A Critical Review of the Study of Neuroprotective Diets to Reduce Cognitive Decline
Abstract
:1. Introduction
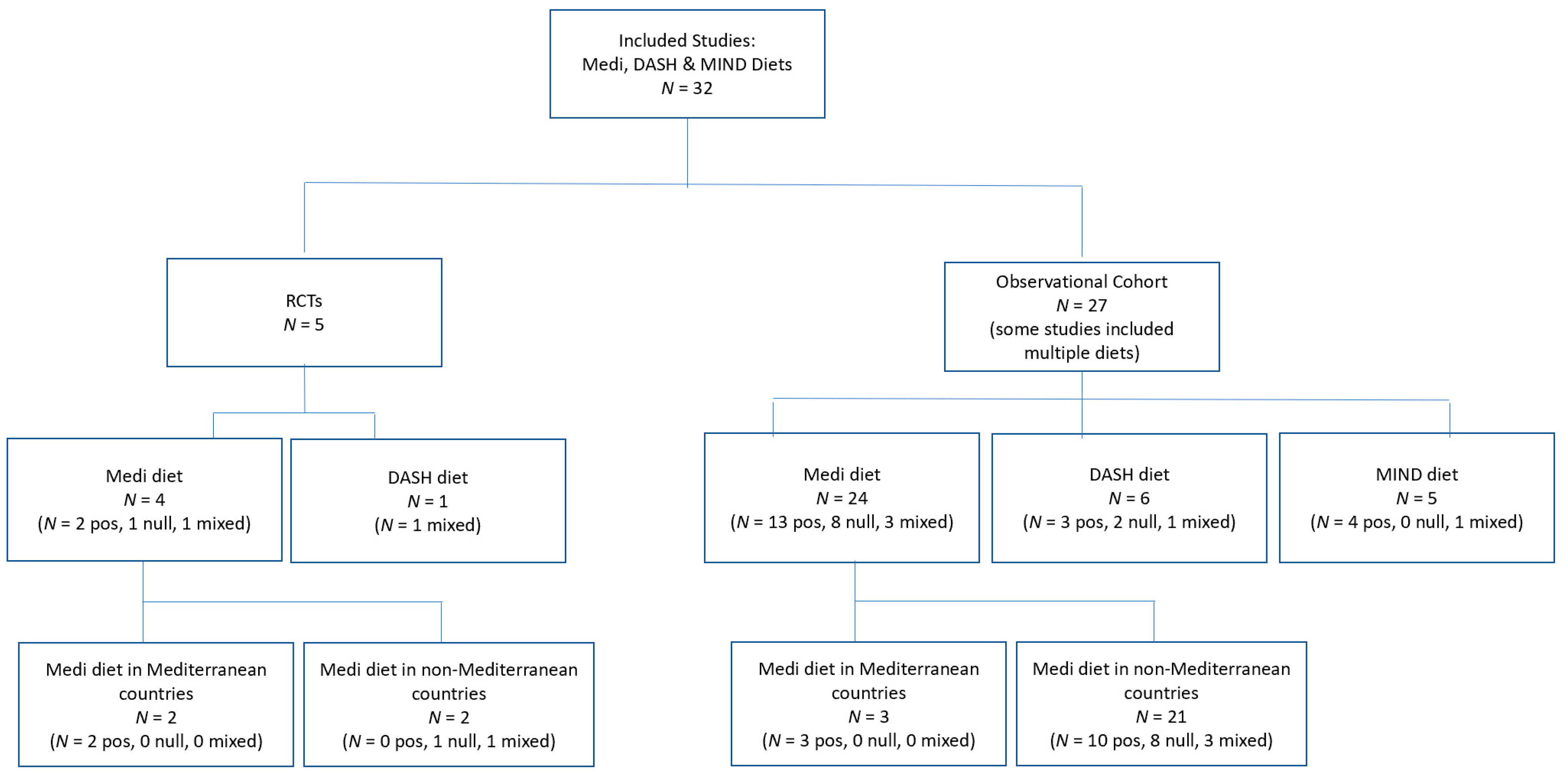
2. Materials and Methods
3. Gaps and Challenges in Current Research
3.1. Challenges in Measuring Dietary Impact on Cognition
3.1.1. Dietary Intake Measures
3.1.2. Dietary Pattern Metrics
3.1.3. Cognitive Assessment Measures
3.2. Gaps Common to Clinical Trials and Observational Studies
3.2.1. Length of Follow-Up
3.2.2. Attrition
3.2.3. Statistical Power and Sample Size
4. Beyond Study Limitations to Practical Recommendations
4.1. The Role of Pragmatic Trials in the Study of Neuroprotective Diets
4.2. New Standards in Explanatory RCTs for Neuroprotective Diets
4.2.1. Dietary Intake Measures
4.2.2. Dietary Pattern Metrics
4.2.3. Cognitive Assessment Measures
4.2.4. Length of Follow-Up
4.2.5. Adherence
4.2.6. Attrition
4.2.7. Statistical Power and Sample Size
4.2.8. Additional Recommendations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. The Top 10 Causes of Death. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 21 May 2021).
- Liu, X.; Morris, M.C.; Dhana, K.; Ventrelle, J.; Johnson, K.; Bishop, L.; Hollings, C.S.; Boulin, A.; Laranjo, N.; Stubbs, B.J.; et al. Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) study: Rationale, design and baseline characteristics of a randomized control trial of the MIND diet on cognitive decline. Contemp. Clin. Trials 2021, 102, 106270. [Google Scholar] [CrossRef]
- Alzheimer’s Association. Alzheimer’s Facts and Figures. 2021. Available online: https://www.alz.org/alzheimers-dementia/facts-figures (accessed on 21 May 2021).
- World Health Organization. Dementia: A Public Health Priority. Available online: https://www.who.int/mental_health/publications/dementia_report_2012/en/ (accessed on 9 June 2021).
- Cummings, J.L.; Tong, G.; Ballard, C. Treatment Combinations for Alzheimer’s Disease: Current and Future Pharmacotherapy Options. J. Alzheimer’s Dis. 2019, 67, 779–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lloret, A.; Esteve, D.; Lloret, M.; Cervera-Ferri, A.; Lopez, B.; Nepomuceno, M.; Monllor, P. When Does Alzheimer’s Disease Really Start? The Role of Biomarkers. Int. J. Mol. Sci. 2019, 20, 5536. [Google Scholar] [CrossRef] [Green Version]
- Changing the Trajectory of Alzheimer’s Disease Report. Available online: https://www.alz.org/media/documents/changing-the-trajectory-r.pdf (accessed on 9 June 2021).
