1. Introduction
Humans have relied on nature throughout the ages for their basic needs of food and health. At present, there is an increasing interest in exploring new sources of plant bio-actives for applications in both the food and pharmaceutical industries [
1]. Fruits and vegetables are the most important food sources, which supply essential nutrients and also contain an array of phytochemicals, such as phenolics and flavonoids, to maintain good health [
2]. Fruits provide an opportunity for local growers to access the specialized markets where consumers show a preference for exotic characteristics and the presence of nutrients in food, capable of preventing degenerative diseases [
3]. A number of reactive oxygen species (ROS), including superoxide anion, hydroxyl and hydrogen peroxide radicals, are produced in the human body by numerous enzymatic systems through oxygen consumption. These reactive oxygen species cause cancer, cardiovascular diseases, aging and neurodegenerative disorders [
4]. The ingestion of fruits and vegetables has been connected with a distinguished health-protecting factor against diseases caused by oxidative stress [
5,
6].
Health benefits of fruits and vegetables have been attributed partly to the compounds having antioxidant capacity and an ability to overcome oxidative stress by neutralizing the overproduction of oxidant species [
7,
8]. It has been reported that the additive and synergistic effects provided by the complex mixture of phytochemicals present in fruits and vegetables cannot be achieved through micronutrient supplements [
9]. Polyphenolic compounds, including phenolic acids, xanthones, gallotannins, carotenoids and vitamins (E and C), are important anti-radical, anti-mutagenic and anti-carcinogen agents [
10,
11]. They reduce the risk of chronic diseases, because of their safety, effectiveness and the presence of hydroxyl groups, which enable these compounds to have more diverse biological activities [
12]. Phenolics antioxidants, such as hydroxyl benzoic acid, and their derivatives are potent free radical scavengers of singlet oxygen possibly concerning DNA damage and tumor promotion [
13]. Phenolic acids are predominant compounds in the pulp of mango [
14]. Consumption of ripened mango is better, as it contains a high content of phenolic acids, which play a significant role in quenching and neutralizing the free radicals to improve consumers’ health [
15,
16]. Gallic acid is one the important anticancer agents, particularly against human prostate cancer cells
in vitro and
in vivo [
17]. Ferulic acid is an important phenolic compound in fruits and vegetables, which is generated as a result of phenylalanine and tyrosine metabolism [
18]. By virtue of effectively scavenging harmful radicals and suppressing radiation-induced oxidative reactions, ferulic acid serves as an important antioxidant, protects the body against different inflammatory diseases and is essential in preserving the physiological integrity of cells exposed to both air and impinging UV radiation [
19].
In vitro and
in vivo studies have revealed that chlorogenic acid is one of the most abundant polyphenols in the human diet, which exhibits significant anti-edematogenic, anti-nociceptive, antioxidant and anti-carcinogenic activities [
20]. It has been reported that protocatechuic acid showed anti-proliferative activity against HL-60 cells by inducing apoptosis and is associated with the phosphorylation and suppression of Bcl-2 protein [
21]. Caffeic acid has been proven as an inhibitor of hypertension and cardiotoxicity in rats by enhanced blood pressure, cardiac injury markers, restoration of the oxidant/antioxidant status, as well as decreasing histopathological changes [
22].
Mangifera indica L. (mango), “the king of fruits” belonging to the family Anacardiaceae, is one of the most popular fruits in tropical regions. Mango has been cultivated for 4000 years and ranks only second to pineapple in quantity and value among internationally-traded tropical fruits. In Mainland China, mango was first introduced from India in 645 A.D. by Tang Xuangzang (Tang Dynasty), and its commercial cultivation was started in the 1980s. Now, China has become the seventh mango cultivation country in the world, with annual production of about 1,061,800 tones on 133,100 hectares [
23]. Mango is considered as a good source of dietary compounds, such as ascorbic acid, phenolic compounds and carotenoids [
15,
24,
25], which are beneficial to health due to their antioxidant capacity [
26,
27]. The pulp of mango is effective for leukemia, prostate, breast and colon cancers
in vitro. Peels are the major by-products of different fruits and are good sources of phytochemicals and bioactive compounds [
28,
29,
30]. Mango peel, which comprises 15%–20% of the fruit, is an edible tissue and a major by-product of the mango processing industry. Peel of unripe mango is used in making chutney and pickle, while that of the ripe fruit, due to its leathery nature, is not so satisfactory in taste, therefore being generally removed and discarded. In the food processing industry, mango peel is removed for technological and sensory advantages and usually ends up as a waste by-product [
31]. Mango peel has been found to be a good source of polyphenols, carotenoids, dietary fiber, vitamin E and vitamin C [
28,
31], and it showed significant antioxidant properties [
32,
33].
