- Article
Association of Seroprevalence of Respiratory Pathogens and Herd-Level Management Factors with Inflammatory Markers in Dairy Cattle
- Anri Timonen,
- Rohish Kaura and
- Toomas Orro
- + 4 authors
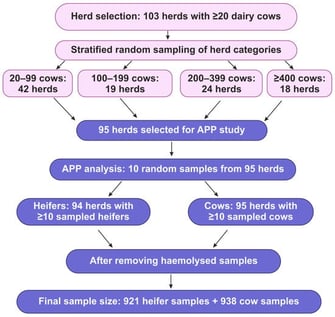
This cross-sectional study investigated the associations between the acute-phase proteins (APP) serum amyloid A (SAA) and haptoglobin (Hp), herd-level factors, and the seroprevalence of respiratory pathogens in Estonian dairy herds. Serum samples were analysed from 938 cows (95 herds) and 921 heifers (94 herds). Seroprevalence was tested for bovine herpesvirus 1 (BHV-1), bovine respiratory syncytial virus (BRSV), bovine parainfluenza virus 3, bovine viral diarrhoea virus, bovine coronavirus, bovine adenovirus, and Mycoplasma bovis (M. bovis). Farm visits included questionnaires on herd management practices. Linear random-intercept regression models showed higher serum SAA concentrations in cows from farms with BHV-1 seroprevalence of >50% and on BRSV-positive farms (p < 0.05), while farms employing a veterinarian had lower serum SAA concentrations. Cows had higher serum Hp concentrations in M. bovis-positive herds (p = 0.030). In heifers, serum SAA concentrations increased with low to moderate BHV-1 seroprevalence, decreased with higher M. bovis seroprevalence, and were higher in free-stall or mixed housing compared to tie-stall housing. Heifers’ serum Hp concentrations were lower in BHV-1-positive herds, but higher in herds with breeding bulls and larger herd sizes. To conclude, APP may reflect the herd health status and management-related effects on animals, supporting their use in herd-level monitoring.
19 February 2026