Electromagnetic Biostimulation of Living Cultures for Biotechnology, Biofuel and Bioenergy Applications
Abstract
:1. Introduction
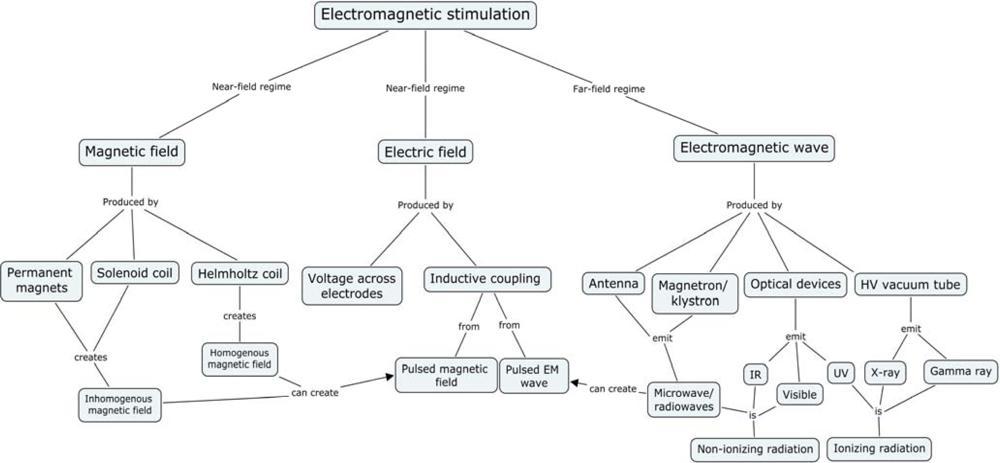
2. Electromagnetic Experiments
- Predominantly magnetic fields: Near-field regime (Permanent, slowly changing, and pulsed fields from magnetic coils)
- Predominantly electric fields: Near-field regime (Permanent or slowly changing)
- Fields with both electric and magnetic components, with ratios between 0.1 and 10: Far-field regime (typical EMF oscillation frequency is 100 kHz or more)
- Fields from (I, II, or III) with unique spatial and/or temporal topology
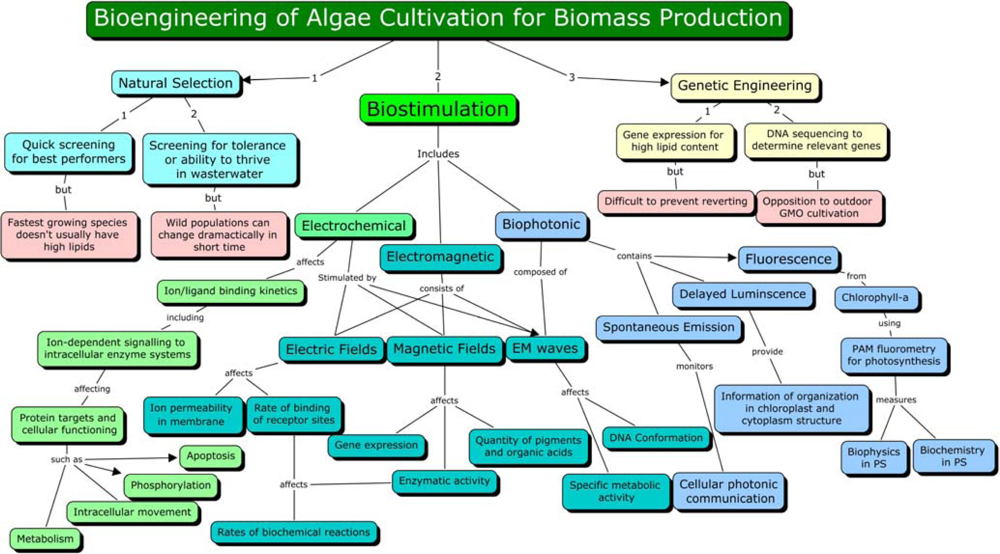
3. Biostimulation by Electromagnetic Fields
3.1. Group I: Treatments Involving Magnetic Field Predominance
3.1.1. Growth
3.1.2. Photosynthesis and Cell Constituents
3.1.3. Other Physiological Processes
3.1.3.1. Ethanol Fermentation
3.1.3.2. Anti-Oxidant Defense System
3.1.3.3. Biodegradation
3.1.4. Genetic Machinery and Molecular Mechanisms
3.2. Group II: Treatments Involving Electric Field Predominance
3.3. Group III: Treatments Involving both Electric and Magnetic Fields in Far-Field Regime
3.4. Group IV: Treatments with Spatial/Temporal Topology
3.4.1. Spatial Superposition
3.4.2. Spatial and Temporal Superposition
3.4.3. Multipolar Electromagnetic Systems
4. Mechanism of Electromagnetic Effects
4.1. Ionization and Free Radical Release
4.2. Electrochemical Models
4.2.1. Ion Cyclotron Resonance Concept
4.2.2. Stochastic Resonance Amplification
4.2.3. Long Range Molecular Organization
4.2.4. Josephson Semiconductor Model
4.2.5. Protein Symmetry
4.2.6. Physical Signals in Intermolecular Communication
4.2.7. Electromagnetic Cell Functions
4.2.8. Quantum Physics and Coherence in Biology
4.2.9. Bioelectromagnetics for Non-Chemical Communication and Signaling
4.2.10. Endogenous EMF Modeling
4.2.11. Role of Water
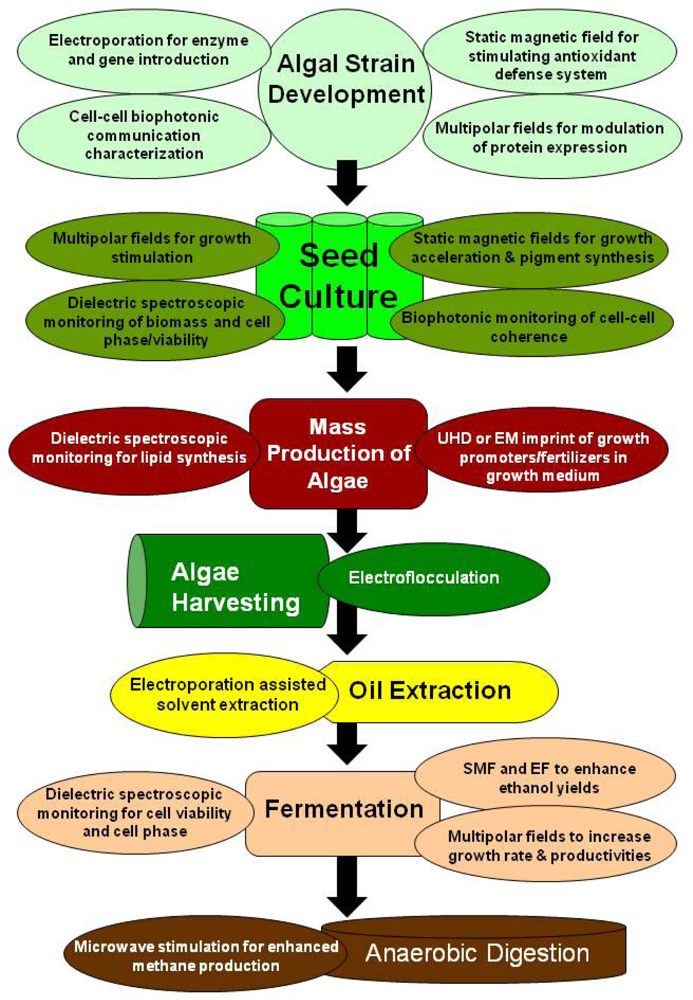
5. Electromagnetic Applications for Production of Algae Biofuels
6. Conclusions
Acknowledgments
References
- Pilla, AA; Markov, MS. Bioeffects of weak electromagnetic fields. Rev Environ Health 1994, 10, 155–169. [Google Scholar]
- Velizarov, S. Electric and magnetic fields in microbial biotechnology: Possiblities, limitations, and perspectives. Electro- Magnetobiol 1999, 18, 185–212. [Google Scholar]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv 2007, 25, 294–306. [Google Scholar]
- Rai, S. Causes and mechanism(s) of ner bioeffects. Electromagn. Biol. Med 1997, 16, 59–67. [Google Scholar]
- Banik, S; Bandyopadhyay, S; Ganguly, S; Dan, D. Effect of microwave irradiated Methanosarcina barkeri DSM-804 on biomethanation. Bioresour. Technol 2005, 97, 819–823. [Google Scholar]
- Del Re, B; Bersani, F; Agostini, C; Mesirca, P; Giorgi, G. Various effects on transposition activity and survival of Escherichia coli cells due to different ELF-MF signals. Radiat. Environ. Biophys 2004, 43, 265–270. [Google Scholar]
- Dutta, S; Verma, M; Blackman, C. Frequency-dependent alterations in enolase activity in Escherichia coli caused by exposure to electric and magnetic fields. Bioelectromagnetics 1994, 15, 377–383. [Google Scholar]
- Justo, OR; Pérez, VH; Alvarez, DC; Alegre, RM. Growth of Escherichia coli under extremely low-frequency electromagnetic fields. Appl. Biochem. Biotechnol 2006, 134, 155–163. [Google Scholar]
- Belyaev, IY; Alipov, YD; Matronchik, AY. Cell density dependent response of E. Coli cells to weak ELF magnetic fields. Bioelectromagnetics 1998, 19, 300–309. [Google Scholar]
- Chang, YHD; Grodzinsky, AJ; Wang, DIC. Augmentation of mass transfer through electrical means for hydrogel-entrapped Escherichia coli cultivation. Biotechnol. Bioeng 1995, 48, 149–157. [Google Scholar]
- Cellini, L; Grande, R; Campli, ED; Bartolomeo, SD; Giulio, MD; Robuffo, I; Trubiani, O; Mariggio, MA. Bacterial response to the exposure of 50 Hz electromagnetic fields. Bioelectromagnetics 2008, 29, 302–311. [Google Scholar]
- Zrimec, A; Jerman, I; Lahajnar, G. Alternating electric fields stimulate ATP synthesis in Escherichia coli. Cell. Mol. Biol. Lett 2002, 7, 172–174. [Google Scholar]
- Zavalin, A; Collins, WE; Morgan, S. A Compensation Zone of Multipolar System of EM Fields Stimulates Bacterial Growth. Proceeds of the 24th Meeting of Bioelectromagnetics Society, Quebec, Canada; 2002; pp. 8–9. [Google Scholar]
- Zavalin, A; Lensky, V; McCarrol, P; Westbrook, R; Collins, WE; Morgan, S. Biostimulation of microorganisms exposed to multipolar systems of mutually compensated EMF. Bioelectromagnetics 2009. [Google Scholar]
- Erygin, GD; Pchedlkina, VV; Kulikova, AK; Rurinova, NG; Bezborodov, AM; Gogolev, MN. Influence on microorganism growth and development of nutrient medium treatment with magnetic field. Prikl. Biokhim. Mikrobiol 1988, 24, 257–263. [Google Scholar]
- Ramon, C; Martin, JT; Powell, MR. Low-level, magnetic-field-induced growth modification on Bacillus subtilis. Biolelectromagnetics 1987, 8, 275–282. [Google Scholar]
- Moore, RL. Biological effects of magnetic fields: Studies with microorganisms. Can. J. Microbiol 1979, 25, 1145–1151. [Google Scholar]
- Hönes, I; Pospischil, A; Berg, H. Electrostimulation of proliferation of the denitrifying bacterium Pseudomonas stutzeri. Bioelectrochem. Bioenerg 1998, 44, 275–277. [Google Scholar]
- Kerns, G; Bauer, E; Berg, H. Electrostimulation of cellulase fermentation by pulsatile electromagnetically induced currents. Bioelectrochem. Bioenerg 1993, 32, 89–94. [Google Scholar]
- Grosse, H-H. Electrostimulation during fermentation. Bioelectrochem. Bioenerg 1988, 20, 279–285. [Google Scholar]
- Thiemann, W; Wagner, E. Die Wirkung eines homogenen Magnetfeldes auf das Wachstum von Micrococcus denitrificans. Z. Naturforsch 1970, 25b, 1020–1023. [Google Scholar]
- Lei, C; Berg, H. Electromagnetic window effects on proliferation rate of Corynebacterium glutamicum. Bioelectrochem. Bioenerg 1998, 45, 261–265. [Google Scholar]
- Rao, TBMLR; Sonolikar, RL; Saheb, SP. Influence of magnetic field on the performance of bubble columns and airlift bioreactor with submersed microorganisms. Chem. Eng. Sci 1997, 52, 4155–4160. [Google Scholar]
- Bustard, M; Rollan, A; McHale, AP. The effect of pulse voltage and capacitance on biosorption of uranium by biomass derived from whiskey distillery spent wash. Bioprocess Eng 1998, 18, 59–62. [Google Scholar]
- Utsunomiya, T; Yamane, Y-I; Watanabe, M; Sasaki, K. Stimulation of porphyrin production by application of an external magnetic field to a photosynthetic bacterium, Rhodobacter sphaeroides. J. Biosci. Bioeng 2003, 95, 401–404. [Google Scholar]
- Hirano, M; Ohta, A; Abe, K. Magnetic field effects on photosynthesis and growth of the cyanobacterium spirulina platensis. J. Ferment. Bioeng 1998, 86, 313–316. [Google Scholar]
