Improving the Utilization of Flammulina velutipes Waste during Biochar-Amended Composting: Emphasis on Bacterial Communities
Abstract
1. Introduction
2. Materials and Methods
2.1. Compost Treatment, Raw Materials and Sample Collection
2.2. Determination Items and Methods
2.3. DNA Extraction and High-Throughput Sequencing
2.4. Bioinformatic and Statistical Analysis
3. Results
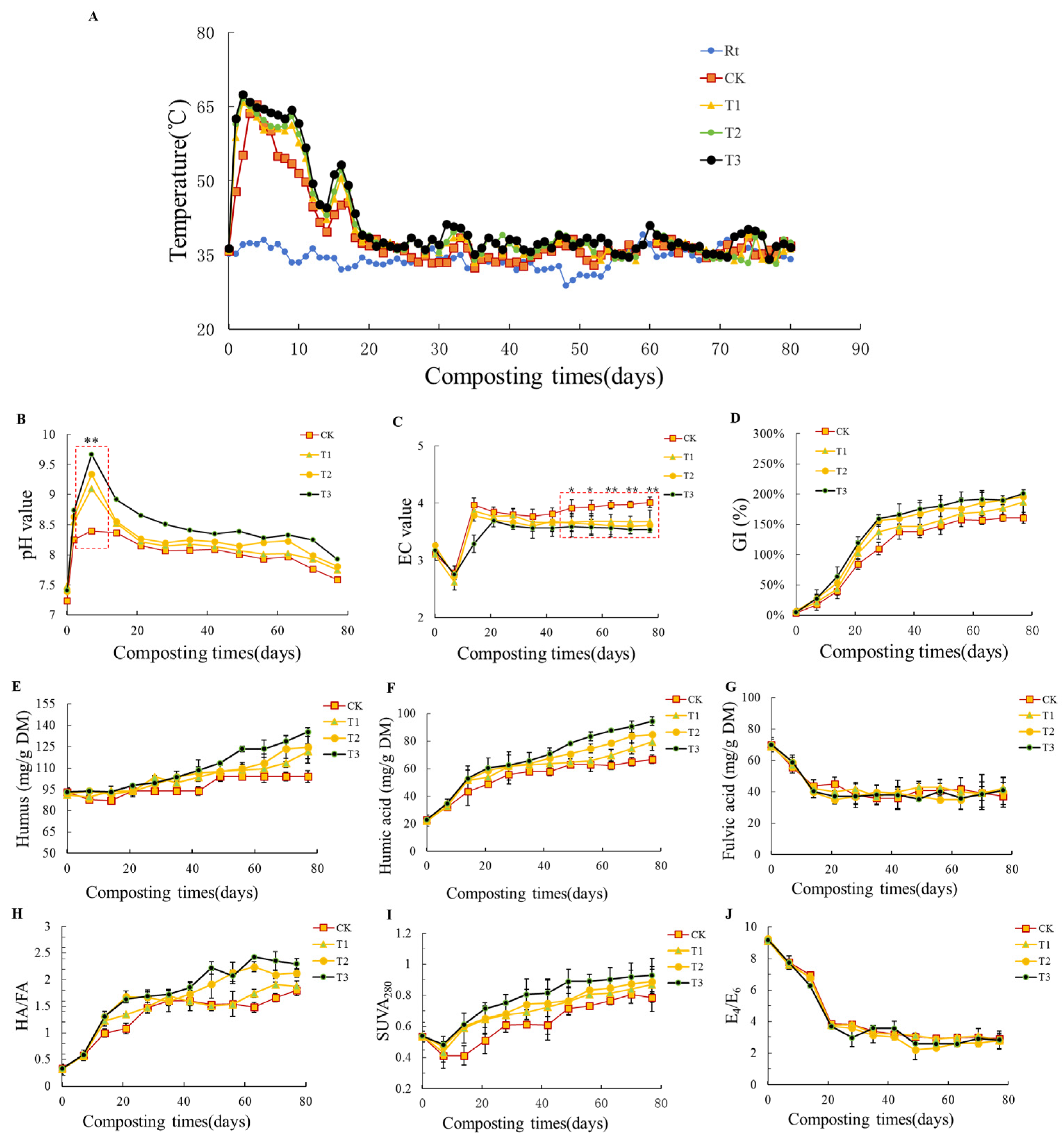
3.1. Effects of Biochar on Physiochemical Properties and Humification during Composting
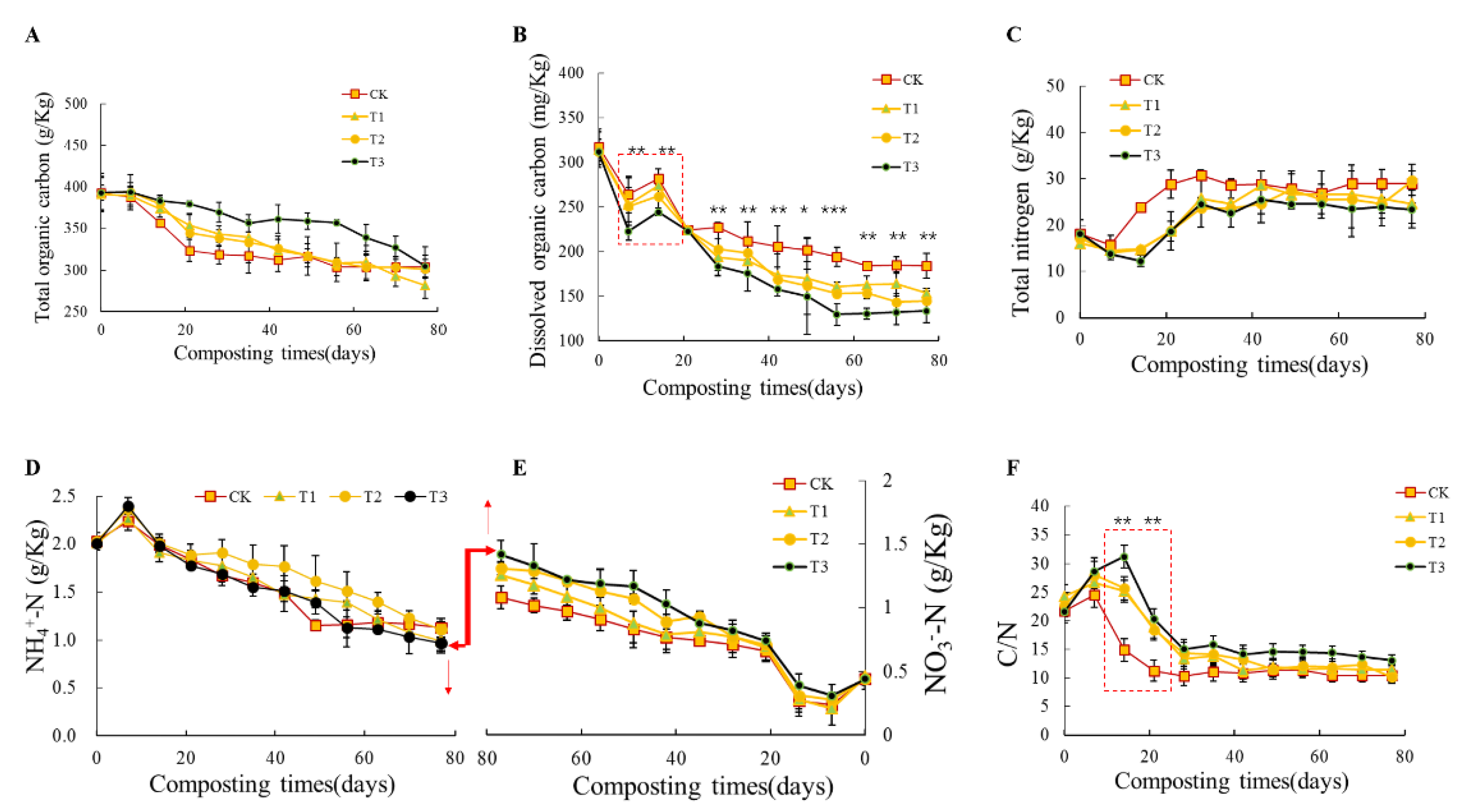
3.2. Effects of Biochar Addition on C and N Conversion
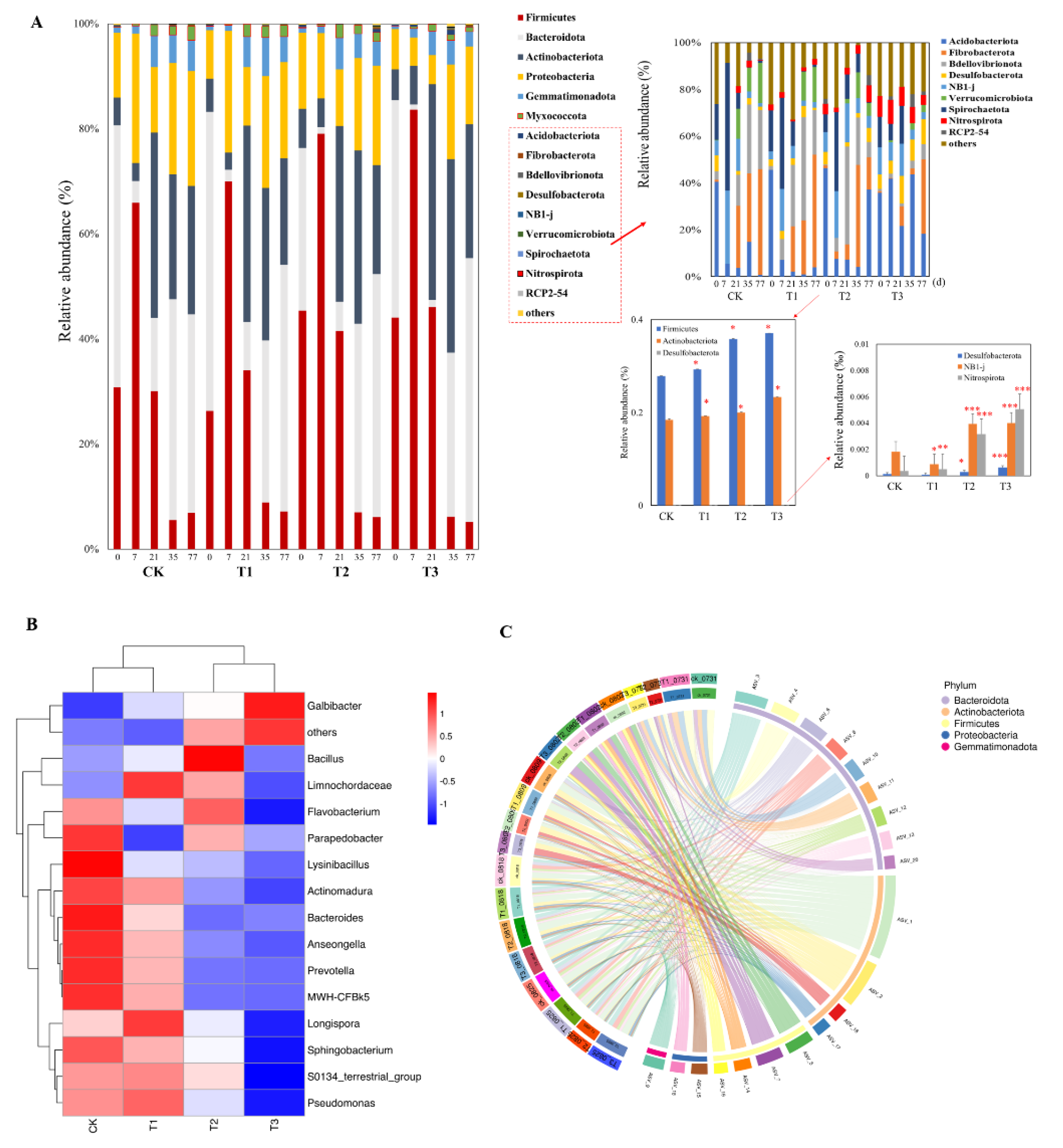
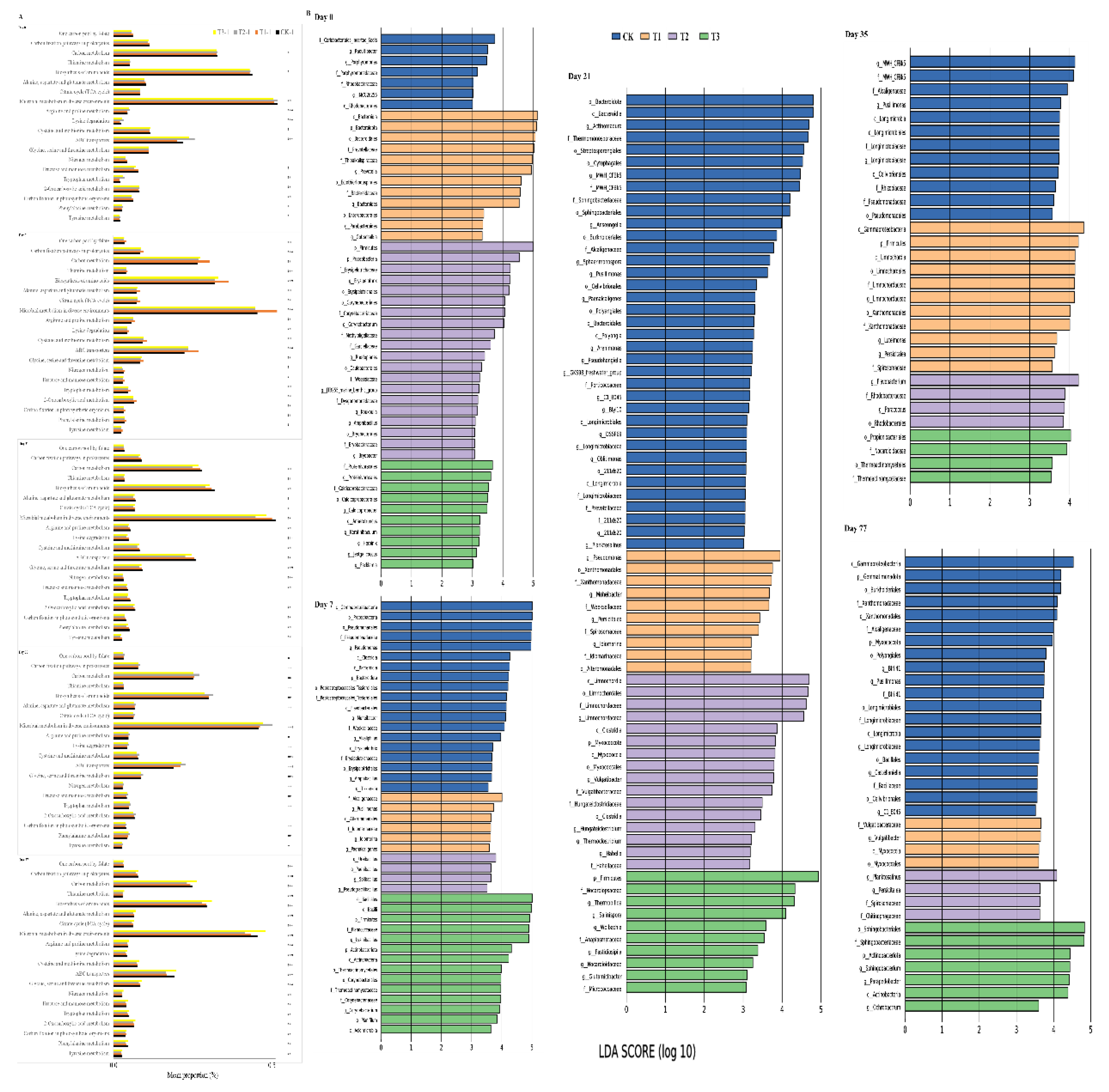
3.3. Effects of Biochar on Bacterial Community Diversity and Composition
3.4. Relationship between Bacterial Community and Composting Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chou, W.T.; Sheih, I.C.; Fang, T.J. The applications of polysaccharides from various mushroom wastes as prebiotics in different systems. J. Food Sci. 2013, 78, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Ye, J.; Xue, C.; Wang, Y.; Liao, W.; Mao, L.; Yuan, M.; Lian, S. Structural characteristics and bioactive properties of a novel polysaccharide from Flammulina velutipes. Carbohydr. Polym. 2018, 197, 9. [Google Scholar] [CrossRef]
- Islam, M.S.; Kasim, S.; Alam, K.M.; Amin, A.M.; Geok Hun, T.; Haque, M.A. Changes in chemical properties of banana pseudostem, mushroom media waste, and chicken manure through the co-composting process. Sustainability 2021, 13, 8458. [Google Scholar] [CrossRef]
- Atallah, E.; Zeaiter, J.; Ahmad, M.N.; Leahy, J.J.; Kwapinski, W. Hydrothermal carbonization of spent mushroom compost waste compared against torrefaction and pyrolysis. Fuel Process. Technol. 2021, 216, 106795. [Google Scholar] [CrossRef]
- Al Qadr Imad Wan-Mohtar, W.A.; Mahmud, N.; Supramani, S.; Ahmad, R.; Zain, N.A.M.; Hassan, N.A.M.; Peryasamy, J.; Halim-Lim, S.A. Fruiting-body-base flour from an oyster mushroom—A waste source of antioxidative flour for developing potential functional cookies and steamed-bun. AIMS Agric. Food 2018, 3, 481–492. [Google Scholar] [CrossRef]
