CMV-Specific Immune Response—New Patients, New Insight: Central Role of Specific IgG during Infancy and Long-Lasting Immune Deficiency after Allogenic Stem Cell Transplantation
Abstract
:1. Introduction
2. Results
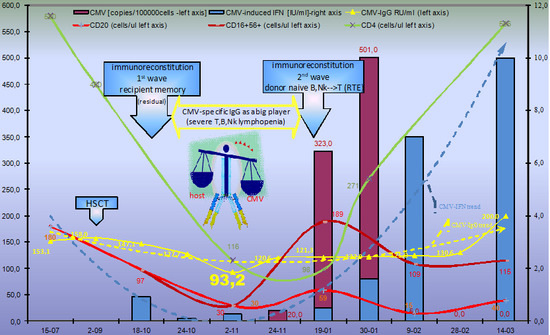
2.1. Evolution of CMV Infectious Process in Immunodeficient HSCT Recipient
2.1.1. Humoral CMV Specific Immunity
2.1.2. Cellular Immune Response
2.2. Evolution of the CMV Infectious Process in Immunocompetent HSCT Donor
2.3. Evolution of CMV Infectious Process in Immunocompetent Infant
3. Discussion
3.1. Humoral Immune Response
3.1.1. CMV—Serostatus
3.1.2. CMV-IgG Level
3.2. Cellular Immune Response
3.3. Innate Immune Response
4. Material and Methods
4.1. Material
4.2. Methods
Interferon Gamma IFNγ Release Assay (IGRA)
- (1)
- CMV-peptide-laden human leukocyte antigen (pHLA) class I, presented in Supplementary Materials.
- (2)
- Tube 2 contained phytohemagglutinin (PHA), i.e., pan-T cell mitogen as a positive control.
- (3)
- Tube 3 contained sterile phosphate-buffered as a negative control.
5. Conclusions
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
Ethics Approval and Consent to Participate
Consent for Publication
Abbreviations
| ADCC | Antibody-dependent cell-mediated cytotoxicity |
| CMV-IgG | Anti-cytomegalovirus immunoglobulin G |
| AI | Avidity index |
| CD94/NKG2C | Ca++dependent (C-type) lectin superfamily of type II transmembrane receptor of natural killer cells |
| TEMRA | CD45RA-positive effector memory T cells |
| gH | CMV glycoprotein H |
| CMV serostatus constellation | D+/R-(donor positive/recipient negative) D-/R+ (donor negative /recipient positive) etc |
| CsA | Cyclosporine A |
| CMV | Cytomegalovirus |
| DLI | Donor lymphocyte infusion |
| EBV | Epstein–Barr virus |
| ELISA | Enzyme-linked immunosorbent assay |
| EBMT | European Group for Blood and Marrow Transplantation |
| GVHD | Graft-versus-host (GvH) disease |
| G-CSF | Granulocyte colony-stimulating factor |
| IGRA | Interferon gamma release assay |
| HSCT | Hematopoietic stem cell transplantation |
| HLA | Human leukocyte antigens |
| HSV | Herpes simplex virus |
| KIRs | Killer-cell immunoglobulin-like receptors |
| MAC | Membrane attack complex |
| NK | Natural killer cells |
| PBSCT | Peripheral blood stem cell transplantation |
| PHA | Phytohemagglutinin |
| Poly(I:C) | Polyinosinic-polycytidylic acid |
| RT-PCR | Real-time quantitative polymerase chain reaction |
| Fc receptors | Receptors for the constant regions of their H chain |
| TEMRA | Terminally differentiated T-cell |
| NMDP | US National Marrow Donor Program |
| VZV | Varicella-zoster virus |
References
- Wróbel, M.J.; Greczka, G.; Szyfter, W. The risk factor profile of children covered bythe Polish universal neonatal hearing screening program and its impact on hearingloss incidence. Int. J. Pediatr. Otorhinolaryngol. 2014, 78, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Lucchese, G.; Kanduc, D. Cytomegalovirus infection: The neurodevelopmental peptide signatures. Curr. Drug. Discov. Technol. 2018, 15, 251–262. [Google Scholar] [CrossRef]
- Ragnaud, J.M.; Morlat, P.; Gin, H.; Dupon, M.; Delafaye, C.; du Pasquier, P.; Aubertin, J. Clinical, biological and developmental aspects of cytomegalovirus infection in immunocompetent patients: Apropos of 34 hospitalized patients. Rev. Med. Interne 1994, 15, 13–18. [Google Scholar] [CrossRef]
