Journal Description
Hemato
Hemato
- formerly Bloods - is an international, peer-reviewed, open access journal on hematology, published quarterly online by MDPI. The Spanish Society of Hematology and Hemotherapy (SEHH) and the Nuclear Medicine Discovery (Nu.Me.D.) are affiliated with Hemato and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within ESCI (Web of Science), Scopus, EBSCO, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 28.1 days after submission; acceptance to publication is undertaken in 7.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
Latest Articles
Effects of Autoimmune Disorders on Myelodysplastic Syndrome Outcomes: A Systematic Review
Hemato 2024, 5(2), 208-219; https://doi.org/10.3390/hemato5020017 - 15 Jun 2024
Abstract
►
Show Figures
Background: Autoimmune disorders (ADs) are prevalent among patients with myelodysplastic syndrome (MDS), yet their impact on MDS outcomes, including overall survival (OS), mortality, and transformation to acute myeloid leukemia (AML), is not well defined. Methods: We conducted a systematic review of
[...] Read more.
Background: Autoimmune disorders (ADs) are prevalent among patients with myelodysplastic syndrome (MDS), yet their impact on MDS outcomes, including overall survival (OS), mortality, and transformation to acute myeloid leukemia (AML), is not well defined. Methods: We conducted a systematic review of articles published up to April 2024, sourced from PubMed, Web of Science, Embase, and Google Scholar, focusing on the influence of ADs on survival and AML transformation rates in MDS patients. The methodological quality of each study was assessed using the Newcastle Ottawa Scale. Results: From 8 studies that met the inclusion criteria, ADs were present in 17.5% (3074/17,481) of MDS patients. Data analysis indicated mortality rates ranging from 15.3% to 67% in MDS patients with ADs and 12% to 69% in those without. The rate of AML transformation varied from 0% to 23% in patients with ADs compared to 4% to 30% in those without. Conclusions: The influence of ADs on survival and AML transformation in MDS patients appears variable. This systematic review highlights the need for further large-scale prospective studies to clarify the relationship between ADs and MDS outcomes.
Full article
Open AccessOpinion
Tumour Microenvironment Contribution to Checkpoint Inhibitor Therapy in Classic Hodgkin Lymphoma
by
Annunziata Gloghini and Antonino Carbone
Hemato 2024, 5(2), 199-207; https://doi.org/10.3390/hemato5020016 - 3 Jun 2024
Abstract
Classic Hodgkin lymphoma (cHL) is a B-cell lymphoma in which tumour cells, the so-called Hodgkin Reed–Sternberg (HRS) cells, are admixed with non-malignant cell types that are a functional part of the disease. Immune cells, fibroblasts, specialised mesenchymal cells, and microvasculature together make up
[...] Read more.
Classic Hodgkin lymphoma (cHL) is a B-cell lymphoma in which tumour cells, the so-called Hodgkin Reed–Sternberg (HRS) cells, are admixed with non-malignant cell types that are a functional part of the disease. Immune cells, fibroblasts, specialised mesenchymal cells, and microvasculature together make up the tumour microenvironment and have functional interactions with tumour cells. HRS cells are surrounded by T and B cells admixed with plasma cells, macrophages, eosinophils, and mast cells. A cross-talk occurs between HRS cells and immune cells of the TME. This cross-talk is mediated either by a large network of cytokines and chemokines expressed by HRS cells or molecules produced by different cell types of the TME, i.e., CD30/CD30L, CD40/CD40L, OX40L/OX40, Il- 3/Il-3R, CCR5/CCL5, CD74 macrophage migration inhibitory factor/macrophages, and PD-L1/PD-1. The over-expression of CD30 and CD40, members of the TNF receptor family, is a hallmark of HRS cells. This review highlights the current development of newer therapeutic strategies as a means of immune checkpoint blockade and suggests that further research should explore innovative molecules aimed at targeting components of HL that are involved in cancer cell growth and/or immune escape. Hopefully, this will influence sensitivity or resistance to checkpoint inhibitor therapy in an individual patient.
Full article
(This article belongs to the Section Lymphomas)
►▼
Show Figures

Figure 1
Open AccessFeature PaperReview
CAR-T Therapy in Multiple Myeloma: Looking Beyond
by
Gianluca Maiorana, Giusy Antolino, Giacinto La Verde and Agostino Tafuri
Hemato 2024, 5(2), 180-198; https://doi.org/10.3390/hemato5020015 - 31 May 2024
Abstract
Multiple Myeloma is a hematological neoplasm that, over the recent few years, has benefited from numerous therapeutic options. Among the latter, CAR-T stands out as the most recent and one of the most promising treatments currently available. Despite its recent introduction, multiple CAR-T
[...] Read more.
Multiple Myeloma is a hematological neoplasm that, over the recent few years, has benefited from numerous therapeutic options. Among the latter, CAR-T stands out as the most recent and one of the most promising treatments currently available. Despite its recent introduction, multiple CAR-T products have already been approved, and research regarding cellular therapy is rapidly increasing. We conducted a comprehensive search and review of the available literature, including published studies and abstracts from recent meetings (ASH, ASCO, ASTCT, IMS), regarding Multiple Myeloma and CAR-T therapy. We describe the discovery and research regarding promising targets like the B-Cell Maturation Antigen (BCMA) and others, the origin and nature of CAR-T cells, and the recent introduction of anti-BCMA CAR-Ts Idecabtagene-vicleucel and Ciltacabtagene-autoleucel, which are currently the only approved CAR-T products for MM. Additionally, we discuss non-BCMA-targeting CAR-Ts and their clinical implications. Given the significant impact of cellular therapy, we provide an overview of its limitations and possible adverse implications, as well as related resistance mechanisms. Finally, we describe the current research aimed at improving CAR-T therapy in MM, including structural innovations and new therapeutic approaches, such as in the earlier lines of treatment and maintenance therapy.