- Li, Z-Y; Guo, S-Y; Lin, L; Cai, M-Y. Effects of electromagnetic field on the batch cultivation and nutritional compostion of Spirulina platensis in an air-lift photobioreactor. Bioresour. Technol 2007, 98, 700–705. [Google Scholar]
- Pakhomov, AG; Akyel, Y; Pakhomova, ON; Stuck, BE; Murphy, MR. Current state and implications of research on biological effects of millimeter waves: A Review of the literature. Bioelectromagnetics 1998, 19, 393–413. [Google Scholar]
- Singh, SS; Tiwari, SP; Abraham, J; Rai, S; Rai, AK. Magnetobiological effects on a cyanobacterium, Anabaena doliolum. Electromagn. Biol. Med 1994, 13, 227–235. [Google Scholar]
- Wang, H-Y; Zeng, X-B; Guo, S-Y; Li, Z-T. Effects of magnetic field on the antioxidant defense system of recirculation-cultured Chlorella vulgaris. Bioelectromagnetics 2008, 29, 39–46. [Google Scholar]
- Takahaski, F; Kamezaki, T. Effect of magnetism of growth of Chlorella. Hakkokogaku 1985, 63, 71–74. [Google Scholar]
- Yamaoka, Y; Takimura, O; Fuse, H; Kamimura, K. Effect of magnetism on growth of Dunaliella salina. Res. Photosynth 1992, 3, 87–90. [Google Scholar]
- Sommerfeld, M; Chen, W; Hu, Q; Giorgi, D; Navapanich, T; Ingram, M; Erdman, R. Application of Electroporation for Lipid Extraction from Microlalgae; Algae Biomass Summit: Seattle, WA, USA, 2008. [Google Scholar]
- Nimitan, E; Topola, N. Influence of magnetic fields on the dehydrogenase activity of Saccharomyces cerevisiae. Analele S. Univ. Al. I. Cuza’ din Iasi. Serie noua. Biologie 1972, 18, 259–264. [Google Scholar]
- Perez, V; Reyes, A; Justo, O; Alvarez, D. Bioreactor coupled with electromagnetic field generator: Effects of extremely low frequency electromagnetic fields on ethanol production by Saccharomyces cerevisiae. Biotechnol. Prog 2007, 23, 1091–1094. [Google Scholar]
- Fiedler, U; Grobner, U; Berg, H. Electrostimulation of yeast proliferation. Bioelectrochem. Bioenerg 1995, 38, 423–425. [Google Scholar]
- Mehedintua, M; Berg, H. Proliferation response of yeast Saccharomyces cerevisiae on electromagnetic field parameters. Bioelectrochem. Bioenerg 1997, 43, 67–70. [Google Scholar]
- Nakanishi, K; Tokuda, H; Soga, T; Yoshinaga, T; Takeda, M. Effect of electric current on growth and alcohol production by yeast cells. J. Ferment. Bioeng 1998, 85, 250–253. [Google Scholar]
- Grundler, W; Keilmann, F; Fröhlich, H. Resonant growth rate response of yeast cells irradiated by weak microwaves. Phys. Lett. A 1977, 62, 463–466. [Google Scholar]
- Russel, DN; Webb, SJ. Metabolic response of Danaüs archippus and Saccharomyces cerevisiae to weak oscillatory magnetic fields. Int. J. Biometereol 1981, 25, 257–262. [Google Scholar]
- Schaarschmidt, B; Lamprecht, L; Müller, K. Influence of a magnetic field on the UV-sensitivity in yeast. Z. Naturforsch 1974, 29c, 447–448. [Google Scholar]
- Pereira, MR; Nutini, LG; Fardon, JC; Cook, ES. Cellular respiration in intermittent magnetic fields. Proc. Soc. Exp. Biol. Med 1967, 124, 573–576. [Google Scholar]
- McCabe, A; Barron, N; McHale, L; McHale, AP. Increased efficiency of substrate utilization by exposure of the thermotolerant yeast strain, Kluyveromyces marxianus IMB3 to electric-field stimulation. Biotechnol. Tech 1995, 9, 133–136. [Google Scholar]
- Goodman, EM; Greenebaum, B; Marron, MT. Effects of extremely low frequency electromagnetic fields on physarum polycephalum. Radiat. Res 1976, 66, 531–540. [Google Scholar]
- Marron, MT; Goodman, EM; Greenebaum, B. Effects of weak electromagnetic fields on Physarum polycephalum: Mitotic delay in heterokaryons and decreased respiration. Experientia 1978, 34, 589–591. [Google Scholar]
- Marron, MT; Goodman, EM; Greenebaum, B; Tipnis, P. Effects of sinusoidal 60-Hz electric and magnetic fields on ATP and oxygen levels in the slime mold, Physarum polycephalum. Bioelectromagnetics 1986, 7, 307–314. [Google Scholar]
- Genkov, D; Cvetkova, A; Atmadzov, P. The effect of the constant magnetic field upon the growth and development of T. vaginalis. Folia Med. (Plovdiv) 1974, 16, 95–99. [Google Scholar]
- Dihel, LE; Smith-Sonneborn, J; Middaugh, CR. Effects of extremely low frequency electromagnetic field on the cell division rate and plasma membrane of Paramecium tetraurelia. Biolelectromagnetics 1985, 6, 61–71. [Google Scholar]
- Tabrah, FL; Guernsey, DL; Chou, S-C; Batkin, S. Effect of alternating magnetic fields (60–100 Gauss, 60 Hz) on Tetrahymena pyriformis. J. Life Sci 1978, 8, 73–77. [Google Scholar]
- Pirt, MW; Pirt, SJ. Production of biomass and starch by Chlorella in chemostat culture. J. Appl. Chem. Biotechnol 1977, 1917, 643–650. [Google Scholar]
- Binhi, VN. Theoretical concepts in magnetobiology. Electromagn. Biol. Med 2001, 20, 43–58. [Google Scholar]
- Nakasono, S; Saiki, H. Effect of ELF magnetic fields on protein synthesis in Escherichia coli K12. Radiat. Res 2000, 154, 208–216. [Google Scholar]
- Nakasono, S; Laramee, C; Saiki, H; McLeod, KJ. Effect of power-frequency magnetic fields on genome-scale gene expression in Saccharomyces cerevisiae. Radiat. Res 2003, 160, 25–37. [Google Scholar]
- Gao, W; Liu, Y; Zhou, J; Pan, H. Effects of a strong static magnetic field on bacterium Shewanella oneidensis: An assessment by using whole genome microarray. Bioelectromagnetics 2005, 26, 558–563. [Google Scholar]
- Fologea, D; Vassu-Dimov, T; Stoica, I; Csutak, O; Radu, M. Increase of Saccharomyces cerevisiae plating efficiency after treatment with bipolar electric pulses. Bioelectrochem. Bioenerg 1998, 46, 285–287. [Google Scholar]
- Belyaev, IY; Alipov, YD; Shcheglov, VS. Chromosome DNA as a target of Resonant interaction between Esherichia coli cells and low-intensity millimeter waves. Electro-Magnetobiol 1992, 11, 97–108. [Google Scholar]
- Engstrom, S; Markov, M; McLean, M; Holcomb, R; Marko, J. Effects of non-uniform static magnetic fields on the rate of myosin phosphorylation. Bioelectromagnetics 2002, 23, 475–479. [Google Scholar]
- Smith, S; McLeod, BR; Liboff, AR; Cooksey, K. Calcium cyclotron resonance and diatom mobility. Bioelectromagnetics 1987, 8, 215–227. [Google Scholar]
- Blackman, C; Benane, SG; House, DE; Elliott, DJ. Importance of alignment between local DC magnetic field and an oscillating magnetic field in response to brain tissue in vitro and in vivo. Bioelectromagnetics 1990, 11, 159–167. [Google Scholar]
- Reese, J; Frazier, ME; Morris, JE; Buschbom, RL; DL, M. Evaluation of changes in diatom mobility after exposure to 16-Hz electromagnetic fields. Bioelectromagnetics 1991, 12, 21–25. [Google Scholar]
- Blackman, C; Blanchard, JP; Benane, SG; House, DE. Effect of AC and DC magnetic field orientation on nerve cells. Biochem. Biophys. Res. Commun 1996, 220, 807–811. [Google Scholar]
- Pilla, AA; Kaufman, JJ; Ryaby, JT. Electrochemical kinetics at the cell membrane: A physicochemical link for electromagnetic bioeffects. In Mechanistic Approaches to Interaction of Electric and Electromagnetic Fields with Living Systems; Blank, M, Findl, E, Eds.; 1987; pp. 39–61. [Google Scholar]