- Rashad, F.M.; Kattan, M.H.E.; Fathy, H.M.; El-Fattah, D.A.A.; Tohamy, M.E.; Farahat, A.A. Recycling of agro-wastes for ganoderma lucidum mushroom production and ganoderma post mushroom substrate as soil amendment. Waste Manag. 2019, 88, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, B.; Mnkeni, P.N.S. Bio-optimization of the carbon-to-nitrogen ratio for efficient vermicomposting of chicken manure and waste paper using eisenia fetida. Environ. Sci. Pollut. Res. Int. 2016, 23, 16965–16976. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ma, J.; Yong, X.; Luo, L.; Wong, J.W.C.; Zhang, Y.; Wu, H.; Zhou, J. Effect of biochar combined with a biotrickling filter on deodorization, nitrogen retention, and microbial community succession during chicken manure composting. Bioresour. Technol. 2022, 343, 126137. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, M.W., Jr.; Liang, P.; Jiang, X.; Doyle, M.P.; Erickson, M.C. Microbiological analysis of composts produced on south carolina poultry farms. J. Appl. Microbiol. 2010, 108, 2067–2076. [Google Scholar] [CrossRef]
- Sharma, S.; Basu, S.; Shetti, N.P.; Kamali, M.; Walvekar, P.; Aminabhavi, T.M. Waste-to-energy nexus: A sustainable development. Environ. Pollut. 2020, 267, 27. [Google Scholar] [CrossRef]
- Li, J.; Chen, Q.; Li, H.; Li, S.; Liu, Y.; Yang, L.; Han, X. Impacts of different sources of animal manures on dissemination of human pathogenic bacteria in agricultural soils. Environ. Pollut. 2020, 266, 13. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Mhatre, A.; Pandit, R.; Lali, A.M. Synergistic biorefinery of Scenedesmus obliquus and Ulva lactuca in poultry manure towards sustainable bioproduct generation. Bioresour. Technol. 2020, 297, 122462. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Hussain, I.; Ghaffar, A.; Rahman, M.H.U.; Saleem, M.Z.; Yonas, M.W.; Hussnain, H.; Ikram, R.M.; Arslan, M. Organic amendments and conservation tillage improve cotton productivity and soil health indices under arid climate. Sci. Rep. 2022, 12, 18157. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yan, J.; Zhang, X.; Zhang, S.; Chen, Y. Organic manure input improves soil water and nutrients use for sustainable maize (Zea mays. L) productivity on the loess plateau. PLoS ONE 2020, 15, e0238042. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, S.S.; Sharma, V.; Shukla, A.K.; Verma, V.; Kaur, M.; Singh, P.; Gaber, A.; Hossain, A. Effect of addition of organic manures on basmati yield, nutrient content and soil fertility status in north-western India. Heliyon 2023, 9, e14514. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, X.; Yuan, M.; Wu, G.; Sun, Y. Effects of partial replacement of nitrogen fertilizer with organic fertilizer on rice growth, nitrogen utilization efficiency and soil properties in the Yangtze River basin. Life 2023, 13, 624. [Google Scholar] [CrossRef] [PubMed]
- Bonanomi, G.; Alioto, D.; Minutolo, M.; Marra, R.; Cesarano, G.; Vinale, F. Organic amendments modulate soil microbiota and reduce virus disease incidence in the tswv-tomato pathosystem. Pathogens 2020, 9, 379. [Google Scholar] [CrossRef] [PubMed]
- Ajibade, S.; Nnadozie, E.C.; Iwai, C.B.; Ghotekar, S.; Chang, S.W.; Ravindran, B.; Kumar Awasthi, M. Biochar-based compost: A bibliometric and visualization analysis. Bioengineered 2022, 13, 15013–15032. [Google Scholar] [CrossRef]
- Chung, W.J.; Chang, S.W.; Chaudhary, D.K.; Shin, J.; Kim, H.; Karmegam, N.; Govarthanan, M.; Chandrasekaran, M.; Ravindran, B. Effect of biochar amendment on compost quality, gaseous emissions and pathogen reduction during in-vessel composting of chicken manure. Chemosphere 2021, 283, 131129. [Google Scholar] [CrossRef]
- Ravindran, B.; Mupambwa, H.A.; Silwana, S.; Mnkeni, P.N.S. Assessment of nutrient quality, heavy metals and phytotoxic properties of chicken manure on selected commercial vegetable crops. Heliyon 2017, 3, e00493. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, B.; Nguyen, D.D.; Chaudhary, D.K.; Chang, S.W.; Kim, J.; Lee, S.R.; Shin, J.; Jeon, B.H.; Chung, S.; Lee, J. Influence of biochar on physico-chemical and microbial community during swine manure composting process. J. Environ. Manag. 2019, 232, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Chia, W.Y.; Chew, K.W.; Le, C.F.; Lam, S.S.; Chee, C.S.C.; Ooi, M.S.L.; Show, P.L. Sustainable utilization of biowaste compost for renewable energy and soil amendments. Environ. Pollut. 2020, 267, 16. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Tan, X.; Lu, G.; Liu, Y.; Naidu, R.; He, W. Soil properties influence kinetics of soil acid phosphatase in response to arsenic toxicity. Ecotoxicol. Environ. Saf. 2018, 147, 266–274. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, X.; Zhang, T.; He, W.; Song, F. Effects of the long-term application of atrazine on soil enzyme activity and bacterial community structure in farmlands in China. Environ. Pollut. 2020, 262, 24. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, I.; Dhal, P.K. Impact of elevated phosphogypsum on soil fertility and its aerobic biotransformation through indigenous microorganisms (imo’s) based technology. J. Environ. Manag. 2021, 297, 16. [Google Scholar] [CrossRef] [PubMed]
- López-Cano, I.; Roig, A.; Cayuela, M.L.; Alburquerque, J.A.; Sánchez-Monedero, M.A. Biochar improves n cycling during composting of olive mill wastes and sheep manure. Waste Manag. 2016, 49, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, G.; Sun, H.; Zhou, S.; Zou, G. Straw biochar hastens organic matter degradation and produces nutrient-rich compost. Bioresour. Technol. 2016, 200, 876–883. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Xu, X.; Qiu, X.; Zhang, J. Biochar influences the succession of microbial communities and the metabolic functions during rice straw composting with pig manure. Bioresour. Technol. 2019, 272, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Kafle, G.K.; Chen, L. Comparison on batch anaerobic digestion of five different livestock manures and prediction of biochemical methane potential (bmp) using different statistical models. Waste Manag. 2016, 48, 492–502. [Google Scholar] [CrossRef]
- Darby, B.; Goodale, C.; Chin, N.; Fuss, C.; Lang, A.; Ollinger, S.; Lovett, G. Depth patterns and connections between gross nitrogen cycling and soil exoenzyme activities in three northern hardwood forests. Soil. Biol. Biochem. 2020, 147, 107836. [Google Scholar] [CrossRef]