Full article
(This article belongs to the Section Plasma Cell Disorders)
►▼
Show Figures

Figure 1
Open AccessOpinion
CAR-T and Bispecific Antibodies: The New Standard for Relapsed and Refractory Multiple Myeloma, or Reserved for Late-Line Salvage Therapy?
by
Paula Rodriguez-Otero and Thomas Martin
Hemato 2024, 5(2), 171-179; https://doi.org/10.3390/hemato5020014 - 24 May 2024
Abstract
The treatment of relapsed and refractory multiple myeloma has improved substantially in the last 5–10 years based on the development and use of several novel classes of drugs and drug combinations. These advances have led to improvements in progression-free and overall survival as
[...] Read more.
The treatment of relapsed and refractory multiple myeloma has improved substantially in the last 5–10 years based on the development and use of several novel classes of drugs and drug combinations. These advances have led to improvements in progression-free and overall survival as well as quality of life. The general tendency has been to advance drugs/combinations that have performed well in advanced disease to the earlier line settings (frontline, first/early relapse). There are several triplet drug combinations that, when used as part of first or early relapse, can provide remission durations of 3 years or longer. More recently, impressive responses have been seen with the use of targeted immunotherapeutics (chimeric antigen receptor T-cells and bispecific antibodies) in heavily pretreated patients with MM. These treatments, however, have been associated with some new and occasionally severe toxicities, including cytokine release syndrome, immune effector cell-associated neurotoxicity syndrome, and severe infections, including opportunistic infections and profound cytopenias. These potential toxicities bring into question whether these immune-targeting drugs should remain as late-line therapeutics or whether the high single-agent overall response rates mandate that these agents be used in earlier line settings. Herein, the authors provide a point and counterpoint about the future use of these agents.
Full article
(This article belongs to the Special Issue Controversies in Multiple Myeloma: A Theme Issue in Honor of Prof. Dr. Jesús San Miguel)
Open AccessReview
B- and T-/NK-Cell Lymphomas in the 2022 International Consensus Classification of Mature Lymphoid Neoplasms and Comparison with the WHO Fifth Edition
by
Elaine S. Jaffe and Antonino Carbone
Hemato 2024, 5(2), 157-170; https://doi.org/10.3390/hemato5020013 - 17 Apr 2024
Abstract
The World Health Organization (WHO) “Classification of Tumours of Haematopoietic and Lymphoid Tissues”, published in 2001 and subsequently updated in 2008 and 2017, defined disease entities based on morphologic and phenotypic characteristics, clinical features, and genomic findings. Recently, the criteria for the diagnosis
[...] Read more.
The World Health Organization (WHO) “Classification of Tumours of Haematopoietic and Lymphoid Tissues”, published in 2001 and subsequently updated in 2008 and 2017, defined disease entities based on morphologic and phenotypic characteristics, clinical features, and genomic findings. Recently, the criteria for the diagnosis of many lymphoma entities have been refined in a proposal by the International Consensus Classification (ICC). Some provisional categories have now been recognized as “definite” entities, while other categories have undergone major revision. This article reports on the major revisions in the criteria and definition of B- and T-/NK-cell lymphomas by the ICC system.
Full article
(This article belongs to the Section Lymphomas)
►▼
Show Figures
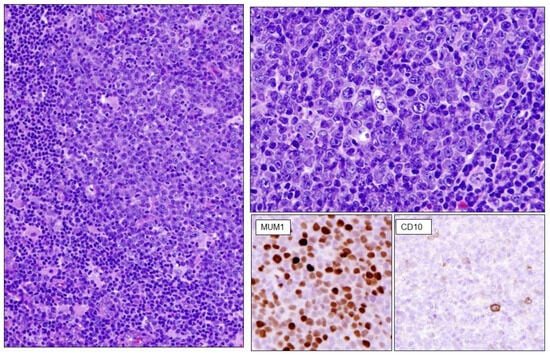
Figure 1
Open AccessOpinion
The Role of Autologous Stem Cell Transplantation in the Treatment of Newly Diagnosed Multiple Myeloma: Is It Time to Rethink the Paradigm in the Era of Targeted Therapy?
by
Paul G. Richardson
Hemato 2024, 5(2), 144-156; https://doi.org/10.3390/hemato5020012 - 9 Apr 2024
Abstract
High-dose melphalan (HDM) plus autologous stem cell transplant (ASCT) remains a standard-of-care treatment approach for eligible patients with newly diagnosed multiple myeloma (NDMM) based on demonstrated superiority in terms of progression-free survival (PFS) versus nontransplant approaches. Very high rates of minimal residual disease
[...] Read more.
High-dose melphalan (HDM) plus autologous stem cell transplant (ASCT) remains a standard-of-care treatment approach for eligible patients with newly diagnosed multiple myeloma (NDMM) based on demonstrated superiority in terms of progression-free survival (PFS) versus nontransplant approaches. Very high rates of minimal residual disease (MRD)-negative responses are also being seen with novel triplet and quadruplet induction regimens plus HDM-ASCT. However, recent clinical trials have shown no overall survival benefit with transplant versus nontransplant approaches. Furthermore, HDM is associated with several important downsides, including acute and long-term toxicities, transient decreases in quality of life, the need for hospitalization, an increased mutational burden at relapse, and an elevated risk of second primary malignancies. In this context, given the highly heterogeneous nature of MM in the NDMM patient population, as well as the continued emergence of novel agents and treatment approaches, there is an increasing rationale for considering deferred HDM-ASCT approaches in selected patients. Approaches under investigation include MRD-adapted therapy and the use of novel immune-based therapies as alternatives to HDM-ASCT. Ongoing developments in understanding the pathobiology and prognostic factors in NDMM, plus immune profiling and routine MRD evaluation, will result in novel, HDM-sparing treatment paradigms, enabling further improvement in patient outcomes.
