Point-of-Care Method T2Bacteria®Panel Enables a More Sensitive and Rapid Diagnosis of Bacterial Blood Stream Infections and a Shorter Time until Targeted Therapy than Blood Culture
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Study Design and Methods
- In-hospital mortality
- Need of mechanical ventilation, vasopressors or Continuous Renal Replacement Therapy
- Length of hospital stay
- Change of HRS at admission and day 7 of hospital stay
- Length of clinical recovery
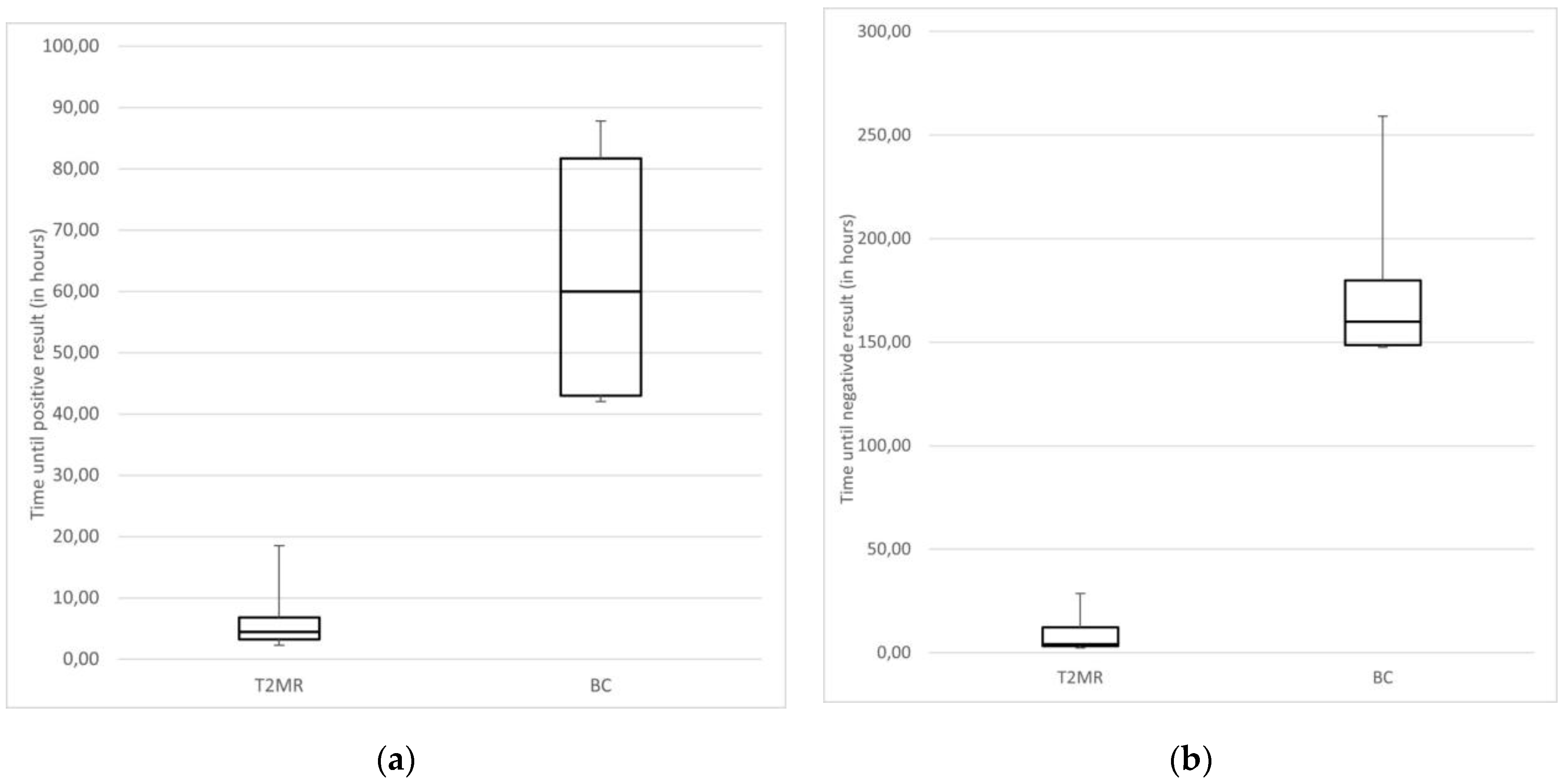
- Time until positive or negative result using BC and T2MR
- Frequency and time until targeted therapy
2.3. Statistical Analysis
Sample Size
2.4. Compliance with Ethical Standards
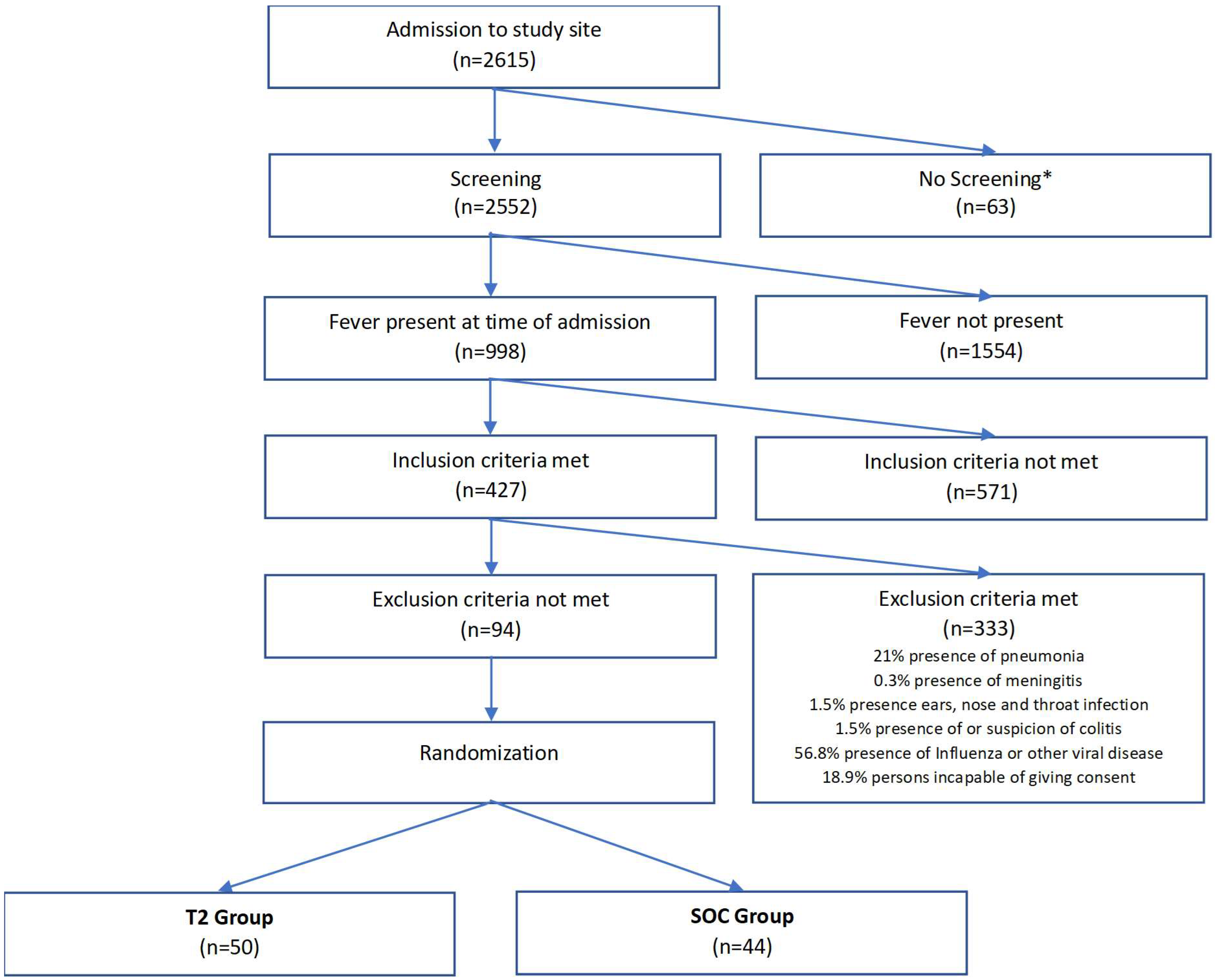
3. Results
3.1. Comparison T2 and BC Group
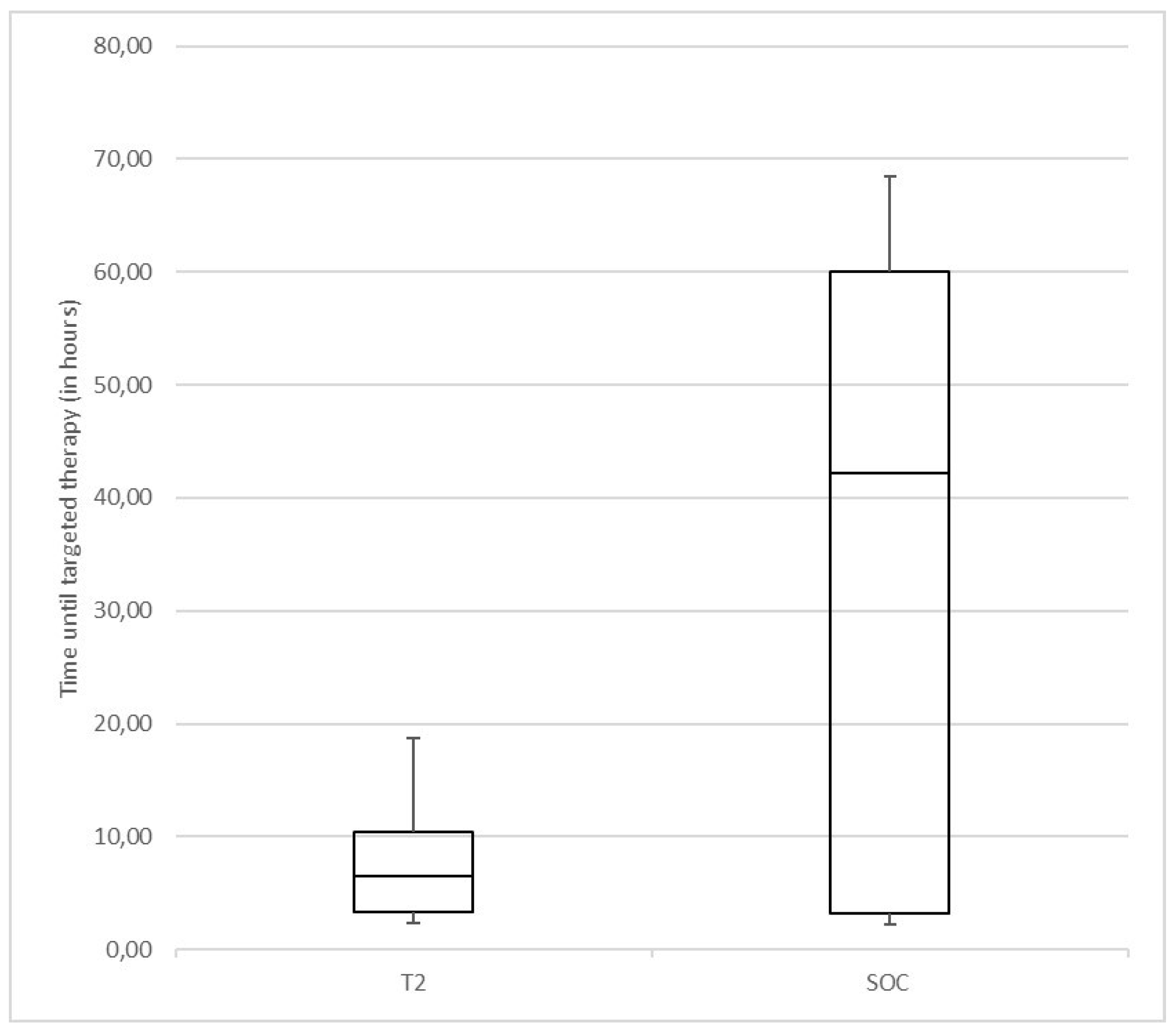
3.2. Antimicrobial Therapy
3.3. Clinical Outcome
3.4. Time to Result Comparison between T2Bacteria®Panel and BC
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Buehler, S.S.; Madison, B.; Snyder, S.R.; Derzon, J.H.; Cornish, N.E.; Saubolle, M.A.; Weissfeld, A.S.; Weinstein, M.P.; Liebow, E.B.; Wolk, D.M. Effectiveness of Practices To Increase Timeliness of Providing Targeted Therapy for Inpatients with Bloodstream Infections: A Laboratory Medicine Best Practices Systematic Review and Meta-analysis. Clin. Microbiol. Rev. 2016, 29, 59–103. [Google Scholar] [CrossRef]
- Cohen, J.; Vincent, J.L.; Adhikari, N.K.J.; Machado, F.R.; Angus, D.C.; Calandra, T.; Jaton, K.; Giulieri, S.; Delaloye, J.; Opal, S.; et al. Sepsis: A roadmap for future research. Lancet Infect. Dis. 2015, 15, 581–614. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Ellis, P.; Arabi, Y.; Roberts, D.; Light, B.; Parrillo, J.E.; Dodek, P.; Wood, G.; Kumar, A.; Simon, D.; et al. Initiation of inappropriate antimicrobial therapy results in a fivefold reduction of survival in human septic shock. Chest 2009, 136, 1237–1248. [Google Scholar] [CrossRef] [PubMed]
- Laupland, K.B.; Church, D.L. Population-Based Epidemiology and Microbiology of Community-Onset Bloodstream Infections. Clin. Microbiol. Rev. 2014, 27, 647–664. [Google Scholar] [CrossRef] [PubMed]