- Lensky, V. Generation of multipolar electromagnetic energy. US Patent Application: 20,080,112,1112006. [Google Scholar]
- Rein, G. Utilization of a cell culture bioassay for measuring quantum field generated from a modified caduceus coil. Proceedings of the 26th International Society of Energy Conversion and Engineering Conference, Boston, MA, USA, 1991; pp. 400–403.
- Ho, M-W; French, A; Haffegee, J; Saunders, PT. Can weak magnetic fields (or potentials) affect pattern formation? In Bioelectrodynamics and Biocommunication; Ho, M-W, Popp, FA, Warnke, U, Eds.; World Scientific: Singapore, 1994; pp. 195–212. [Google Scholar]
- Rein, G. Bioinformation within the biofield: Beyond bioelectromagnetics. J. Altern. Complem. Med 2004, 10, 59–68. [Google Scholar]
- Repacholi, MH; Greenebaum, B. Interaction of static and extremely low frequency electric and magnetic fields with living systems: Health effects and research needs. Bioelectromagnetics 1999, 20, 133–201. [Google Scholar]
- Liboff, AR. Interactions between Electromagnetic Fields and Cells; Plenum Press: New York, NY, USA, 1985. [Google Scholar]
- Pazur, A; Schimek, C; Galland, P. Magnetoreception in microorganisms ad fungi. Cent. Eur. J. Biol 2007, 2, 597–659. [Google Scholar]
- Kaiser, F. External signals and internal oscillation dynamics: Biophysical aspects and modelling approaches for interactions of weak electromagnetic fields at the cellular level. Bioelectrochem. Bioenerg 1996, 41, 3–18. [Google Scholar]
- McLeod, BR; Liboff, A; Smith, S. Electromagnetic gating in ion channels. J. Theor. Biol 1992, 158, 15–32. [Google Scholar]
- Del Guidice, E; De Ninno, A; Fleischmann, M; Mengoli, G; Milani, M; Talpo, G; Vitiello, G. Coherent quantum electrodynamics in living matter. Electromagn. Biol. Med 2005, 24, 199–210. [Google Scholar]
- Pilla, AA. Electrochemical information transfer at living cell membranes. Ann. N. Y. Acad. Sci 1974, 238, 149–167. [Google Scholar]
- Wojtczak, L; Romanowski, S. Simple model of intermembrane communiation by means of collective excitations modified by an electric field. Bioelectrochem. Bioenerg 1996, 41, 47–51. [Google Scholar]
- Pilla, AA; Muehsam, DJ; Markov, MS; Sisken, BF. EMF signals and ion/ligand binding kinetics: Prediction of bioeffective waveform parameters. Bioelectrochem. Bioenerg 1999, 48, 27–34. [Google Scholar]
- Pilla, AA; Muehsam, DJ; Markov, MS. A dynamical systems/Larmor precession model for weak magnetic field bioeffects: Ion binding and orientation of bound water molecules. Bioelectrochem. Bioenerg 1997, 43, 239–249. [Google Scholar]
- Markov, MS; Pilla, AA. Weak static magnetic field modulation of myosin phosphorylation in a cell-free preparation: Calcium dependence. Bioelectrochem. Bioenerg 1997, 43, 233–238. [Google Scholar]
- Binhi, VN. Interference of ion quantum states within protein explains weak magnetic field’s effect on biosystems. Electro- Magnetobiol 1997, 16, 203–214. [Google Scholar]
- Solov’yov, IA; Chandler, DE; Schulten, K. Magnetic field effects in Arabidopsis thaliana Cryptochrome-1. Biophys. J 2007, 92, 2711–2726. [Google Scholar]
- Ritz, T; Adem, S; Schulten, K. A model for photoreceptor-based magnetoreception in birds. Biophys. J 2000, 78, 707–718. [Google Scholar]
- Ahmad, M; Galland, P; Ritz, T; Wiltschko, R; Wiltschko, W. Magnetic intensity affects cryptochrome-dependent responses in Arabidopsis thaliana. Planta 2006, 225, 177–180. [Google Scholar]
- Hunter, P. A quantum leap in biology. EMBO 2006, 7, 971–974. [Google Scholar]
- Wiltschko, R; Wiltschko, W. Magnetoreception. Bioessays 2006, 28, 157–168. [Google Scholar]
- Wiesenfeld, K; Moss, F. Stochastic resonance and the benefits of noise: From ice ages to crayfish and SQUIDs. Nature 1995, 373, 33–36. [Google Scholar]
- Kruglikov, IL; Dertinger, H. Stochastic resonance as a possible mechanism of amplification of weak electric signals in living cells. Bioelectromagnetics 1994, 15, 539–547. [Google Scholar]
- Pollack, GH. The role of aqueous interfaces in the cell. Adv. Colloid Interface Sci 2003, 103, 173–196. [Google Scholar]
- Tyner, KM; Kopelman, R; Philbert, MA. “Nanosized voltmeter” enables cellular-wide electric field mapping. Biophys. J 2007, 93, 1163–1174. [Google Scholar]
- Collins, PJ. Liquid Crystal’s Delicate Phase of Matter; Princeton University Press: Princeton, NJ, USA, 1990. [Google Scholar]
- Ho, M-W. The Rainbow and the Worm-The Physics of Organisms, 2nd ed; World Scientific Publishing Co: Singapore, 1998; p. 272. [Google Scholar]
- Ho, M-W. Bioenergetics and the coherence of organisms. Neuronetwork World 1995, 5, 733–750. [Google Scholar]
- Del Guidice, E; Doglia, S; Milani, M; Smith, CW; Vitiello, G. Magnetic flyx quantization and Josephson behaviour in living systems. Phys. Scr 1989, 40, 786–791. [Google Scholar]
- Costato, M; Milani, M; Spinoglio, L. Quantum mechancs: A breakthrough into biological system dynamics. Bioelectrochem. Bioenerg 1996, 41, 27–30. [Google Scholar]
- Fröhlich, H; Kremer, F. Coherent excitations in biological systems. In Biological Coherence and Response to External Stimuli; Fröhlich, H, Ed.; Springer-Verlag: Berlin, Germany, 1983. [Google Scholar]
- Merlino, A; Sica, F; Mazzarella, L. Approximate values for force constant and wave number associated with a low-frequency concerted motion in proteins can be evaluated by a comparison of X-ray structures. J. Phys. Chem. B 2007, 111, 5483–5486. [Google Scholar]
- Goodsell, D; Olson, A. Structural symmetry and protein function. Annu. Rev. Biophys. Biomol. Struct 2000, 29, 105–153. [Google Scholar]
- Trushin, MV. Studies on distant regulation of bacterial growth and light emission. Microbiology 2003, 149, 363–368. [Google Scholar]
- Pokorný, J; Hašek, J; Jelínek, F. Electromagnetic field on microtubules: Effects on transfer of mass particles and electrons. JBP 2005b, 31, 501–514. [Google Scholar]
- Presman, AS. Electromagnetic Fields and Life; Plenum Press: New York, NY, USA, 1970; p. 336. [Google Scholar]
- Pokorný, J; Hašek, J; Jelínek, F. Endogenous Electric field and organization of living matter. Electromagn. Biol. Med 2005a, 24, 185–197. [Google Scholar]
- Smith, CW. Quanta and coherence effects in water and living systems. J. Altern. Complem. Med 2004, 10, 69–78. [Google Scholar]
- Cifra, M; Vaniš, J; Kučera, O; Hašek, J; Frýdlova, I; Jelínek, F; Šaroch, J; Pokorný, J. Electrical Vibrations of Yeast Cell Membrane. In Progress In Electromagnetics Research Symposium; Prague; Czech Republic, 2007; pp. 215–220. [Google Scholar]
- Cifra, M; Pokorný, J; Jelínek, F; Hašek, J. Measurements of Yeast Cell Electrical Oscillations around 1 kHz. Progress In Electromagnetics Research Symposium, Cambridge, MA, USA, 2008; pp. 780–784.