- Wilson, R.S.; Segawa, E.; Boyle, P.A.; Anagnos, S.E.; Hizel, L.P.; Bennett, D.A. The Natural History of Cognitive Decline in Alzheimer’s Disease. Psychol. Aging 2012, 27, 1008–1017. [Google Scholar] [CrossRef] [Green Version]
- Hu, N.; Yu, J.; Tan, L.; Wang, Y.; Sun, L.; Tan, L. Nutrition and the Risk of Alzheimer’s Disease. BioMed Res. Int. 2013, 2013, 524820–524822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández-Sanz, P.; Ruiz-Gabarre, D.; García-Escudero, V. Modulating Effect of Diet on Alzheimer’s Disease. Diseases 2019, 7, 12. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Liu, Y.; Sun, M. Autophagy and Alzheimer’s Disease. Cell Mol. Neurobiol. 2017, 37, 377–388. [Google Scholar] [CrossRef]
- Ma, Q.; Galasko, D.R.; Ringman, J.M.; Vinters, H.V.; Edland, S.D.; Pomakian, J.; Ubeda, O.J.; Rosario, E.R.; Teter, B.; Frautschy, S.A.; et al. Reduction of SorLA/LR11, a Sorting Protein Limiting β-Amyloid Production, in Alzheimer Disease Cerebrospinal Fluid. Arch. Neurol. 2009, 66, 448–457. [Google Scholar] [CrossRef] [Green Version]
- Cardoso, C.; Afonso, C.; Bandarra, N.M. Dietary DHA and health: Cognitive function ageing. Nutr. Res. Rev. 2016, 29, 281–294. [Google Scholar] [CrossRef]
- Sydenham, E.; Dangour, A.D.; Lim, W.; Sydenham, E. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst. Rev. 2012, 2012, CD005379. [Google Scholar] [CrossRef] [Green Version]
- Walker, K.A.; Ficek, B.N.; Westbrook, R. Understanding the Role of Systemic Inflammation in Alzheimer’s Disease. ACS Chem. Neurosci. 2019, 10, 3340–3342. [Google Scholar] [CrossRef] [Green Version]
- Nunomura, A.; Castellani, R.J.; Zhu, X.; Moreira, P.I.; Perry, G.; Smith, M.A. Involvement of Oxidative Stress in Alzheimer Disease. J. Neuropathol. Exp. Neurol. 2006, 65, 631–641. [Google Scholar] [CrossRef] [Green Version]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND diet associated with reduced incidence of Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [Green Version]
- Wiȩckowska-Gacek, A.; Mietelska-Porowska, A.; Chutorański, D.; Wydrych, M.; Długosz, J.; Wojda, U. Western Diet Induces Impairment of Liver-Brain Axis Accelerating Neuroinflammation and Amyloid Pathology in Alzheimer’s Disease. Front. Aging Neurosci. 2021, 13, 654509. [Google Scholar] [CrossRef]
- Román, G.C.; Jackson, R.E.; Gadhia, R.; Román, A.N.; Reis, J. Mediterranean diet: The role of long-chain ω-3 fatty acids in fish; polyphenols in fruits, vegetables, cereals, coffee, tea, cacao and wine; probiotics and vitamins in prevention of stroke, age-related cognitive decline, and Alzheimer disease. Rev. Neurol. 2019, 175, 724–741. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.; Corella, D.; Lapetra, J.; Angel Munoz, M.; Martinez, J.A.; Saez, G.; Serra-Majem, L.; Pinto, X.; Mit Javala, M.T.; Tur, J.A.; et al. Cohort Profile: Design and methods of the PREDIMED study. Int. J. Epidemiol. 2012, 41, 377–385. [Google Scholar] [CrossRef] [Green Version]
- Chiavaroli, L.; Viguiliouk, E.; Nishi, S.K.; Blanco Mejia, S.; Rahelić, D.; Kahleová, H.; Salas-Salvadó, J.; Kendall, C.W.; Sievenpiper, J.L. DASH Dietary Pattern and Cardiometabolic Outcomes: An Umbrella Review of Systematic Reviews and Meta-Analyses. Nutrients 2019, 11, 338. [Google Scholar] [CrossRef] [Green Version]
- Kahleova, H.; Salas-Salvadó, J.; Rahelić, D.; Kendall, C.W.; Rembert, E.; Sievenpiper, J.L. Dietary Patterns and Cardiometabolic Outcomes in Diabetes: A Summary of Systematic Reviews and Meta-Analyses. Nutrients 2019, 11, 2209. [Google Scholar] [CrossRef] [Green Version]
- Ozemek, C.; Laddu, D.R.; Arena, R.; Lavie, C.J. The role of diet for prevention and management of hypertension. Curr. Opin. Cardiol. 2018, 33, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Anstey, K.J.; Kingston, A.; Kiely, K.M.; Luszcz, M.A.; Mitchell, P.; Jagger, C. The influence of smoking, sedentary lifestyle and obesity on cognitive impairment-free life expectancy. Int. J. Epidemiol. 2014, 43, 1874–1883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumgart, M.; Snyder, H.M.; Carrillo, M.C.; Fazio, S.; Kim, H.; Johns, H. Summary of the evidence on modifiable risk factors for cognitive decline and dementia: A population-based perspective. Alzheimer’s Dement. 2015, 11, 718–726. [Google Scholar] [CrossRef] [Green Version]
- Gottesman, R.F.; Schneider, A.L.C.; Zhou, Y.; Coresh, J.; Green, E.; Gupta, N.; Knopman, D.S.; Mintz, A.; Rahmim, A.; Sharrett, A.R.; et al. Association Between Midlife Vascular Risk Factors and Estimated Brain Amyloid Deposition. JAMA J. Am. Med. Assoc. 2017, 317, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Attems, J.; Jellinger, K.A. The overlap between vascular disease and Alzheimer’s disease—Lessons from pathology. BMC Med. 2014, 12, 206. [Google Scholar] [CrossRef] [Green Version]
- Livingston, G.; Huntley, J.; Sommerlad, A.; Ames, D.; Ballard, C.; Banerjee, S.; Brayne, C.; Burns, A.; Cohen-Mansfield, J.; Cooper, C.; et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 2020, 396, 413–446. [Google Scholar] [CrossRef]
- Caracciolo, B.; Xu, W.; Collins, S.; Fratiglioni, L. Cognitive decline, dietary factors and gut–brain interactions. Mech. Ageing Dev. 2014, 136–137, 59–69. [Google Scholar] [CrossRef]
- Solfrizzi, V.; Custodero, C.; Lozupone, M.; Imbimbo, B.P.; Valiani, V.; Agosti, P.; Schilardi, A.; D’Introno, A.; La Montagna, M.; Calvani, M.; et al. Relationships of dietary patterns, foods, and micro- and macronutrients with Alzheimer’s disease and late-life cognitive disorders: A systematic review. J. Alzheimer’s Dis. 2017, 59, 815–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jennings, A.; Cunnane, S.C.; Minihane, A.M. Can nutrition support healthy cognitive ageing and reduce dementia risk? BMJ 2020, 369, m2269. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Trichopoulou, A.; Kyrozis, A.; Kyrozis, A.; Rossi, M.; Rossi, M.; Katsoulis, M.; Katsoulis, M.; Trichopoulos, D.; Trichopoulos, D.; et al. Mediterranean diet and cognitive decline over time in an elderly Mediterranean population. Eur. J. Nutr. 2015, 54, 1311–1321. [Google Scholar] [CrossRef]
- Martínez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; Estruch, R.; Salas-Salvadó, J.; San Julián, B.; Sanchez-Tainta, A.; Ros, E.; Valls-Pedret, C.; Martinez-Gonzalez, M. Mediterranean diet improves cognition: The PREDIMED-NAVARRA randomised trial. J. Neurol. Neurosurg. Psychiatr. 2013, 84, 1318–1325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; de la Torre, R.; Martínez-González, M.Á.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Int. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef] [Green Version]
- Wengreen, H.; Munger, R.G.; Cutler, A.; Quach, A.; Bowles, A.; Corcoran, C.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K. Prospective study of Dietary Approaches to Stop Hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: The Cache County Study on Memory, Health and Aging. Am. J. Clin. Nutr. 2013, 98, 1263–1271. [Google Scholar] [CrossRef]