- Gao, X.; Xu, Z.; Li, Y.; Zhang, L.; Li, G.; Nghiem, L.D.; Luo, W. Bacterial dynamics for gaseous emission and humification in bio-augmented composting of kitchen waste. Sci. Total Environ. 2021, 801, 149640. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhao, Y.; Zhao, W.; Yang, T.; Zhang, X.; Xie, X.; Cui, H.; Wei, Z. Effect of precursors combined with bacteria communities on the formation of humic substances during different materials composting. Bioresour. Technol. 2017, 226, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Zhang, X.; Yu, C.; Shi, P.; Zhou, Y.; Li, Y.; Yang, P.; Shen, Z. Response of soil respiration to grazing in an alpine meadow at three elevations in Tibet. Sci. World J. 2014, 9, 265142. [Google Scholar] [CrossRef] [PubMed]
- Jindo, K.; Sánchez-Monedero, M.A.; Hernández, T.; García, C.; Furukawa, T.; Matsumoto, K.; Sonoki, T.; Bastida, F. Biochar influences the microbial community structure during manure composting with agricultural wastes. Sci. Total Environ. 2012, 416, 476–481. [Google Scholar] [CrossRef]
- Zhang, J.; Lü, F.; Shao, L.; He, P. The use of biochar-amended composting to improve the humification and degradation of sewage sludge. Bioresour. Technol. 2014, 168, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wei, Y.; Fan, Y.; Zhang, F.; Tan, W.; He, X.; Xi, B. Roles of bacterial community in the transformation of dissolved organic matter for the stability and safety of material during sludge composting. Bioresour. Technol. 2018, 267, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Jindo, K.; Sonoki, T.; Matsumoto, K.; Canellas, L.; Roig, A.; Sanchez-Monedero, M.A. Influence of biochar addition on the humic substances of composting manures. Waste Manag. 2016, 49, 545–552. [Google Scholar] [CrossRef] [PubMed]
- Duan, M.; Zhang, Y.; Zhou, B.; Qin, Z.; Wu, J.; Wang, Q.; Yin, Y. Effects of bacillus subtilis on carbon components and microbial functional metabolism during cow manure–straw composting. Bioresour. Technol. 2020, 303, 122868. [Google Scholar] [CrossRef]
- Guo, H.; Gu, J.; Wang, X.; Nasir, M.; Yu, J.; Lei, L.; Wang, J.; Zhao, W.; Dai, X. Beneficial effects of bacterial agent/bentonite on nitrogen transformation and microbial community dynamics during aerobic composting of pig manure. Bioresour. Technol. 2020, 298, 122384. [Google Scholar] [CrossRef]