- Rodríguez-Baño, J.; Muniain, M.A.; Borobio, M.V.; Corral, J.L.; Ramírez, E.; Perea, E.J.; Perez-Cano, R. Cytomegalovirus mononucleosis as a cause of prolonged fever and prominent weight loss in immunocompetent adults. Clin. Microbiol. Infect. 2004, 10, 468–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Snydman, D.R. Editorial commentary: The complexity of latent cytomegalovirus infection in stem cell donors. Clin. Infect. Dis. 2014, 59, 482–483. [Google Scholar] [CrossRef] [PubMed]
- Boeckh, M.; Nichols, W.G. The impact of cytomegalovirus serostatus of donor and recipient before hematopoietic stem cell transplantation in the era of antiviral prophylaxis and preemptive therapy. Blood 2004, 103, 2003–2008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zdziarski, P.; Gamian, A.; Dworacki, G. A case report of lymphoid intestitial pneumonia in common variable immunodeficiency: Oligoclonal expansion of effector lymphocytes with preferential cytomegalovirus-specific immune response and lymphoproliferative disease promotion. Medicine (Baltimore) 2017, 96, e7031. [Google Scholar] [CrossRef]
- D’Orsogna, L.J.; Roelen, D.L.; Doxiadis, I.I.; Claas, F.H. Alloreactivity from human viral specific memory T-cells. Transpl. Immunol. 2010, 23, 149–155. [Google Scholar] [CrossRef]
- Jaskula, E.; Dlubek, D.; Tarnowska, A.; Lange, J.; Mordak-Domagala, M.; Suchnicki, K.; Sedzimirska, M.; Borowik, A.; Mizia, S.; Lange, A. Anti-CMV-IgG positivity of donors is beneficial for alloHSCT recipients with respect to the better short-term immunological recovery and high level of CD4+CD25high lymphocytes. Viruses 2015, 7, 1391–1408. [Google Scholar] [CrossRef]
- Klenovsek, K.; Weisel, F.; Schneider, A.; Appelt, U.; Jonjic, S.; Messerle, M.; Bradel-Tretheway, B.; Winkler, T.H.; Mach, M. Protection from CMV infection in immunodeficient hosts by adoptive transfer of memory B cells. Blood 2007, 110, 3472–3479. [Google Scholar] [CrossRef] [Green Version]
- Yong, M.K.; Cameron, P.U.; Slavin, M.A.; Cheng, A.C.; Morrissey, C.O.; Bergin, K.; Spencer, A.; Ritchie, D.; Lewin, S.R. Low T-Cell responses to mitogen stimulation predicts poor survival in recipients of allogeneic hematopoietic stem cell transplantation. Front. Immunol. 2017, 8, 1506. [Google Scholar] [CrossRef]
- Jin, F.; Lin, H.; Gao, S.; Wang, H.; Yan, H.; Guo, J.; Hu, Z.; Jin, C.; Wang, Y.; Wang, Z.; et al. Characterization of IFNγ-producing natural killer cells induced by cytomegalovirus reactivation after haploidentical hematopoietic stem cell transplantation. Oncotarget 2017, 8, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.K.; Kennedy, G.A.; Cromer, D.; Davenport, M.P.; Walker, S.; Jones, L.I.; Crough, T.; Durrant, S.T.; Morton, J.A.; Butler, J.P.; et al. Clinical assessment of anti-viral CD8+ T cell immune monitoring using QuantiFERON-CMV(R) assay to identify high risk allogeneic hematopoietic stem cell transplant patients with CMV infection complications. PLoS ONE 2013, 8, e74744. [Google Scholar] [CrossRef]
- Westall, G.P.; Mifsud, N.A.; Kotsimbos, T. Linking CMV serostatus to episodes of CMV reactivation following lung transplantation by measuring CMV-specific CD8+ T.-cell immunity. Am. J. Transplant. 2008, 8, 1749–1754. [Google Scholar] [CrossRef] [PubMed]
- Giorgetti, S.; Raimondi, S.; Pagano, K.; Relini, A.; Bucciantini, M.; Corazza, A.; Fogolari, F.; Codutti, L.; Salmona, M.; Mangione, P.; et al. Effect of tetracyclines on the dynamics of formation and destructuration of beta2-microglobulin amyloid fibrils. J. Biol. Chem. 2011, 286, 2121–2131. [Google Scholar] [CrossRef] [PubMed]