- Pelling, AE; Sehati, S; Gralla, EB; Valentine, JS; Gimzewski, JK. Local nanomechanical motion of the cell qall of Saccharomyces cerevisiae. Science 2004, 305, 1147–1150. [Google Scholar]
- Matsuhashi, M; Pankrushina, AN; Endoh, K; Watanabe, H; Ohshima, H; Tobi, M; Endo, S; Mano, Y; Hyodo, M; Kaneko, T; Otani, S; Yoshimura, S. Bacillus carboniphilus cells respond to growth-promoting physical signals from cells of homologous and heterologous bacteria. J. Gen. Appl. Microbiol 1996, 42, 315–323. [Google Scholar]
- Matsuhashi, M; Pankrushina, AN; Takeuchi, S; Ohshima, H; Miyoi, H; Endoh, K; Murayama, K; Watanabe, H; Endo, S; Tobi, M; Mano, Y; Hyodo, M; Kobayashi, T; Kaneko, T; Otanu, S; Yoshimura, S; Harata, A; Sawada, T. Production of sound waves by bacterial cells and the response of bacterial cells to sound. J. Gen. Appl. Microbiol 1998, 44, 49–55. [Google Scholar]
- Brookes, JC; Hartoutsiou, F; Horsfield, AP; Stoneham, AM. Could humans recognize odor by phonon assisted tunneling. Phys. Rev. Lett 2007, 98, 1–4. [Google Scholar]
- Turin, L. A method for the calculation of odor character from molecular structure. J. Theor. Biol 2002, 216, 367–385. [Google Scholar]
- Leegwater, JA. Coherent versus incoherent energy transfer and trapping in photosynthetic antenna complexes. J. Phys. Chem 1996, 100, 14403–14409. [Google Scholar]
- Frauenfelder, H; Wolynes, PG; Austin, RH. Biological Physics. Rev. Mod. Phys 1999, 71, 419–430. [Google Scholar]
- Engel, GS; Calhoun, TR; Read, EL; Ahn, T-K; Mancal, T; Cheng, Y-C; Blankenship, RE; Fleming, GR. Evidence for wavelike energy transfer through quantum coherence in photosynthetic systems. Nature 2007, 446, 782–786. [Google Scholar]
- Popp, FA. On the coherence of ultraweak photon emission from living tissues. In Photon Emission from Biological Systems; Jeżowska-Trzebiatowska, B, Kochel, B, Sławiński, J, Stręk, W, Eds.; World Scientific Publishing Co, Inc.: Singapore, 1987. [Google Scholar]
- Bischof, M. Introduction to integrative biophysics. In Integrative Biophysics- Biophotonics; Popp, FA, Beloussov, LV, Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2003; pp. 1–116. [Google Scholar]
- Musumeci, F. Physical basis and application of delayed luminescence. In Integrative Biophysics-Biophotonics; Popp, FA, Beloussov, LV, Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2003; pp. 203–230. [Google Scholar]
- van Wijk, R; van Wijk, EPA. Oscillations in ultraweak photon emission of Acetabularia acetabulum (L.). Indian J. Exp. Biol 2003, 41, 411–418. [Google Scholar]
- Trushin, MV. Distant non-chemical commuication in various biological systems. Riv. Biol./Biol. Forum 2004, 97, 409–442. [Google Scholar]
- Vilenskaya, L; Kirkin, AF. Non-chemical distant interactions between cell in a culture. Parapsychol. USSR 1982, 5, 25–29. [Google Scholar]
- Kaznacheyev, VP. Sverkhslabye Izlucheniia v Mezhkletochnykh Vzaimodeistviiakh (in Russian); Nauka: Novosibirsk, USSR, 1981. [Google Scholar]
- Kaznacheyev, VP. Electromagnetic bioinformation in intercellular interactions. PSI Res 1982, 1, 47–76. [Google Scholar]
- Kaznacheyev, VP; Shurin, SP; Mikhailova, LP; Ignatovish, NV. Apparent information transfer between two groups of cells. Psychoenerg. Syst 1974a, 1, 37–38. [Google Scholar]
- Kaznacheyev, VP; Mikhailova, LP; Shurin, SP. Informational interactions in biological systems caused by electromagnetic radiation of the optical range. Parapsychol. USSR 1974b, 5, 13–24. [Google Scholar]
- Kaznacheyev, VP; Shurin, SP; Mikhailova, LP; Ignatovish, NV. Distant intercellular interactions in a system of two tissue cultures. Psychoenerg. Syst 1976, 1, 141–142. [Google Scholar]
- Bearden, TE. Gravitobiology; Tesla Book Co: Chula Vista, CA, USA, 1991. [Google Scholar]
- Popp, FA. Biophotons- background, experimental results, theoretical approach and applications. In Integrative Biophysics-Biophotonic; Popp, FA, Beloussov, L, Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2003; pp. 387–438. [Google Scholar]
- Schamhart, DHJ; Wijk, RV. Photon emission and the degree of differentiation. In Photon Emission from Biological Systems; Jeżowska-Trzebiatowska, B, Kochel, B, Sławiński, J, Stręk, W, Eds.; World Scientific: Singapore, 1987. [Google Scholar]
- Galle, M; Neurohr, R; Altmann, G; Popp, FA; Nagl, W. Biophoton emission from Daphnia magna: A possible factor in the self-regulation of swarming. Cell. Mol. Life Sci 1991, 47, 457–460. [Google Scholar]
- Zrimec, A; Drinovec, L; Berden-Zrimec, M. Influence of Chemical and Physical Factors on Long-Term Delayed Fluorescence in Dunaliella tertiolecta. Electromagn. Biol. Med 2005, 24, 309–318. [Google Scholar]
- Chang, JJ; Popp, F-A; Yu, WD. Research on cell communication of P. elegans by means of photon emission. Chin. Sci. Bull 1995, 40, 76–79. [Google Scholar]
- Chang, JJ. Biological effects of electromagnetic fields on living cells. In Integrative Biophysics-Biophotonics; Popp, FA, Beloussov, L, Eds.; Kluwer Academic, Publisher: Dordrecht, The Netherlands, 2003; pp. 231–259. [Google Scholar]
- Popp, FA; Chang, JJ; Gu, Q; Ho, MW. Bioelectrodynamics and Biocommunication; Ho, MW, Popp, FA, Warnke, U, Eds.; World Scientific: Singapore, 1994; pp. 293–317. [Google Scholar]
- Farhadi, A; Forsyth, C; Banan, A; Shaikh, M; Engen, P; Fields, JZ; Keshavarzian, A. Evidence for non-chemical, non-electrical intercellular signaling in intenstinal epithelial cells. Bioelectrochemistry 2007, 71, 142–148. [Google Scholar]
- Musumeci, F; Scordino, A; Triglia, A; Blandino, G; Milazzo, I. Intercellular communication during yeast cell growth. Europhys. Lett 1999, 47, 736–742. [Google Scholar]
- Nikoleav, YA. Distant Interaction in the bacterium Pseudomonas fluorescens as a factor of adhesion regulation. Mikrobiologiia 2000, 69, 365–361. [Google Scholar]
- Nikoleav, YA. Distant Interaction between bacterial cells (In Russian). Mikrobiologiia 1992, 61, 1066–1071. [Google Scholar]
