-
 A New Phosphate Sensor Based on Electrochemically Modified All-Solid-State Copper Electrode for Phosphate Ion Detection
A New Phosphate Sensor Based on Electrochemically Modified All-Solid-State Copper Electrode for Phosphate Ion Detection -
 Opioids and Their Analogs: Challenges, Electrochemical Sensors, Biosensors, and Optical Sensors Detection Methods
Opioids and Their Analogs: Challenges, Electrochemical Sensors, Biosensors, and Optical Sensors Detection Methods -
 Ultrahigh-Sensitivity Detection of 17β-Estradiol
Ultrahigh-Sensitivity Detection of 17β-Estradiol -
 A Dynamic Range Preservation Readout Integrated Circuit for Multi-Gas Sensor Array Applications
A Dynamic Range Preservation Readout Integrated Circuit for Multi-Gas Sensor Array Applications
Journal Description
Chemosensors
Chemosensors
is an international, scientific, peer-reviewed, open access journal on the science and technology of chemical sensors and related analytical methods and systems, published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), CAPlus / SciFinder, Inspec, and other databases.
- Journal Rank: JCR - Q1 (Instruments & Instrumentation) / CiteScore - Q2 (Analytical Chemistry)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.9 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
4.2 (2022);
5-Year Impact Factor:
4.2 (2022)
Latest Articles
Hydrothermally Synthesized Cerium Phosphate with Functionalized Carbon Nanofiber Nanocomposite for Enhanced Electrochemical Detection of Hypoxanthine
Chemosensors 2024, 12(5), 84; https://doi.org/10.3390/chemosensors12050084 - 16 May 2024
Abstract
This work presents the detection of hypoxanthine (HXA), a purine derivative that is similar to nucleic acids who overconsumption can cause health issues, by using hydrothermally synthesized cerium phosphate (CePO4) followed by a sonochemical approach for CePO4 decorated with a
[...] Read more.
This work presents the detection of hypoxanthine (HXA), a purine derivative that is similar to nucleic acids who overconsumption can cause health issues, by using hydrothermally synthesized cerium phosphate (CePO4) followed by a sonochemical approach for CePO4 decorated with a functionalized carbon nanofiber (CePO4@f-CNF) nanocomposite. The formation of the nanocomposite was confirmed with X-ray powder diffraction (XRD), scanning electron microscopy (SEM), and energy-dispersive spectroscopy (EDS). A CePO4@f-CNF nanocomposite is used to modify a glassy carbon electrode (GCE) to analyze the electrochemical detection of HXA. Cyclic voltammetry (CV), Electrochemical impedance spectroscopy (EIS), and Differential pulse voltammetry (DPV) were used to examine the electrochemical properties of the composite. As a result, the modified electrode exhibits a larger active surface area (A = 1.39 cm2), a low limit of detection (LOD) at 0.23 µM, a wide linear range (2.05–629 µM), and significant sensitivity. Therefore, the CePO4@f-CNF nanocomposite was used to study the real-time detection in chicken and fish samples, and it depicted significant results.
Full article
(This article belongs to the Special Issue Electrochemical Sensors and Biosensors for Environmental Detection)
Open AccessArticle
Influence of pH on Room-Temperature Synthesis of Zinc Oxide Nanoparticles for Flexible Gas Sensor Applications
by
Fazia Mechai, Ahmad Al Shboul, Mohand Outahar Bensidhoum, Hossein Anabestani, Mohsen Ketabi and Ricardo Izquierdo
Chemosensors 2024, 12(5), 83; https://doi.org/10.3390/chemosensors12050083 - 16 May 2024
Abstract
This research contributes to work on synthesizing zinc oxide nanoparticles (ZnO NPs) at room temperature (RT) and their utilization in flexible gas sensors. RT ZnO NP synthesis with a basicity solution (pH ≈ 13) demonstrates an efficient method for synthesizing well-crystalline ZnO NPs
[...] Read more.
This research contributes to work on synthesizing zinc oxide nanoparticles (ZnO NPs) at room temperature (RT) and their utilization in flexible gas sensors. RT ZnO NP synthesis with a basicity solution (pH ≈ 13) demonstrates an efficient method for synthesizing well-crystalline ZnO NPs (RT.pH13) comparable to those synthesized by the hydrothermal method (hyd.C). The RT.pH13 achieved a high thermal stability with minimal organic reside impurities (~4.2 wt%), 30–80 nm particle size distribution, and a specific surface area (14 m2 g−1). The synthesized pre- and post-calcinated RT.pH13 NPs were then incorporated into flexible sensors for gas sensing applications at ambient conditions (RT and relative humidity of 30–50%). The pre-calcinated ZnO-based sensor (RT.pH13) demonstrated superior sensitivity to styrene and acetic acid and lower sensitivity to dimethyl-6-octenal. The calcinated ZnO-based sensor (RT.pH13.C) exhibited lower sensitivity to styrene and acetic acid, but heightened sensitivity to benzene, acetone, and ethanol. This suggests a correlation between sensitivity and structural transformations following calcination. The investigation of the sensing mechanisms highlighted the role of surface properties in the sensors’ affinity for specific gas molecules and temperature and humidity variations. The study further explored the sensors’ mechanical flexibility, which is crucial for flexible Internet of Things (IoT) applications.
Full article
(This article belongs to the Special Issue Advances in Gas Sensors and their Application)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Synergistic Enhancement Effect of Ag/rGO as SERS Platform for Capture and Trace Detection of Fenvalerate Molecules
by
Minghui Yu, Chongyang Qin, Zhi Yu, Biao Sun, Dejiang Ni, De Zhang and Pei Liang
Chemosensors 2024, 12(5), 82; https://doi.org/10.3390/chemosensors12050082 - 16 May 2024
Abstract
Surface-enhanced Raman scattering (SERS) provides an alternative rapid detection method for pesticide residues in food, but fenvalerate possesses poor affinity to the novel metal substrate, thus restricting its analysis. To break this bottleneck, a SERS-active platform with an Ag/rGO composite structure was engineered
[...] Read more.