Full article
(This article belongs to the Special Issue Controversies in Multiple Myeloma: A Theme Issue in Honor of Prof. Dr. Jesús San Miguel)
►▼
Show Figures
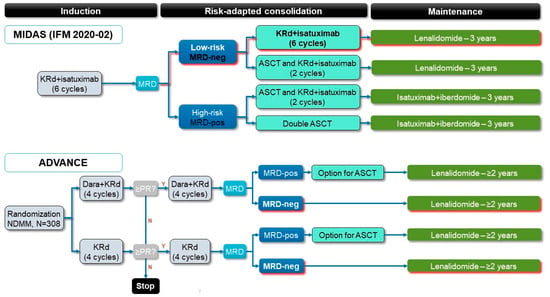
Figure 1
Open AccessArticle
Artificial Intelligence, Lymphoid Neoplasms, and Prediction of MYC, BCL2, and BCL6 Gene Expression Using a Pan-Cancer Panel in Diffuse Large B-Cell Lymphoma
by
Joaquim Carreras and Naoya Nakamura
Hemato 2024, 5(2), 119-143; https://doi.org/10.3390/hemato5020011 - 9 Apr 2024
Abstract
Background: Artificial intelligence in medicine is a field that is rapidly evolving. Machine learning and deep learning are used to improve disease identification and diagnosis, personalize disease treatment, analyze medical images, evaluate clinical trials, and speed drug development. Methods: First, relevant
[...] Read more.
Background: Artificial intelligence in medicine is a field that is rapidly evolving. Machine learning and deep learning are used to improve disease identification and diagnosis, personalize disease treatment, analyze medical images, evaluate clinical trials, and speed drug development. Methods: First, relevant aspects of AI are revised in a comprehensive manner, including the classification of hematopoietic neoplasms, types of AI, applications in medicine and hematological neoplasia, generative pre-trained transformers (GPTs), and the architecture and interpretation of feedforward neural net-works (multilayer perceptron). Second, a series of 233 diffuse large B-cell lymphoma (DLBCL) patients treated with rituximab-CHOP from the Lymphoma/Leukemia Molecular Profiling Project (LLMPP) was analyzed. Results: Using conventional statistics, the high expression of MYC and BCL2 was associated with poor survival, but high BCL6 was associated with a favorable overall survival of the patients. Then, a neural network predicted MYC, BCL2, and BCL6 with high accuracy using a pan-cancer panel of 758 genes of immuno-oncology and translational research that includes clinically relevant actionable genes and pathways. A comparable analysis was performed using gene set enrichment analysis (GSEA). Conclusions: The mathematical way in which neural networks reach conclusions has been considered a black box, but a careful understanding and evaluation of the architectural design allows us to interpret the results logically. In diffuse large B-cell lymphoma, neural networks are a plausible data analysis approach.
Full article
(This article belongs to the Section Lymphomas)
►▼
Show Figures

Figure 1
Open AccessOpinion
Elderly Patients with Newly Diagnosed Multiple Myeloma: Continuous or Fixed Duration Treatment?
by
Salomon Manier and Thierry Facon
Hemato 2024, 5(2), 115-118; https://doi.org/10.3390/hemato5020010 - 1 Apr 2024
Abstract
Multiple myeloma (MM) presents unique challenges in the elderly population due to increased frailty and comorbidities. Balancing treatment efficacy, safety, and quality of life is essential in managing elderly patients. While two-drug regimens were often favored for elderly patients, recent studies show promising
[...] Read more.
Multiple myeloma (MM) presents unique challenges in the elderly population due to increased frailty and comorbidities. Balancing treatment efficacy, safety, and quality of life is essential in managing elderly patients. While two-drug regimens were often favored for elderly patients, recent studies show promising outcomes with anti-CD38 antibody-based therapies, particularly daratumumab and lenalidomide with minimal dexamethasone. Continuous low-intensity treatments have shown improved progression-free survival and overall survival, with significant benefits observed in elderly patients. The DRd combination has now emerged as the standard of care for elderly MM patients, offering a favorable balance of efficacy, safety, and convenience. Ongoing trials are evaluating the addition of bortezomib in an induction phase for fit patients. New-generation immunotherapies hold promise for further refining treatment approaches, potentially leading to treatment discontinuation in select patient populations with sustained minimal residual disease negativity.
Full article
(This article belongs to the Special Issue Controversies in Multiple Myeloma: A Theme Issue in Honor of Prof. Dr. Jesús San Miguel)
►▼
Show Figures
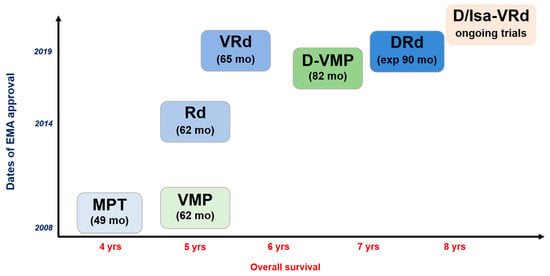
Figure 1
Open AccessBrief Report
Selective IgA Deficiency and Blood Component Transfusion: In Search of the Lost Evidence
by
Pilar Solves, Ana Bataller, Ana Belén Gálvez, Pedro Asensi Cantó, Marta Santiago, María José Moreno, Inés Gómez-Seguí and Javier de la Rubia
Hemato 2024, 5(2), 109-114; https://doi.org/10.3390/hemato5020009 - 26 Mar 2024
Abstract
Background: Selective IgA deficiency (IgA-D) has been historically considered a high-risk entity for developing allergic/anaphylactic reactions after blood transfusion (AATRs). However, it has been suggested that the IgA-D-related anaphylactic transfusion reaction is not evidence-based. Methods: We conducted three different approaches to
[...] Read more.