Total phenolics, vitamin C and antioxidant activity have been reported in the fruit of mango varieties [
15,
24,
25]. However, comparative assessment of total phenolic content, phenolic acids and
in vitro antioxidant capacity in the pulp and peel of mango cultivars predominantly cultivated in China has rarely been reported before. In this context, the present study was designed to evaluate and correlate phenolic content and antioxidant activity and to assess the disparity in phenolic compounds and antioxidant capacity in the pulp and peel of nine cultivars of mango.
3. Materials and Methods
3.1. Chemicals and Material
Ascorbic acid (ASA), aluminum chloride, chloranil, tetrahydrofuran (THF), catechin hydrate, vanillin, Folin-Ciocalteu reagent, dichlorofluorescein diacetate (DCFH-DA), 2,2ʹ-azobis-amidinopropane (ABAP), gallic acid and 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) were purchased from Sigma Chemical Co. (St. Louis, MO, USA). Potassium dihydrogen phosphate (KH2PO4), sodium borohydride (NaBH4), ethanol, acetone, acetic acid, hydrochloric acid (HCl), di-potassium hydrogen phosphate (K2HPO4), sodium carbonate, sodium bicarbonate (NaHCO3) and acetonitrile were purchased from Aladdin Co. (Shanghai, China). Protocatechuic acid, chlorogenic acid, (+)-catechin, caffeic acid, p-coumaric acid, ferulic acid and formic acid of HPLC grade were purchased from Sigma-Aldrich, Inc. (St. Louis, MO, USA). Methanol and acetonitrile of HPLC grade were purchased from Anpel Scientific instrument Co., Ltd. (Shanghai, China).
Fresh and fully-ripened mango fruits of nine different cultivars, including Luzon (Lvsong), Narcissus (Shuixian), Royal (Guifei), Big Tainong (Da Tainong), Keitt (Kaite), Australian mango (Aozhou), Thai mango (Xiangya), Small Tainong (Xiao Tainong) and Egg mango (Jidan), were purchased from the supermarkets of Guangzhou city in Guangdong province of China and transported instantly to the laboratory for analysis (
Figure 4). All fruits were properly cleaned with de-ionized water and stored at −40 °C until analysis.
Figure 4.
Mango cultivars used in the present analysis. M1, Luzon mango (Lvsong); M2, Narcissus mango (Shuixian); M3, Royal mango (Guifei); M4, Big Tainong mango (Da Tainang); M5, Keitt mango (Kaite); M6, Australian mango (Aozhou); M7, Thai mango (Xiangya); M8, Small Tainong mango (Xiao Tainang); M9, Egg mango (Jidan).
Figure 4.
Mango cultivars used in the present analysis. M1, Luzon mango (Lvsong); M2, Narcissus mango (Shuixian); M3, Royal mango (Guifei); M4, Big Tainong mango (Da Tainang); M5, Keitt mango (Kaite); M6, Australian mango (Aozhou); M7, Thai mango (Xiangya); M8, Small Tainong mango (Xiao Tainang); M9, Egg mango (Jidan).
3.2. Moisture Content
The moisture content in the pulp and peel was determined by a modified oven-dried method [
46]. Briefly, 10 g of pulp and 5 g of peel samples were dried in an oven at 105 °C until constant weight. Each drying test was performed in triplicate, and data were presented as the mean ± standard deviation (SD) of triplicates.
3.4. Determination of Total Phenolic Content
The Folin-Ciocalteu colorimetric method, as described earlier [
49], with modifications [
50], was used to determine the total phenolic content in the peel and pulp samples. All extracts were diluted with Milli-Q water to get readings falling within the range of the standard curve concentration: 0.0–600.0 µg gallic acid/mL. One hundred microliters of gallic acid solution or extracts were added to 0.4 mL of Milli-Q water in each test tube, followed by the addition of Folin-Ciocalteu reagent (0.1 mL). The solutions were allowed to react for 6 min to ensure the complete and speedy reaction of the Folin-Ciocalteu reagent with oxidizable phenolates in the sample. Then, 1 mL of 7% sodium carbonate solution was added to neutralize the mixture, followed by the addition of 0.8 mL Milli-Q water to adjust the final volume to 2.4 mL. The samples were mixed and allowed to stand for 90 min at room temperature. After color development, absorbance was measured at 760 nm on a DU 730 Nucleic Acid/Protein analyzer (BECKMAN, Inc., Fullerton, CA, USA). Total phenolic contents were calculated based on the standard curve of known gallic acid concentrations, and final values were expressed as milligrams of gallic acid equivalent per 100 grams on a fresh weight basis (mg GAE/100 g FW). Data were presented as the mean ± SD for triplicates analyses.