- Qiu, X.; Zhou, G.; Zhang, J.; Wang, W. Microbial community responses to biochar addition when a green waste and manure mix are composted: A molecular ecological network analysis. Bioresour. Technol. 2019, 273, 666–671. [Google Scholar] [CrossRef]
- Li, F.; Yu, H.; Li, Y.; Wang, Y.; Shen Resource, J.; Hu, D.; Feng, B.; Han, Y. The quality of compost was improved by low concentrations of fulvic acid owing to its optimization of the exceptional microbial structure. Bioresour. Technol. 2021, 342, 25. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Kong, F.; Lu, L.; Wang, P.; Jiang, Z. Biochar—An effective additive for improving quality and reducing ecological risk of compost: A global meta-analysis. Sci. Total Environ. 2022, 806, 151439. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.Y.; Zhang, J.; Zhong, X.Z.; Tan, L.; Tang, Y.Q.; Kida, K. Production of nitrate-rich compost from the solid fraction of dairy manure by a labscale composting system. Waste Manag. 2016, 51, 55–64. [Google Scholar] [CrossRef]
- Yang, Y.; Awasthi, M.K.; Bao, H.; Bie, J.; Lei, S.; Lv, J. Exploring the microbial mechanisms of organic matter transformation during pig manure composting amended with bean dregs and biochar. Bioresour. Technol. 2020, 313, 6. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, M.K.; Wang, Q.; Chen, H.; Wang, M.; Ren, X.; Zhao, J.; Li, J.; Guo, D.; Li, D.S.; Awasthi, S.K.; et al. Evaluation of biochar amended biosolids co-composting to improve the nutrient transformation and its correlation as a function for the production of nutrient-rich compost. Bioresour. Technol. 2017, 237, 156–166. [Google Scholar] [CrossRef]
- Kalam, S.; Basu, A.; Ahmad, I.; Sayyed, R.Z.; El-Enshasy, H.A.; Dailin, D.J.; Suriani, N.L. Recent understanding of soil acidobacteria and their ecological significance: A critical review. Front. Microbiol. 2020, 30, 580024. [Google Scholar] [CrossRef] [PubMed]
- Metzner, R.; Nomura, T.; Kitaoka, N.; Ando, A.; Ogawa, J.; Kato, Y. Cobalt-dependent inhibition of nitrite oxidation in Nitrobacter winogradskyi. J. Biosci. Bioeng. 2019, 128, 463–467. [Google Scholar] [CrossRef]
- Taha, M.; Foda, M.; Shahsavari, E.; Aburto-Medina, A.; Adetutu, E.; Ball, A. Commercial feasibility of lignocellulose biodegradation: Possibilities and challenges. Curr. Opin. Biotechnol. 2016, 38, 190–197. [Google Scholar] [CrossRef]