- Kaznacheyev, VP; Mikhailova, LP; Shurin, SP. Informational interactions in biological systems caused by electromagnetic radiation of the optical range. In Parapsychology in the USSR; Vilenskaya, L, Ed.; 1982; Volume 5, pp. 13–24. [Google Scholar]
- Chang, JJ; Popp, FA; Yu, WD. Biocommunication and bioluminescence of Lampyride. In Biophotonics: Non-equilibrium and Coherent Systems in Biology, Biophysics and Biotechnology; Bioinform Services CO: Moscow, Russia, 1995; pp. 267–280. [Google Scholar]
- Stapp, HP. Nonlocal character of quantum theory. AmJPh 1997, 65, 300–304. [Google Scholar]
- Whittaker, ET. On the partial differential equations of mathematical physics. Math Ann 1903, 57, 333–355. [Google Scholar]
- Whittaker, ET. On an expression of the electromagnetic field due to electrons by means of two scalar potential functions. Proc. London Math. Soc 1904, 1, 367–372. [Google Scholar]
- Barret, TW. Topological Foundations of Electromagnetism, 196th ed; World Scientific Publishing Co: Singapore, 2008; Volume 26. [Google Scholar]
- Aharonov, Y; Bohm, D. Significance of electromagnetic potentials in the quantum theory. Phys. Rev 1959, 115, 485–491. [Google Scholar]
- Aharonov, Y; Bohm, D. Further considerations on electromagnetic potentials in the quantum theory. Phys. Rev 1961, 123, 1511–1524. [Google Scholar]
- Osakabe, N; Matsuda, T; Kawasaki, T; Endo, J; Tonomura, A; Yano, S; Yamada, H. Experimental confirmation of Aharonov-Bohm effect using a toroidal magnetic field confined by a superconductor. Phys. Rev. A 1986, 34, 815–822. [Google Scholar]
- Hyland, GJ. Bio-Electromagnetism. In Integrative Biophysics-Biophotonics; Popp, FA, Beloussov, LV, Eds.; Kluwer Academic, Publsiher: Dordrecht, The Netherland, 2003; pp. 117–148. [Google Scholar]
- Phillips, M. “Classical electrodynamics” in principles of electrodynamics and relativity. In Encyclopedia of Physics; Flugge, S, Ed.; Springer Verlag: Berlin, Germany, 1962; Volume 4. [Google Scholar]
- Bearden, TE. Energy from the Vacuum: Concepts and Principles; Cheniere Press: Santa Barbara, CA, USA, 2002. [Google Scholar]
- Šrobár, F; Pokorný, J. Topology of mutual relationships implicit in the Fröhlich model. Bioelectrochem. Bioenerg 1996, 41, 31–33. [Google Scholar]
- Guillot, J-C; Robert, J. The multipolar Hamiltonian in QED for moving atoms and ions. J. Phys. A: Math. Gen 2002, 35, 5023–5039. [Google Scholar]
- Shahverdiev, EM. Chaos synchronization in the multi-feedback ikeda model. In Chaotic Dynamics: Adaptation and Self-Organizing Systems; arXiv:nlin/0405004v1Ithaca: New York, NY, USA, 2004; pp. 1–5. [Google Scholar]
- Raab, RE; De Lange, OL. Multipole Theory In Electromagnetism: Classical, Quantum, and Symmetry Aspects, with Applications; Oxford University Press: Oxford, UK, 2005; p. 235. [Google Scholar]
- Conti, D; Tomassini, A. Special symplectic six-manifolds. QJMat 2007, 58, 297–231. [Google Scholar]
- Lobyshev, VI. Water is a sensor to weak forces including electromagnetic fields of low intensity. Electromagn. Biol. Med 2005, 24, 449–461. [Google Scholar]
- Chaplin, MF. The memory of water: An overview. Homeopathy 2007, 96, 143–150. [Google Scholar]
- Markov, MS. Direct and Indirect Action of Constant Magnetic Field on Biological Subjects. Sixth International Conference on Magnet Technology (MT-6); ALFA Bratislava: Bratislava, Czechoslovakia, 1977; pp. 384–389. [Google Scholar]
- Goldsworthy, A. Biological effects of physically conditioned water. Water Res 1999, 33, 1618–1626. [Google Scholar]
- Tschulakow, AV; Yan, Y; Klimek, W. A new approach to the memory of water. Homeopathy 2005, 94, 241–247. [Google Scholar]
- Elia, V; Niccoli, M. Thermodynamics of extremely diluted aqueous solutions. Ann. N. Y. Acad. Sci 1999, 879, 241–248. [Google Scholar]
- Rey, L. Thermoluminescence of ultra-high dilutions of lithium chloride and sodium chloride. Physica A 2003, 323, 67–74. [Google Scholar]
- Lenger, K; Bajpai, RP; Drexel, M. Delayed Luminescence of high homeopathic potencies on sugar globuli. Homeopathy 2008, 97, 134–140. [Google Scholar]
- Elia, V; Baiano, S; Duro, I; Napoli, E; Niccoli, M; Nonatelli, L. Permanent physico-chemical properties of extremely diluted aqueous solutions of homeopathic medicines. Homeopathy 2004, 93, 144–150. [Google Scholar]
- Assumpção, R. Electrical impedance of HV plasma images of high dilutions of sodium chloride. Homeopathy 2008, 97, 129–133. [Google Scholar]
- Sukul, NC; Ghosh, S; Sukul, A; Sinhababu, SP. Variation in fourier transform infrared spectra of some homeopathic potencies and their diluent media. J. Altern. Complem. Med 2005, 11, 807–812. [Google Scholar]
- Sinitsyn, N; Petrosyan, VL; Yolkin, VA; Devyatkov, ND; Gulaev, YV; Betskii, OV. Particular role of an “MM wave-water medium” system in the nature. Biomed. Electron 1998, 1, 5–23. [Google Scholar]
- Geletyuk, VI; Kazachenko, VN; Chemeris, NK; Fesenko, EE. Dual effects of microwaves on signle Ca2+ activated K+ channels in cultured kidney cells. Vero. FEBS Lett 1995, 359, 85–88. [Google Scholar]
- Fesenko, EE; Gluvstein, AY. Changes in the state of water, induced by radio-frequency electromagnetic fields. FEBS Lett 1995, 367, 53–55. [Google Scholar]
- Sukul, NC; De, A; Sukul, A; Sinhababu, SP. Potentized Mercuric chloride and Mercuric iodide enhance α-amylase activity in vitro. Homeopathy 2002, 91, 217–220. [Google Scholar]
- Citro, M; Smith, CW; Scott-Morley, A; Pongratz, W; Endler, PC. Transfer of information from molecules by means of electronic amplification-preliminary results. In Ultra-High Dilution; Endler, PC, Schulte, J, Eds.; Kluwer Academic Publishers: Dordrecht, The Netherland, 1994; pp. 209–214. [Google Scholar]
- Cardella, C; De Magistris, L; Florio, E; Smith, CW. Permanent changes in the physico-chemical properties of water following exposure to resonant circuits. J. Sci. Explor 2001, 15, 501–518. [Google Scholar]
- Jerman, I; Ružič, R; Krašovec, R; Škarja, M; Mogilnicki, L. Electrical transfer of molecule information into water, its storage, and bioeffects on plants and bacteria. Electromagn. Biol. Med 2005, 24, 341–353. [Google Scholar]