Surface-enhanced Raman scattering (SERS) provides an alternative rapid detection method for pesticide residues in food, but fenvalerate possesses poor affinity to the novel metal substrate, thus restricting its analysis. To break this bottleneck, a SERS-active platform with an Ag/rGO composite structure was engineered using a facile method for fenvalerate detection. Ag nanoparticles with a 60 nm diameter can grow evenly on the top and bottom of rGO layers under intense ultrasonic oscillation, and rGO in hybrid material acts as an ideal hotspot holder between the gaps of Ag nanoparticles, not only allowing the interaction area to be enhanced both electromagnetically and chemically but also enabling the capture and enrichment of fenvalerate pesticide molecules into the “hotspot” area to improve detection sensitivity. Ag/rGO composite substrate possesses superior SERS performance with an ultralow detectable concentration of 4-aminothiophenol (10−10 M) and good reproducibility, endowing the material with a better enhancement effect than pure Ag nanoparticles. When used as the SERS substrate for fenvalerate detection, Ag/rGO composite material showed excellent performance in both experiments and theoretical calculation, with the limit of detection (LOD) of fenvalerate being as low as 1.69 × 10−5 mg/kg and a detection model with an R2 of 99.2%, demonstrating its exciting potential as a SERS substrate for pesticides detection.
Full article
(This article belongs to the Special Issue Recent Advances in Optical Chemo- and Biosensors)
►▼
Show Figures

Figure 1
Open AccessReview
Conducting Polymers in Amperometric Sensors: A State of the Art over the Last 15 Years with a Focus on Polypyrrole-, Polythiophene-, and Poly(3,4-ethylenedioxythiophene)-Based Materials
by
Maria I. Pilo, Gavino Sanna and Nadia Spano
Chemosensors 2024, 12(5), 81; https://doi.org/10.3390/chemosensors12050081 - 11 May 2024
Abstract
Conducting polymers are used in a wide range of applications, especially in the design and development of electrochemical sensors. Their main advantage, in this context, is their ability to efficiently modify an electrode surface using the direct polymerization of a suitable monomer in
[...] Read more.
Conducting polymers are used in a wide range of applications, especially in the design and development of electrochemical sensors. Their main advantage, in this context, is their ability to efficiently modify an electrode surface using the direct polymerization of a suitable monomer in an electrochemical cell, or by physical coating. Additionally, the conducting polymers can be mixed with further materials (metal nanoparticles, carbonaceous materials) to enhance conductivity and analytical features (linear range, limit of detection, sensitivity, and selectivity). Due to their characteristics, conducting polymer-based amperometric sensors are applied to the determination of different organic and inorganic analytes. A view of recent advances in this field focusing on pyrrole, thiophene, and 3,4-ethylenedioxythiophene as starting materials is reported.
Full article
(This article belongs to the Special Issue Recent Advances in Electrode Materials for Electrochemical Sensing)
►▼
Show Figures

Figure 1
Open AccessArticle
A Naked-Eye Colorimetric Ratio Method for the Selective and Sensitive Detection of L-Cys Based on a Silver Nanoflakes–Chromium (III) Ion System
by
Xi Zhang, Yunyi Zhang, Yuwei Gu, Junyu Zhou, Ming Li and Jian Qi
Chemosensors 2024, 12(5), 80; https://doi.org/10.3390/chemosensors12050080 - 11 May 2024
Abstract
As a necessary sulfhydryl amino acid, L-cysteine (L-Cys) maintains many physiological functions in the biological system. However, abnormal L-Cys levels can cause a variety of diseases. In our work, a highly sensitive and selective assay has been developed for sensing L-Cys using the
[...] Read more.
As a necessary sulfhydryl amino acid, L-cysteine (L-Cys) maintains many physiological functions in the biological system. However, abnormal L-Cys levels can cause a variety of diseases. In our work, a highly sensitive and selective assay has been developed for sensing L-Cys using the morphological transformation of silver-based materials induced by Cr3+. In this sensing system, Cr3+ could etch the silver nanoflakes into silver nanoparticles, accompanied by a change in absorbance, which decreases at 395 nm, creates a new peak at 538 nm, and keeps increasing the absorbance with the addition of Cr3+ concentration. Meanwhile, under the naked eye, the solution color changes from bright yellow to dark purple. Because of the strong affinity between L-Cys and Cr3+, L-Cys could inhibit the induction of Cr3+ on silver-based materials, thereby preventing changes in the configuration, absorption spectrum, and color of silver-based materials. Taking advantage of this point, we can quantitatively detect the concentration of L-Cys. A linear relationship between the absorbance ratio (A538 nm/A395 nm) and L-Cys concentration was found in the range of 0.1–0.9 μM, and the detection limit was 41.2 nM. The strategy was applied to measure L-Cys spiked in beer and urine samples, with recovery from 93.80 to 104.03% and 93.33% to 107.14% and RSD from 0.89 to 2.40% and 1.80% to 6.78%, respectively. This detection strategy demonstrates excellent selectivity and sensitivity, which makes it a practical and effective method for the detection of L-Cys in real samples.
Full article
(This article belongs to the Special Issue Low-Cost Chemosenors for Applications in Environment, Health, Food, and Industry Process Control)
►▼
Show Figures

Figure 1
Open AccessArticle
Synthesis, Spectral Characteristics, Sensing Properties and Microbiological Activity of New Water-Soluble 4-Sulfo-1,8-naphthalimides
by
Awad I. Said, Desislava Staneva, Evgenia Vasileva-Tonkova, Petar Grozdanov, Ivanka Nikolova, Radostina Stoyanova, Albena Jordanova and Ivo Grabchev
Chemosensors 2024, 12(5), 79; https://doi.org/10.3390/chemosensors12050079 - 9 May 2024
Abstract
A new water-soluble polyamidoamine (PAMAM) dendrimer modified with 4-sulfo-1,8-naphthalimide (DSNI) and its monomeric structural analogue (MSDI) were synthesized. Their photophysical properties were investigated in organic solvents of different polarities and aqueous solutions. The effect of pH on fluorescence intensity was determined. It was
[...] Read more.