- Tangney, C.C. DASH and Mediterranean-Type Dietary Patterns to Maintain Cognitive Health. Curr. Nutr. Rep. 2014, 3, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Adair, L.S.; Plassman, B.L.; Batis, C.; Edwards, L.J.; Popkin, B.M.; Mendez, M.A. Dietary Patterns and Cognitive Decline Among Chinese Older Adults. Epidemiology 2015, 26, 758–768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berendsen, A.A.M.; Kang, J.H.; van de Rest, O.; Feskens, E.J.M.; de Groot, L.C.P.G.M.; Grodstein, F. The Dietary Approaches to Stop Hypertension Diet, Cognitive Function, and Cognitive Decline in American Older Women. J. Am. Med. Dir. Assoc. 2016, 18, 427–432. [Google Scholar] [CrossRef]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Barnes, L.L.; Bennett, D.W.; Aggarwal, N.T. MIND diet slows cognitive decline with aging. Alzheimer’s Dement. 2015, 11, 1015–1022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosking, D.E.; Eramudugolla, R.; Cherbuin, N.; Anstey, K.J. MIND not Mediterranean diet related to 12-year incidence of cognitive impairment in an Australian longitudinal cohort study. Alzheimer’s Dement. 2019, 15, 581–589. [Google Scholar] [CrossRef]
- Chen, X.; Maguire, B.; Brodaty, H.; O’Leary, F. Dietary Patterns and Cognitive Health in Older Adults: A Systematic Review. J. Alzheimer’s Dis. 2019, 67, 583–619. [Google Scholar] [CrossRef] [PubMed]
- Warnberg, J.; Gomez-Martinez, S.; Romeo, J.; Diaz, L.; Marcos, A. Nutrition, Inflammation, and Cognitive Function. Ann. N. Y. Acad. Sci. 2009, 1153, 164–175. [Google Scholar] [CrossRef]
- Ma, Y.; Hébert, J.R.; Li, W.; Bertone-Johnson, E.; Olendzki, B.; Pagoto, S.L.; Tinker, L.; Rosal, M.C.; Ockene, I.S.; Ockene, J.K.; et al. Association between dietary fiber and markers of systemic inflammation in the Women’s Heath Initiative Observational Study. Nutrition 2008, 24, 941–949. [Google Scholar] [CrossRef] [Green Version]
- Morrison, D.J.; Preston, T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 2016, 7, 189–200. [Google Scholar] [CrossRef] [Green Version]
- Eichelmann, F.; Schwingshackl, L.; Fedirko, V.; Aleksandrova, K. Effect of plant-based diets on obesity-related inflammatory profiles: A systematic review and meta-analysis of intervention trials. Obes. Rev. 2016, 17, 1067–1079. [Google Scholar] [CrossRef]
- Ghosh, D.; Scheepens, A. Vascular action of polyphenols. Mol. Nutr. Food Res. 2009, 53, 322–331. [Google Scholar] [CrossRef]
- Gupta, A.; Singh, A.K.; Kumar, R.; Jamieson, S.; Pandey, A.K.; Bishayee, A. Neuroprotective Potential of Ellagic Acid: A Critical Review. Adv. Nutr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Viguiliouk, E.; Glenn, A.J.; Nishi, S.K.; Chiavaroli, L.; Seider, M.; Khan, T.; Bonaccio, M.; Iacoviello, L.; Mejia, S.B.; Jenkins, D.J.A.; et al. Associations between Dietary Pulses Alone or with Other Legumes and Cardiometabolic Disease Outcomes: An Umbrella Review and Updated Systematic Review and Meta-analysis of Prospective Cohort Studies. Adv. Nutr. 2019, 10, S308–S319. [Google Scholar] [CrossRef] [PubMed]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive Oil and Red Wine Antioxidant Polyphenols Inhibit Endothelial Activation: Antiatherogenic Properties of Mediterranean Diet Phytochemicals. Arterioscl. Thromb. Vas. 2003, 23, 622–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romeu, M.; Rubió, L.; Sánchez-Martos, V.; Castañer, O.; de la Torre, R.; Valls, R.M.; Ras, R.; Pedret, A.; Catalán, U.; López de las Hazas, M.d.C.; et al. Virgin Olive Oil Enriched with Its Own Phenols or Complemented with Thyme Phenols Improves DNA Protection against Oxidation and Antioxidant Enzyme Activity in Hyperlipidemic Subjects. J. Agric. Food Chem. 2016, 64, 1879–1888. [Google Scholar] [CrossRef] [Green Version]
- Assunção, M.; Santos-Marques, M.; de Freitas, V.; Carvalho, F.; Andrade, J.P.; Lukoyanov, N.V.; Paula-Barbosa, M. Red wine antioxidants protect hippocampal neurons against ethanol-induced damage: A biochemical, morphological and behavioral study. Neuroscience 2007, 146, 1581–1592. [Google Scholar] [CrossRef] [PubMed]
- Micallef, M.; Lexis, L.; Lewandowski, P. Red wine consumption increases antioxidant status and decreases oxidative stress in the circulation of both young and old humans. Nutr. J. 2007, 6, 27. [Google Scholar] [CrossRef] [Green Version]
- Jardim, F.R.; de Rossi, F.T.; Nascimento, M.X.; da Silva Barros, R.G.; Borges, P.A.; Prescilio, I.C.; de Oliveira, M.R. Resveratrol and Brain Mitochondria: A Review. Mol. Neurobiol. 2018, 55, 2085–2101. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean Diet and Survival in a Greek Population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcella, E.R.; Johanna, T.D.; Nicola, M.M.; James, B.M.; Rogers, G.; Paul, F.J. The Development of the Mediterranean-Style Dietary Pattern Score and Its Application to the American Diet in the Framingham Offspring Cohort1-3. J. Nutr. 2009, 139, 1150–1156. [Google Scholar] [CrossRef]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-Style Diet and Risk of Coronary Heart Disease and Stroke in Women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef] [Green Version]
- Joyce, B.T.; Wu, D.; Hou, L.; Dai, Q.; Castaneda, S.F.; Gallo, L.C.; Talavera, G.A.; Sotres-Alvarez, D.; Van Horn, L.; Beasley, J.M.; et al. DASH diet and prevalent metabolic syndrome in the Hispanic Community Health Study/Study of Latinos. Prev. Med. Rep. 2019, 15, 100950. [Google Scholar] [CrossRef] [PubMed]
- Leshner, A. (Ed.) Preventing Cognitive Decline and Dementia: A Way Forward; National Academies Press: Washington, DC, USA, 2017. [Google Scholar]
- Van den Brink, A.C.; Brouwer-Brolsma, E.M.; Berendsen, A.A.M.; van de Rest, O. The Mediterranean, Dietary Approaches to Stop Hypertension (DASH), and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) Diets Are Associated with Less Cognitive Decline and a Lower Risk of Alzheimer’s Disease-A Review. Adv. Nutr. 2019, 10, 1040–1065. [Google Scholar] [CrossRef]
- Knight, A.; Bryan, J.; Wilson, C.; Hodgson, J.M.; Davis, C.R.; Murphy, K.J. The Mediterranean Diet and Cognitive Function among Healthy Older Adults in a 6-Month Randomised Controlled Trial: The MedLey Study. Nutrients 2016, 8, 579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wardle, J.; Rogers, P.; Judd, P.; Taylor, M.A.; Rapoport, L.; Green, M.; Nicholson Perry, K. Randomized trial of the effects of cholesterol-lowering dietary treatment on psychological function. Am. J. Med. 2000, 108, 547–553. [Google Scholar] [CrossRef]
- Smith, P.J.; Blumenthal, J.A.; Babyak, M.A.; Craighead, L.; Welsh-Bohmer, K.; Browndyke, J.N.; Strauman, T.A.; Sherwood, A. Effects of the Dietary Approaches to Stop Hypertension Diet, Exercise, and Caloric Restriction on Neurocognition in Overweight Adults with High Blood Pressure. Hypertension 2010, 55, 1331–1338. [Google Scholar] [CrossRef]