- Timsit, J.F.; Ruppé, E.; Barbier, F.; Tabah, A.; Bassetti, M. Bloodstream infections in critically ill patients: An expert statement. Intensive Care Med. 2020, 46, 266–284. [Google Scholar] [CrossRef] [PubMed]
- Faron, M.L.; Buchan, B.W.; Ledeboer, N.A. Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry for Use with Positive Blood Cultures: Methodology, Performance, and Optimization. J. Clin. Microbiol. 2017, 55, 3328–3338. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, R.; Teng, C.B.; Cunningham, S.A.; Ihde, S.M.; Steckelberg, J.M.; Moriarty, J.P.; Shah, N.D.; Mandrekar, J.N.; Patel, R. Randomized Trial of Rapid Multiplex Polymerase Chain Reaction–Based Blood Culture Identification and Susceptibility Testing. Clin. Infect. Dis. 2015, 61, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, S.A.; Gardner, C.; Das, S. Diagnosis and Management of Bloodstream Infections With Rapid, Multiplexed Molecular Assays. Front. Cell. Infect. Microbiol. 2022, 12, 859935. [Google Scholar] [CrossRef]
- Bach, K.; Edel, B.; Höring, S.; Bartoničkova, L.; Glöckner, S.; Löffler, B.; Bahrs, C.; Rödel, J. Performance of the eazyplex® BloodScreen GN as a simple and rapid molecular test for identification of Gram-negative bacteria from positive blood cultures. Eur. J. Clin. Microbiol. Infect. Dis. 2022, 41, 489–494. [Google Scholar] [CrossRef]
- Parize, P.; Muth, E.; Richaud, C.; Gratigny, M.; Pilmis, B.; Lamamy, A.; Mainardi, J.-L.; Cheval, J.; de Visser, L.; Jagorel, F.; et al. Untargeted next-generation sequencing-based first-line diagnosis of infection in immunocompromised adults: A multicentre, blinded, prospective study. Clin. Microbiol. Infect. 2017, 23, 574.e1–574.e6. [Google Scholar] [CrossRef]
- Blauwkamp, T.A.; Thair, S.; Rosen, M.J.; Blair, L.; Lindner, M.S.; Vilfan, I.D.; Kawli, T.; Christians, F.C.; Venkatasubrahmanyam, S.; Wall, G.D.; et al. Analytical and clinical validation of a microbial cell-free DNA sequencing test for infectious disease. Nat. Microbiol. 2019, 4, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.J.; Pappas, P.G.; Vazquez, J.; Judson, M.A.; Kontoyiannis, D.P.; Thompson, G.R., III.; Garey, K.W.; Reboli, A.; Greenberg, R.N.; Apewokin, S.; et al. Detecting Infections Rapidly and Easily for Candidemia Trial, Part 2 (DIRECT2): A Prospective, Multicenter Study of the T2Candida Panel. Clin. Infect. Dis. 2018, 66, 1678–1686. [Google Scholar] [CrossRef] [PubMed]