A new water-soluble polyamidoamine (PAMAM) dendrimer modified with 4-sulfo-1,8-naphthalimide (DSNI) and its monomeric structural analogue (MSDI) were synthesized. Their photophysical properties were investigated in organic solvents of different polarities and aqueous solutions. The effect of pH on fluorescence intensity was determined. It was found that the dendrimer emits blue fluorescence in an acidic medium, which is quenched in an alkaline environment. This phenomenon is due to the possibility of suppression of nonradiative photoinduced electron transfer in acidic media. The influence of different metal ions (Cu2+, Pb2+, Sn2+, Sr2+, Mg2+, Ba2+, Co2+, Hg2+, Zn2+, Ni2+, Fe3+, Al3+) and anions (CN−, S2−, S2O52−, HPO42−, H2PO4−, F−, CH3COO−, NO2−, CO32−, SO42−) on the intensity of the emitted fluorescence was studied. Quenching was only found in the presence of Cu2+. This makes the dendrimer suitable for determining copper ions in water solutions in the presence of other metal ions and anions. Additionally, DSNI was used as a ligand to obtain a stable copper complex, the structure of which was investigated by electron paramagnetic resonance (EPR), infrared spectrum, and elemental analysis. Two copper ions were found to form a complex with one dendrimer. The in vitro microbiological activity of the new compounds against bacteria Pseudomonas aeruginosa and two viruses HRSV-2 and HAdV-5 was investigated. With a view to obtaining antibacterial and anti-viral textiles, cotton fabrics were treated with the three compounds, and then their activity against the same microbial strains was investigated. It was found that the microbiological activity was preserved after the application of the new compounds to the cotton fabrics.
Full article
(This article belongs to the Special Issue Chemosensors in Biological Challenges, Volume II)
►▼
Show Figures

Graphical abstract
Open AccessReview
Chemiresistive Materials for Alcohol Vapor Sensing at Room Temperature
by
Anna Maria Laera and Michele Penza
Chemosensors 2024, 12(5), 78; https://doi.org/10.3390/chemosensors12050078 - 7 May 2024
Abstract
The development of efficient sensors able to detect alcoholic compounds has great relevance in many fields including medicine, pharmaceuticals, food and beverages, safety, and security. In addition, the measurements of alcohols in air are significant for environmental protection because volatile alcohols can have
[...] Read more.
The development of efficient sensors able to detect alcoholic compounds has great relevance in many fields including medicine, pharmaceuticals, food and beverages, safety, and security. In addition, the measurements of alcohols in air are significant for environmental protection because volatile alcohols can have harmful effects on human health not only through ingestion, but also through inhalation or skin absorption. The analysis of alcohols in breath is a further expanding area, being employed for disease diagnoses. The analyses performed by using chromatography, mass-spectrometry, nuclear magnetic resonance, ultraviolet-visible spectroscopy, Fourier-transform infrared spectroscopy, or Raman spectroscopy often require complex sampling and procedures. As a consequence, many research groups have focused their efforts on the development of efficient portable sensors to replace conventional methods and bulky equipment. The ability to operate at room temperature is a key factor in designing portable light devices suitable for in situ real-time monitoring. In the present review, we provide a survey of the recent literature on the most efficient chemiresistive materials for alcohol sensing at room temperature. Remarkable gas-sensing performances have mainly been obtained by using metal oxides semiconductors (MOSs), metal organic frameworks (MOFs), 2D materials, and polymers. Among 2D materials, we mainly consider graphene-based materials, graphitic carbon nitride, transition metal chalcogenides, and MXenes. We discuss scientific advances and innovations published in the span of the last five years, focusing on sensing mechanisms.
Full article
(This article belongs to the Special Issue Innovative Nanomaterials-Based Chemosensor Devices for Air Quality Monitoring)
►▼
Show Figures

Figure 1
Open AccessArticle
Liquid-Phase Exfoliated Graphene and Polytetrafluoroethylene for Highly Durable and Reusable Chemical Leak Detection Sensors
by
Najaf Rubab, Eunbee Sohn, Won-Seok Kang and TaeYoung Kim
Chemosensors 2024, 12(5), 77; https://doi.org/10.3390/chemosensors12050077 - 7 May 2024
Abstract
Graphene-based chemical sensors hold promise across diverse applications owing to their exceptional sensitivity and selectivity. However, achieving their long-term durability and reusability while preserving high sensitivity remains a significant challenge, particularly in harsh environments where exposure to strong chemicals is inevitable. This paper
[...] Read more.
Graphene-based chemical sensors hold promise across diverse applications owing to their exceptional sensitivity and selectivity. However, achieving their long-term durability and reusability while preserving high sensitivity remains a significant challenge, particularly in harsh environments where exposure to strong chemicals is inevitable. This paper presents a novel approach to address this challenge by synergistically integrating liquid-phase exfoliated graphene (LPEG) with polytetrafluoroethylene (PTFE) within a single sensing strip. Through a comprehensive experimental investigation, we demonstrate the fabrication of highly durable and reusable chemical leak detection sensors by combining LPEG and PTFE. Furthermore, we explore the sensing mechanism, highlighting the roles of LPEG and PTFE in enhancing sensitivity and selectivity, along with durability and reusability. Performance evaluation reveals the sensors’ robustness against mechanical and chemical degradation, coupled with excellent recyclability. This innovative approach holds promise for applications in environmental monitoring, industrial safety, and healthcare, thus advancing the field of graphene-based chemical leak detection sensors.
Full article
(This article belongs to the Special Issue Novel Materials for Sensing, Imaging and Energy Conversion/Storage)
►▼
Show Figures

Figure 1
Open AccessArticle
Influence of Silsesquioxane-Containing Ultra-Thin Polymer Films on Metal Oxide Gas Sensor Performance for the Tunable Detection of Biomarkers
by
Oleg Lupan, Mihai Brinza, Julia Piehl, Nicolai Ababii, Nicolae Magariu, Lukas Zimoch, Thomas Strunskus, Thierry Pauporte, Rainer Adelung, Franz Faupel and Stefan Schröder
Chemosensors 2024, 12(5), 76; https://doi.org/10.3390/chemosensors12050076 - 5 May 2024
Abstract
Certain biomarkers in exhaled breath are indicators of diseases in the human body. The non-invasive detection of such biomarkers in human breath increases the demand for simple and cost-effective gas sensors to replace state-of-the-art gas chromatography (GC) machines. The use of metal oxide
[...] Read more.