3.5. Estimation of Total Flavonoid Content
Total flavonoid content was estimated by the sodium borohydride/chloranil method (SBCM) as established in our laboratory [
30]. Briefly, 1 mL of each extract was added into test tubes (15 × 150 mm), then kept under nitrogen gas until dried and reconstituted with 1 mL of tetrahydrofuran/ethanol (THF/EtOH, 1:1,
v/
v). Freshly-prepared catechin hydrate (0.3–10.0 mM) in 1 mL of THF/EtOH (1:1,
v/
v) was used as the standard for analysis. Zero-point-five milliliters of each (NaBH
4 (50 mM) and AlCl
3 (74.6 mM)) solution were added into all test tubes with samples or standards and shaken on an orbital shaker at room temperature for 30 min. Additionally, 0.5 mL of NaBH
4 (50.0 mM) solution were added into each test tube and shaken for another 30 min under the same condition. After shaking, 2.0 mL of chilled acetic acid (0.8 M) were thoroughly mixed, and the mixture was kept in the dark for 15 min. Then, 1 mL chloranil solution (20.0 mM) was added in each tube, and the mixture was heated at 95 °C in a shaking bath for 60 min. The reaction solutions were cooled with tap water, and the final volume was kept at 4 mL using methanol. One milliliter of 16% vanillin solution (
w/
v) was added into each tube, followed by the addition of 2 mL HCl (12 M), then mixed thoroughly and kept in the dark for 15 min. The reaction solutions were centrifuged at 2500 rpm for 10 min, and absorbance was immediately measured at 490 nm against a blank using a DU 730 Nucleic Acid/Protein analyzer (BECKMAN, Inc.). Total flavonoid content in each sample was calculated, using the standard curve of catechin hydrate concentration. The final value was expressed as milligrams of catechin equivalent per 100 gram of fresh weight (mg CE/100 g FW), and data were reported as the mean ± SD for triplicate analyses.
3.6. Determination of Total Anthocyanin Content
Total anthocyanin content was determined following the method as explained by [
41]. Acetone extracts of pulp and peel samples in triplicate were mixed carefully with 0.025 M potassium chloride buffer (pH = 1) in 1:6 ratio. The absorbance was measured at 515 and 700 nm against distilled water blank (BECKMAN). Afterword, the extracts were mixed with sodium acetate buffer (pH = 4.5); absorbance was measured at the same wavelengths, and the total content of anthocyanins was calculated using the formula as follows:
where
A is absorbance = (
A515 −
A700) pH 1.0 − (
A515 −
A700) pH 4.5;
MW is the molecular weight for cyanidin 3-glucoside = 449.2; ε is the molar absorptivity of cyanidin 3-glucoside = 26,900; and
C is the concentration of the buffer in mg/mL. Anthocyanin content was expressed as milligrams of cyanidin 3-glucoside equivalent per 100 g on fresh weight basis (mg CGE/100 g FW), and data were reported as the mean ± SD for triplicates analyses.