Background: Selective IgA deficiency (IgA-D) has been historically considered a high-risk entity for developing allergic/anaphylactic reactions after blood transfusion (AATRs). However, it has been suggested that the IgA-D-related anaphylactic transfusion reaction is not evidence-based. Methods: We conducted three different approaches to collect evidence about epidemiology, AATRs, and transfusion management of patients with IgA-D at La Fe University Hospital. Firstly, we analysed the prevalence of IgA-D in a population of patients diagnosed with acute leukaemia, The second approach consisted of collecting transfusion data from IgA-D patients. Finally, we reviewed the IgA levels of patients recorded in the hemovigilance system suffering an AATR. Results: IgA-D prevalence was 1 in 334 patients. At least one blood component was transfused to 23 patients diagnosed with IgA-D. Plasma was transfused to eight IgA-D patients, while six patients received red blood cells, platelets, and plasma. No adverse reactions were reported in any patient. AATRs occurred in 325 men and 264 women with a median age of 52 years. Severe reactions occurred in 56 patients (1/14,520 components). Mean IgA levels were 215 mg/dL (4–5570) for mild reactions and 214 mg/dL (14–824) for severe reactions (p = ns). Washed platelets were administered to two patients who developed severe and repeated AATRs. Both had normal IgA levels. Conclusions: Since the AATRs related to IgA-D are extremely low, as reported in current hemovigilance systems, IgA-D should not be considered a high-risk entity to develop AATRs. On the contrary, our findings support standard transfusion management of IgA-D patients.
Full article
Open AccessReview
Review of Peripheral Blood Eosinophilia: Workup and Differential Diagnosis
by
Michael Dennis Weaver, Bianca Glass, Chance Aplanalp, Gauri Patel, Jeshrine Mazhil, Isabella Wang and Samir Dalia
Hemato 2024, 5(1), 81-108; https://doi.org/10.3390/hemato5010008 - 16 Mar 2024
Cited by 1
Abstract
►▼
Show Figures
Eosinophils are a type of granulocyte key to immune system modulation seen in a number of disease processes. Nearly every major organ system can be connected to peripheral eosinophilia through a number of different disease processes, ranging from benign conditions to malignancy. In
[...] Read more.
Eosinophils are a type of granulocyte key to immune system modulation seen in a number of disease processes. Nearly every major organ system can be connected to peripheral eosinophilia through a number of different disease processes, ranging from benign conditions to malignancy. In this paper, we review both common and rare causes of peripheral eosinophilia, their symptoms, and a framework for the workup of peripheral eosinophilia of unknown etiology.
Full article
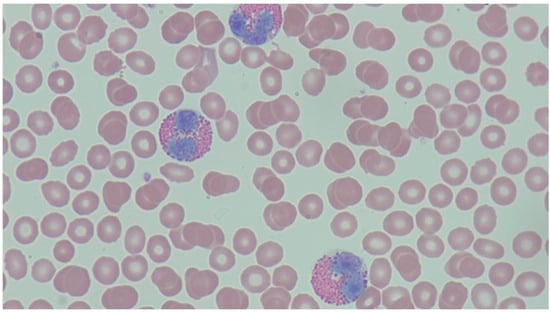
Figure 1
Open AccessArticle
Immune Checkpoint Inhibition in Pediatric Oncology Patients: A Single-Institution Experience
by
Natalia Wojciechowska, Kaci Orr, Karen Albritton, Kenneth Heym, Kelly Vallance, Lauren Murray, Rocio Aguilar and Anish Ray
Hemato 2024, 5(1), 66-80; https://doi.org/10.3390/hemato5010007 - 6 Mar 2024
Abstract
►▼
Show Figures
Immunotherapy has emerged as a promising treatment approach in oncology, as it is specifically designed to boost the strength and accuracy of the immune system, allowing it to target tumor cells but spare non-tumor tissue. This treatment not only demonstrates potential for improved
[...] Read more.
Immunotherapy has emerged as a promising treatment approach in oncology, as it is specifically designed to boost the strength and accuracy of the immune system, allowing it to target tumor cells but spare non-tumor tissue. This treatment not only demonstrates potential for improved clinical outcomes but may also be associated with fewer adverse effects compared to traditional therapies. Despite its early success, the application of immunotherapy has largely been limited to adult cancer patients, with slow adoption noted in the treatment of pediatric cancer patients. Our objective is to demonstrate a single institution’s experience with immunotherapy in pediatric cancer patients and to discuss the use of these treatment modalities in this unique patient population. We performed a retrospective chart review and identified patients who received immune checkpoint inhibitors (ICIs) and/or underwent immunohistochemistry (IHC) testing for programmed death ligand 1 (PD-L1), quantification of tumor mutational burden (TMB), and classification of microsatellite instability (MSI) status. In total, we identified seven pediatric cancer patients who received therapy with ICIs. Four of these patients demonstrated positive PD-L1 expression, high TMB, and/or MSI-high status. These patients were treated with nivolumab alone or in combination with ipilimumab or brentuximab. The diagnoses included: multifocal epithelioid and spindle cell hemangioma (n = 1); metastatic melanoma (n = 2); histiocytic sarcoma (n = 1); rectal adenocarcinoma in the setting of constitutional mismatch repair deficiency syndrome (CMMRD) (n = 1); and Hodgkin lymphoma (n = 2). The patients received between four and nineteen cycles of immunotherapy. Immunotherapy-related adverse events included: mild allergic reaction; prodromal symptoms; anemia; neutropenia; transaminitis; endocrinopathies; and self-limiting neuritis. Of the seven patients, three are still being treated with immunotherapy (the patients with rectal adenocarcinoma, metastatic melanoma, and multifocal epithelioid and spindle cell hemangioma) with positive treatment responses observed on imaging, one is being treated with other modalities (the patient with Hodgkin lymphoma), two have achieved remission (the patients with metastatic melanoma and Hodgkin lymphoma), and one has relapsed (the patient with histiocytic sarcoma). The three patients who completed their immunotherapy regimens have been followed for 1 month, 4 months, and 10 months, respectively. This report of a single-institution experience with immunotherapy in pediatric cancer patients highlights the positive impact immunotherapy can have, especially when utilized to treat relapsed/refractory malignancies, as tumor regression or stabilization of disease burden was achieved in six of the patients described (CR = 2; PR = 4). Further research is needed to accurately identify pediatric oncology patients who could benefit from immunotherapy.
