- Review
Fueling the Fire: How Glutamine Metabolism Sustains Leukemia Growth and Resistance
- Giovannino Silvestri
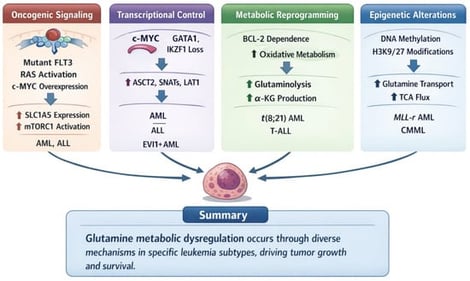
Glutamine metabolism has emerged as one of the most critical bioenergetic and biosynthetic programs sustaining leukemic cell growth, survival, stemness and therapeutic resistance. In both acute and chronic leukemias, including acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL), malignant cells display a strong dependency on extracellular glutamine to support mitochondrial respiration, anabolic biosynthesis and redox homeostasis. This dependency is reinforced by oncogenic signaling networks, post-transcriptional metabolic regulation and microenvironmental adaptation within the bone marrow niche. Therapeutic strategies targeting glutamine utilization, including glutaminase inhibition, transporter blockade and enzymatic glutamine depletion, have demonstrated robust antileukemic activity in preclinical models, and early clinical efforts have begun to explore glutamine-directed interventions in myeloid neoplasms. However, metabolic plasticity, microenvironment-derived nutrient buffering and systemic toxicity remain significant limitations to clinical translation. This review provides a detailed synthesis of the biochemical framework of glutamine metabolism in leukemia, the molecular mechanisms enforcing glutamine addiction, the downstream functional consequences on proliferation, redox balance and leukemic stem cell biology, the current landscape of therapeutic strategies and emerging directions aimed at overcoming resistance and improving clinical efficacy.
4 February 2026