Certain biomarkers in exhaled breath are indicators of diseases in the human body. The non-invasive detection of such biomarkers in human breath increases the demand for simple and cost-effective gas sensors to replace state-of-the-art gas chromatography (GC) machines. The use of metal oxide (MOX) gas sensors based on thin-film structures solves the current limitations of breath detectors. However, the response at high humidity levels, i.e., in the case of exhaled human breath, significantly decreases the sensitivity of MOX sensors, making it difficult to detect small traces of biomarkers. We have introduced, in previous work, the concept of a hybrid gas sensor, in which thin-film-based MOX gas sensors are combined with an ultra-thin (20–30 nm) polymer top layer deposited by solvent-free initiated chemical vapor deposition (iCVD). The hydrophobic top layer enables sensor measurement in high-humidity conditions as well as the precise tuning of selectivity and sensitivity. In this paper, we present a way to increase the hydrogen (H2) sensitivity of hybrid sensors through chemical modification of the polymer top layer. A poly(1,3,5,7-tetramethyl-tetravinylcyclotetrasiloxane) (PV4D4) thin film, already applied in one of our previous studies, is transformed into a silsesquioxane-containing top layer by a simple heating step. The transformation results in a significant increase in the gas response for H2 ~709% at an operating temperature of 350 °C, which we investigate based on the underlying sensing mechanism. These results reveal new pathways in the biomedical application field for the analysis of exhaled breath, where H2 indicates gastrointestinal diseases.
Full article
(This article belongs to the Special Issue Innovative Nanomaterials-Based Chemosensor Devices for Air Quality Monitoring)
►▼
Show Figures

Graphical abstract
Open AccessArticle
A Foldable Thermoplastic Microdevice Integrating Isothermal Amplification and Schiff-Reaction-Based Colorimetric Assay for the Detection of Infectious Pathogens
by
Hee Mang Kim and Nae Yoon Lee
Chemosensors 2024, 12(5), 75; https://doi.org/10.3390/chemosensors12050075 - 3 May 2024
Abstract
In this study, we introduce a plastic-based foldable microdevice that integrates loop-mediated isothermal amplification (LAMP) and a colorimetric assay based on the Schiff reaction to detect the genes of infectious bacteria. The device comprises two sides: a sample zone containing amplification chambers and
[...] Read more.
In this study, we introduce a plastic-based foldable microdevice that integrates loop-mediated isothermal amplification (LAMP) and a colorimetric assay based on the Schiff reaction to detect the genes of infectious bacteria. The device comprises two sides: a sample zone containing amplification chambers and a detection zone for the colorimetric assay. The detection zone contains poly(methyl methacrylate) structures for transferring the colorimetric reagent-soaked glass micro-fiber paper into the sample chambers. Specific genes of Staphylococcus aureus (S. aureus) and Streptococcus pneumoniae (S. pneumoniae), the most common bacterial infection causes, were amplified by LAMP assay. The S. aureus gene was detected up to 10 fg/μL and the S. pneumoniae gene up to 0.1 pg/μL. The amplified target genes were visually identified using a colorimetric assay with Schiff’s reagent, which showed clear color discrimination through a reaction with aldehyde groups derived from the DNA in the amplicons. The introduced method, integrating amplification and detection processes in a single device, is expected to be utilized in point-of-care testing analysis for the simple and rapid detection of infectious pathogens.
Full article
(This article belongs to the Section (Bio)chemical Sensing)
►▼
Show Figures

Figure 1
Open AccessArticle
A High-Precision Monitoring Method Based on SVM Regression for Multivariate Quantitative Analysis of PID Response to VOC Signals
by
Xiujuan Feng, Zengyuan Liu, Yongjun Ren and Chengliang Dong
Chemosensors 2024, 12(5), 74; https://doi.org/10.3390/chemosensors12050074 - 3 May 2024
Abstract
In the moist environment of soil-water-air, there is a problem of low accuracy in monitoring volatile organic compounds (VOCs) using a photoionization detector (PID). This study is based on the PID water-soil-gas VOC online monitor developed by this group, online monitoring of the
[...] Read more.
In the moist environment of soil-water-air, there is a problem of low accuracy in monitoring volatile organic compounds (VOCs) using a photoionization detector (PID). This study is based on the PID water-soil-gas VOC online monitor developed by this group, online monitoring of the concentration of different constituents of VOCs in different production enterprises of the petroleum and chemical industries in Shandong Province, with the concentration of the laboratory test, to build a relevant model. The correlation coefficient about the PID test concentration and the actual concentration correlation coefficient was obtained through the collection of a large number of data trainings. Based on the application of PID in VOC monitoring, the establishment of a PID high-precision calibration model is important for the precise monitoring of VOCs. In this paper, multiple quantitative analyses were conducted, based on SVM regression of PID response to VOC signals, to study the high-precision VOC monitoring method. To select the response signals of PID under different concentrations of environmental VOCs measured by the research group, first, the PID response to VOC signals was modeled using the support vector machine principle to verify the effect of traditional SVM regression. For the problem of raw data redundancy, calculate the time-domain and frequency-domain characteristics of the PID signal, and conduct the principal component analysis of the time-domain of the PID signal. In order to make the SVM regression more generalized and robust, the selection of kernel function parameters and penalty factor of SVM is optimized by genetic algorithm. By comparing the accuracy of PID calibration models such as PID signal feature extraction, SVM regression, and principal component analysis SVM regression, the superiority of photoionization detector using the signal feature extraction PCA-GA-SVM method to monitor VOCs is verified.
Full article
(This article belongs to the Special Issue Chemical Sensors for Volatile Organic Compound Detection, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessArticle
Biosensor-Based Assessment of Pesticides and Mineral Fertilizers’ Influence on Ecotoxicological Parameters of Soils under Soya, Sunflower and Wheat
by
Ludmila Khmelevtsova, Maria Klimova, Shorena Karchava, Tatiana Azhogina, Elena Polienko, Alla Litsevich, Elena Chernyshenko, Margarita Khammami, Ivan Sazykin and Marina Sazykina
Chemosensors 2024, 12(5), 73; https://doi.org/10.3390/chemosensors12050073 - 2 May 2024
Abstract
Pesticides and fertilizers used in agriculture can negatively affect the soil, increasing its toxicity. In this work, a battery of whole-cell bacterial lux-biosensors based on the E. coli MG1655 strain with various inducible promoters, as well as the natural luminous Vibrio aquamarinus VKPM
[...] Read more.
