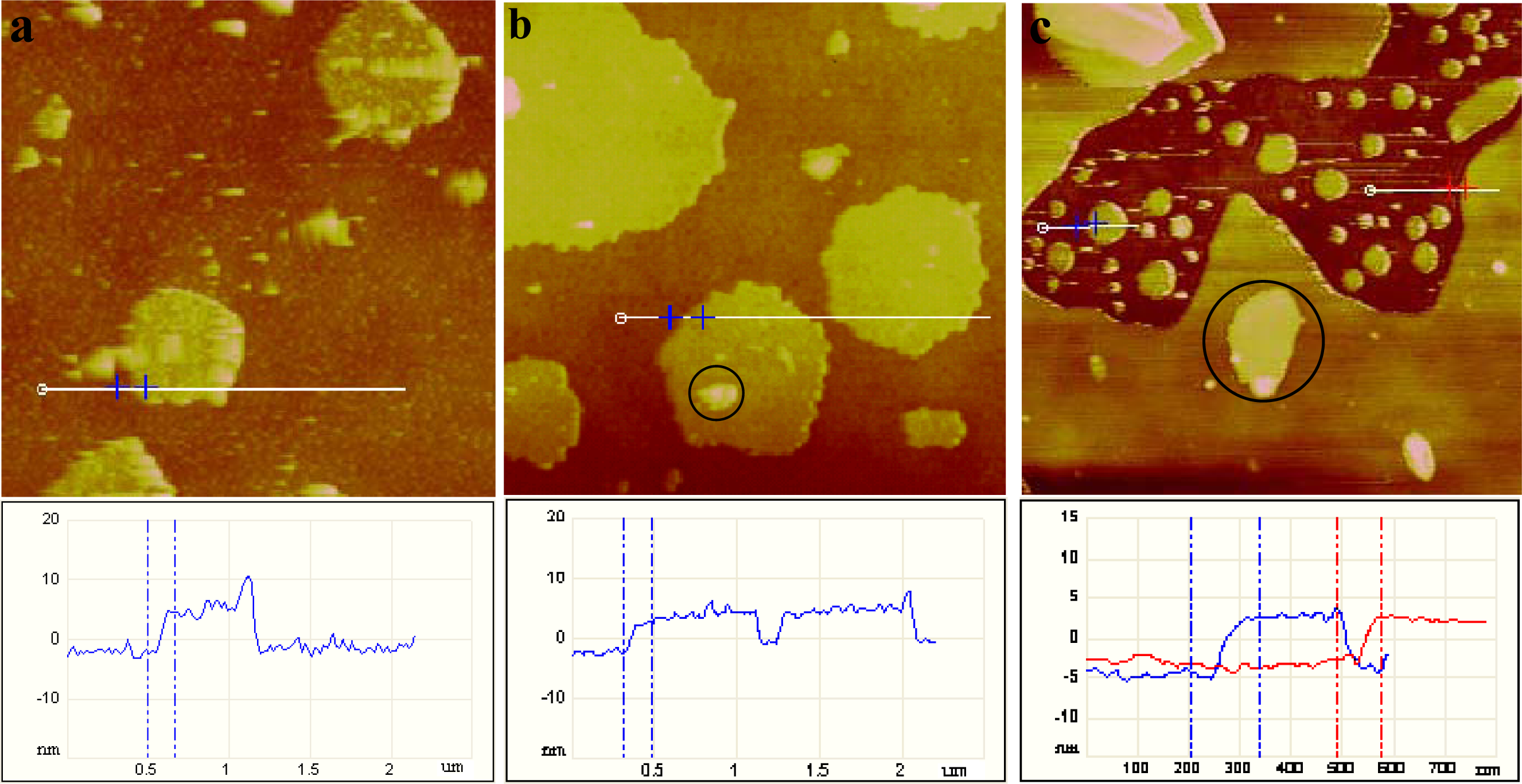
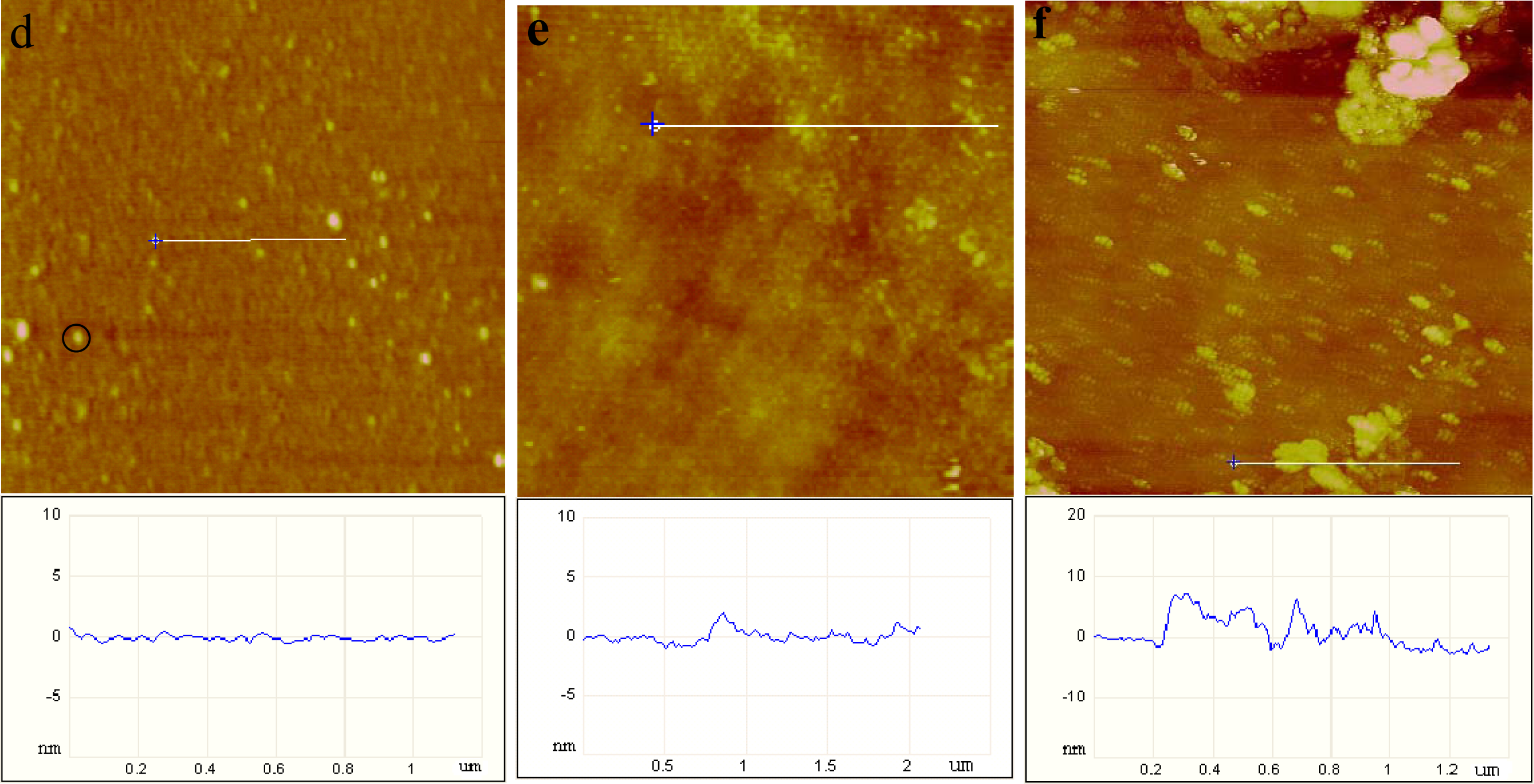
2.1. AFM Characterization
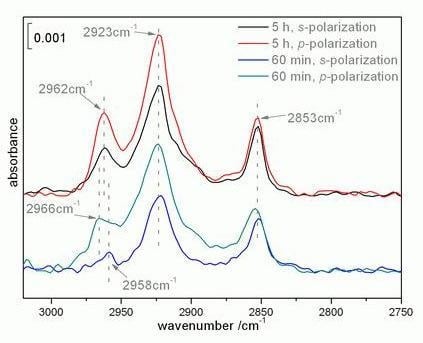
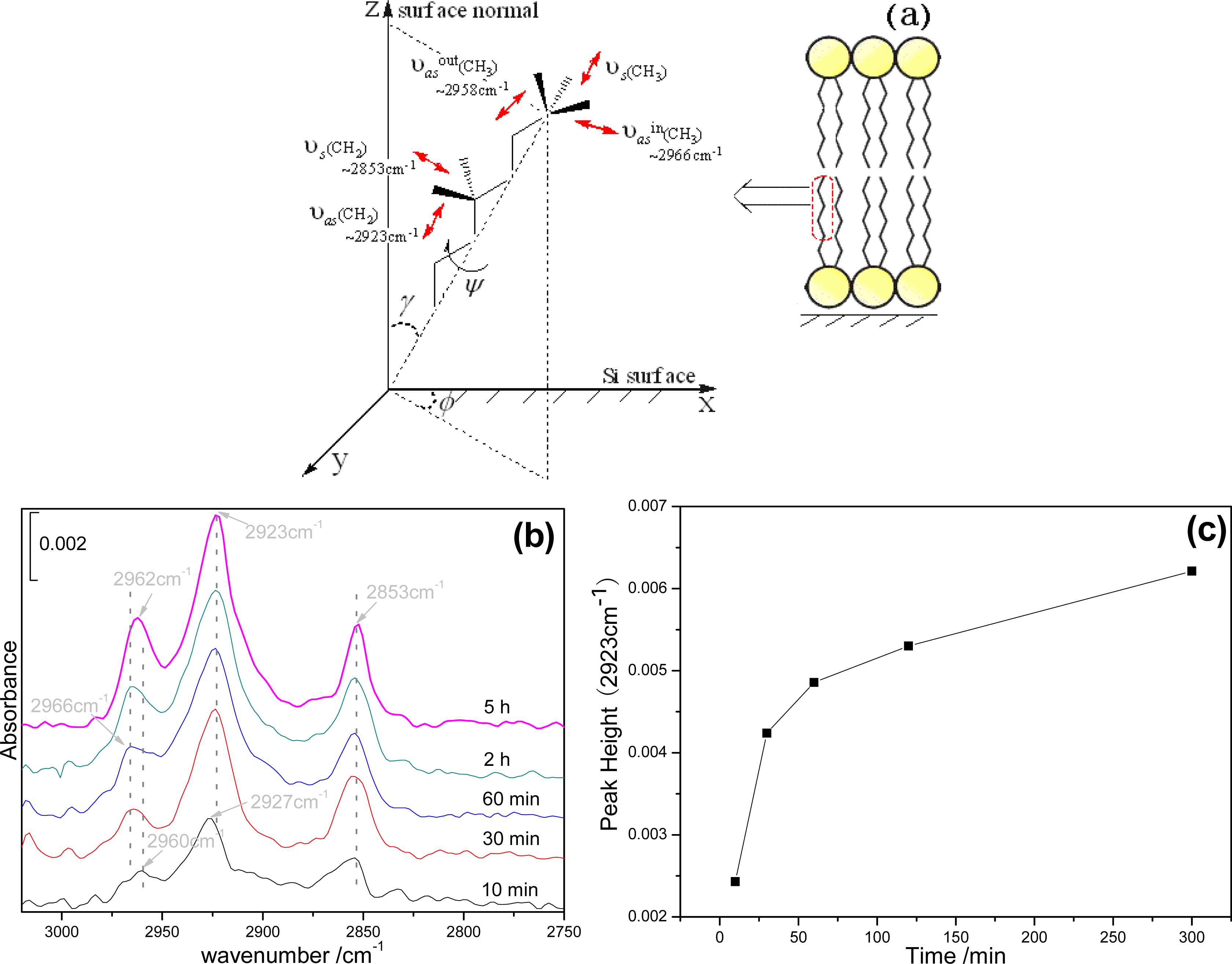
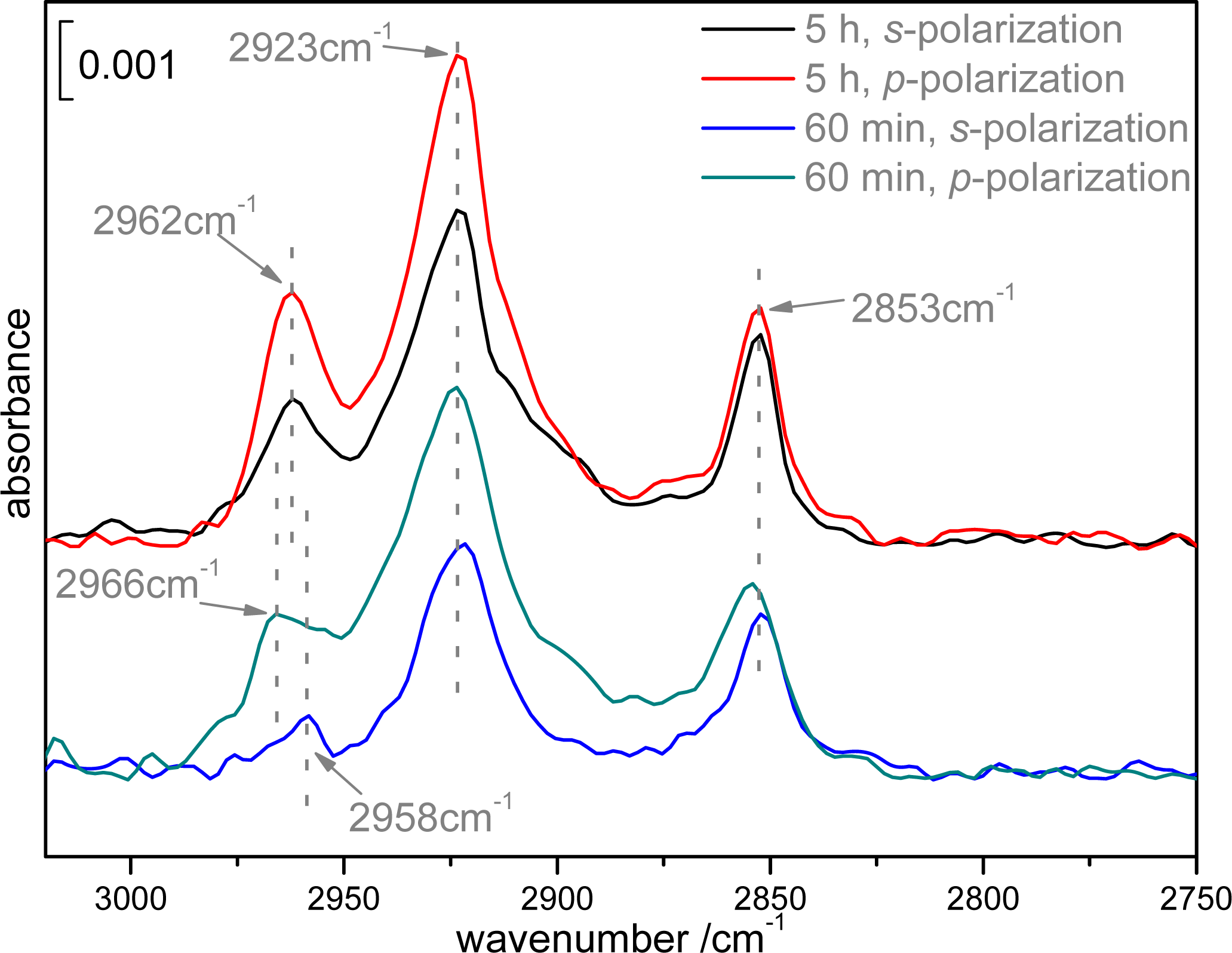
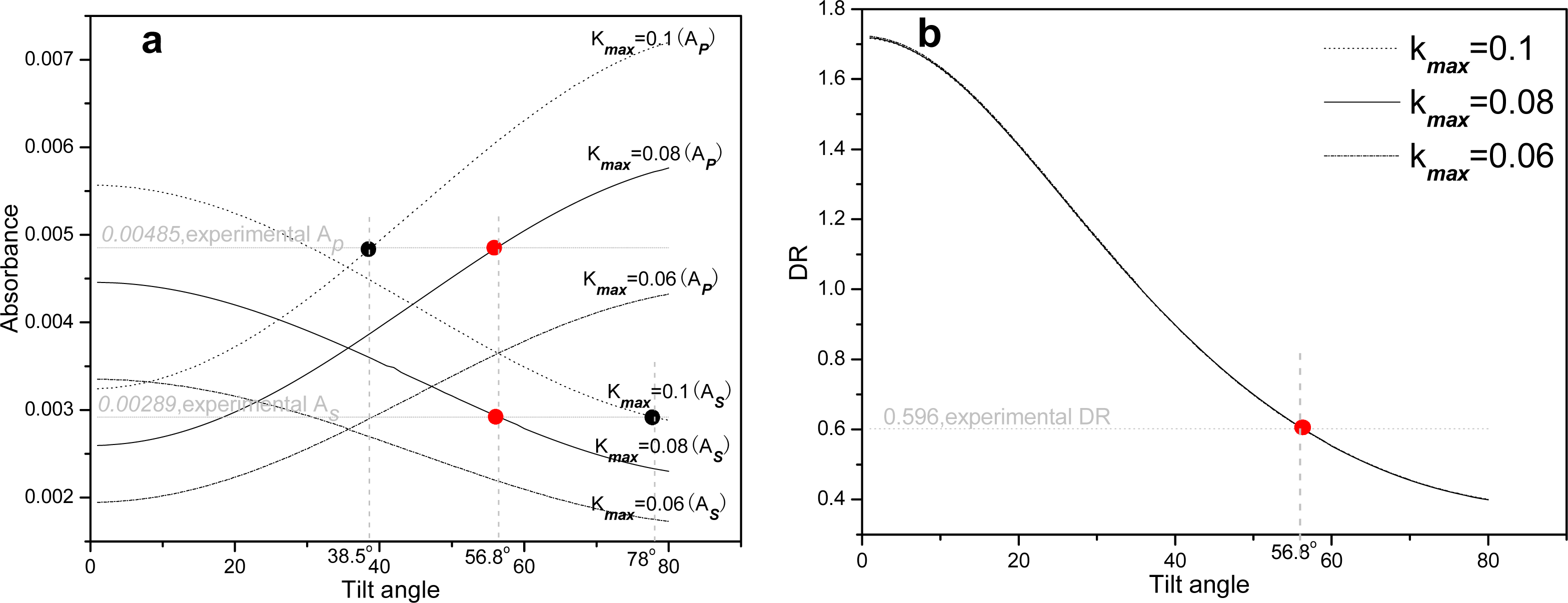
2.2. Infrared spectroscopy analysis
3. Experimental Section
4. Conclusions
Acknowledgments
References and Notes
- Barenholz, Y; Gibbes, D; Litman, BJ; Goll, J; Thompson, TE; Carlson, FD. A simple method for the preparation of homogeneous phospholipid vesicles. Biochemistry 1977, 16, 2806–2810. [Google Scholar]
- Kahya, N; Scherfeld, D; Bacia, K; Poolman, B; Schwille, P. Probing lipid mobility of raft-exhibiting model membranes by fluorescence correlation spectroscopy. J. Biol. Chem 2003, 278, 28109–28115. [Google Scholar]
- Atanasov, V; Knorr, N; Duran, RS; Ingebrandt, S; Offenhausser, A; Knoll, W; Koper, I. Membrane on a chip: A functional tethered lipid bilayer membrane on silicon oxide surfaces. Biophys. J 2005, 89, 1780–1788. [Google Scholar]
- Keller, CA; Kasemo, B. Surface specific kinetics of lipid vesicle adsorption measured with a quartz crystal microbalance. Biophys. J 1998, 75, 1397–1402. [Google Scholar]