- Mylonakis, E.; Clancy, C.J.; Ostrosky-Zeichner, L.; Garey, K.W.; Alangaden, G.J.; Vazquez, J.A.; Groeger, J.S.; Judson, M.A.; Vinagre, Y.-M.; Heard, S.O.; et al. T2 magnetic resonance assay for the rapid diagnosis of candidemia in whole blood: A clinical trial. Clin. Infect. Dis. 2015, 60, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Seitz, T.; Holbik, J.; Hind, J.; Gibas, G.; Karolyi, M.; Pawelka, E.; Traugott, M.; Wenisch, C.; Zoufaly, A. Rapid Detection of Bacterial and Fungal Pathogens Using the T2MR versus Blood Culture in Patients with Severe COVID-19. Microbiol. Spectr. 2022, 10, e0014022. [Google Scholar] [CrossRef] [PubMed]
- Mylonakis, E.; Zacharioudakis, I.M.; Clancy, C.J.; Hong Nguyen, M.; Pappas, P.G. Efficacy of T2 Magnetic Resonance Assay in Monitoring Candidemia after Initiation of Antifungal Therapy: The Serial Therapeutic and Antifungal Monitoring Protocol (STAMP) Trial. J. Clin. Microbiol. 2018, 56, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, P.; Vena, A.; Machado, M.; Gioia, F.; Martínez-Jiménez, M.C.; Gómez, E.; Muñoz, P.; Martínez-Jiménez, M.C.; Gómez, E.; Origüen, J.; et al. T2Candida MR as a predictor of outcome in patients with suspected invasive candidiasis starting empirical antifungal treatment: A prospective pilot study. J. Antimicrob. Chemother. 2018, 73, iv6–iv12. [Google Scholar] [CrossRef]
- Bilir, S.P.; Ferrufino, C.P.; Pfaller, M.A.; Munakata, J. The economic impact of rapid Candida species identification by T2Candida among high-risk patients. Future Microbiol. 2015, 10, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Hong Nguyen, M.; Clancy, C.J.; William Pasculle, A.; Pappas, P.G.; Alangaden, G.; Pankey, G.A.; Schmitt, B.H.; Rasool, A.; Weinstein, M.P.; Widen, R.; et al. Performance of the T2Bacteria Panel for Diagnosing Bloodstream Infections: A Diagnostic Accuracy Study. Ann. Intern. Med. 2019, 170, 845–852. [Google Scholar] [CrossRef] [PubMed]