Pesticides and fertilizers used in agriculture can negatively affect the soil, increasing its toxicity. In this work, a battery of whole-cell bacterial lux-biosensors based on the E. coli MG1655 strain with various inducible promoters, as well as the natural luminous Vibrio aquamarinus VKPM B-11245 strain, were used to assess the effects of agrochemical soil treatments. The advantages of using biosensors are sensitivity, specificity, low cost of analysis, and the ability to assess the total effect of toxicants on a living cell and the type of their toxic effect. Using the V. aquamarinus VKPM B-11245 strain, the synergistic effect of combined soil treatment with pesticides and mineral fertilizers was shown, which led to an increase in the overall (integral) toxicity of soils higher than that of the individual application of substances. Several probable implementation mechanisms of agrochemical toxic effects have been discovered. DNA damage caused by both SOS response induction and alkylation, oxidative stress due to increased superoxide levels, and damage to cellular proteins and membranes are among them. Thus, the usage of biosensors makes it possible to assess the cumulative effect of various toxicants on living organisms without using expensive chemical analyses.
Full article
(This article belongs to the Special Issue Chemiluminescent and Bioluminescent Sensors)
►▼
Show Figures
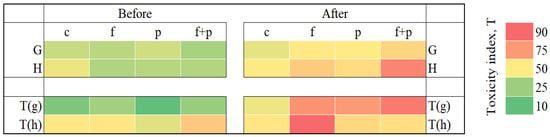
Figure 1
Open AccessReview
Research Progress on Molecularly Imprinted Materials for the Screening and Identification of Organic Pollutants
by
Jialing Song, Xuanhao Lin, Liang Ying Ee and Sam F. Y. Li
Chemosensors 2024, 12(5), 72; https://doi.org/10.3390/chemosensors12050072 - 2 May 2024
Abstract
Organic pollutants, distinguished by their persistence and bioaccumulation in the environment, pose significant ecological and health threats that surpass those of traditional pollutants. Crucial to understanding their environmental behavior, health risks, and mitigation strategies, is the screening and identification of these pollutants. This
[...] Read more.
Organic pollutants, distinguished by their persistence and bioaccumulation in the environment, pose significant ecological and health threats that surpass those of traditional pollutants. Crucial to understanding their environmental behavior, health risks, and mitigation strategies, is the screening and identification of these pollutants. This process indispensably employs functional materials, among which molecularly imprinted polymers (MIPs) prove to be particularly advantageous because of their specific recognition capabilities and extensive application range. This review presents cutting-edge techniques and strategies for the fabrication of MIPs, including surface imprinting techniques and dummy molecular strategies. It encapsulates the last five years’ advancements in MIP research within the domains of sample pretreatment, as well as optical and electrochemical sensing analysis. The objective of this discourse is to potentially foster the evolution of MIP technology and establish the groundwork for its transition from lab-scale to commercial production.
Full article
(This article belongs to the Special Issue A Comprehensive Review on Chemical Sensors: Materials, Physico-Chemical Properties and Devices)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Sprayable Diacetylene-Containing Amphiphile Coatings for Visual Detection of Gas-Phase Hydrogen Peroxide
by
Priyanka Shiveshwarkar and Justyn Jaworski
Chemosensors 2024, 12(5), 71; https://doi.org/10.3390/chemosensors12050071 - 1 May 2024
Abstract
Colorimetric chemical sensing of target gases, such as hydrogen peroxide vapors, is an evolving area of research that implements responsive materials that undergo molecule-specific interaction, resulting in a visible color change. Due to the intuitive nature of an observable color change, such sensing
[...] Read more.
Colorimetric chemical sensing of target gases, such as hydrogen peroxide vapors, is an evolving area of research that implements responsive materials that undergo molecule-specific interaction, resulting in a visible color change. Due to the intuitive nature of an observable color change, such sensing systems are particularly desirable as they can be widely deployed at low cost and without the need for complex analytical instrumentation. In this work, we describe our development of a new spray-on sensing material that can provide a colorimetric response to the presence of a gas-phase target, specifically hydrogen peroxide vapor. By providing a cumulative response over time, we identified that part per million concentrations of hydrogen peroxide vapor can be detected. Specifically, we make use of iron chloride-containing formulations to enable the catalysis of hydrogen peroxide to hydroxyl radicals that serve to initiate polymerization of the diacetylene-containing amphiphile, resulting in a white to blue color transition. Due to the irreversible nature of the color change mechanism, the cumulative exposure to hydrogen peroxide over time is demonstrated, enabling longitudinal assessment of target exposure with the same coatings. The versatility of this approach in generating a colorimetric response to hydrogen peroxide vapor may find practical applications for environmental monitoring, diagnostics, or even industrial safety.