- Majewski, J; Wong, JY; Park, CK; Seitz, M; Israelachvili, JN; Smith, GS. Structural studies of polymer-cushioned lipid bilayers. Biophys. J 1998, 75, 2363–2367. [Google Scholar]
- Sackmann, E. Supported membranes: scientific and practical applications. Science 1996, 271, 43–48. [Google Scholar]
- Dahlin, A; Zach, M; Rindzevicius, T; Kall, M; Sutherland, DS; Hook, F. Localized surface plasmon resonance sensing of lipid-membrane-mediated biorecognition events. J. Am. Chem. Soc 2005, 127, 5043–5048. [Google Scholar]
- Castellana, ET; Cremer, PS. Solid supported lipid bilayers: From biophysical studies to sensor design. Surf. Sci. Rep 2006, 61, 429–444. [Google Scholar]
- Worsfold, O; Voelcker, NH; Nishiya, T. Biosensing using lipid bilayers suspended on porous silicon. Langmuir 2006, 22, 7078–7083. [Google Scholar]
- Furukawa, K; Nakashima, H; Kashimura, Y; Torimitsu, K. Microchannel device using self-spreading lipid bilayer as molecule carrier. Lab Chip 2006, 6, 1001–1006. [Google Scholar]
- Kim, P; Lee, SE; Jung, HS; Lee, HY; Kawai, T; Suh, KY. Soft lithographic patterning of supported lipid bilayers onto a surface and inside microfluidic channels. Lab Chip 2006, 6, 54–59. [Google Scholar]
- Malmstadt, N; Nash, MA; Purnell, RF; Schmidt, JJ. Automated formation of lipid-bilayer membranes in a microfluidic device. Nano Lett 2006, 6, 1961–1965. [Google Scholar]