- AWMF Leitlinienregister. Kalkulierte parenterale Initialtherapie Bakterieller Erkrankungen bei Erwachsenen—Update 2018. Available online: https://register.awmf.org/de/leitlinien/detail/082-006 (accessed on 21 February 2024).
- Jordana-Lluch, E.; Rivaya, B.; Marcó, C.; Giménez, M.; Quesada, M.D.; Escobedo, A.; Batlle, M.; Martró, E.; Ausina, V. Molecular diagnosis of bloodstream infections in onco-haematology patients with PCR/ESI-MS technology. J. Infect. 2017, 74, 187–194. [Google Scholar] [CrossRef]
- De Angelis, G.; Posteraro, B.; De Carolis, E.; Menchinelli, G.; Franceschi, F.; Tumbarello, M.; De Pascale, G.; Spanu, T.; Sanguinetti, M. T2Bacteria magnetic resonance assay for the rapid detection of ESKAPEc pathogens directly in whole blood. J. Antimicrob. Chemother. 2018, 73, iv20–iv26. [Google Scholar] [CrossRef]
- Caliendo, A.M.; Gilbert, D.N.; Ginocchio, C.C.; Hanson, K.E.; May, L.; Quinn, T.C.; Tenover, F.C.; Alland, D.; Blaschke, A.J.; Bonomo, R.A.; et al. Better tests, better care: Improved diagnostics for infectious diseases. Clin. Infect. Dis. 2013, 57 (Suppl. 3), S139–S170. [Google Scholar] [CrossRef] [PubMed]
- Tafelski, S.; Nachtigall, I.; Adam, T.; Bereswill, S.; Faust, J.; Tamarkin, A.; Trefzer, T.; Deja, M.; Idelevich, E.A.; Wernecke, K.-D.; et al. Randomized controlled clinical trial evaluating multiplex polymerase chain reaction for pathogen identification and therapy adaptation in critical care patients with pulmonary or abdominal sepsis. J. Int. Med. Res. 2015, 43, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Gies, F.; Tschiedel, E.; Felderhoff-Müser, U.; Rath, P.M.; Steinmann, J.; Dohna-Schwake, C. Prospective evaluation of SeptiFast Multiplex PCR in children with systemic inflammatory response syndrome under antibiotic treatment. BMC Infect. Dis. 2016, 16, 378. [Google Scholar] [CrossRef]