Full article
(This article belongs to the Special Issue Revolutionizing the Future: Cutting-Edge Chemical Sensor Technologies in the USA—2024 Insights and Innovations)
►▼
Show Figures
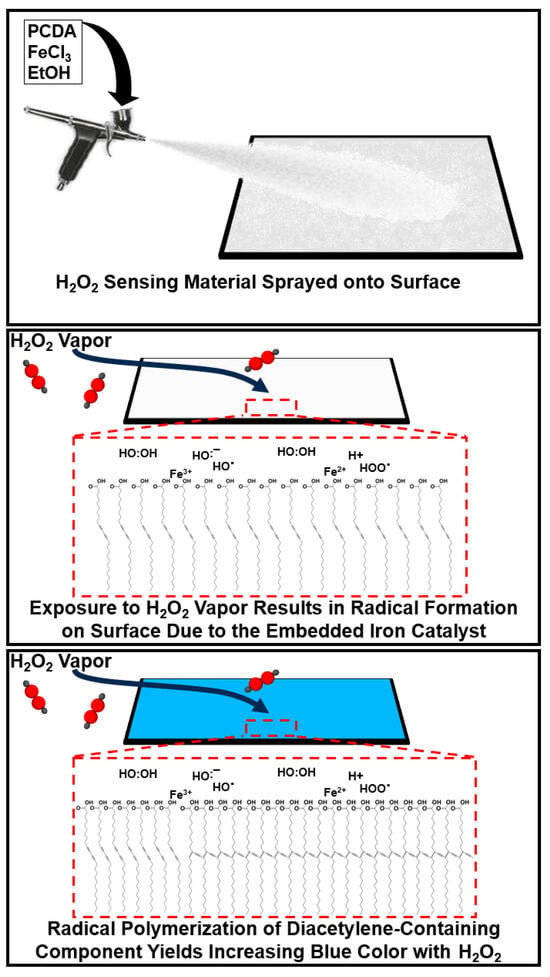
Figure 1
Open AccessArticle
Controlling Fluorescence Wavelength in the Synthesis of TGA-Capped CdTe Quantum Dots
by
Catarina S. M. Martins, Ana L. Silva, Luís Pleno de Gouveia, Ihsan Çaha, Oleksandr Bondarchuk, Alec P. LaGrow, Francis Leonard Deepak and João A. V. Prior
Chemosensors 2024, 12(4), 70; https://doi.org/10.3390/chemosensors12040070 - 21 Apr 2024
Abstract
Quantum dots (QDs) are semiconductor materials, with a size range between 1–10 nm, showcasing unique size-dependent physical and chemical properties. Such properties have potentiated their use in areas like medical imaging and biosensing. Herein, we present an open-air approach for synthesis of QDs,
[...] Read more.
Quantum dots (QDs) are semiconductor materials, with a size range between 1–10 nm, showcasing unique size-dependent physical and chemical properties. Such properties have potentiated their use in areas like medical imaging and biosensing. Herein, we present an open-air approach for synthesis of QDs, reducing the need for controllable atmospheric conditions. Furthermore, we present a predictive mathematical model for maximum emission wavelength (λmax) control. Through a straightforward microwave-based aqueous synthesis of TGA-CdTe QDs, we investigated the influence of time, temperature, and Te:Cd and TGA:Cd molar ratios on λmax, using a chemometric experimental design approach. CdTe-QDs were characterized by UV-Vis and fluorescence spectroscopies. Additionally, Fourier-Transform Infrared spectroscopy, X-ray photoelectron spectroscopy, Transmission Electron Microscopy, and Energy Dispersive X-ray were conducted. Stable QDs with fluorescence ranging from green to red (527.6 nm to 629.2 nm) were obtained. A statistical analysis of the results revealed that time and temperature were the most significant factors influencing λmax. After fine-tuning the variables, a mathematical model with 97.7% of prediction accurately forecasted experimental conditions for synthesizing TGA-CdTe QDs at predefined λmax. Stability tests demonstrated that the QDs retained their optical characteristics for over a month at 4 °C, facilitating diverse applications.
Full article
(This article belongs to the Section Optical Chemical Sensors)
►▼
Show Figures

Figure 1
Open AccessArticle
Electrochemical Sensor for the Evaluation of Doxorubicin from Novel Pharmaceutical Formulations and Serum
by
Alexandra Pusta, Mihaela Tertis, Irina Bura, Diana Bogdan, Maria Suciu, Simona Mirel and Cecilia Cristea
Chemosensors 2024, 12(4), 69; https://doi.org/10.3390/chemosensors12040069 - 19 Apr 2024
Abstract
This study focuses on addressing the challenges associated with doxorubicin (DOX), an anthracycline chemotherapeutic widely used in cancer treatment. Despite its efficacy, DOX is linked to severe side effects that limit its clinical applications. Novel pharmaceutical formulations aim to mitigate these issues, providing
[...] Read more.
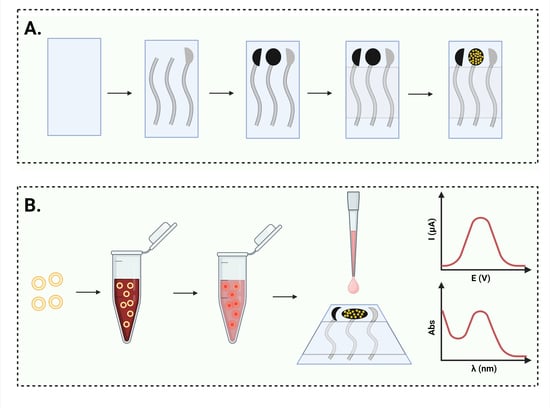
This study focuses on addressing the challenges associated with doxorubicin (DOX), an anthracycline chemotherapeutic widely used in cancer treatment. Despite its efficacy, DOX is linked to severe side effects that limit its clinical applications. Novel pharmaceutical formulations aim to mitigate these issues, providing better safety profiles. The development of these formulations requires analytical methods that can accurately and quickly quantify DOX. A cost-effective and portable electrochemical sensor for DOX detection was developed utilizing in-house printed carbon electrodes decorated with gold nanoparticles. DOX was detected using differential pulse voltammetry. The sensor demonstrated an accurate quantification of DOX from novel pharmaceutical formulations and serum, presenting a dynamic range of 1 to 500 μg/mL and a low detection limit of 0.3 μg/mL. The method, successfully applied to characterize DOX-loaded nanosomes, offers a valuable alternative in the early stages of formulation development, reducing costs and saving time, while maintaining accuracy.
Full article
(This article belongs to the Special Issue Application of Carbon Nanotubes in Sensing)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Rapid and Sensitive Detection of Influenza B Virus Employing Nanocomposite Spheres Based on Ag-Doped ZnIn2S4 Quantum Dots
by
Jia-Xuan Hu, Li-Bang Zhu, Sheng-Tong Wu and Shou-Nian Ding
Chemosensors 2024, 12(4), 68; https://doi.org/10.3390/chemosensors12040068 - 19 Apr 2024
Abstract
Lateral flow immunoassay (LFIA) technology serves a significant role as a simple and rapid biosensor in the detection of influenza viruses. The focus of this study is the development of a rapid and convenient screening method for influenza B virus (IBV) proteins using
[...] Read more.