- Voeikov, VL. The possible role of active oxygen in the memory of water. Homeopathy 2007, 96, 196–201. [Google Scholar]
- Elia, V; Napoli, E; Germano, R. The “Memory of Water”: An almost deciphered enigma. Dissipative structures in extremely dilute aqueous solutions. Homeopathy 2007, 96, 163–169. [Google Scholar]
- Arani, R; Bono, I; Del Guidice, E; Preparata, G. QED coherence and the thermodynamics of water. Int. J. Mod. Phys. B 1995, 9, 1813–1841. [Google Scholar]
- Strauch, B; Patel, MK; Navarro, JA; Berdichevsky, M; Yu, H-L; Pilla, AA. Pulsed magnetic fields accelerate cutaneous wound healing in rats. Plast. Reconstr. Surg 2007, 120, 425–430. [Google Scholar]
- Kloth, LC; Berman, JE; Sutton, CH. Effect of pulsed radio frequency stimulation on wound healing: A double-blind pilot study. In Electricity and Magnetism in Biology and Medicine; Bersani, F, Ed.; Plenum: New York, NY, USA, 1999; pp. 875–878. [Google Scholar]
- Aaron, RK; Lennox, D; Bunce, GE; Ebert, T. The conservative treatment of osteonecrosis of the femoral head: A comparison of core decompression and pulsing electromagnetic fields. Clin. Orthop 1989, 249, 199–208. [Google Scholar]
- Salzberg, CA; Cooper, SA; Cooper, P; Perez, P. The effects of non-thermal pulsed electromagnetic energy on wound healing; a double blind pilot study. OWM 1995, 41, 42–48. [Google Scholar]
- Zizic, TM; Hoffman, KC; Holt, PA; Hungerford, DS; O’Dell, JR; Jacobs, MA; Lewis, CG; Deal, CL; Caldwell, JR; Cholewczynski, JG. The treatment of osteoarthritis of the knee with pulsed electrical stimulation. J. Rheumatol 1995, 22, 1757–1761. [Google Scholar]
- Pennington, GM; Danley, DL; Sumko, MH. Pulsed non-thermal, high-frequency electromagnetic energy (Diapulse) in the treatment of grade I and grade II ankle sprains. Mil. Med 1993, 158, 101–104. [Google Scholar]
- Kenkre, JE; Hobbs, FD; Carter, YH. A randomized controlled trial of electromagnetic therapy in the primary care management of venous leg ulceration. Fam. Pract 1996, 13, 236–241. [Google Scholar]
- Morejón, LP; Castro palacio, JC; Velázquez Abad, L; Govea, AP. Stimulation of Pinus tropicalis M. seeds by magnetically treated water. Int. Agrophysics 2007, 21, 173–177. [Google Scholar]
- Vashisth, A; Nagarajan, S. Exposure of seeds to static magnetic field enhances germination and early growth characteristics in chickpea (Cicer arietinum L.). Bioelectromagnetics 2008, 29, 571–578. [Google Scholar]
- Ozdemir, S; Dede, OH; Koseoglu, G. Electromagnetic water treatment and water quality effect on germination, rooting, and plant growth on flower. Asian J. Wat. Environ. Pollut 2005, 2, 9–13. [Google Scholar]
- Pietruszewski, S. Effects of magnetic biostimulation of wheat seeds on germination, yields, and proteins. Int. Agrophysics 1996, 10, 51–55. [Google Scholar]
- Carbonell, MV; Martinez, E; Amaya, JM. Stimulation of germination in rice (Oryza sative L.) by a static magnetic field. Electro- Magnetobiol 2000, 19, 121–128. [Google Scholar]
- Martinez, E; Carbonell, MV; Amaya, JM. A static magnetic field of 125 mT stimulates the initial growth stages of barley. Electro- Magnetobiol 2000, 19, 271–277. [Google Scholar]
- Souza, AD; Garcia, D; Sueiro, L; Gilart, F; Poras, E; Licea, L. Pre-sowing magnetic treatments of tomato seeds increase the growth and yield of plants. Bioelectromagnetics 2006, 27, 247–257. [Google Scholar]
- Miyake, M; Asada, Y. Direct electroporation of clostridial hydrogenase into cyanobacterial cells. Biotechnol. Tech 1997, 11, 787–790. [Google Scholar]
- Joersbo, M; Brunstedt, J. Electroporation: Mechanism and transient expression, stable transformation and biological effects in plant protoplasts. Physiol. Plant 1991, 81, 256–264. [Google Scholar]
- Holt, PK; Barton, GW; Mitchell, CA. The future for electrocoagulation as a localised water treatment technology. Chemosphere 2005, 59, 355–367. [Google Scholar]
- Poelman, E; De Pauw, N; Jeurissen, B. Potential of electrolytic flocculation for recovery of micro-algae. Resour. Conserv. Recycl 1997, 19, 1–10. [Google Scholar]
- Azarian, GH; Mesdaghinia, AR; Vaezi, F; Nabizadeh, R; Nematollahi, D. Algae removal by electro-coagulation process, application for treatment of the effluent from an industrial wastewater treatment plant. Iran. J. Publ. Health 2007, 36, 57–64. [Google Scholar]
- Islam, S; Suidan, MT. Electrolytic denitrification: Long term performance and effect of current intensity. Water Res 1998, 32, 528–536. [Google Scholar]
- Kuroda, M; Watanabe, T; Umedu, Y. Simultaneous oxidation and reduction treatments of polluted water by a bio-electro reactor. Water Sci. Technol 1996, 34, 101–108. [Google Scholar]
- Kuroda, M; Watanabe, T; Umedu, Y. Simultaneous COD removal and denitrification of wastewater by bio-electro reactors. Water Sci. Technol 1997, 35, 161–168. [Google Scholar]
- Schreiber, U. Pulse-amplitude modulation (PAM) fluorometry and saturation pulse method: An overview. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Papageorgiou, GC, Govindjee, Eds.; Springer: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Papazi, A; Makridis, P; Divanach, P; Kotzabasis, K. Bioenergetic changes in the microalgal photosynthetic apparatus by extremely high CO2 concentrations induce an intense biomass production. Physiol. Plant 2008, 132, 338–349. [Google Scholar]
- Triglia, A; La Malfa, G; Musumeci, F; Leonardi, C; Scordino, A. Delayed luminescence as an indicator of tomato fruit quality. J. Food Sci 1998, 63, 512–515. [Google Scholar]
- Gheorghiu, E. Measuring living cells using dielectric spectroscopy. Bioelectrochem. Bioenerg 1996, 40, 133–139. [Google Scholar]
- Asami, K; Yonezawa, T; Wakamatsu, H; Koyanagi, N. Dielectric spectroscopy of biological cells. Bioelectrochem. Bioenerg 1996, 40, 141–145. [Google Scholar]
- Asami, K; Gheorghiu, E; Yonezawa, T. Real-time monitoring of yeast cell division by dielectric spectroscopy. Biophys. J 1999, 76, 3345–3348. [Google Scholar]
- Maskow, T; Rollich, A; Fetzer, I; Ackermann, J-U; Harms, H. On-line monitoring of lipid storage in yeasts using impedance spectroscopy. J. Biotechnol 2008, 135, 64–70. [Google Scholar]