- Box, M.J.; Sullivan, E.L.; Ortwine, K.N.; Parmenter, M.A.; Quigley, M.M.; Aguilar-Higgins, L.M.; MacIntosh, C.L.; Goerke, K.F.; Lim, R.A. Outcomes of rapid identification for gram-positive bacteremia in combination with antibiotic stewardship at a community-based hospital system. Pharmacotherapy 2015, 35, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, S.; Bender, J.M.; Sposto, R.; Gentry, M.; Takemoto, C.; Bard, J.D. Impact of a Rapid Blood Culture Assay for Gram-Positive Identification and Detection of Resistance Markers in a Pediatric Hospital. Arch. Pathol. Lab. Med. 2016, 140, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Eby, J.C.; Richey, M.M.; Platts-Mills, J.A.; Mathers, A.J.; Novicoff, W.M.; Cox, H.L. A Healthcare Improvement Intervention Combining Nucleic Acid Microarray Testing With Direct Physician Response for Management of Staphylococcus aureus Bacteremia. Clin. Infect. Dis. 2018, 66, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Weng, T.P.; Lo, C.L.; Lin, W.L.; Lee, J.C.; Li, M.C.; Ko, W.C.; Lee, N.Y. Integration of antimicrobial stewardship intervention with rapid organism identification improve outcomes in adult patients with bloodstream infections. J. Microbiol. Immunol. Infect. 2023, 56, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Cavalieri, S.J.; Kwon, S.; Vivekanandan, R.; Ased, S.; Carroll, C.; Anthone, J.; Schmidt, D.; Baysden, M.; Destache, C.J. Effect of antimicrobial stewardship with rapid MALDI-TOF identification and Vitek 2 antimicrobial susceptibility testing on hospitalization outcome. Diagn. Microbiol. Infect. Dis. 2019, 95, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Huang, Z.; Li, W.; Fang, X.; Zhang, W. Can metagenomic next-generation sequencing identify the pathogens responsible for culture-negative prosthetic joint infection? BMC Infect. Dis. 2020, 20, 253. [Google Scholar] [CrossRef]
- Voigt, C.; Silbert, S.; Widen, R.H.; Marturano, J.E.; Lowery, T.J.; Ashcraft, D.; Pankey, G. The T2Bacteria Assay Is a Sensitive and Rapid Detector of Bacteremia That Can Be Initiated in the Emergency Department and Has Potential to Favorably Influence Subsequent Therapy. J. Emerg. Med. 2020, 58, 785–796. [Google Scholar] [CrossRef]
- T2 Biosystems 101 Hartwell Avenue Lexington M 02421 P +1-781-457-1200 F +1-781-357-3080. T2Resistance Panel T2 Biosystems. 2021. Available online: https://www.t2biosystems.com/products-technology/pipeline/t2resistance-panel/ (accessed on 1 April 2024).
