Hydroxyalkyl Amination of Agarose Gels Improves Adsorption of Bisphenol A and Diclofenac from Water: Conceivable Prospects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Methods
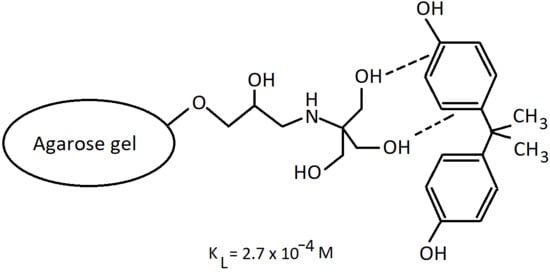
2.2.1. Chemical Modification of Agarose Gels
2.2.2. pH Measurements and Potentiometric Titration of TRIS-Agarose Gels
2.2.3. Bisphenol A Batch Adsorption Kinetics
2.2.4. Adsorption Isotherms of Bisphenol A
2.2.5. Column Adsorption of Bisphenol A and Diclofenac
2.2.6. Fourier-Transform Infrared Spectroscopy (FTIR) Spectrometry
2.2.7. Scanning Electron Microscopy
3. Results and Discussion
3.1. Characteristics of TRIS- and EA-Agarose Gels
3.2. Kinetics of Bisphenol A Adsorption and the Equilibrium Adsorbed Amounts
3.3. Removal of Bisphenol A and Diclofenac from Water by Filtration through TRIS-Zetarose 4FF Columns
3.4. Future Trends
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Suresh, S.; Srivastava, V.C.; Mishra, I.M. Adsorption of catechol, resorcinol, hydroquinone, and their derivatives: A review. Int. J. Energy Environ. Eng. 2012, 3, 32. [Google Scholar] [CrossRef]
- Cordova Villegas, L.G.; Mashhadi, N.; Chen, M.; Mukherjee, D.; Taylor, K.E.; Biswas, N. A short review of techniques for phenol removal from wastewater. Curr. Pollut. Rep. 2016, 2, 157–167. [Google Scholar] [CrossRef]
- Djebbar, M.; Djafri, F.; Bouchekara, M.; Djafri, A. Adsorption of phenol on natural clay. Appl. Water Sci. 2012, 2, 77–86. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Y.; Li, S.; Yu, S. Sorption and removal of tetrabromobisphenol A from solution by graphene oxide. Chem. Eng. J. 2013, 222, 94–100. [Google Scholar] [CrossRef]
- Javadi, E.; Baghdadi, M.; Taghavi, L.; Ahmad Panahi, H. Removal of 4-nonylphenol from surface water and municipal wastewater effluent using three-dimensional graphene oxide–chitosan aerogel beads. Int. J. Environ. Res. 2020, 14, 513–526. [Google Scholar] [CrossRef]
- Ersan, G.; Apul, O.G.; Perreault, F.; Karanfil, T. Adsorption of organic contaminants by graphene nanosheets: A review. Water Res. 2017, 126, 385–398. [Google Scholar] [CrossRef]
- Cusumano, J.A.; Low, M.J.D. Interaction between surface hydroxyl groups and adsorbed molecules: III. The nature of the adsorbate-hydroxyl interaction. J. Catal. 1971, 23, 214–227. [Google Scholar] [CrossRef]
- Pozharskii, A.F.; Dyablo, O.V.; Pogosova, O.G.; Ozeryanskii, V.A.; Filarowski, A.; Vasilikhina, K.M.; Dzhangiryan, N.A. Modelling biologically important NH···π Interactions using peri-disubstituted naphthalenes. J. Org. Chem. 2020, 85, 12468–12481. [Google Scholar] [CrossRef]
- Gu, M.; Su, Z.-G.; Janson, J.-C. The separation of polyphenols by isocratic hydrogen bond adsorption chromatography on a cross-linked 12% agarose gel. Chromatographia 2006, 64, 247–253. [Google Scholar] [CrossRef]
- Liu, D.; Ma, Y.; Wang, Y.; Su, Z.; Gu, M.; Janson, J.-C. One-step separation and purification of hydrolysable tannins from Geranium wilfordii maxim by adsorption chromatography on cross-linked 12% agarose gel. J. Sep. Sci. 2011, 34, 995–998. [Google Scholar] [CrossRef]
- Nakamura, S.; Tsuji, Y.; Yoshizawa, K. Role of hydrogen-bonding and OH−π Interactions in the adhesion of epoxy resin on hydrophilic surfaces. ACS Omega 2020, 5, 26211–26219. [Google Scholar] [CrossRef]
- Porath, J. Superhydroxylated Adsorbents and Use Thereof. PCT/SE2013/051410, WO 2014/084790 A1. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014084790 (accessed on 22 January 2024).