- Cannon, B; Weaver, N; Pu, QS; Thiagarajan, V; Liu, SR; Huang, JY; Vaughn, MW; Cheng, KH. Cholesterol modulated antibody binding in supported lipid membranes as determined by total Internal reflectance microscopy on a microfabricated high-throughput glass chip. Langmuir 2005, 21, 9666–9674. [Google Scholar]
- Larsson, C; Bramfeldt, H; Wingren, C; Borrebaeck, C; Hook, F. Gravimetric antigen detection utilizing antibody-modified lipid bilayers. Anal. Biochem 2005, 345, 72–80. [Google Scholar]
- Groves, JT; Boxer, SG. Electric field-induced concentration gradients in planar supported bilayers. Biophys. J 1995, 69, 1972–1975. [Google Scholar]
- Mou, JX; Yang, J; Shao, ZF. Atomic force microscopy of cholera toxin boligomers bound to bilayers of biologically relevant lipids. J. Mol. Biol 1995, 5, 507–512. [Google Scholar]
- Williams, LM; Evans, SD; Flynn, TM; Marsh, A; Knowles, PF; Bushby, RJ; Boden, N. Kinetics of the unrolling of small unilamellar phospholipid vesicles onto self-assembled monolayers. Langmuir 1997, 13, 751–757. [Google Scholar]
- Jenkins, ATA; Hu, J; Wang, YZ; Schiller, S; Foerch, R; Knoll, W. Pulsed plasma deposited maleic anhydride thin films as supports for lipid bilayers. Langmuir 2000, 16, 6381–6384. [Google Scholar]