- Lanzanò, L; Grasso, R; Gulino, M; Bellia, P; Falciglia, F; Scordino, A; Tudisco, S; Triglia, A; Musumeci, F. Corresponding measurements of delayed luminescence and impedance spectroscopy on acupuncture points. Indian J. Exp. Biol 2008, 46, 364–370. [Google Scholar]
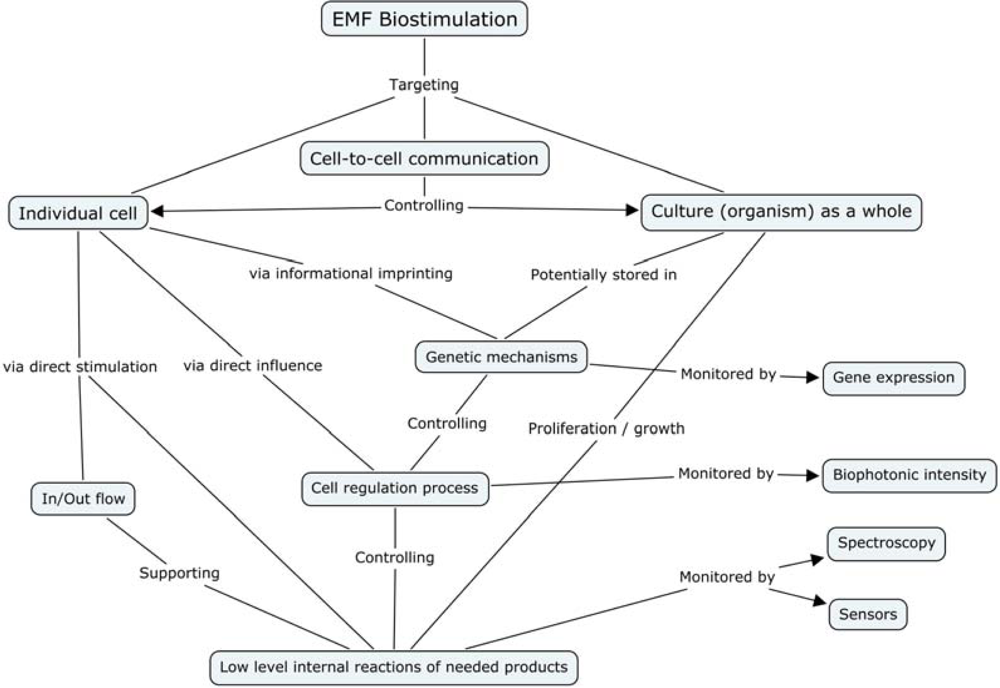
- Cell Membrane
- - Magnetic field oscillations may increase membrane permeability under ion cyclotron resonance
- - Increased circulation and selective enhancement of ion flow may affect the rate of biochemical reactions
- - Alter the rate of binding of calcium ions to enzymes or receptor sites
- - Change distribution of protein and lipid domains, and conformational changes in lipid-protein associations
- - Change internal molecular distribution of electronic charge inside lipid molecule in the membrane bilayer
- - May play the primary role in the stochastic resonance amplification process
- Chloroplast
- - May modulate the quantity of pigments, such as chlorophyll, phycocyanin, and beta-carotene
- Nucleus/DNA
- - Magnetic field affects specific gene expression
- - Individual DNA sequences may function as antennae
- - Leads to changes in DNA conformation
- - May activate different DNA sequences depending on field intensity
- - Can affect enzyme activity
- Proteins:
- - Breathing motions are the source and receiver of multipole EMF
- - Potential coupling mechanism for external multipolar influences
- Protoplasm
- - Static magnetic fields influence the speed of protoplasm movement, miotic activity, and quantity of organic acids in plants
- Whole Cell
- - Biophotonic emission and interaction with nearby cells
- - Endogenous electric field modulation may alter natural processes
- Cell Membrane
- - Magnetic field oscillations may increase membrane permeability under ion cyclotron resonance
- - Increased circulation and selective enhancement of ion flow may affect the rate of biochemical reactions
- - Alter the rate of binding of calcium ions to enzymes or receptor sites
- - Change distribution of protein and lipid domains, and conformational changes in lipid-protein associations
- - Change internal molecular distribution of electronic charge inside lipid molecule in the membrane bilayer
- - May play the primary role in the stochastic resonance amplification process
- Chloroplast
- - May modulate the quantity of pigments, such as chlorophyll, phycocyanin, and beta-carotene
- Nucleus/DNA
- - Magnetic field affects specific gene expression
- - Individual DNA sequences may function as antennae
- - Leads to changes in DNA conformation
- - May activate different DNA sequences depending on field intensity
- - Can affect enzyme activity
- Proteins:
- - Breathing motions are the source and receiver of multipole EMF
- - Potential coupling mechanism for external multipolar influences
- Protoplasm
- - Static magnetic fields influence the speed of protoplasm movement, miotic activity, and quantity of organic acids in plants
- Whole Cell
- - Biophotonic emission and interaction with nearby cells
- - Endogenous electric field modulation may alter natural processes
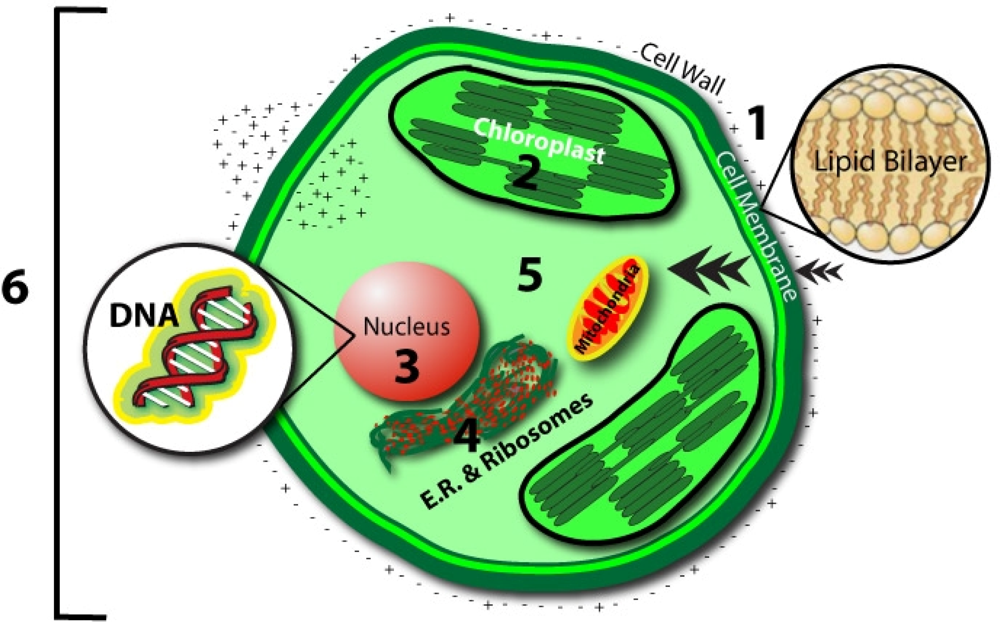
| Organism | Class* | EM Intensity | Biological effect | Reference |
|---|---|---|---|---|
| Archaea | ||||
| Methanosarcina barkeri | MW | 13.5–36.5 GHz | Increase in growth, cell count and size and methane production | [5] |
| Eubacteria | ||||
| PMF | 0.05–1 mT | Stimulated transposition activity & reduced cell viability | [6] | |
| AC MF | 16, 60 Hz | Enolase activity stimulation; Suppression of enolase activity | [7] | |
| 0.05–1 mT | Reduced transposition activity & enhanced cell viability | [6] | ||
| E. coli | OMF | 100 mT | Exposure time dependent stimulation or inhibition of cell viability | [8] |
| 30 μT | Cell density dependent changes in AVTD | [9] | ||
| DC EF | NA | Increase in growth, removal of inhibitory compounds in medium | [10] | |
| AC MF | 0.1–1 mT @ 50 Hz | Significant morphotype changes & alteration during cell division | [11] | |
| ACEF | 2.5–50 V/cm @ 0.05–100 kHz | Stimulation of membrane bound ATP synthesis, optimum at 100 Hz | [12] | |
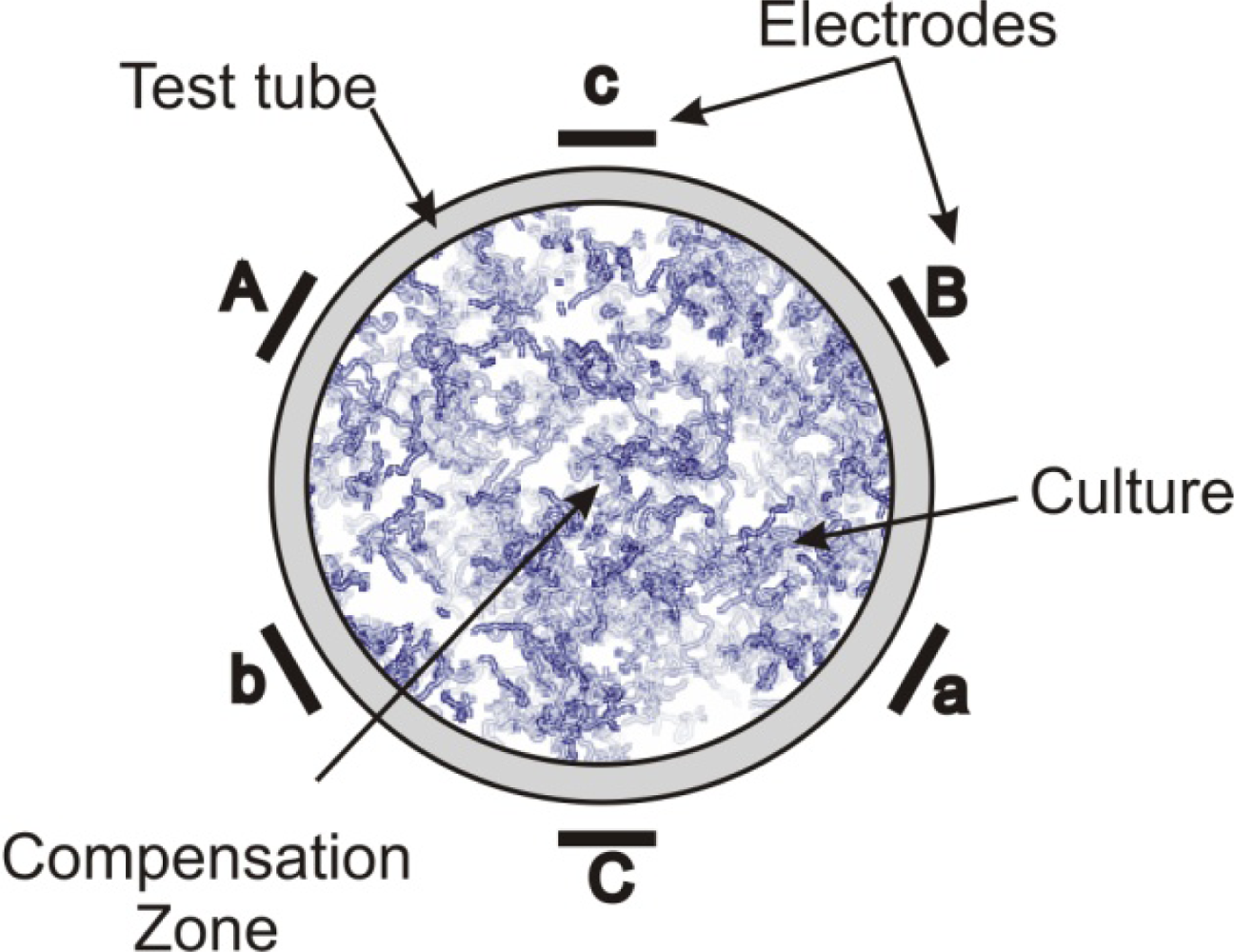
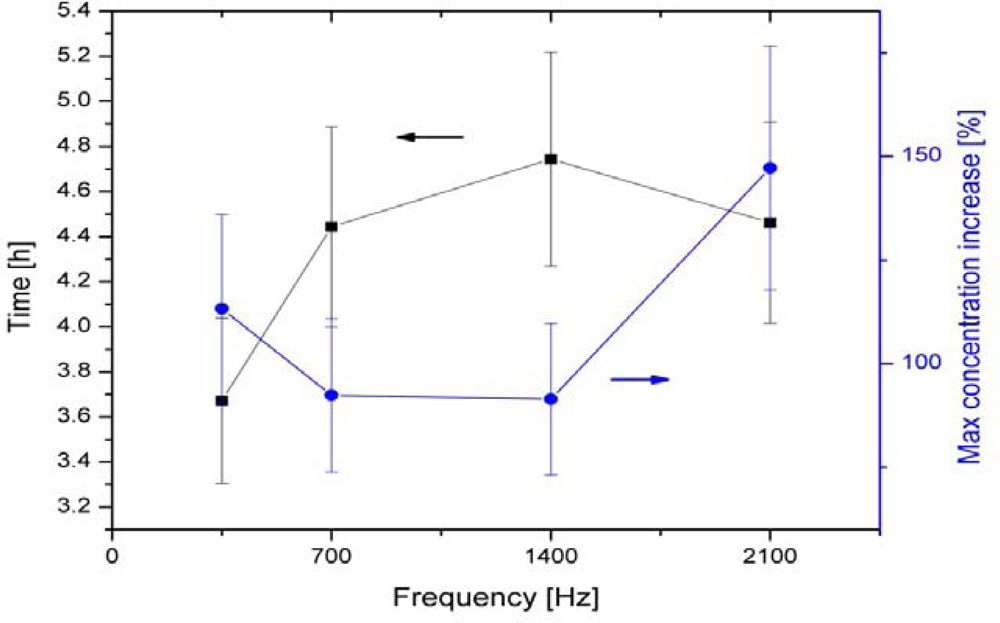
| 6-polar ACEF | 0.35–2.1 kHz for test tubes 60 Hz for Petri dishes | Increase in growth in test tubes (147 ± 24%) and colonies (42–179%) | [13,14] | |
| Bacillus cereus | 6-polar ACEF | 1 kHz | Increase in growth in tubes (196 ± 29%) and colonies | [13,14] |
| B. mucilaginosus | SMF | ~0.39 T | Increase in growth | [15] |
| B. subtilis | AC MF | 0.8, 2.5 mT, 0.8 and 1 kHz | Growth increase and interestingly a loss of intercellular cohesion | [16] |
| Paper to be seen | AC MF | 0–0.3 Hz @ 5−90 mT | Elevated or even diminished growth rates for Bacillus subtilis, Candida albicans, Halobacterium, Salmonella typhimurium, and Staphylococci | [17] |
| Pseudomonas stutzeri | PMF | 0.6–1.3 mT | Increase in growth | [18] |
| Trichoderma reesei | PMF | 1.5 mV cm−1 | Increase in growth, cellulase activity and secretion | [19] |
| Streptomyces noursei | PMF | 1.5 mV cm−1 | Increased antibiotic production, O2 evolution, glucose uptake | [20] |
| Salmonella typhimurium | OMF | 15 mT@ 0.3Hz | Growth stimulation, Mutation reversion rate unaffected | [17] |
| Micrococcus denitrificans | SMF | 500–800 mT | Growth inhibition followed by stimulation after 6 h | [21] |
| Corynebacterium glutamicum | AC MF | 4.9 mT, 50 Hz | Increase in ATP levels by about 30% | [22] |
| Natural Flora | SMF | 22 mT | Enhanced degradation of phenolic waste liquors | [23] |
| Natural Flora | PEF | 1.25 – 3.25 kVcm−1 | Enhanced biosorption of uranium | [24] |
| Bacteria & yeast | OMF | 15 mT@ 0.3 Hz | Larger increase (30%) in growth in gram –ve (Psuedomonas aeruginosa, Halobacterium halobium) than gram +ve (Bacillus subtilis, Staphylococcus epidermidis) and yeast (Candida albicans) | [17] |
| Rhodobacter sphaeroides | AC/DC MF | 0.13–0.3 T | Increase in porphyrin synthesis, Enhanced expression of 5-aminolevulinic acid dehydratase | [25] |
