- Article
Optimal Timing for Neonatal Hearing Screening in Well-Babies
- Lisanne Vonk,
- Paula van Dommelen and
- Catharina (Kitty) P. B. van der Ploeg
- + 3 authors
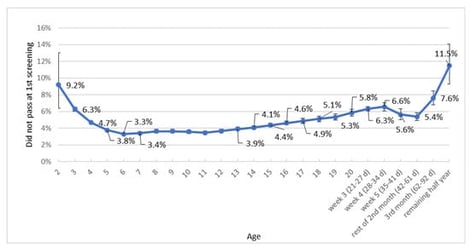
In The Netherlands, preventive child healthcare (PCHC) has been carrying out neonatal hearing screening in well-babies since 2006. The aim of this study was to examine the relationship between the age of newborns and the false positive referral rate of the first hearing screening using a transient evoked otoacoustic emission (OAE) test, to identify the most efficient timing for OAE screening. Additionally, we investigated the relationship between the type of OAE screening device (Echoscreen (ES)I/II versus ESIII) and the referral rate during the first screening. We used data from the Dutch universal well-baby neonatal hearing screening programme by PCHC between 2013 and 2023. Multilevel logistic regression analyses were performed to estimate the probability of a referral in 2023 for newborns screened in 2022 and 2023. We included a total of 1,650,506 newborns for 2013–2022 and 323,194 newborns for 2022–2023. The lowest false positive referral rates were found between days five and thirteen, ranging from 3.3 to 3.9%. ESIII significantly increased the probability of a referral compared to ESI/II (odds ratio = 1.84, 95% confidence interval = 1.65–2.06). In conclusion, the timing of neonatal hearing screening significantly impacts the false positive referral rate. Furthermore, the likelihood of a referral is significantly higher when using the ESIII compared to the ESI/II.
15 February 2026