- Article
Prediction and Validation of Phase II Glucuronide Conjugates in Urine Using Combined Non-Targeted and Targeted LC–HRMS/MS Workflows and Their Validation for over 200 Drugs
- Camila Bardy,
- Luis Manuel Menéndez-Quintanal and
- Jose Manuel Matey
- + 3 authors
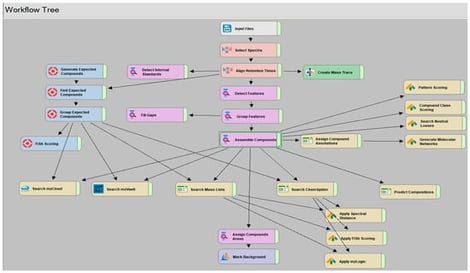
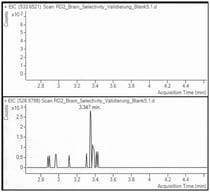
High-resolution mass spectrometry (HRMS) enables non-targeted detection of drugs and metabolites in complex matrices. Phase II metabolites—especially glucuronides—are often the only detectable biomarkers in late or postmortem samples but are underrepresented in commercial libraries. This work pursued the prediction of phase II-glucuronide conjugates in diluted urine samples by non-targeted/targeted LC-HRMS workflows. A simply “dilute-and-shoot” qualitative UHPLC-HRMS/MS method (Q Exactive HF, ddMS2) was integrated with Compound Discoverer® software for data processing. The workflow incorporated predictive strategies such as exact mass suspect lists, Structured Query Language (SQL)-based filters, compound-class and diagnostic neutral-loss rules (including the characteristic loss of 176.0321 Da for glucuronides) and MS/MS confirmation using both in-house and public spectral libraries. An additional part of the application’s performance assessment involved its validation for diluted urine sample. A qualitative validated method for more than two hundred drugs in urine samples was performed, including the method’s selectivity/specificity, limit of identification, matrix effects, and potential carryover. Most analytes fulfilled the qualitative acceptance criteria, with more than 60% successfully identified at a concentration of at least 2.5 ng/mL. Matrix effects were within acceptable limits for most compounds, and no severe ion suppression was observed. A non-targeted workflow was applied to real forensic samples (n = 16), allowing a reduction of approximately 66,800 detected features to 225 glucuronide candidates, while a targeted workflow based on exact mass lists yielded 31 high-confidence identifications. Characteristic neutral losses and diagnostic fragment ions led to the tentative identification of some glucuronide phase II metabolites such as mirtazapine–glucuronide, morphine-6–glucuronide, and glucuronide conjugates of benzodiazepines and synthetic opioids. In conclusion, the integration of biotransformation knowledge with HRMS-based predictive filtering allows for the efficient and hydrolysis-free detection of glucuronide metabolites, thereby extending detection windows and enhancing toxicological interpretation in complex forensic scenarios. This adaptable and library-independent workflow also facilitates retrospective data mining, making it suitable for the identification of emerging substances and newly characterized metabolites.
26 February 2026