- Nguyen, M.H.; Clancy, C.C.; Pasculle, W.; Pappas, P.; Alangaden, G.; Pankey, G.A.; Schmitt, B.H.; Rasool, A.; Weinstein, M.B.; Widen, R.; et al. In-depth analysis of T2Bacteria positive results in patients with concurrent negative blood culture: A case series. BMC Infect. Dis. 2020, 20, 326. [Google Scholar] [CrossRef] [PubMed]
| T2 (n = 50) | SOC (n = 44) | p-Value | |
|---|---|---|---|
| Sex | p = 0.295 | ||
| female (%) | 23 (46) | 25 (56.8) | |
| male (%) | 27 (54) | 19 (43.2) | |
| Median age in years (min–max) | 69.00 (19–95) | 71.50 (30–94) | p = 0.178 |
| MedianCharlson Comorbidity Index (min–max) | 4 (0–10) | 5 (0–11) | p = 0.404 |
| Suspected infection at admission | |||
| Urine tract infection (%) | 62 (31) | 59.1 (26) | |
| Skin infection (%) | 20 (10) | 27.3 (12) | |
| Signs of abscesses (%) | 0 (0) | 0 (0) | |
| Clinical suspected spondylodiscitis (%) | 2 (1) | 6.8 (3) | |
| Recent abuse of IV-Drugs (%) | 4 (2) | 2.3 (1) | |
| Endocarditis (%) | 2 (1) | 2.3 (1) | |
| Presence of intravascular devices (%) | 2 (1) | 0 (0) | |
| Presence of peritoneal dialysis (%) | 0 (0) | 0 (0) | |
| Colonization with ESKAPE spp. (%) | 4 (2) | 4.5 (2) | |
| Intraabdominal infection (%) | 8 (4) | 9.1 (4) |
| T2 (n = 50) | SOC (n = 44) | p-Value | |
|---|---|---|---|
| Temperature measured in ear (in °C) | |||
| Mean (+/−SD) | 38.7 (0.55) | 38.6 (0.57) | p = 0.315 |
| Duration of fever (in days) | |||
| Mean (+/−SD) | 2.3 (1.56) | 2.4 (3.01) | p = 0.851 |
| HRS (in %) | |||
| 2 points | 72 (36) | 70.5 (31) | |
| 3 points | 16 (8) | 25 (11) | p = 0.340 |
| 4 points | 12 (6) | 4.5 (2) | |
| qSOFA (in %) | |||
| 0 points | 56 (28) | 45.5 (20) | |
| 1 points | 30 (15) | 38.6 (17) | p = 0.583 |
| 2 points | 14 (7) | 15.9 (7) | |
| CRP (mg/l) | |||
| Mean (+/−SD) | 165.1 (116.7) | 139.4 (101.1) | p = 0.260 |
| WBC (G/L) | |||
| Mean (+/−SD) | 13.6 (5.7) | 13.8 (5.9) | p = 0.814 |
| Species | T2Bacteria®Panel (n) | BC + | BC − | Surrogate + | Surrogate − |
|---|---|---|---|---|---|
| E. coli | POS (7) | 3 | 4 | 6 | 0 * |
| NEG (43) | 0 | 43 | 0 | 38 | |
