- Article
The Effect of Lipoproteins on Platelet-Associated PCSK9 of Healthy Normolipidemic Volunteers: An In Vitro Study
- Aikaterini N. Tsouka,
- Ioannis K. Koutsaliaris and
- Alexandros D. Tselepis
- + 1 author
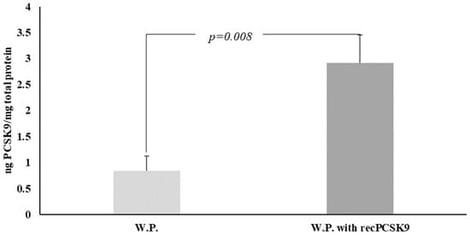
Background: Proprotein convertase subtilisin/kexin type 9 (PCSK9) promotes low-density lipoprotein receptor degradation and thereby regulates plasma LDL cholesterol levels. Although PCSK9 is primarily produced by the liver, it has been detected in platelets; however, the origin and functional relevance of platelet-associated PCSK9 remain unclear. Methods: Washed platelets (WPs) were isolated from normolipidemic subjects. Endogenous platelet PCSK9 content was quantified by ELISA, and PCSK9 molecular forms were assessed by immunoblotting. The WPs were incubated with recombinant PCSK9 (recPCSK9), and platelet aggregation in response to arachidonic acid (AA) or thrombin (Thr) was evaluated. The effects of LDL- or HDL-bound PCSK9 on platelet aggregation were also examined. Results: Platelets from normolipidemic subjects contained measurable amounts of PCSK9 (0.84 ± 0.27 ng/mg protein), which increased to 2.91 ± 0.53 ng/mg protein following incubation with recPCSK9. Exposure to recPCSK9 significantly enhanced AA- and Thr-induced platelet aggregation. In contrast, LDL and HDL inhibited platelet aggregation independently of their PCSK9 content. Conclusions: Human platelets contain endogenous PCSK9 and can accumulate additional PCSK9 from the extracellular environment. Exogenous PCSK9 enhances platelet aggregation, supporting a potential prothrombotic role for circulating PCSK9 even in normolipidemic individuals. These findings provide new insight into the complex interplay between PCSK9, lipoproteins, and platelet function.
25 February 2026