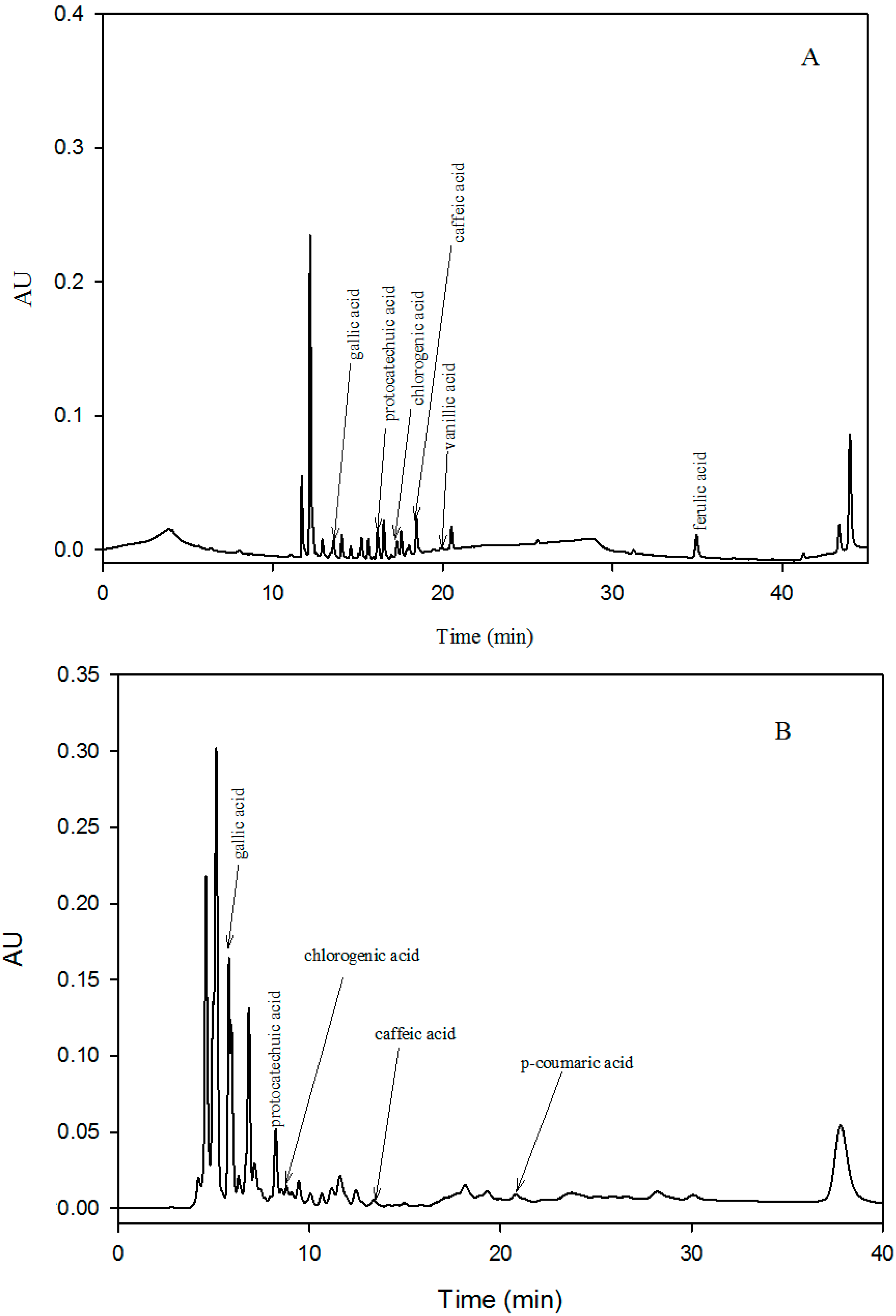
3.7. Identification and Quantification of Phenolic Acids
Phenolic acids in the pulp and peel extracts of mango cultivars were determined by the method explained by [
15]. Samples were injected automatically into an HPLC system (Waters Corp., Milford, MA, USA) equipped with a photodiode array detector. Absorption spectra for the main peaks were recorded at 280 and 320 nm. The HPLC system was equipped with a C18 reverse phase column (250 mm × 4.6 mm, 5 μm);the mobile phase was composed of 1% formic acid (A) and acetonitrile (B), and the isocratic elution gradient was 20% (B) in 40 min at a flow rate of 0.6 mL/min at 25 °C. The injection volume of the sample was 20 µL. Peaks were identified on the basis of retention time and chromatographs of the standards. Phenolic acids were identified and quantified on the basis of calibration curves and were expressed as mg phenolics per 100 g of FW. Data were reported as the mean ± SD for triplicate analyses.