| S. aureus | POS (4) | 3 | 1 | 1 | 0 ** |
| NEG (46) | 0 | 46 | 0 | 40 | |
| K. pneumoniae | POS (2) | 0 | 2 | 1 | 1 |
| NEG (48) | 0 | 48 | 0 | 42 | |
| A. baumannii | POS (2) | 0 | 2 | 0 | 1 * |
| NEG (48) | 0 | 48 | 0 | 42 | |
| P. aeruginosa | POS (2) | 1 | 1 | 1 | 0 * |
| NEG (48) | 0 | 48 | 0 | 42 | |
| E. faecium | POS (0) | 0 | 0 | 0 | 0 |
| NEG (50) | 0 | 50 | 0 | 44 | |
| Fusobacterium necrophorum | POS (0) | 0 | 0 | 0 | 0 |
| NEG (50) | 1 | 49 | 0 | 44 | |
| S. epidermidis | POS (0) | 0 | 0 | 0 | 0 |
| NEG (50) | 1 | 49 | 0 | 44 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clodi-Seitz, T.; Baumgartner, S.; Turner, M.; Mader, T.; Hind, J.; Wenisch, C.; Zoufaly, A.; Presterl, E. Point-of-Care Method T2Bacteria®Panel Enables a More Sensitive and Rapid Diagnosis of Bacterial Blood Stream Infections and a Shorter Time until Targeted Therapy than Blood Culture. Microorganisms 2024, 12, 967. https://doi.org/10.3390/microorganisms12050967
Clodi-Seitz T, Baumgartner S, Turner M, Mader T, Hind J, Wenisch C, Zoufaly A, Presterl E. Point-of-Care Method T2Bacteria®Panel Enables a More Sensitive and Rapid Diagnosis of Bacterial Blood Stream Infections and a Shorter Time until Targeted Therapy than Blood Culture. Microorganisms. 2024; 12(5):967. https://doi.org/10.3390/microorganisms12050967
Chicago/Turabian StyleClodi-Seitz, Tamara, Sebastian Baumgartner, Michael Turner, Theresa Mader, Julian Hind, Christoph Wenisch, Alexander Zoufaly, and Elisabeth Presterl. 2024. "Point-of-Care Method T2Bacteria®Panel Enables a More Sensitive and Rapid Diagnosis of Bacterial Blood Stream Infections and a Shorter Time until Targeted Therapy than Blood Culture" Microorganisms 12, no. 5: 967. https://doi.org/10.3390/microorganisms12050967
APA StyleClodi-Seitz, T., Baumgartner, S., Turner, M., Mader, T., Hind, J., Wenisch, C., Zoufaly, A., & Presterl, E. (2024). Point-of-Care Method T2Bacteria®Panel Enables a More Sensitive and Rapid Diagnosis of Bacterial Blood Stream Infections and a Shorter Time until Targeted Therapy than Blood Culture. Microorganisms, 12(5), 967. https://doi.org/10.3390/microorganisms12050967