- Zhang, B.; Wang, Y.; Gao, M.; Gu, M.; Wang, C. Tris(hydroxymethyl)aminomethane-functionalized agarose particles: Parameters affecting the binding of bovine serum albumin. J. Sep. Sci. 2012, 35, 1406–1410. [Google Scholar] [CrossRef] [PubMed]
- Bui, N.T.H.; Jiang, W.; Sparrman, T.; Irgum, K. Synthesis of poly(N-[tris(hydroxymethyl)methyl] acrylamide) functionalized porous silica for application in hydrophilic interaction chromatography. J. Sep. Sci. 2012, 35, 3257–3269. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.E.; Halthur, T.; Ljunggren, L. Flow permeable composites of lignin and poly (vinyl alcohol): Towards removal of bisphenol A and erythromycin from water. J. Environ. Chem. Eng. 2016, 4, 1432–1441. [Google Scholar] [CrossRef]
- Abe, M.; Sugimura, K.; Nishio, Y. Rapid allylation of cellulose without heating in tetra-n-butylphosphonium hydroxide aqueous solution. Cellulose 2020, 27, 6887–6896. [Google Scholar] [CrossRef]
- Teramoto, N.; Motoyama, T.; Yosomiya, R.; Shibata, M. Synthesis and properties of thermoplastic propyl-etherified amylose. Eur. Polym. J. 2002, 38, 1365–1369. [Google Scholar] [CrossRef]
- Corrales, J.; Kristofco, L.A.; Steele, W.B.; Yates, B.S.; Breed, C.S.; Williams, E.S.; Brooks, B.W. Global assessment of bisphenol A in the environment: Review and analysis of its occurrence and bioaccumulation. Dose-Response 2015, 13, 1559325815598308. [Google Scholar] [CrossRef]
- Rivera-Tovar, P.R.; Pérez-Manríquez, J.; Mariotti-Celis, M.S.; Escalona, N.; Pérez-Correa, J.R. Adsorption of low molecular weight food relevant polyphenols on cross-linked agarose gel. J. Mol. Liq. 2022, 347, 117972. [Google Scholar] [CrossRef]
- Hacıosmanoğlua, G.G.; Doğruel, T.; Genç, S.; Oner, E.T.; Cana, Z.S. Adsorptive removal of bisphenol A from aqueous solutions using phosphonated levan. J. Hazard. Mater. 2019, 374, 43–49. [Google Scholar] [CrossRef]
- Dehghani, M.H.; Ghadermazi, M.; Bhatnagar, A.; Sadighara, P.; Jahed-Khaniki, G.; Heibati, B.; McKay, G. Adsorptive removal of endocrine disrupting bisphenol A from aqueous solution using chitosan. J. Environ. Chem. Eng. 2016, 4, 2647–2655. [Google Scholar] [CrossRef]
- Chen, P.; Yang, Y.; Bhattacharya, P.; Wang, P.; Ke, P.C. A Tris-dendrimer for hosting diverse chemical species. J. Phys. Chem. C 2011, 115, 12789–12796. [Google Scholar] [CrossRef]
- Godiya, C.B.; Kumar, S.; Xiao, Y. Amine functionalized egg albumin hydrogel with enhanced adsorption potential for diclofenac sodium in water. J. Hazard. Mater. 2020, 393, 122417. [Google Scholar] [CrossRef]
- Franco, P.; Cardea, S.; Tabernero, A.; De Marco, I. Porous aerogels and adsorption of pollutants from water and air: A review. Molecules 2021, 26, 4440. [Google Scholar] [CrossRef]
- Seida, Y.; Tokuyama, H. Hydrogel adsorbents for the removal of hazardous pollutants—Requirements and available functions as adsorbent. Gels 2022, 8, 220. [Google Scholar] [CrossRef]
- Ivanov, A.E.; Ljunggren, L. Thin poly (vinyl alcohol) cryogels: Reactive groups, macropores and translucency in microtiter plate assays. Heliyon 2019, 5, e02913. [Google Scholar] [CrossRef] [PubMed]
- Baimenov, A.Z.; Berillo, D.A.; Moustakas, K.; Inglezakis, V.J. Efficient removal of mercury (II) from water by use of cryogels and comparison to commercial adsorbents under environmentally relevant conditions. J. Hazard. Mater. 2020, 399, 123056. [Google Scholar] [CrossRef] [PubMed]