- Berendsen, A.; Kang, J.H.; Feskens, E.J.M.; de Groot, C.P.G.M.; Grodstein, F.; van de Rest, O. Association of long-term adherence to the mind diet with cognitive function and cognitive decline in American women. J. Nutr. Health Aging 2018, 22, 222–229. [Google Scholar] [CrossRef]
- Bhushan, A.; Bhushan, A.; Fondell, E.; Fondell, E.; Ascherio, A.; Ascherio, A.; Yuan, C.; Yuan, C.; Grodstein, F.; Grodstein, F.; et al. Adherence to Mediterranean diet and subjective cognitive function in men. Eur. J. Epidemiol. 2018, 33, 223–234. [Google Scholar] [CrossRef]
- Cherbuin, N.; Anstey, K.J. The Mediterranean Diet Is Not Related to Cognitive Change in a Large Prospective Investigation: The PATH Through Life Study. Am. J. Geriatr. Psychiatry 2012, 20, 635–639. [Google Scholar] [CrossRef]
- Galbete, C.; Toledo, E.; Toledo, J.B.; Bes-Rastrollo, M.; Buil-Cosiales, P.; Marti, A.; Guillén-Grima, F.; Martínez-González, M.A. Mediterranean diet and cognitive function: The SUN project. J. Nutr. Health Aging 2015, 19, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Gardener, S.; Gu, Y.; Rainey-Smith, S.; Keogh, J.B.; Clifton, P.M.; Mathieson, S.L.; Taddei, K.; Mondal, A.; Ward, V.K.; Scarmeas, N.; et al. Adherence to a Mediterranean diet and Alzheimer’s disease risk in an Australian population. Transl. Psychiatry 2012, 2, e164. [Google Scholar] [CrossRef] [Green Version]
- Gardener, S.L.; Rainey-Smith, S.; Barnes, M.B.; Sohrabi, H.R.; Weinborn, M.; Lim, Y.Y.; Harrington, K.; Taddei, K.; Gu, Y.; Rembach, A.; et al. Dietary patterns and cognitive decline in an Australian study of ageing. Mol. Psychiatry 2015, 20, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Luchsinger, J.A.; Stern, Y.; Scarmeas, N. Mediterranean Diet, Inflammatory and Metabolic Biomarkers, and Risk of Alzheimer’s Disease. J. Alzheimer’s Dis. 2010, 22, 483–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haring, B.; Wu, C.; Mossavar-Rahmani, Y.; Snetselaar, L.; Brunner, R.; Wallace, R.B.; Neuhouser, M.L.; Wassertheil-Smoller, S. No Association between Dietary Patterns and Risk for Cognitive Decline in Older Women with 9-Year Follow-Up: Data from the Women’s Health Initiative Memory Study. J. Acad. Nutr. Diet. 2016, 116, 921–930.e1. [Google Scholar] [CrossRef] [Green Version]
- Kesse-Guyot, E.; Andreeva, V.A.; Lassale, C.; Ferry, M.; Jeandel, C.; Hercberg, S.; Galan, P. Mediterranean diet and cognitive function: A French study. Am. J. Clin. Nutr. 2013, 97, 369–376. [Google Scholar] [CrossRef] [Green Version]
- Koyama, A.; Houston, D.K.; Simonsich, E.M.; Lee, J.S.; Ayonayon, H.N.; Shahar, D.R.; Rosano, C.; Satterfield, S.; Yaffe, K. Association between the mediterranean diet and cognitive decline in a biracial population. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 354–359. [Google Scholar] [CrossRef] [Green Version]
- Olsson, E.; Karlström, B.; Kilander, L.; Byberg, L.; Cederholm, T.; Sjögren, P. Dietary patterns and cognitive dysfunction in a 12-year follow-up study of 70 year old men. J. Alzheimer’s Dis. 2015, 43, 109–119. [Google Scholar] [CrossRef]
- Roberts, R.O.; Geda, Y.E.; Cerhan, J.R.; Knopman, D.S.; Cha, R.H.; Christianson, T.J.H.; Pankratz, V.S.; Ivnik, R.J.; Boeve, B.F.; O’Connor, H.M.; et al. Vegetables, Unsaturated Fats, Moderate Alcohol Intake, and Mild Cognitive Impairment. Dement. Geriatr. Cogn. Disord. 2010, 29, 413–423. [Google Scholar] [CrossRef] [Green Version]
- Samieri, C.; Okereke, O.I.; Devore, E.E.; Grodstein, F. Long-Term Adherence to the Mediterranean Diet Is Associated with Overall Cognitive Status, but Not Cognitive Decline, in Women. J. Nutr. 2013, 143, 493–499. [Google Scholar] [CrossRef] [Green Version]
- Shakersain, B.; Rizzuto, D.; Larsson, S.C.; Faxén-Irving, G.; Fratiglioni, L.; Xu, W. The Nordic Prudent Diet Reduces Risk of Cognitive Decline in the Swedish Older Adults: A Population-Based Cohort Study. Nutrients 2018, 10, 229. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, T.; Talegawkar, S.A.; Jin, Y.; Colpo, M.; Ferrucci, L.; Bandinelli, S. Adherence to a Mediterranean Diet Protects from Cognitive Decline in the Invecchiare in Chianti Study of Aging. Nutrients 2018, 10, 2007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tangney, C.C.; Kwasny, M.J.; Hong, L.I.; Wilson, R.S.; Evans, D.A.; Morris, M.C. Adherence to a Mediterranean-type dietary pattern and cognitive decline in a community population. Am. J. Clin. Nutr. 2011, 93, 601–607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tangney, C.C.; Li, H.; Wang, Y.; Barnes, L.; Schneider, J.A.; Bennett, D.A.; Morris, M.C. Relation of DASH- and Mediterranean-like dietary patterns to cognitive decline in older persons. Neurology 2014, 83, 1410–1416. [Google Scholar] [CrossRef] [Green Version]
- Titova, O.E.; Ax, E.; Brooks, S.J.; Sjögren, P.; Cederholm, T.; Kilander, L.; Kullberg, J.; Larsson, E.; Johansson, L.; Åhlström, H.; et al. Mediterranean diet habits in older individuals: Associations with cognitive functioning and brain volumes. Exp. Gerontol. 2013, 48, 1443–1448. [Google Scholar] [CrossRef] [Green Version]
- Tsivgoulis, G.; Judd, S.; Letter, A.J.; Alexandrov, A.V.; Howard, G.; Nahab, F.; Unverzagt, F.W.; Moy, C.; Howard, V.J.; Kissela, B.; et al. Adherence to a Mediterranean diet and risk of incident cognitive impairment. Neurology 2013, 80, 1684–1692. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vercambre, M.; Grodstein, F.; Berr, C.; Kang, J.H. Mediterranean Diet and Cognitive Decline in Women with Cardiovascular Disease or Risk Factors. J. Acad. Nutr. Diet. 2012, 112, 816–823. [Google Scholar] [CrossRef] [Green Version]
- Cade, J.E.; Warthon-Medina, M.; Albar, S.; Alwan, N.A.; Ness, A.; Roe, M.; Wark, P.A.; Greathead, K.; Burley, V.J.; Finglas, P.; et al. DIET@NET: Best Practice Guidelines for dietary assessment in health research. BMC Med. 2017, 15, 202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, P.; Morris, M.C.; Barnes, L.L. Racial Differences in Dietary Relations to Cognitive Decline and Alzheimer’s Disease Risk: Do We Know Enough? Front. Hum. Neurosci. 2020, 14, 359. [Google Scholar] [CrossRef]
- Tangney, C.; Sarkar, D.; Staffileno, B.A. Comparison of three DASH scoring paradigms and prevalent hypertension among older Hispanics. J. Hum. Hypertens 2016, 30, 210–215. [Google Scholar] [CrossRef]
- Carroll, R.J.; Midthune, D.; Subar, A.F.; Shumakovich, M.; Freedman, L.S.; Thompson, F.E.; Kipnis, V. Taking Advantage of the Strengths of 2 Different Dietary Assessment Instruments to Improve Intake Estimates for Nutritional Epidemiology. Am. J. Epidemiol. 2012, 175, 340–347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shim, J.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Health 2014, 36, e2014009. [Google Scholar] [CrossRef]
- Freedman, L.S.; Midthune, D.; Arab, L.; Prentice, R.L.; Subar, A.F.; Willett, W.; Neuhouser, M.L.; Tinker, L.F.; Kipnis, V. Combining a Food Frequency Questionnaire with 24-Hour Recalls to Increase the Precision of Estimation of Usual Dietary Intakes—Evidence From the Validation Studies Pooling Project. Am. J. Epidemiol. 2018, 187, 2227–2232. [Google Scholar] [CrossRef] [Green Version]