Lateral flow immunoassay (LFIA) technology serves a significant role as a simple and rapid biosensor in the detection of influenza viruses. The focus of this study is the development of a rapid and convenient screening method for influenza B virus (IBV) proteins using a fluorescence lateral flow biosensor based on Ag-doped ZnIn2S4 quantum dots (Ag: ZIS QDs) as signal reporters. These Ag: ZIS QDs-emitting orange fluorescence are loaded onto dendritic mesoporous silica nanoparticles (DMSNs) and are further coated with a layer of silica shell to form a core–shell structured composite nanomaterial (SiO2 @ Ag: ZIS QDs @ DMSNs). The orange fluorescence effectively eliminates the interference of blue background fluorescence, significantly enhancing the detection sensitivity. This technology demonstrates outstanding performance in the immediate detection of IBV, with a minimum detection limit of 1 ng/mL, compared to the traditional colloidal gold strip with a detection limit of 6 ng/mL. Furthermore, both intra-assay and inter-assay coefficients of variation (CV) are less than 9%. This method holds promise for wide application in early diagnosis, epidemiological investigation, and epidemic surveillance of IBV.
Full article
(This article belongs to the Special Issue Rapid Point-of-Care Testing Technology and Application)
►▼
Show Figures

Figure 1
Open AccessArticle
High Sensitivity Hydrogen Sensor via the Coupling of Tamm Plasmon Polaritons and Defect Mode
by
Feng Zhang, Weifeng Yin and Jianxia Zhang
Chemosensors 2024, 12(4), 67; https://doi.org/10.3390/chemosensors12040067 - 18 Apr 2024
Abstract
Optical hydrogen sensors offer high sensitivity, high accuracy, and non-invasive sensing capabilities, making them promising devices in various fields, including the construction of hydrogen fuel cells, storage and transportation, and aerospace. However, to achieve better sensitivity and faster reaction times, such sensors are
[...] Read more.
Optical hydrogen sensors offer high sensitivity, high accuracy, and non-invasive sensing capabilities, making them promising devices in various fields, including the construction of hydrogen fuel cells, storage and transportation, and aerospace. However, to achieve better sensitivity and faster reaction times, such sensors are often constructed as nano-arrays or nano-gratings, leading to increased manufacturing costs and complexity. In this study, we propose and demonstrate a highly sensitive hydrogen sensor based on a multilayer structure. The proposed structure consists of a Pd metal film and a photonic crystal with a defect layer, in which the photonic crystal is designed by an alternating arrangement of Ta2O5 and SiO2, and the material comprising the defect layer is SiO2. With a sensitivity of up to 16,020 at 670 nm, the proposed sensor relies on the coupling of Tamm plasmon polaritons and defect modes. The electric field distribution inside the structure is also provided in order to reveal its physical mechanism. Furthermore, we investigate the effects of the thickness of the defect layer and the angle of incident light on the sensor’s performance. The study results show that the sensor has good fault tolerance in either scenario. The findings of this study open up new possibilities for hydrogen sensor applications.
Full article
(This article belongs to the Section Nanostructures for Chemical Sensing)
►▼
Show Figures
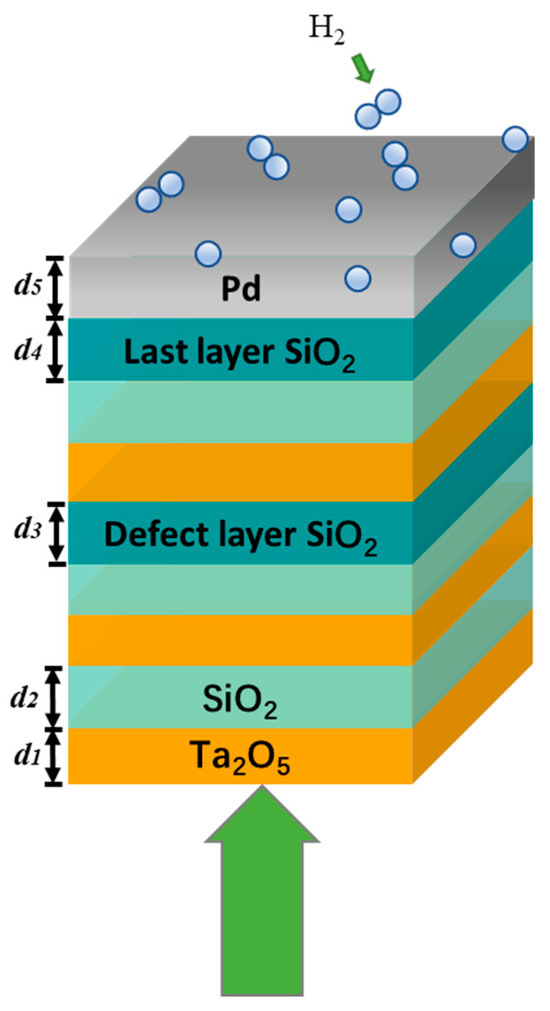
Figure 1
Open AccessArticle
AuNPs/Ti3C2 Signal-Enhanced Surface Plasmon Resonance Imaging Biosensor for Ultrasensitive Detection of miRNA
by
Yirui Qin, Li Jiang, Rengang Sun, Yunzhu Fang, Boya Shi and Shangzhong Jin
Chemosensors 2024, 12(4), 66; https://doi.org/10.3390/chemosensors12040066 - 17 Apr 2024
Abstract
MicroRNA-21 is a potential cancer biomarker that is highly expressed in many cancer cells. Therefore, it is important to perform highly sensitive detection of miRNA-21. In this study, we designed a surface plasmon resonance imaging (SPRi) sensor based on an AuNPs/Ti3C
[...] Read more.