- Nigro, G.; Adler, S.P.; Parruti, G.; Anceschi, M.M.; Coclite, E.; Pezone, I.; Di Renzo, G.C. Immunoglobulin therapy of fetal cytomegalovirus infection occurring in the first half of pregnancy—A case-control study of the outcome in children. J. Infect. Dis. 2012, 205, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Santini, P.A.; Matthews, A.J.; Chiu, A.; Plebani, A.; He, B.; Chen, K.; Cerutti, A. Viral double-stranded RNA triggers Ig class switching by activating upper respiratory mucosa B cells through an innate TLR3 pathway involving BAFF. J. Immunol. 2008, 181, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.A.; Stevenson, K.; Kim, H.T.; Cutler, C.; Ballen, K.; McDonough, S.; Reynolds, C.; Herrera, M.; Liney, D.; Ho, V.; et al. Clearance of CMV viremia and survival after double umbilical cord blood transplantation in adults depends on reconstitution of thymopoiesis. Blood 2010, 115, 4111–4119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, J.; Song, W. Alemtuzumab versus antithymocyte globulin induction therapies in kidney transplantation patients: A systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore) 2017, 96, e7151. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.F.; Grebe, S.O.; Neumann, M.C.; Heymanns, J.; Radsak, K.; Sprenger, H.; Lange, H. Persistent long-term changes in lymphocyte subsets induced by polyclonal antibodies. Transplantation 1997, 64, 1432–1437. [Google Scholar] [CrossRef]
- Ljungman, P.; Brand, R.; Hoek, J.; Camara, R.; Cordonnier, C.; Einsele, H.; Styczynski, J.; Ward, K.N.; Cesaro, S.; Infectious Diseases Working Party of the EBMT. Donor cytomegalovirus status influences the outcome of allogeneic stem cell transplant: A study by the European Group for blood and marrow transplantation. Clin. Infect. Dis. 2014, 59, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Dollard, S.C.; Staras, S.A.; Amin, M.M.; Schmid, D.S.; Cannon, M.J. National prevalence estimates for cytomegalovirus IgM and IgG avidity and association between high IgM antibody titer and low IgG avidity. Clin. Vaccine Immunol. 2011, 18, 1895–1899. [Google Scholar] [CrossRef] [PubMed]
- Revello, M.G.; Fabbri, E.; Furione, M.; Zavattoni, M.; Lilleri, D.; Tassis, B.; Quarenghi, A.; Cena, C.; Arossa, A.; Montanari, L.; et al. Role of prenatal diagnosis and counseling in the management of 735 pregnancies complicated by primary human cytomegalovirus infection: A 20-year experience. J. Clin. Virol. 2011, 50, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Wallach, J. Interpretation of Diagnostics Tests; Wolters Kluwer: Alfon am Rhein, The Netherlands, 2015; 631p. [Google Scholar]
- Szomolanyi-Tsuda, E.; Brehm, M.A.; Welsh, R.M. Acquired Immunity Against Virus Infections. In The Immune Response to Infection; Kaufmann, S.H.E., Rouse, B.T., Sacks, D.L., Eds.; ASM Press: Washington, DC, USA, 2011; pp. 239–254. [Google Scholar]
- Foley, B.; Cooley, S.; Verneris, M.R.; Curtsinger, J.; Luo, X.; Waller, E.K.; Anasetti, C.; Weisdorf, D.; Miller, J.S. Human cytomegalovirus (CMV)-induced memory-like NKG2C(+) NK cells are transplantable and expand in vivo in response to recipient CMV antigen. J. Immunol. 2012, 189, 5082–5088. [Google Scholar] [CrossRef]
- Foley, B.; Cooley, S.; Verneris, M.R.; Pitt, M.; Curtsinger, J.; Luo, X.; Lopez-Vergčs, S.; Lanier, L.L.; Weisdorf, D.; Miller, J.S. Cytomegalovirus reactivation after allogeneic transplantation promotes a lasting increase in educated NKG2C+ natural killer cells with potent function. Blood 2012, 119, 2665–2674. [Google Scholar] [CrossRef]