- Mikhalovsky, S.M.; Savina, I.N.; Dainiak, M.; Ivanov, A.E.; Galaev, I.Y. Biomaterials/Cryogels. In Comprehensive Biotechnology, 2nd ed.; Moo-Young, M., Butler, M., Webb, C., Moreira, A., Grodzinski, B., Cui, Z.F., Agathos, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; Volume 5, Chapter 5.03; pp. 11–22. [Google Scholar] [CrossRef]
- Lozinsky, V.I. A brief history of polymeric cryogels. Adv. Polym. Sci. 2014, 263, 1–48. [Google Scholar] [CrossRef]
- Busquets, R.; Ivanov, A.E.; Mbundi, L.; Hörberg, S.; Kozynchenko, O.P.; Cragg, P.J.; Savina, I.N.; Whitby, R.L.D.; Mikhalovsky, S.V.; Tennison, S.R.; et al. Carbon-cryogel hierarchical composites as effective and scalable filters for removal of trace organic pollutants from water. J. Environ. Manag. 2016, 182, 141–148. [Google Scholar] [CrossRef]
- Kumari, J.; Kumar, A. Development of polymer based cryogel matrix for transportation and storage of mammalian cells. Sci. Rep. 2017, 7, 41551. [Google Scholar] [CrossRef]
- Dragan, E.S.; Dinu, M.V. Advances in porous chitosan-based composite hydrogels: Synthesis and applications. React. Funct. Polym. 2020, 146, 104372. [Google Scholar] [CrossRef]
- Tuong, T.L.; Aymes, A.; Framboisier, X.; Ioannou, I.; Kapel, R. Adsorption of phenolic compounds from an aqueous by-product of sunflower protein extraction/purification by macroporous resins. J. Chromatogr. Sep. Tech. 2020, 11, 435. [Google Scholar] [CrossRef]
- Kodjapashis, M.P.; Zentelis, A.D.; Zagklis, D.P.; Sygouni, V.; Paraskeva, C.A. Resin adsorption of phenolic compounds from olive leaf and coffee residue extracts: Batch and packed column adsorption experimental investigation and mathematical modeling. Separations 2023, 10, 313. [Google Scholar] [CrossRef]
- Smith, C.J.; Perfetti, T.A.; Morton, M.J.; Rodgman, A.; Garg, R.; Selassie, C.D.; Hansch, C. The relative toxicity of substituted phenols reported in cigarette mainstream smoke. Toxicol. Sci. 2002, 69, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Sami, H.; Srivastava, A.; Ghatak, A. Cryotropic Hydrogels and Their Use as Filters. PCT/SE2010/050285, WO2010/107375 A1, EP2411113A1. Available online: https://patents.google.com/patent/EP2411113A1/en (accessed on 22 January 2024).








| Type of Agarose Gel | Bromohydrin, μmol/g Wet Gel; μmol/mL | TRIS or EA Content (*), μmol/g Wet Gel; μmol/mL | %N | TRIS or EA Content (**), μmol/g Wet Gel; μmol/mL |
|---|---|---|---|---|
| TRIS-Sepharose 4B | 220; 150 | 80; 55 | 0.28 | 18; 12 |
| TRIS-ZetaCell-CL6B | 290; 190 | 140; 92 | 0.48 | 31; 20 |
| TRIS-Zetarose FF4 | 290; 190 | 75; 49 | 1.03 | 46; 30 |
| EA-Zetarose FF4 | 290; 190 | 144; 94 | 1.55 | 67; 44 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ljunggren, L.; Ivanova, S.; Ivanov, A.E. Hydroxyalkyl Amination of Agarose Gels Improves Adsorption of Bisphenol A and Diclofenac from Water: Conceivable Prospects. AppliedChem 2024, 4, 42-55. https://doi.org/10.3390/appliedchem4010004
Ljunggren L, Ivanova S, Ivanov AE. Hydroxyalkyl Amination of Agarose Gels Improves Adsorption of Bisphenol A and Diclofenac from Water: Conceivable Prospects. AppliedChem. 2024; 4(1):42-55. https://doi.org/10.3390/appliedchem4010004
Chicago/Turabian StyleLjunggren, Lennart, Svetlana Ivanova, and Alexander E. Ivanov. 2024. "Hydroxyalkyl Amination of Agarose Gels Improves Adsorption of Bisphenol A and Diclofenac from Water: Conceivable Prospects" AppliedChem 4, no. 1: 42-55. https://doi.org/10.3390/appliedchem4010004