- Radd-Vagenas, S.; Kouris-Blazos, A.; Singh, M.F.; Flood, V.M. Evolution of Mediterranean diets and cuisine: Concepts and definitions. Asia Pac. J. Clin. Nutr. 2017, 26, 749–763. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Pitsavos, C.; Stefanadis, C. Dietary patterns: A Mediterranean diet score and its relation to clinical and biological markers of cardiovascular disease risk. Nutr. Metab. Cardiovasc. Dis. 2006, 16, 559–568. [Google Scholar] [CrossRef]
- Morris, M.C. Nutrition and risk of dementia: Overview and methodological issues: Nutrition and dementia. Ann. N. Y. Acad. Sci. 2016, 1367, 31–37. [Google Scholar] [CrossRef] [Green Version]
- Moore, L.V.; Thompson, F.E. Adults Meeting Fruit and Vegetable Intake Recommendations—United States, 2013. MMWR Morb. Mortal. Wkly. Rep. 2015, 64, 709–713. [Google Scholar] [PubMed]
- Folsom, A.R.; Parker, E.D.; Harnack, L.J. Degree of Concordance with DASH Diet Guidelines and Incidence of Hypertension and Fatal Cardiovascular Disease. Am. J. Hypertens 2007, 20, 225–232. [Google Scholar] [CrossRef] [Green Version]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS—ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 2011, 77, 333. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Ashford, J.W. Advances in screening instruments for Alzheimer’s disease. Aging Med. 2019, 2, 88–93. [Google Scholar] [CrossRef] [Green Version]
- De Roeck, E.E.; De Deyn, P.P.; Dierckx, E.; Engelborghs, S. Brief cognitive screening instruments for early detection of Alzheimer’s disease: A systematic review. Alzheimer’s Res. Ther. 2019, 11, 21. [Google Scholar] [CrossRef] [Green Version]
- Bennett, D.A.; Schneider, J.A.; Buchman, A.S.; Barnes, L.L.; Boyle, P.A.; Wilson, R.S. Overview and findings from the rush Memory and Aging Project. Curr. Alzheimer Res. 2012, 9, 646–663. [Google Scholar] [CrossRef]
- Zwarenstein, M.; Oxman, A. Why are so few randomized trials useful, and what can we do about it? J. Clin. Epidemiol. 2006, 59, 1125–1126. [Google Scholar] [CrossRef]
- Röhr, S.; Zülke, A.; Luppa, M.; Brettschneider, C.; Weißenborn, M.; Kühne, F.; Zöllinger, I.; Samos, F.Z.; Bauer, A.; Döhring, J.; et al. Recruitment and Baseline Characteristics of Participants in the AgeWell.de Study-A Pragmatic Cluster-Randomized Controlled Lifestyle Trial against Cognitive Decline. Int. J. Environ. Res. Public Health 2021, 18, 408. [Google Scholar] [CrossRef] [PubMed]
- Patsopoulos, N.A. A pragmatic view on pragmatic trials. Dialogues Clin. Neurosci. 2011, 13, 217–224. [Google Scholar]
- Kivipelto, M.; Mangialasche, F.; Snyder, H.M.; Allegri, R.; Anieu, S.; Arai, H.; Baker, L.; Belleville, S.; Brodaty, H.; Brucki, S.M.; et al. World-Wide FINGERS Network: A global approach to risk reduction and prevention of dementia. Alzheimer’s Dement. 2020, 16, 1078–1094. [Google Scholar] [CrossRef] [PubMed]
- Zülke, A.; Luck, T.; Pabst, A.; Hoffmann, W.; Thyrian, J.R.; Gensichen, J.; Kaduszkiewicz, H.; König, H.; Haefeli, W.E.; Czock, D.; et al. AgeWell.de—Study protocol of a pragmatic multi-center cluster-randomized controlled prevention trial against cognitive decline in older primary care patients. BMC Geriatr. 2019, 19, 203. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. Automated Self-Administered 24-Hour (ASA24®) Dietary Assessment Tool. Available online: https://epi.grants.cancer.gov/asa24/ (accessed on 1 May 2021).
- Karr, J.E.; Graham, R.B.; Hofer, S.M.; Muniz-Terrera, G. When Does Cognitive Decline Begin? A Systematic Review of Change Point Studies on Accelerated Decline in Cognitive and Neurological Outcomes Preceding Mild Cognitive Impairment, Dementia, and Death. Psychol. Aging 2018, 33, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, S.; Lee, F.; Burnett, D.J.; Kan, A.; Bonnel, E.L.; Allen, L.H.; Adams, S.H.; Keim, N.L. Challenges in Designing and Delivering Diets and Assessing Adherence: A Randomized Controlled Trial Evaluating the 2010 Dietary Guidelines for Americans. Curr. Dev. Nutr. 2020, 4, nzaa022. [Google Scholar] [CrossRef]
- Salthouse, T.A. Selectivity of Attrition in Longitudinal Studies of Cognitive Functioning. J. Gerontol. B Psychol. Sci. Soc. Sci. 2014, 69, 567–574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Beijsterveldt, C.E.M.; van Boxtel, M.P.J.; Bosma, H.; Houx, P.J.; Buntinx, F.; Jolles, J. Predictors of attrition in a longitudinal cognitive aging study: The Maastricht Aging Study (MAAS). J. Clin. Epidemiol. 2002, 55, 216–223. [Google Scholar] [CrossRef] [Green Version]
- King, A.C.; Winter, S.J.; Chrisinger, B.W.; Hua, J.; Banchoff, A.W. Maximizing the promise of citizen science to advance health and prevent disease. Prev. Med. 2019, 119, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Rhee, T.G.; Choi, Y.C.; Ouellet, G.M.; Ross, J.S. National Prescribing Trends for High-Risk Anticholinergic Medications in Older Adults. J. Am. Geriatr. Soc. 2018, 66, 1382–1387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, A.; Bhattacharya, G.; Balaram, K.; Tampi, D.; Rajesh, R.T. Benzodiazepine use among older adults. Neurodegener. Dis. Manag. 2020, 11, 5–8. [Google Scholar] [CrossRef] [PubMed]
| Dietary Component | Vascular Protection | Anti-Inflammatory Protection | Antioxidant Protection | Neural Protection |
|---|---|---|---|---|
| Whole Grains [2,13,29,41,42,43,44,45,46] | X | X | X | X |
| Vegetables (MIND diet emphasizes leafy greens in addition to other vegetables) [2,29,39,41,42,43,44,45,46,47] | X | X | X | |
| Fruits (MIND diet emphasizes berries over other fruits) [2,29,39,41,42,43,44,45,46,47] | X | X | X | |
| Fatty Fish (e.g., salmon, tuna, sardines) [2,13,14,31,41,42] | X | X | X | X |
| Legumes/Beans [2,41,45,48] | X | X | X | X |
| Nuts [2,29,41,46,47] | X | X | X | X |
| Olive Oil (Medi, MIND diets) [33,49,50] | X | X | ||
| Red Wine (Medi, MIND diets) [29,46,49,51,52,53] | X | X |
| Mediterranean [17,54,55] | DASH [56,57] | MIND [17] | |
|---|---|---|---|
| HIGH | Olive Oil (exclusive) | - | - |
| AMOUNTS | Fish | - | - |
| Grains (non-refined) | Grains | Grains (whole) | |
| Fruits | Fruits | Berries | |
| Vegetables | Vegetables | Green leafy vegetables | |
| - | - | Other vegetables | |
| Potatoes | - | - | |
| Legumes | Legumes | - | |
| Beans | - | Beans | |
| Nuts | Nuts | Nuts | |
| Seeds | Seeds | - | |
| - | Low-fat diary | - | |
| MODERATE | Poultry | Poultry | Poultry |
| AMOUNTS | - | Fish | Fish |
| Alcohol | - | Alcohol | |
| - | - | Olive oil (primary) | |
| Full-fat dairy | - | - | |
| SMALL | Red meat | Red meat | Red meat |
| AMOUNTS | Processed meat | - | - |
| Sweets | Sweets | Sweets/pastries | |
| - | Saturated fat as a | - | |
| % of total fat | |||
| - | Sodium <2400 mg/d | - | |
| RESTRICTED | - | - | Cheese |
| - | - | Butter/margarine | |
| - | - | Fast/fried foods |