| Cyanobacteria | ||||
| Spirulina platensis | SMF | 10 mT | Increase in growth (50%), O2, sugar, phycocyanin | [26] |
| 250 mT | Increase in growth (22%), CNP-Uptake, Chl, minerals | [27] | ||
| MW | 7.1 mm @ 2.2mWcm−2 | Increased growth (50%) | [28] | |
| Anabaena doliolum | SMF | 300 mT | Increase in growth, pigments, carbohydrate and protein | [29] |
| Algae | ||||
| Chlorella vulgaris | SMF | 10–35 mT | Increase in growth (100%); Stimulated antioxidant defense | [30] |
| Chlorella sp. | SMF | 6–58 mT | Increase in growth (NA) | [31] |
| Dunaliella salina | SMF | 10–23 mT | Increase in growth (90%), and β-carotene | [32] |
| Scenedesmus sp. | PEF | NA | Enhanced oil extraction- Solvent+Electroporation | [33] |
| Yeast | ||||
| Saccharomyces cerevisiae | PMF | ~ 4.7 μT | Increased activity of alcohol dehydrogenase | [34] |
| OMF+SMF | 20 mT + 8 mT | Increase in ethanol, sugar utilization | [35] | |
| S. cerevisiae | OMF | 0.28–12 mT | Increase in growth | [36] |
| OMF | 0.2–12 mT @ 50 Hz | Increase in growth (25 +/− 5%) | [37] | |
| AC/DC EF | 100/10 mA | Increase in growth, organic acid production, cell budding | [38] | |
| MW | 42GHz@ < 3 mWcm−2 | Frequency dependent increase or decrease in growth | [39] | |
| 6-polar ACEF | 1 kHz | Increase in gas production (195 ± 20%) | [13,14] | |
| AC MF | 0.5 μT, 100 200 Hz | 30% reduction in respiration | [40] | |
| Saccharomyces sp. | Better UV survival in those given magnetic pretreatment | [41] | ||
| [42] | ||||
| Respiration stimulation | ||||
| S. fragilis | SMF | ~0.26 T | Increase in growth (27–36%) | [15] |
| Kluyveromyces marxianus | PEF | 0.25 kV | Increased ethanol production and cellobiose utilization | [43] |
| Physarum polycephalum | ELF EMF | 45,60,75 Hz | Delayed mitosis by 0.5 to 2 h | [44] |
| AC MF | 0.1 mT, 60 Hz | Lower ATP levels but no decreased respiration | [45,46] | |
| 0.2 mT and 60 and 75 Hz | Reduced respiration | |||
| Protozoa | ||||
| Trichomonas vaginalis | SMF | Field strength dependent growth stimulation/inhibition | [47] | |
| Ciliophora | ||||
| Paramecium tetraurelia | AC MF | 1.8 mT, 72 Hz | Ca2+ specific increase in cell division rates, absent in the presence of a Ca2+ blocker, Alterations in membrane fluidity | [48] |
| Tetrahymena pyriformis | AC MF | 10 mT, 60 Hz | Delayed cell division and increased oxygen uptake | [49] |
| Culture | Experiment | Effect | Reference |
|---|---|---|---|
| Daphnia | D.I. & S.E. | Established destructive interference found at natural population density | [125] |
| D. tertiolecta | D.I. & D.L. | Changes in external environment demonstrated dose/intensity dependent decay curves | [126] |
| P. elegans | D.I. w/E-Field | E-field stimulated distant culture’s photonic activity and synchronization | [127,128] |
| Gonyaulax sp. | D.I. | Established destructive interference and synchronization of photon pulses | [129] |
| XC tumor cells | D.I. | Dense cell culture stimulated growth rate of isolated culture via optical contact | [116] |
| Epithelial cells | D.I. w/H2O2 | Reduction in protein, increased nuclear activation, and structural damage | [130] |
| E. coli | D.I. | Synchronized growth parameters when in optical contact of Vis-IR. | [96] |
| S. cerevisiae | D.I. | Stimulation of cellular subdivision via optical coupling with culture of same type | [131] |
| P. fluorescens | D.I. | Long range interactions of an isolated culture diminished adhesion between cells of another culture | [132] |
| V. costicola | D.I. | Isolated treated culture stimulated growth of second culture of same species | [133] |
| Fibroblasts | D.I. w/Viruses | Three viral effects transferred to 72–78% of distant isolated cells | [134] |
| D.I. w/HgCl2 | Effects transferred to 78% of distant isolated cells | ||
| D.I. w/Rad | UV radiation effects transferred to 82% of distant isolated cells | ||
| L. pekennisis | S.E. | Measured coherent emission from 200–800 nm which differed between male and female specimens | [135] |
| Biomedical | Influence | Application | Reference |
|---|---|---|---|
| PEMF | Chronic wound healing, and non-union fracture healing | [172] | |
| Chronic wound healing | [173] | ||
| Treatment of osteonecrosis | [174] | ||
| Treatment of pressure ulcers in spinal-cord injuries | [175] | ||
| Treatment of osteoarthritis of the knee | [176] | ||
| Treatment of grade I & II ankle sprains | [177] | ||
| Treatment of venous leg ulceration | [178] | ||
| Agricultural | Influence | Application | Reference |
| SMF | Treated water to stimulate germination in Pinus tropicalis seeds | [179] | |
| Treated chickpea seeds increased germination, seedling and root length & size | [180] | ||
| Treated water increased plant height, branch number, and shoot dry weight | [181] | ||
| Treated wheat seeds increased germination, yields, and protein | [182] | ||
| Treated rice seeds and water increased rate and % of germination | [183] | ||
| Treated barley seeds and water increased length and weight | [184] | ||
| OMF | Treated tomato seeds for increased growth, yields, and disease resistance | [185] | |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hunt, R.W.; Zavalin, A.; Bhatnagar, A.; Chinnasamy, S.; Das, K.C. Electromagnetic Biostimulation of Living Cultures for Biotechnology, Biofuel and Bioenergy Applications. Int. J. Mol. Sci. 2009, 10, 4515-4558. https://doi.org/10.3390/ijms10104515
Hunt RW, Zavalin A, Bhatnagar A, Chinnasamy S, Das KC. Electromagnetic Biostimulation of Living Cultures for Biotechnology, Biofuel and Bioenergy Applications. International Journal of Molecular Sciences. 2009; 10(10):4515-4558. https://doi.org/10.3390/ijms10104515
Chicago/Turabian StyleHunt, Ryan W., Andrey Zavalin, Ashish Bhatnagar, Senthil Chinnasamy, and Keshav C. Das. 2009. "Electromagnetic Biostimulation of Living Cultures for Biotechnology, Biofuel and Bioenergy Applications" International Journal of Molecular Sciences 10, no. 10: 4515-4558. https://doi.org/10.3390/ijms10104515