- Dammermann, W.; Bochmann, D.; Bentzien, F.; Komorowski, L.; Steinhagen, K.; Ullrich, S.; van Lunzen, J.; Lüth, S. CMV specific cytokine release assay in whole blood is optimized by combining synthetic CMV peptides and toll like receptor agonists. J. Immunol. Methods. 2014, 414, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Spindler, N.; Rücker, P.; Pötzsch, S.; Diestel, U.; Sticht, H.; Martin-Parras, L.; Winkler, T.H.; Mach, M. Characterization of a discontinuous neutralizing epitope on glycoprotein B of human cytomegalovirus. J. Virol. 2013, 87, 8927–8939. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.; Boan, P.; Baumwol, J.; Chakera, A.; MacQuillan, G.; Swaminathan, S.; Lavender, M.; Flexman, J.; James, I.; John, M. Analysis of the QuantiFERON-CMV assay, CMV viraemia and antiviral treatment following solid organ transplantation in Western Australia. Pathology 2018, 50, 554–561. [Google Scholar] [CrossRef]
- Yong, M.K.; Cameron, P.U.; Slavin, M.; Morrissey, C.O.; Bergin, K.; Spencer, A.; Ritchie, D.; Cheng, A.C.; Samri, A.; Carcelain, G.; Autran, B.; Lewin, S.R. Identifying cytomegalovirus complications using the quantiferon-CMV assay after allogeneic hematopoietic stem cell transplantation. J. Infect Dis. 2017, 215, 1684–1694. [Google Scholar] [CrossRef]
- Nahum, A.; Dadi, H.; Bates, A.; Roifman, C.M. The biological significance of TLR3 variant, L412F, in conferring susceptibility to cutaneous candidiasis, CMV and autoimmunity. Autoimmun. Rev. 2012, 11, 341–347. [Google Scholar] [CrossRef]



| Feature | Recipient (−14 Day) | Donor | |
|---|---|---|---|
| Age | 56 y | 64y | |
| Gender | Female | Male | |
| Blood group | A Rh-positive | B Rh-positive | |
| Serostatus (IgM) | CMV, EBV, VZV, HSV-negative | EBV, HSV-negativeCMV (−) →(+) 5 | |
| Serostatus (IgG) | CMV, EBV, HSV-positive | CMV 0,12 (−) →63.05 (+) 5 HSV, EBV -positive | |
| Body weight (kg) | 58 | 86 | |
| Lowest WBC | 250 (+10 day) | 2800 | |
| Lowest lymphocyte count | 190 (+31 day) | 650 | |
| CMV-IgG AWIDITY 1 (%) | High (till +60 day) Intermediate (60–84 day) Low (84–150 day) High (>180 day) | Low (till + 115 day) Intermediate 6 | |
| Flow Cytometry 2 | 19–01 | 30–01 | During CMV viremia |
| CD4+CD27+CCR7+CD45RA+ Naive | 0.05 | 0.23 | NT |
| CD4+CD27+CCR7+CD45RA- Central memory | 0.69 | 2.22 | NT |
| CD4+CD27-CCR7-CD45RA- Effector memory | 5.30 | 4.16 | NT |
| CD4+CD27-CCR7-CD45RA+ CD45RA + effector memory T cells (TEMRA) | 0.41 | 0.15 | NT |
| Pentamer Analysis 3,4 (% of CD8 lymphocytes) | |||
| CD8high + CMV pp65+ | 2.32 | 6.91 | 0.08 |
| CD8high+CD57 + CMV pp65+ | 0.9 | 2.32 | 0.00 |
| β2-microoglobulin4 (mg/L) | 6.4 | 9.2 | 3.0 |
| Immune Response | Primary | Secondary (Days) | |
|---|---|---|---|
| Infection | |||
| Primary (by mucosa, skin) | Typical IgM→IgG | DLI 2, Breastfeeding MFT | |
| Secondary (i.e., reactivation *) | HSCT 1, neonatal CMV (without IgM **) solid organ transplantation | Typical (IgG only) | |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zdziarski, P. CMV-Specific Immune Response—New Patients, New Insight: Central Role of Specific IgG during Infancy and Long-Lasting Immune Deficiency after Allogenic Stem Cell Transplantation. Int. J. Mol. Sci. 2019, 20, 271. https://doi.org/10.3390/ijms20020271
Zdziarski P. CMV-Specific Immune Response—New Patients, New Insight: Central Role of Specific IgG during Infancy and Long-Lasting Immune Deficiency after Allogenic Stem Cell Transplantation. International Journal of Molecular Sciences. 2019; 20(2):271. https://doi.org/10.3390/ijms20020271
Chicago/Turabian StyleZdziarski, Przemyslaw. 2019. "CMV-Specific Immune Response—New Patients, New Insight: Central Role of Specific IgG during Infancy and Long-Lasting Immune Deficiency after Allogenic Stem Cell Transplantation" International Journal of Molecular Sciences 20, no. 2: 271. https://doi.org/10.3390/ijms20020271