MicroRNA-21 is a potential cancer biomarker that is highly expressed in many cancer cells. Therefore, it is important to perform highly sensitive detection of miRNA-21. In this study, we designed a surface plasmon resonance imaging (SPRi) sensor based on an AuNPs/Ti3C2 composite for real-time and highly sensitive detection of miRNA-21. The fixation of the capture polyA-DNA probes was completed by the freezing method, which improved the detection efficiency. DNA−AuNPs/Ti3C2 conjugates were added to amplify the SPRi signal. The signal amplification combines the large specific surface area of Ti3C2 and the electronic coupling between the local surface plasmon resonance (LSPR) of AuNPs and the plasmon wave on the surface of the Au chip, thereby enhancing the SPRi response signal. Using this sensing strategy, the detection limit for miRNA-21 can reach 6.13 fM, with a wide dynamic range between 10 fM and 10 nM. In addition, the sensor has excellent selectivity for miRNA-21 and miRNAs with similar sequences, and receives minimal interference when applied to complex matrices. Based on these results, we believe that this study provides a simple and highly sensitive method for miRNA detection, which has great potential for the quantitative detection of miRNA in biomedical research and early clinical diagnosis.
Full article
(This article belongs to the Special Issue Advanced Surface Plasmon Resonance Sensors)
►▼
Show Figures
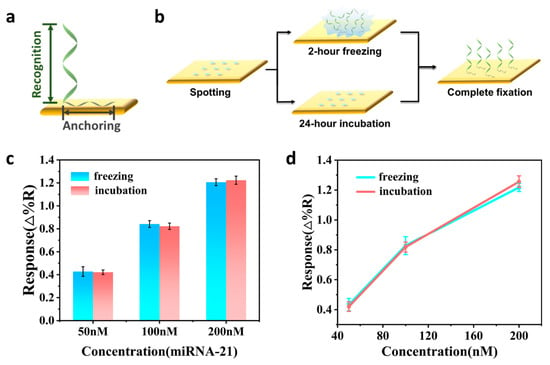
Figure 1
Open AccessArticle
Study of the Gas Sensing Performance of Ni-Doped Perovskite-Structured LaFeO3 Nanospheres
by
Fanli Meng, Zhenhua Yu, Renze Zhang, Hongliang Gao and Zhenyu Yuan
Chemosensors 2024, 12(4), 65; https://doi.org/10.3390/chemosensors12040065 - 16 Apr 2024
Abstract
This study synthesizes Ni-doped perovskite-structured LaFeO3 composite materials via a one-step hydrothermal method, characterizes the morphology and structure of the materials, and tests their gas sensing performance. The test results show that compared to pure LaFeO3 material, the gas sensing performance
[...] Read more.
This study synthesizes Ni-doped perovskite-structured LaFeO3 composite materials via a one-step hydrothermal method, characterizes the morphology and structure of the materials, and tests their gas sensing performance. The test results show that compared to pure LaFeO3 material, the gas sensing performance of Ni-doped LaFeO3 material is improved in all aspects. Specifically, LFO-Ni2% exhibits a response as high as 102 towards 100 ppm of triethylamine at 190 °C, along with better selectivity and stability. Furthermore, the gas sensing mechanism is investigated. On one hand, doping with an appropriate proportion of Ni can lead to the formation of more-complete and smaller-sized microsphere structures with pores. This is beneficial for the adsorption of oxygen from the air onto the material surface, as well as for the diffusion of the target gas to the surface of the material, thereby enhancing gas sensitivity performance. On the other hand, the doped Ni enters the interior of the LaFeO3 crystal, replacing some of the cations in LaFeO3, increasing the concentration of charge carriers in the material, and reducing the material’s resistance. The sample can adsorb more oxygen, promoting the reaction between adsorbed oxygen and the target gas, and thereby improving the gas sensitivity performance of the sample.
Full article
(This article belongs to the Special Issue Functional Nanomaterial-Based Gas Sensors)
►▼
Show Figures
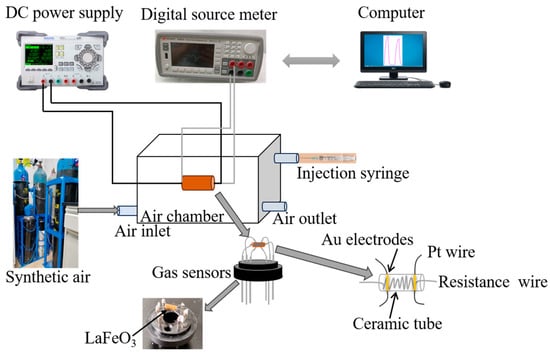
Figure 1

Journal Menu
► ▼ Journal Menu-
- Chemosensors Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Molecules, Biomimetics, Chemosensors, Life, AI, Sci
Recent Advances in Chemical Artificial Intelligence
Topic Editors: Pier Luigi Gentili, Jerzy Górecki, David C Magri, Pasquale StanoDeadline: 1 March 2025

Conferences
Special Issues
Special Issue in
Chemosensors
Fluorescent Probe and Biosensing
Guest Editors: Peng Yin, Sheng YangDeadline: 30 May 2024
Special Issue in
Chemosensors
Nanomaterials-Based Sensors
Guest Editors: Simas Rackauskas, Agne SulciuteDeadline: 30 June 2024
Special Issue in
Chemosensors
Preparation and Application of Photoelectrochemical Sensors
Guest Editors: Mei Yan, Jing ZhangDeadline: 20 July 2024
Special Issue in
Chemosensors
Dedicated to Professor Giorgio Sberveglieri on the Occasion of His 75th Birthday for His Outstanding Contributions to the Field of Chemical Sensors
Guest Editors: Estefanía Núñez Carmona, Veronica SberveglieriDeadline: 30 July 2024
Topical Collections
Topical Collection in
Chemosensors
Women Special Issue in Chemosensors and Analytical Chemistry
Collection Editors: Teresa Corrales, Nicole Jaffrezic-Renault, Eleonora Alfinito
Topical Collection in
Chemosensors
Recent Trend in Chromatography for Pharmaceutical Analysis
Collection Editors: Cláudia Maria Rosa Ribeiro, Maria Elizabeth Tiritan
Topical Collection in
Chemosensors
Optical Chemosensors and Biosensors
Collection Editor: Ambra Giannetti
Topical Collection in
Chemosensors
Sustainable Metal Oxide Materials for Sensing Applications
Collection Editors: Ana Rovisco, Elisabetta Comini