- Kalb, E; Frey, S; Tamm, LK. Formation of supported planar bilayers by fusion of vesicles to supported phospholipid monolayers. Biochim. Biophys. Acta 1992, 31, 307–316. [Google Scholar]
- Reimhult, E; Zach, M; Hook, F; Kasemo, B. A multitechnique study of liposome adsorption on Au and lipid bilayer formation on SiO2. Langmuir 2006, 22, 3313–3319. [Google Scholar]
- Pfeiffer, I; Seantier, B; Petronis, S; Sutherland, D; Kasemo, B; Zach, M. Influence of nanotopography on phospholipid bilayer formation on silicon dioxide. J. Phys. Chem. B 2008, 112, 5175–5181. [Google Scholar]
- Solletti, JM; Botreau, M; Sommer, F; Brunat, WL; Kasas, S; Duc, TM; Celio, MR. Elaboration and characterization of phospholipid langmuir-blodgett films. Langmuir 1996, 12, 5379–5386. [Google Scholar]
- Naumann, CA; Prucker, O; Lehmann, T; Ruhe, J; Knoll, W; Frank, CW. The polymer-supported phospholipid bilayer: Tethering as a new approach to substrate-membrane stabilization. Biomacromolecules 2002, 3, 27–35. [Google Scholar]
- Mcconnell, HM; Watts, TH; Weis, RM; Brian, AA. Supported planar membranes in studies of cell-cell recognition in the immune system. Biochim. Biophys. Acta 1986, 864, 95–106. [Google Scholar]