| Type | Author, Study Name | Year #Participants Country Follow-Up | Dietary Pattern | Purpose | Findings | Outcome | Dietary Intake | Dietary Pattern Scoring | Cognitive Scoring |
|---|---|---|---|---|---|---|---|---|---|
| RCT | Martinez-Lapiscina et al. PREDIMED NAVARA [33] | 2013 N = 522 Spain 6.5 years | Medi | Compared the effects of two interventions of a Mediterranean diet vs. a low-fat diet on cognition or incidences of dementia. One intervention was supplemented with extra virgin olive oil (EVOO) and one supplemented with nuts. | Participants in both Medi Diet groups showed higher mean global cognitive function scores vs. the control group, with more significant differences in the Medi Diet + EVOO group. Medi Diet + EVOO group showed mean global cognitive function scores with significant differences versus the control group (p = 0.005 for MMSE, p = 0.001 for CDT). MedDiet + Nuts group versus the control group showed mean global cognitive differences vs. the control group (p = 0.015 for MMSE and p = 0.048 for CDT). | Positive correlation | FFQ | 14-item Medi diet questionnaire | MMSE + CDT |
| RCT | Valls-Pedret et al. PREDIMED-NAVARRA [34] | 2015 N = 447 Spain 4.1 years | Medi | Compared the effects of two interventions of a Mediterranean diet vs. a low-fat diet on cognition or incidences of dementia. One intervention was supplemented with extra virgin olive oil (EVOO) and one supplemented with nuts. | Both the Mediterranean diet plus olive oil and the Mediterranean diet plus nuts demonstrated improved composite measures of cognitive function, although the results varied based on the cognitive assessment. Similarly adjusted cognitive composites (mean z scores with 95% CIs) for changes above baseline of the memory composite were 0.04 (−0.10 to 0.17) for the Mediterranean diet plus olive oil; 0.10 (−0.04 to 0.24; p = 0.04 vs. controls) for the Mediterranean diet plus nuts; and −0.16 (−0.32 to −0.01) for the control diet. Respective changes from baseline of the frontal cognition composite were 0.23 (0.02 to 0.43; p = 0.004 vs. controls); 0.03 (−0.26 to 0.32); and −0.33 (−0.57 to −0.09). Changes from baseline of the global cognition composite were 0.04 (−0.12 to 0.20; p = 0.008 vs. controls) for the Mediterranean diet plus olive oil; −0.04 (−0.27 to 0.19) for the Mediterranean diet plus nuts; and −0.37 (−0.56 to −0.17) for the control diet. All cognitive composites significantly (p < 0.05) decreased from baseline in controls. | Positive correlation | FFQ | 14-item Medi diet questionnaire | Battery |
| RCT | Knight et al. MedLey Study [60] | 2016 N = 137 Australia 0.5 years | Medi | Compared an intervention arm (Medi Diet) vs. a control arm (habitual diet) on cognition and cognitive decline. | No effect of Medi diet vs. habitual diet on cognition. No difference in executive function (p = 0.33); speed of processing (p = 0.15); memory (p = 0.50); visual-spatial ability (p = 0.48); or overall age-related cognitive performance (p = 0.19). | Null | FFQ | An Australian version based on the food content of 15 Medi diets and the nutrient content of 8 Medi diets | Battery |
| RCT | Wardle et al. (No Study Name) [61] | 2000 N = 176 UK 0.2 years | Medi | Compared adults with elevated serum cholesterol levels assigned to one of two intervention groups (low-fat diet or Mediterranean diet) and a control group (wait-list control) to determine effects of diet on serum cholesterol and cognitive function. | There were no between-group differences on three measures of cognitive function, but for a fourth measure, which involved the task with the greatest processing load, the two intervention groups did significantly worse (p < 0.001) than the control group. The change in performance was correlated with the change in total serum cholesterol level (p = 0.01). | Mixed findings | Dietary diaries | Omitted, except for guidelines to increase vegetables, fruits, and oily fish; reduce dietary fat to 30% of energy; substitute predominantly mono-unsaturated fats for saturated fats | Battery |
| RCT | Smith et al. ENCORE [62] | 2010 N = 124 0.3 years US | DASH | Compared two intervention arms (DASH diet only; DASH diet + weight management) vs. a control arm to determine effects on executive-function-memory-learning (EFML). | DASH diet alone participants showed no improvement in EFML, but better psychomotor speed (p < 0.036) than the usual diet control. DASH diet combined with behavioral weight management program showed greater improvements in EFML (p < 0.008) and psychomotor speed (p < 0.023) than the control. | Mixed findings | Not stated (but measured change against the Healthy Eating Index) | Omitted | Battery |
| Obs | Berendsen Nurses Health Study [38] | 2017 N = 16,144 US Years = 6.0 | DASH | Cognition and cognitive decline | Greater adherence to long-term DASH score was associated with better average cognitive function (p = 0.009 for global cognition) but not associated with change in cognitive function over 6 years. | Mixed findings | FFQ | Med 0–9 | Battery |
| Obs | Berendsen Nurses Health Study [63] | 2017 N = 16,058 US 6.0 years | MIND | Cognition and cognitive decline | Greater adherence to MIND diet was positive for verbal memory score in extreme quintiles (p = 0.006), but not with cognitive decline over 6 years in global cognition, verbal memory, or TICS score. | Mixed findings | FFQ | MIND 0–15 | Battery |
| Obs | Bhushan Health Professionals’ Follow-Up Study [64] | 2017 N = 27,842 US 16.0 years | Medi | Cognition and cognitive decline | Long-term adherence to Medi diet was strongly related to lower subjective cognitive decline as measured by a 6-question Subjective Cognitive Function score (SCF). Compared with men having a Medi diet score in the lowest quintile, those in the highest quintile had 36% lower odds of a poor SCF score (p < 0.001) and 24% lower odds of a moderate SCF score (p < 0.001). | Positive correlation | FFQ | Med 0–9 | SCF |
| Obs | Cherbuin PATH Through Life study [65] | 2012 N = 1528 Australia 4.0 years | Medi | Cognitive decline, incidence of mild cognitive impairment (MCI) or dementia | There were no protective benefits of Medi diet. Instead, a high intake of mono-unsaturated fatty acids and excessive caloric intake were predictive of MCI (p < 0.01). | Null | FFQ | Med 0–9 | Clinical Dementia Rating battery |
| Obs | Galbete et al. SUN [66] | 2015 N = 823 Spain 2.0 years | Medi | Cognition and cognitive decline | Higher cognitive decline was observed in participants with low to moderate adherence to the Medi diet than among those with better adherence (adjusted difference = −0.56 points in the Telephone Interview for Cognitive Status-modified (TICS-m) score, 95% CI = −0.99 − 0.13). | Positive correlation | FFQ | Med 0–9 | TICs-m battery |