- Chen, L.; Fang, K.; Zhou, J.; Yang, Z.P.; Dong, X.F.; Dai, G.H.; Zhang, H.B. Enrichment of soil rare bacteria in root by an invasive plant Ageratina adenophora. Sci. Total Environ. 2019, 683, 202–209. [Google Scholar] [CrossRef]
- Wang, B.; Wang, Y.; Wei, Y.; Chen, W.; Ding, G.; Zhan, Y.; Liu, Y.; Xu, T.; Xiao, J.; Li, J. Impact of inoculation and turning for full-scale composting on core bacterial community and their co-occurrence compared by network analysis. Bioresour. Technol. 2022, 345, 126417. [Google Scholar] [CrossRef]
- Ding, S.; Zhou, D.; Wei, H.; Wu, S.; Xie, B. Alleviating soil degradation caused by watermelon continuous cropping obstacle: Application of urban waste compost. Chemosphere 2021, 262, 128387. [Google Scholar] [CrossRef] [PubMed]
- Overholt, W.A.; Trumbore, S.; Xu, X.; Bornemann, T.L.; Probst, A.J.; Krueger, M.; Herrmann, M.; Thamdrup, B.; Bristow, L.A.; Taubert, M. Carbon fixation rates in groundwater similar to those in oligotrophic marine systems. Nat. Geosci. 2022, 15, 18. [Google Scholar] [CrossRef]
- Wu, J.; Zhao, Y.; Qi, H.; Zhao, X.; Yang, T.; Du, Y.; Zhang, H.; Wei, Z. Identifying the key factors that affect the formation of humic substance during different materials composting. Bioresour. Technol. 2017, 244, 1193–1196. [Google Scholar] [CrossRef] [PubMed]
- Hwang, O.; Lee, S.R.; Cho, S.; Ro, K.S.; Spiehs, M.; Woodbury, B.; Silva, P.J.; Han, D.W.; Choi, H.; Kim, K.Y. Efficacy of different biochars in removing odorous volatile organic compounds (vocs) emitted from swine manure. ACS Sustain. Chem. Eng. 2018, 6, 14239–14247. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, B.; Creamer, A.E.; Cao, C.; Li, Y. Adsorption of vocs onto engineered carbon materials: A review. J. Hazard. Mater. 2017, 338, 102–123. [Google Scholar] [CrossRef] [PubMed]
- Qiu, M.; Liu, L.; Ling, Q.; Cai, Y.; Yu, S.; Wang, S.; Fu, D.; Hu, B.; Wang, X. Biochar for the removal of contaminants from soil and water: A review. Biochar 2022, 4, 19. [Google Scholar] [CrossRef]
- Czekała, W.; Malińska, K.; Cáceres, R.; Janczak, D.; Dach, J.; Lewicki, A. Co-composting of poultry manure mixtures amended with biochar—The effect of biochar on temperature and c-co2 emission. Bioresour. Technol. 2016, 200, 921–927. [Google Scholar] [CrossRef]