Full article
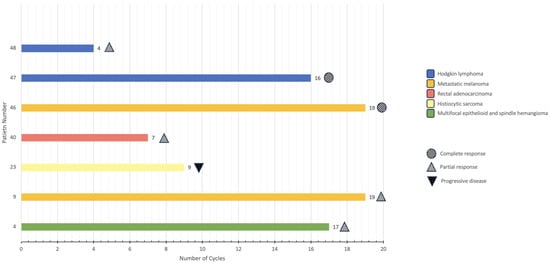
Figure 1
Open AccessArticle
Haemostaseological Changes of VWF and FVIII during Pregnancy and the Oestrus Cycle in a Porcine Model of Von Willebrand Disease
by
Rabea Möller, Katharina Kaiser, Ulrich Baulain, Björn Petersen, Carsten Detering, Mahnaz Ekhlasi-Hundrieser, Richard Zimmermann, Christian Mühlfeld, Mario von Depka Prondzinski, Christiane Pfarrer and Stefanie Lehner
Hemato 2024, 5(1), 48-65; https://doi.org/10.3390/hemato5010006 - 1 Feb 2024
Abstract
►▼
Show Figures
Pregnancy and the oestrus cycle are challenging for female patients suffering from von Willebrand disease (VWD). Therefore, our study aimed to investigate the changes in von Willebrand factor (VWF) and factor VIII (FVIII) during pregnancy and the oestrus cycle in our porcine model
[...] Read more.
Pregnancy and the oestrus cycle are challenging for female patients suffering from von Willebrand disease (VWD). Therefore, our study aimed to investigate the changes in von Willebrand factor (VWF) and factor VIII (FVIII) during pregnancy and the oestrus cycle in our porcine model of von Willebrand disease compared with the wild-type. Plasma analyses regarding primary hemostasis, secondary hemostasis, and VWF multimers, as well as immunohistochemistry analyses of VWF in the uterus and ovary, were performed. For levels of VWF and FVIII activities, significant elevations were seen in the last trimester. Primary hemostasis improved towards the end of pregnancy. In the oestrus cycle, significantly lower VWF values can be seen in the immunohistochemistry of the ovaries during the oestrus, while values were highest in the metoestrus. VWF multimer patterns in pigs were similar to the ones in human VWD patients. In summary, the course of VWF and FVIII during pregnancy and the oestrus cycle in porcine VWD were investigated for the first time. The porcine model seems to be suitable for haemostaseological studies on VWD. This provides an advantage for investigating reproduction-related bleeding and understanding the underlying mechanisms of post-partum hemorrhage or miscarriage in women with VWD.
Full article
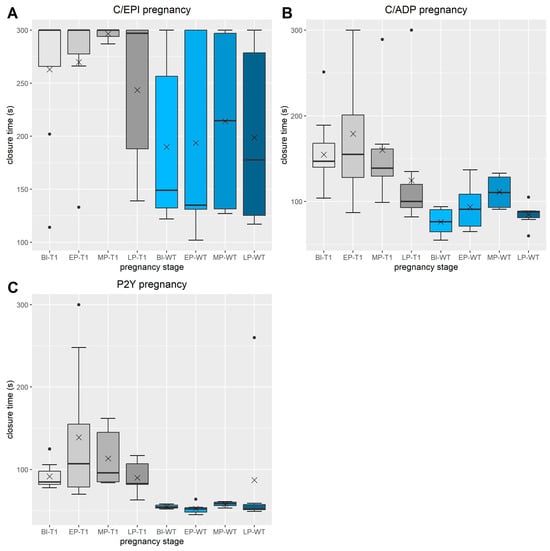
Figure 1
Open AccessReview
Parenteral Iron Therapy for Pediatric Patients
by
Elpis Mantadakis, Sonia Alexiadou and Panagiota Zikidou
Hemato 2024, 5(1), 35-47; https://doi.org/10.3390/hemato5010005 - 19 Jan 2024
Abstract
Iron deficiency (ID) is by far the most common nutritional disorder in developing and developed countries. When left untreated, ID leads to anemia. Although the usually recommended treatment for iron deficiency anemia (IDA) is oral iron therapy with countless products, such therapy necessitates
[...] Read more.
Iron deficiency (ID) is by far the most common nutritional disorder in developing and developed countries. When left untreated, ID leads to anemia. Although the usually recommended treatment for iron deficiency anemia (IDA) is oral iron therapy with countless products, such therapy necessitates administration for >3–6 months with questionable patient compliance since most oral iron products have an unpleasant metallic aftertaste and cause intestinal side effects. In addition, in certain gastrointestinal conditions, such as inflammatory bowel diseases or untreated gluten-sensitive enteropathy, oral iron therapy is contraindicated or unsuccessful. Intravenous iron is considered safe in adults, where adverse events are mild and easily managed. The experience with parenteral iron in children is much more limited, and many pediatricians appear reluctant to use it because of uncorroborated fears of serious anaphylactic reactions. In the current article, we thoroughly review the available pediatric literature on the use of all commercially available parenteral iron products except ferumoxytol, which was recently removed from the market. We conclude that parenteral iron appears to be safe in children; it works faster than oral iron, and the newer third-generation products allow replacement of the total iron deficit in a single sitting.