| Obs | Gardener et al. AIBL [67] | 2012 N = 970 Australia 1.5 years | Medi | Cognition and cognitive decline within healthy control (HC) subjects, those with mild cognitive impairment (MCI), and those with Alzheimer’s Disease (AD) | Lower adherence to the Medi diet was associated with MCI and AD. Compared with HC subjects, subjects with AD had a lower mean Medi diet score (p < 0.001); and compared with HC subjects, subjects with MCI had a lower mean Medi diet score (p < 0.05). Compared with the reference HC category, each additional unit in the Medi diet score was associated with 13–19% lower odds of being in the MCI category, and 19–26% lower odds of being in the AD category. | Positive correlation | FFQ | Med 0–9 modified for Australian population | MMSE |
| Obs | Gardener et al. AIBL [68] | 2015 N = 527 Australia 3.0 years | Medi | Cognition change based on three dietary patterns: AusMeDi (modified Mediterranean); Western diet; Prudent diet | Results for global cognitive score and visuospatial functioning not significant. In a subset of APOE ε4 allele carriers, higher adherence to AusMeDi diet was associated with better performance in executive function in (p < 0.01). | Null | FFQ | Med 0–9 modified for Australian population | Battery |
| Obs | Gu et al. WHICAP II [69] | 2010 N = 1219 US 4.0 years | Medi | Effect of adherence to the Mediterranean diet (MeDi) and Alzheimer’s disease (AD) | Better adherence to Medi was significantly associated with lower risk for AD. Compared to those in the lowest tertile of Medi, subjects in the highest tertile had 34% less risk of developing AD (p-for-trend = 0.04) | Positive correlation | FFQ | Med 0–9 | Battery |
| Obs | Haring et al. WHI Memory study [70] | 2016 N = 6425 US 9.11 years | Medi DASH | Cognitive decline, incidence of mild cognitive impairment (MCI) or Parkinson’s Disease (PD) | Medi and DASH dietary patterns were not associated with cognitive decline in older women (p = 0.30 for Medi diet, p = 0.23 for DASH diet). Adherence to these healthy dietary patterns did not modify the risk for cognitive decline in women with hypertension. | Null: Med Null: DASH | FFQ | Med 0–9 | Battery |
| Obs | Hosking et al. PATH Through Life study [40] | 2019 N = 1220 Australia 12.0 years | Medi MIND | Cognitive impairment (MCI or dementia) | In adjusted logistic regression models, the MIND diet (OR = 0.47, 95% CI 0.24, 0.91), but not the Medi diet, was associated with reduced odds of 12-year cognitive impairment. For every 1-point increase in MIND score, the odds of impairment decreased by 19%. There were no associations between Medi and incidence of MCI/dementia. | Pos: MIND Null: Med | FFQ | Med 0–9 Med 0–55 MIND 0–15 | Battery |
| Obs | Kesse-Guyot et al. SUNVIMAX [71] | 2013 N = 3083 France 2.0 years | Medi | Cognition | Adherence to Medi did not provide a beneficial effect on cognitive function. However, within the battery of six neuropsychological tests, a lower phonemic fluency score (p = 0.048) and lower backward digit span score (p = 0.03) were observed with decreasing Medi score. | Mixed findings | 24HR | Med 0–9 | Battery |
| Obs | Koyama et al. Health ABC Study [72] | 2015 N = 2326 US 7.9 years | Medi | Cognition and cognitive decline | Adherence to Medi shows mixed results between Black and white participants. In Black participants, high Medi scores had lower mean rate of cognitive decline on the 3MS score, which measures several cognitive domains (p = 0.01). No similar association was found in white participants (p = 0.14). Mechanisms for differences undetermined. | Mixed findings | FFQ | Med 0–9 | 3MS |
| Obs | Morris et al. MAP [39] | 2015 N = 923 US 4.5 years | Medi DASH MIND | Cognition and cognitive decline | High adherence to all three diets demonstrated a reduced risk of AD. Moderate adherence to the MIND diet only also showed decreased AD risk. In adjusted proportional hazards models, the second (HR = 0.65, 95% CI 0.44, 0.98) and highest tertiles (HR = 0.47, 95% CI 0.26, 0.76) of MIND diet scores had lower rates of AD versus tertile 1, whereas only the third tertiles of the DASH (HR = 0.61, 95% CI 0.38, 0.97) and Mediterranean (HR = 0.46, 95% CI 0.26, 0.79) diets were associated with lower AD rates. | Pos: Med Pos: DASH Pos: MIND | FFQ | Med 0–55 Dash 0–10 MIND 15 | Battery |
| Obs | Morris et al. MAP [17] | 2015 N = 960 US 4.7 years | MIND | Cognition and cognitive decline | In adjusted mixed models, the MIND score was positively associated with slower decline in global cognitive score (p < 0.0001) and with each of the five cognitive domains. The difference in decline rates for the top tertile of MIND diet scores vs. the lowest was the equivalent of being 7.5 years younger in age. | Positive correlation | FFQ | MIND 0–15 | Battery |
| Obs | Olsson et al. Sweden ULSAM [73] | 2015 N = 1038 Sweden 12.0 years | Medi | Cognitive decline rates based on changes to the Mini-Mental State Examination (MMSE) score for participants who ate a Swedish Modified Medi Diet (mMDS); the WHO-recommended Healthy Diet Indicator; or a low carbohydrate high protein diet (LCHP) | There were no strong associations with development of cognitive dysfunction for any of the dietary patterns investigated. Neither the continuous Medi score nor the higher tertile of the Medi score was associated with lower risk of AD, dementia, or cognitive impairment. In a secondary analysis, in a subset of participants based on energy intake according to the Goldberg cut-off, the highest tertile of the Medi score was significantly associated with a lower risk of cognitive impairment (OR: 0.32, 95% CI: 0.11, 0.89). | Null | 24HR | Med 0–9 modified for the Swedish population | MMSE |
| Obs | Qin China Health and Nutrition Survey [37] | 2015 N = 1650 China 7.0 years | Medi | Cognition and cognitive decline | Adherence to the Medi diet showed different outcomes based on age group. For participants =/>65, quintile 3 of the Chinese adapted Medi vs. quintile 1 had a slower rate of cognitive decline (β = 0.042). There was no association in adults <65. | Mixed findings | 24HR | Med 0–9 | Battery |
| Obs | Roberts et al. [No Study Name] [74] | 2010 N = 1223 US 2.2 years | Medi | Mild Cognitive Impairment (MCI) | Higher Medi score tertile was not significantly associated with reduced risk of MCI (p = 0.24). | Null | FFQ | Med 0–9 | Battery |
| Obs | Samieri et al. Nurses Health Study [75] | 2013 N = 16,058 US 13.0 years | Medi | Cognition and cognitive decline | Medi was not associated with decline in global cognition or verbal memory. In a secondary approach analyzing a subset of participants age 70+, each higher quintile of long-term Medi score was linearly associated with better multivariable-adjusted mean cognitive scores (p = 0.004, 0.002, and <0.001 for Telephone Interview for Cognitive Status (TICS), global cognition, and verbal memory, respectively.) | Null | FFQ | Med 0–9 modified for US population | Battery |