- Graneli, A; Rydstrom, J; Kasemo, B; Hook, F. Formation of supported lipid bilayer membranes on SiO2 from proteoliposomes containing transmembrane proteins. Langmuir 2003, 19, 842–850. [Google Scholar]
- Reviakine, I; Brisson, A. Formation of supported phospholipid bilayers from unilamellar vesicles investigated by atomic force microscopy. Langmuir 2000, 16, 1806–1815. [Google Scholar]
- Jass, J; Tjärnhage, T; Puu, G. From liposomes to supported, planar bilayer structures on hydrophilic and hydrophobic surfaces: An atomic force microscopy study. Biophys. J 2000, 79, 3153–3163. [Google Scholar]
- Liang, X; Mao, G; Ng, KYS. Probing small unilamellar egg PC vesicles on mica surface by atomic force microscopy. Coll. Surf. B: Biointerf 2004, 34, 41–51. [Google Scholar]
- Richter, RP; Brisson, A. Characterization of lipid bilayers and protein assemblies supported on rough surfaces by atomic force microscopy. Langmuir 2003, 19, 1632–1640. [Google Scholar]
- Richter, RP; Brisson, AR. Following the formation of supported lipid bilayers on mica: A study combining AFM, QCM-D, and ellipsometry. Biophys. J 2005, 88, 3422–3433. [Google Scholar]
- Liu, HB; Venkataraman, NV; Bauert, T; Textor, M; Xiao, SJ. Multiple transmission-reflection infrared spectroscopy for high-sensitivity measurement of molecular monolayers on silicon surfaces. J. Phys. Chem. A 2008, 112, 12372–12377. [Google Scholar]