Full article
(This article belongs to the Section Non Neoplastic Blood Disorders)
Open AccessArticle
Patterns of Blood Transfusion in Sickle Cell Disease Hospitalizations
by
Aditi Sharma, Amit Dahiya, Asif Alavi, Indryas Woldie, Aditya Sharma, Jeffrey Karson and Vijendra Singh
Hemato 2024, 5(1), 26-34; https://doi.org/10.3390/hemato5010004 - 15 Jan 2024
Abstract
Background: Transfusional iron overload causes significant morbidity and mortality in sickle cell disease (SCD). Nevertheless, red blood cell transfusions continue to be essential in its management. This study describes the transfusion patterns among SCD hospitalizations. Methods: Hospitalizations for SCD in the 2017–2018 Nationwide
[...] Read more.
Background: Transfusional iron overload causes significant morbidity and mortality in sickle cell disease (SCD). Nevertheless, red blood cell transfusions continue to be essential in its management. This study describes the transfusion patterns among SCD hospitalizations. Methods: Hospitalizations for SCD in the 2017–2018 Nationwide Readmissions Database were divided into two groups based on whether they received transfusions. Descriptive analysis was performed to compare their demographics and complications. Multivariable logistic regression was performed to determine the factors associated with transfusions. Results: Out of 109,783 hospitalizations, 28,300 were transfused, and 81,483 were not transfused. Females and older individuals were higher in the transfused category than the non-transfused category (59.49% vs. 53.52% and 28.86% vs. 21.27%, respectively; p < 0.001 for both). The wealthiest population was more likely to be in the transfused category (11.27% vs. 8.34%; p < 0.001). Admissions to teaching hospitals, large metropolitan hospitals, and highest-volume hospitals were higher in the non-transfused category vs. transfused category (79.89% vs. 72.17%; p < 0.001, 69.26% vs. 65.35%; p 0.003 and 74.71% vs. 63.51%; p < 0.001, respectively). Most admissions were transfused once, with three or more transfusions being given more in the non-teaching hospitals than the teaching hospitals (1.27% vs. 0.41%; p 0.01). Furthermore, a higher proportion of early transfusions occurred in the non-teaching hospitals (65.6% vs. 57.82% for admission days 1 and 2; p < 0.001). Admission to a teaching hospital was associated with lower blood transfusion odds than a non-teaching hospital. Conclusion: A quarter of admissions for SCD receive a blood transfusion. In addition to performing more frequent and early transfusions, the odds of being transfused are higher in non-teaching hospitals.
Full article
(This article belongs to the Section Non Neoplastic Blood Disorders)
►▼
Show Figures
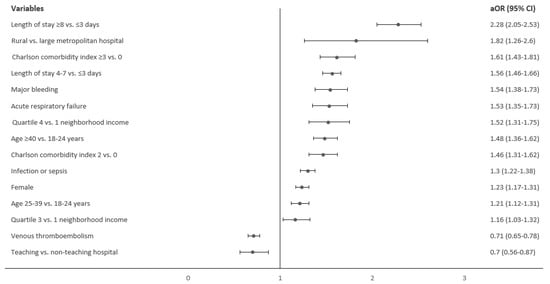
Figure 1
Open AccessCase Report
Demyelinating Polyradiculoneuropathy in Chronic Lymphocytic Leukemia: A Case Report on BTKis versus Venetoclax-Rituximab
by
Alessandro Cellini, Andrea Visentin, Alessandro Salvalaggio, Mario Cacciavillani, Sergio Ferrari and Chiara Briani
Hemato 2024, 5(1), 19-25; https://doi.org/10.3390/hemato5010003 - 27 Dec 2023
Abstract
The dysregulation of the immune system in Chronic Lymphocytic Leukemia (CLL) often allows for the development of immune-mediated diseases. Among them, autoimmune cytopenias are the most common, but cases of chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) have been reported. We herein report on a
[...] Read more.
The dysregulation of the immune system in Chronic Lymphocytic Leukemia (CLL) often allows for the development of immune-mediated diseases. Among them, autoimmune cytopenias are the most common, but cases of chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) have been reported. We herein report on a patient who developed a CIDP while undergoing ibrutinib treatment for CLL, prompting drug discontinuation. Steroid treatment and a rituximab course proved to be ineffective at obtaining long-term control of CIDP, but therapy with venetoclax and rituximab, which was started due to CLL progression, led to the progressive amelioration of the symptoms up to complete remission of the neurological disease.
Full article
(This article belongs to the Section Leukemias)
►▼
Show Figures
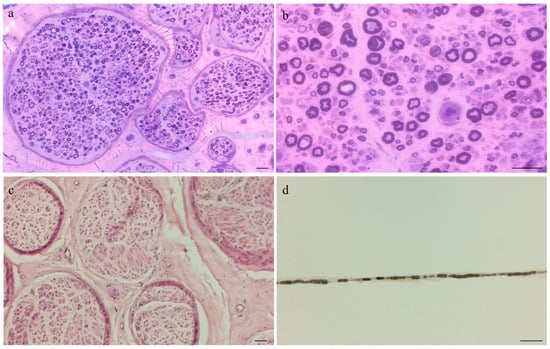
Figure 1
Open AccessReview
Synthetic Lethality Approaches in Acute Lymphoblastic Leukemia
by
Francisco Alejandro Lagunas-Rangel and Venice Chávez-Valencia
Hemato 2024, 5(1), 6-18; https://doi.org/10.3390/hemato5010002 - 26 Dec 2023
Cited by 1
Abstract
Acute lymphoblastic leukemia (ALL), a remarkable cancer that mainly affects children, has seen commendable advances in its treatment. However, the occurrence of relapses after initial treatments poses a major threat and is one of the leading causes of cancer-related mortality in pediatric patients.
[...] Read more.