| Treatment | Time (d) | Chao1 | Shannon | Observed Species | Simpson | PD Whole Tree | ACE |
|---|---|---|---|---|---|---|---|
| CK | 0 | 789 | 6.66 | 783 | 0.97 | 71 | 787 |
| 7 | 466 | 6.45 | 462 | 0.97 | 39 | 465 | |
| 21 | 533 | 6.43 | 526 | 0.96 | 46 | 529 | |
| 35 | 493 | 6.56 | 487 | 0.97 | 44 | 494 | |
| 77 | 527 | 6.65 | 521 | 0.97 | 47 | 527 | |
| T1 | 0 | 661 | 6.28 | 657 | 0.96 | 61 | 660 |
| 7 | 423 | 6.25 | 419 | 0.97 | 35 | 418 | |
| 21 | 467 | 6.40 | 463 | 0.96 | 41 | 468 | |
| 35 | 586 | 6.88 | 581 | 0.98 | 50 | 585 | |
| 77 | 464 | 6.60 | 460 | 0.97 | 41 | 464 | |
| T2 | 0 | 806 | 7.24 | 801 | 0.98 | 70 | 804 |
| 7 | 498 | 6.41 | 492 | 0.97 | 41 | 496 | |
| 21 | 480 | 6.47 | 474 | 0.97 | 42 | 474 | |
| 35 | 441 | 6.45 | 437 | 0.97 | 39 | 437 | |
| 77 | 698 | 6.99 | 694 | 0.98 | 59 | 696 | |
| T3 | 0 | 565 | 6.44 | 562 | 0.97 | 55 | 563 |
| 7 | 658 | 6.30 | 654 | 0.95 | 55 | 656 | |
| 21 | 473 | 6.18 | 468 | 0.96 | 42 | 470 | |
| 35 | 752 | 6.76 | 745 | 0.97 | 61 | 747 | |
| 77 | 583 | 6.31 | 578 | 0.97 | 50 | 579 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, L.; Lin, Y.; Liu, C.; Zhang, H.; Lin, C. Improving the Utilization of Flammulina velutipes Waste during Biochar-Amended Composting: Emphasis on Bacterial Communities. Agronomy 2024, 14, 1046. https://doi.org/10.3390/agronomy14051046
Chen L, Lin Y, Liu C, Zhang H, Lin C. Improving the Utilization of Flammulina velutipes Waste during Biochar-Amended Composting: Emphasis on Bacterial Communities. Agronomy. 2024; 14(5):1046. https://doi.org/10.3390/agronomy14051046
Chicago/Turabian StyleChen, Longjun, Yu Lin, Cenwei Liu, Hui Zhang, and Chenqiang Lin. 2024. "Improving the Utilization of Flammulina velutipes Waste during Biochar-Amended Composting: Emphasis on Bacterial Communities" Agronomy 14, no. 5: 1046. https://doi.org/10.3390/agronomy14051046
APA StyleChen, L., Lin, Y., Liu, C., Zhang, H., & Lin, C. (2024). Improving the Utilization of Flammulina velutipes Waste during Biochar-Amended Composting: Emphasis on Bacterial Communities. Agronomy, 14(5), 1046. https://doi.org/10.3390/agronomy14051046