- Liu, HB; Venkataraman, NV; Spencer, ND; Textor, M; Xiao, SJ. Structural evolution of self-assembled alkanephosphate monolayers on TiO2. Chem. Phys. Chem 2008, 9, 1979–1981. [Google Scholar]
- Schonherr, H; Johnson, JM; Lenz, P; Frank, CW; Boxer, SG. Vesicle adsorption and lipid bilayer formation on glass studied by atomic force microscopy. Langmuir 2004, 20, 11600–11606. [Google Scholar]
- Hill, IR; Levin, IW. Vibrational spectra and carbon–hydrogen stretching mode assignments for a series of N-alkyl carboxylic acids. J. Chem. Phys 1979, 70, 842–851. [Google Scholar]
- Chernyshova, IV; Rao, KH. A new approach to the IR spectroscopic study of molecular orientation and packing in adsorbed monolayers. Orientation and packing of long-chain primary amines and alcohols on quartz. J. Phys. Chem. B 2001, 105, 810–820. [Google Scholar]
- Zawisza, I; Wittstock, G; Boukherroub, R; Szunerits, S. PM IRRAS investigation of thin silica films deposited on gold. Part 1. Theory and proof of concept. Langmuir 2007, 23, 9303–9309. [Google Scholar]
- Porter, MD; Bright, TB; Allara, DL; Chidsey, CED. Spontaneously organized molecular assemblies. 4. Structural characterization of n-alkyl thiol monolayers on gold by optical ellipsometry, infrared spectroscopy, and electrochemistry. J. Am. Chem. Soc 1987, 109, 3559–3568. [Google Scholar]
- Aveyard, R; Binks, BP; Fletcher, PD; Ye, X. Dynamic contact angles and deposition efficiency for transfer of docosanoic acid on to mica from CdCl2 subphases as a function of pH. Thin Solid Films 1992, 210–211, 36–38. [Google Scholar]
- Johnson, DJ; Amm, DT; Laursen, T; Gupta, SK. Langmuir-blodgett deposition of yttrium arachidate. Thin Solid Films 1993, 232, 245–251. [Google Scholar]
- Parikh, AN; Allara, DL. Quantitative determination of molecular structure in multilayered thin films of biaxial and lower symmetry from photon spectroscopies. I. Reflection infrared vibrational spectroscopy. J. Chem. Phys 1992, 96, 927–945. [Google Scholar]
- Ren, Y; Meuse, CW; Hsu, SL; Stidham, HD. Reflectance infrared spectroscopic analysis of monolayer films at the air-water interface. J. Phys. Chem 1994, 98, 8424–8430. [Google Scholar]
- Bin, X; Zawisza, I; Goddard, JD; Lipkowski, J. Electrochemical and PM-IRRAS studies of the effect of the static electric field on the structure of the DMPC bilayer supported at a Au(111) electrode surface. Langmuir 2005, 21, 330–347. [Google Scholar]