Acute lymphoblastic leukemia (ALL), a remarkable cancer that mainly affects children, has seen commendable advances in its treatment. However, the occurrence of relapses after initial treatments poses a major threat and is one of the leading causes of cancer-related mortality in pediatric patients. To address this problem, innovative therapeutic approaches for ALL need to be continuously developed and refined. Synthetic lethality, an interaction between genes in which alteration of only one allows survival, but simultaneous alteration of both leads to inviability, is emerging as a promising therapeutic approach against ALL and other cancers. In this regard, the review aims to examine the documented cases of synthetic lethality in ALL reported to date (2023) and to elucidate the molecular mechanisms underlying this phenomenon. Furthermore, this review explores possible targets that have so far gone unnoticed, justifying their importance in this context.
Full article
(This article belongs to the Section Leukemias)
►▼
Show Figures
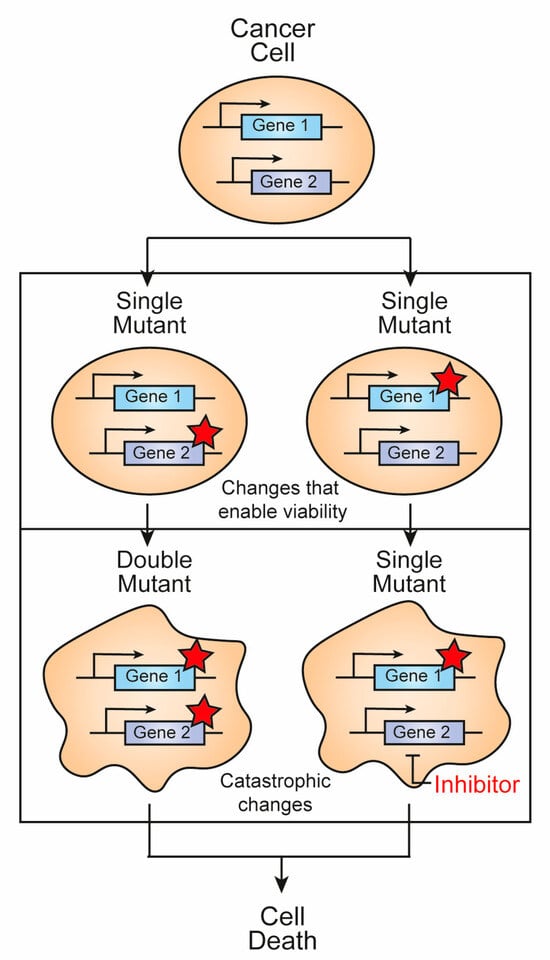
Figure 1
Open AccessEditorial
Hemato Keeps You Updated on the Research in Hematology
by
Antonino Carbone
Hemato 2024, 5(1), 1-5; https://doi.org/10.3390/hemato5010001 - 25 Dec 2023
Abstract
►▼
Show Figures
Hemato (ISSN 2673-6357) is an open access, peer-reviewed journal that publishes original articles and reviews highlighting important advances in the fundamental areas of Hematology [...]
Full article
Figure 1
Open AccessFeature PaperReview
The Role of Platelet Molecules in Risk Stratification of Patients with COVID-19
by
Lívia de Oliveira Sales, Lais Lacerda Brasil de Oliveira, Jean Breno Silveira da Silva, Manoel Odorico de Moraes Filho, Maria Elisabete Amaral de Moraes, Raquel Carvalho Montenegro and Caroline Aquino Moreira-Nunes
Hemato 2023, 4(4), 364-383; https://doi.org/10.3390/hemato4040029 - 30 Nov 2023
Cited by 1
Abstract
The new severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first reported in China and is responsible for Coronavirus disease (COVID-19). Despite being well tolerated by most patients, a fraction of cases evolve into a potentially fatal condition requiring intensive care. In addition
[...] Read more.
The new severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first reported in China and is responsible for Coronavirus disease (COVID-19). Despite being well tolerated by most patients, a fraction of cases evolve into a potentially fatal condition requiring intensive care. In addition to respiratory complications, several studies have reported cases of patients who developed intense thrombosis, including acute myocardial infarction and ischemic stroke, as well as the presence of elevated coagulation markers. Evidence has shown that the virus can interact directly with platelets and modulate their thrombotic and inflammatory functions, with significant prognostic implications. It is important to highlight that the emerging literature shows that when hyperactive these cells can act as pro-viral infections both in transporting their particles and in increasing inflammation, leading to a hyperinflammatory state and consequent clinical worsening. In this review, we searched for studies available in public databases and discussed the interaction of platelet biomarkers in the pathogenesis of COVID-19. In this context, understanding the mechanism of SARS-CoV-2 and these cells in different clinical conditions could help us to understand the coagulation and inflammation profiles of critically ill patients with the disease, guiding faster clinical management and enabling the reuse and targeting of more efficient therapies.
Full article
(This article belongs to the Section Coagulation)
►▼
Show Figures
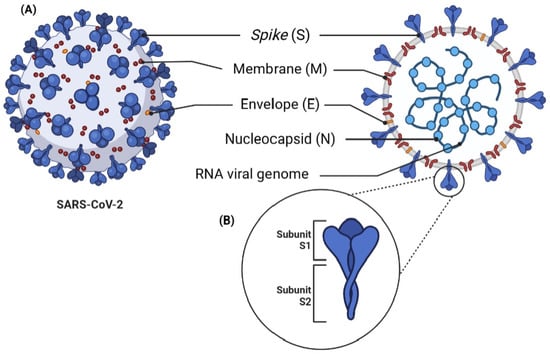
Figure 1
Open AccessArticle
Fibril-Forming Organelles in Mesangial Cells in Renal Biopsies from Patients with Light-Chain-Associated Amyloidosis
by
Guillermo A. Herrera, Jiamin Teng, Chun Zeng, Luis Del Pozo-Yauner, Bing Liu and Elba A. Turbat-Herrera
Hemato 2023, 4(4), 350-363; https://doi.org/10.3390/hemato4040028 - 23 Nov 2023
Abstract
The process of light-chain-associated amyloid (AL-Am) fibril formation in unique organelles (fibril-forming organelles) with lysosomal features has been documented in vitro in renal mesangial cells incubated with amyloidogenic light chains using electron microscopy and lysosomal gradient centrifugation to visualize intricate interactions between monoclonal
[...] Read more.