| Obs | Shakersain et al. SNAC-K [76] | 2018 N = 2223 Sweden 6.0 years | Medi DASH MIND | Analyze a change in global cognition based on the Mini-Mental State Examination (MMSE) when comparing the Nordic Prudent Dietary Pattern (NPDP) to the Medi, MIND, DASH, and Baltic Sea Diets (BSD). | Moderate (p < 0.019) and high (p < 0.001) adherence to MIND diet was related to less cognitive decline as measured by the MMSE. Only high adherence (0.002) to Medi was related to less cognitive decline. No associations were observed between DASH and cognitive decline. Moderate (p < 0.001) and high (p < 0.001) adherence to the NPDP were related to less cognitive decline. No associations were observed between BSD and cognitive decline. | Med: Pos DASH: Null MIND: Pos | FFQ | Med 0–55 Dash 8–40 modified for the Swedish population | MMSE |
| Obs | Tanaka et al. InCHIANTI [77] | 2018 N = 832 Italy 10.1 years | Medi | Global cognition measured by the Mini-Mental State Examination (MMSE) | Adherence to Medi was associated with a lower risk of cognitive decline as measured by the MMSE. Those in the highest adherence group (OR = 0.48, 95% CI: 0.29–0.79) and medium adherence group (OR = 0.64, 95% CI: 0.41–0.99) were less likely to experience cognitive decline. The annual average decline in MMSE scores was 0.4 units; for those in the high and medium adherence group, this decline was attenuated by 0.34 units (p < 0.001) and 0.16 units (p = 0.03), respectively. | Positive correlation | FFQ | Med 0–9 | MMSE |
| Obs | Tangney et al. Chicago Health and Aging Project [78] | 2011 N = 3790 US 7.6 years | Medi | Cognition and cognitive decline | Higher Medi scores were associated with reduced decline in cognitive function (p = 0.0004). Higher Medi diet wine scores were also associated with reduced decline in cognitive function (p = 0.0009). | Positive correlation | FFQ | Med 0–55 | Battery |
| Obs | Tangney et al. MAP [79] | 2014 N = 826 US 4.1 years | Medi DASH | Cognition and cognitive decline | Both the DASH and Mediterranean diet patterns were associated with slower rates of cognitive decline in the same cohort of older persons. A 1-unit difference in the DASH score was associated with a slower rate of global cognitive decline by 0.007 standardized units (standard error of estimate = 0.003, p < 0.03). A 1-unit-higher Medi score was associated with a slower rate of global cognitive decline by 0.002 standardized units (standard error of estimate = 0.001, p < 0.01). | Pos: Med Pos: DASH | FFQ | Med 0–55 | Battery |
| Obs | Titova et al. PIVUS [80] | 2013 N = 194 Sweden 5 years | Medi | Association between dietary habits, cognitive functioning and brain volumes in older adults | Low consumption of meat and meat products was linked to a better performance on the seven-minute screening (7MS) test (p = 0.001) and greater total brain volume (i.e., the sum of white and gray matter, p = 0.03) when controlling for potential confounders (e.g., BMI). Integrating all dietary features into the total modified Medi diet score explained less variance in cognitive functioning and brain volumes than the single dietary component meat intake. | Positive correlation | 7-day food diary | Med 0-8 (adapted from Med 0–9 for the Swedish population) | 7MS score |
| Obs | Trichopoulou et al. EPIC-Greece [32] | 2015 N = 401 Greece 7.0 years | Medi | Cognition and cognitive decline | Decline in cognition measured by MMSE was inversely associated with adherence to Medi, with only vegetable consumption exhibiting a substantial inverse effect. For mild vs. no decline, p = 0.012. For substantial vs. no decline, p = 0.025. | Positive correlation | FFQ | Med 0–9 | MMSE |
| Obs | Tsivgoulis et al. REGARDS [81] | 2012 N = 17,148 US 4+/−1.5 | Medi | Cognition, Incident Cognitive Impairment (ICI) | Higher adherence to the Medi diet was associated with lower likelihood of ICI before (OR = 0.89; 95% CI 0.79–1.00) and after adjustment for potential confounders (OR = 0.87; 95% CI 0.76–1.00). Higher adherence was associated with lower likelihood of ICI in non-diabetic (p = 0.0066) but not diabetic (p = 0.1063) individuals; diabetics were less adherent to the diet. | Positive correlation | FFQ | Med 0–9 | Six-item screener |
| Obs | Vercambre et al. WACS [82] | 2012 N = 2504 US 5.4 years | Medi | Cognitive decline | In both basic- and multivariable-adjusted models, consuming a Mediterranean style diet was not related to cognitive decline. The mean multivariable-adjusted difference 95% CI in rates of change in the global composite score was 0.01 (−0.01, 0.02) between the second and first tertile of the Medi score, and 0.00 (−0.02, 0.01) between the top tertile and first tertile (p = 0.88). | Null | FFQ | Med 0–9 | Battery |
| Obs | Wengreen et al. Cache County Study on Memory, Health, and Aging [35] | 2013 N = 3831 US 11.0 years | MediDASH | Global cognition | Higher DASH and Mediterranean diet scores were associated with higher average cognition measured by Modified Mini-Mental State (3MS) test scores over an 11-year period. Participants in quintile 5 of DASH averaged 0.97 points higher than those in quintile 1 (p = 0.001). The corresponding difference for Mediterranean quintiles was 0.94 (p = 0.001). | Pos: MediPos: DASH | FFQ | Med 0–9 (modified for low alcohol consumption in population) Dash 8–40 | 3MS Battery |
| Purpose/Cognitive Domain | Representative Cognitive Tests |
|---|---|
| Screening | Mini-Mental State Examination (MMSE) |
| Montreal Cognitive Assessment (MCOA) | |
| Executive Functioning | Stroop Test |
| Initial Letter Fluency (ILF) | |
| Excluded Letter Fluency (ELF) | |
| Tower of London (TOL) | |
| Trail Making Test-Part B | |
| NIH Toolbox Flanker Test | |
| Episodic Memory | Rey Auditory Verbal Learning Test (RALVT) |
| Letter-Number Sequencing Subtest from the Wechsler Adult Intelligence Scale (WAIS-IV) | |
| Immediate and Delayed Recall of the East Boston Story | |
| Word List Learning, Recall and Recognition from the Consortium to Establish a Registry for Alzheimer’s Disease (CERAD) | |
| Semantic Memory | Category Fluency (fruits, animals) |
| Multilingual Naming Test | |
| Working Memory | Digit Span Forward (DSF) |
| Digit Span Backward (DSB) | |
| Perceptual Speed | Symbol Digit Modalities Test |
| Number Comparison | |
| Stroop Word Reading | |
| Stroop Color Naming | |
| Symbol Search and Coding Core Subtests from WAIS IV | |
| Trail Making Test-Part A | |
| Visuospatial Ability | Judgment of Line Orientation |
| Standard Progressive Matrices | |
| Benton Visual Retention Test (BVRT) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duplantier, S.C.; Gardner, C.D. A Critical Review of the Study of Neuroprotective Diets to Reduce Cognitive Decline. Nutrients 2021, 13, 2264. https://doi.org/10.3390/nu13072264
Duplantier SC, Gardner CD. A Critical Review of the Study of Neuroprotective Diets to Reduce Cognitive Decline. Nutrients. 2021; 13(7):2264. https://doi.org/10.3390/nu13072264
Chicago/Turabian StyleDuplantier, Sally C., and Christopher D. Gardner. 2021. "A Critical Review of the Study of Neuroprotective Diets to Reduce Cognitive Decline" Nutrients 13, no. 7: 2264. https://doi.org/10.3390/nu13072264