- Li, Y; Michael, G. Formation of membrane domains during the activation of protein kinase C. Biochemistry 1996, 35, 13966–13974. [Google Scholar]
- Bagatolli, LA; Parasassi, TE. Gratton giant phospholipid vesicles: comparison among the whole lipid sample characteristics using different preparation methods: A two photon fluorescence microscopy study. Chem. Phys. Lipids 2000, 105, 135–147. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guo, P.-F.; Huang, W.-Y.; Liu, H.-B.; Xiao, S.-J. AFM and Multiple Transmission-Reflection Infrared Spectroscopy (MTR-IR) Studies on Formation of Air-Stable Supported Lipid Bilayers. Int. J. Mol. Sci. 2009, 10, 1407-1418. https://doi.org/10.3390/ijms10031407
Guo P-F, Huang W-Y, Liu H-B, Xiao S-J. AFM and Multiple Transmission-Reflection Infrared Spectroscopy (MTR-IR) Studies on Formation of Air-Stable Supported Lipid Bilayers. International Journal of Molecular Sciences. 2009; 10(3):1407-1418. https://doi.org/10.3390/ijms10031407
Chicago/Turabian StyleGuo, Peng-Feng, Wen-Yi Huang, Hong-Bo Liu, and Shou-Jun Xiao. 2009. "AFM and Multiple Transmission-Reflection Infrared Spectroscopy (MTR-IR) Studies on Formation of Air-Stable Supported Lipid Bilayers" International Journal of Molecular Sciences 10, no. 3: 1407-1418. https://doi.org/10.3390/ijms10031407