The process of light-chain-associated amyloid (AL-Am) fibril formation in unique organelles (fibril-forming organelles) with lysosomal features has been documented in vitro in renal mesangial cells incubated with amyloidogenic light chains using electron microscopy and lysosomal gradient centrifugation to visualize intricate interactions between monoclonal light chains and endosomes/lysosomes. It is important to determine whether this process also occurs in vivo in the human renal mesangium. The present study analyzes 13 renal biopsies from patients with renal AL-amyloidosis and utilizes ultrastructural labeling techniques to define the nature and function of these organelles. Organelles were labeled for lysosomal-associated membrane protein (LAMP) and CD-68 (a macrophage marker). Furthermore, lambda was also localized inside these structures in transformed mesangial cells with a macrophage phenotype. These 11 cases from renal biopsies with a diagnosis of AL-amyloidosis (5 kappa and 8 lambda light-chain-associated) were examined ultrastructurally. All of the cases exhibited numerous fibrils forming organelles in approximately 40–50% of the remaining mesangial cells. All of the cases revealed mesangial cells engaged in active amyloidogenesis. Fibril-forming organelles are organelles with morphological/immunohistochemical and biochemical characteristics of lysosomes but with a unique, peculiar morphology. Five cases of other glomerular disorders used as controls were also carefully scrutinized for fibril-forming organelles and failed to show any. In the AL-amyloid renal cases, there was an intricate interaction between the fibril-forming organelles and lambda-/kappa-containing amyloid fibrils, supporting the notion that the monoclonal light chains participated in their formation.
Full article
(This article belongs to the Section Plasma Cell Disorders)
►▼
Show Figures
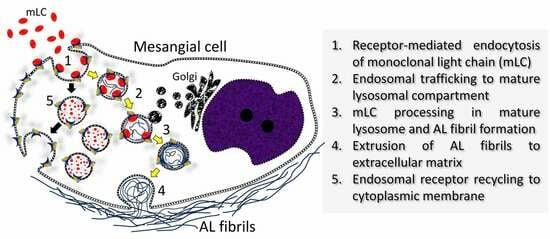
Graphical abstract
Open AccessSystematic Review
Geographic Prevalence Patterns and Modifiable Risk Factors for Monoclonal Gammopathy of Undetermined Significance
by
Karina P. Verma, Rebecca Steuer and Camille V. Edwards
Hemato 2023, 4(4), 331-349; https://doi.org/10.3390/hemato4040027 - 1 Nov 2023
Abstract
Monoclonal gammopathy of undetermined significance (MGUS) is a pre-malignant plasma cell disorder with an etiology that is incompletely understood. Modifiable risk factors and genetic predispositions likely interact to increase MGUS risk in specific individuals and populations. Identifying geographic prevalence patterns and modifiable risk
[...] Read more.
Monoclonal gammopathy of undetermined significance (MGUS) is a pre-malignant plasma cell disorder with an etiology that is incompletely understood. Modifiable risk factors and genetic predispositions likely interact to increase MGUS risk in specific individuals and populations. Identifying geographic prevalence patterns and modifiable risk factors is critical for understanding the etiology of MGUS. The aim of this review was to outline original research on MGUS prevalence across geographic locations and modifiable risk factors. We conducted a systematic review of 39 eligible studies from PubMed®, Embase®, and Web of Science® written in English and published by February 2023. Our protocol was registered in accordance with PROSPERO guidelines. Studies were synthesized using Research Electronic Data Capture and appraised using the National Heart, Lung, and Blood Institute study quality assessment tools. The prevalence of MGUS ranged from 0.24% to 9% across geographic locations. Modifiable risk factors for MGUS include infections, autoimmune diseases, chronic inflammatory conditions, lifestyle factors, environmental exposures, and ionizing radiation. Therefore, the development of MGUS may be related to chronic antigenic stimulation and genetic aberrations that promote clonal proliferation of plasma cells. Prospective studies assessing gene–environment interactions are needed to further define risk factors for MGUS and inform screening and preventative strategies.
Full article
(This article belongs to the Section Plasma Cell Disorders)
►▼
Show Figures
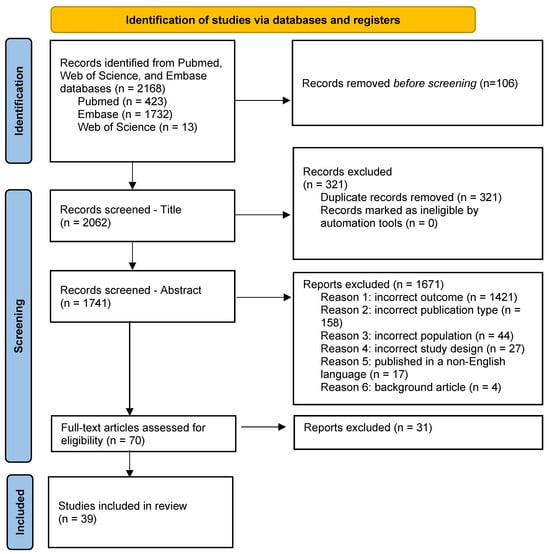
Figure 1

Journal Menu
► ▼ Journal Menu-
- Hemato Home
- Aims & Scope
- Editorial Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
BioMedInformatics, Cancers, Current Oncology, Hemato, Hematology Reports
Myeloma and Leukemia-Challenges and Current Treatment Options
Topic Editors: Giovanni Martinelli, Claudio CerchioneDeadline: 20 June 2024

Conferences
Special Issues
Topical Collections
Topical Collection in
Hemato
Exclusive Papers of the Editorial Board Members (EBMs) of Hemato
Collection Editors: Antonino Carbone, Laurent Garderet