3.8. Antioxidant Capacity Assays
Currently, researchers are paying more attention to natural antioxidants present in fruits, vegetables and whole foods because of their safety and potential nutritional and therapeutic effects [
51]. The antioxidant potential of commonly-consumed tropical and subtropical fruit has been rated in the order of guava > mango > papaya > lemon [
52]. Owing to the complex reactivity of phytochemicals, the antioxidant capacity of food and food extracts cannot be estimated by only a single method. However, at least two test systems have been recommended to establish legitimacy [
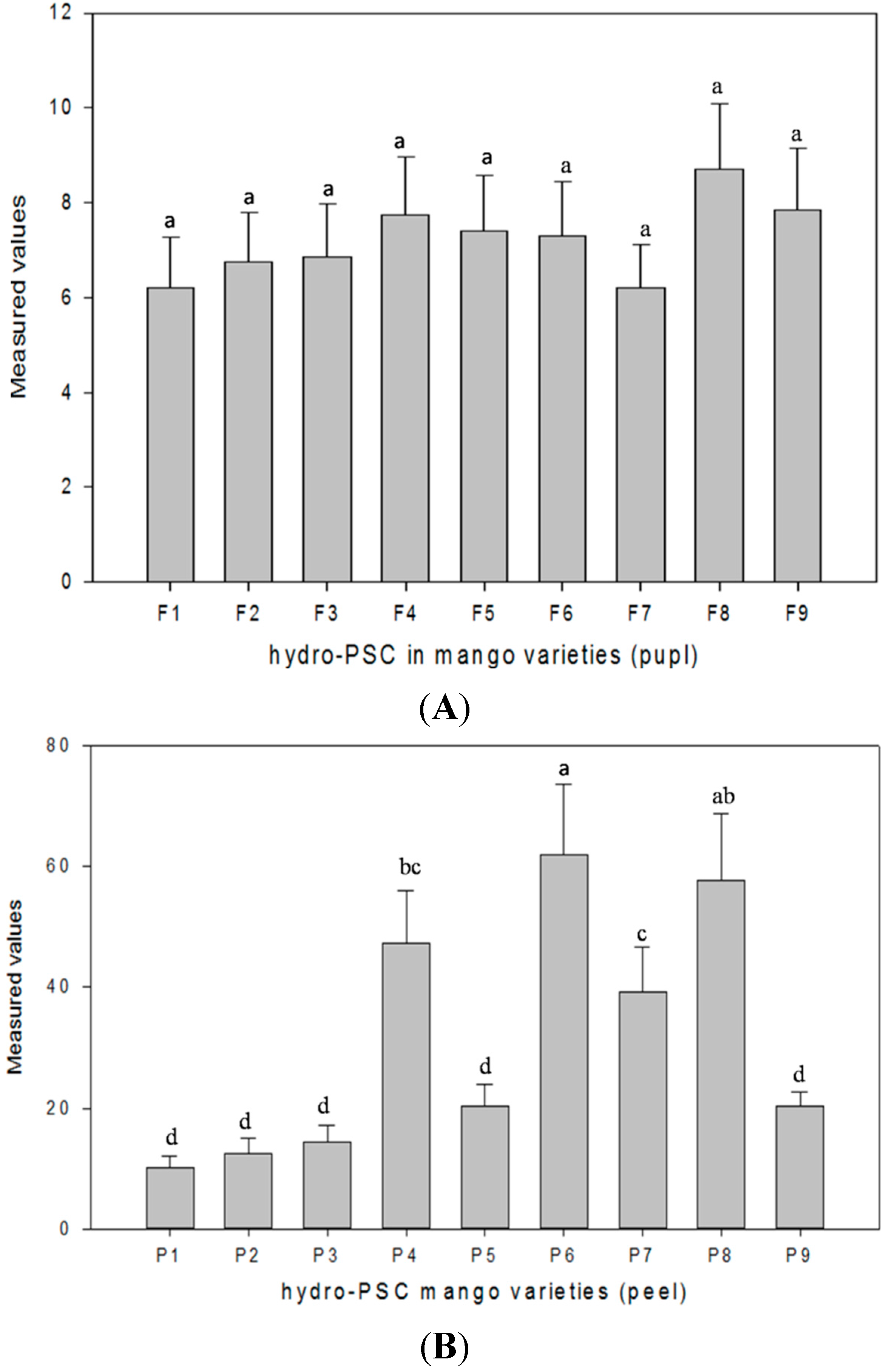
53]. Consequently, the antioxidant capacity in the peel and pulp samples of mango cultivars was evaluated by the peroxyl scavenging capacity (PSC) and oxygen radical antioxidant capacity (ORAC) methods.
3.8.1. Hydrophilic Peroxyl Radical Scavenging Capacity Assay
The peroxyl scavenging capacity (PSC) assay is based on the oxidation of DCFH by peroxyl radicals and is used to determine the antioxidant capacity in hydrophilic and lipophilic extracts of fruits, vegetables, grains and whole food [
47].The hydrophilic peroxyl radical scavenging capacity (hydro-PSC) assay, as explained by [
47], with modifications [
48,
54], was used to assess antioxidant capacity in the pulp and peel of mango cultivars. Seventy five millimolar phosphate buffer (pH 7.4) was used to dilute samples in appropriate concentrations. Ascorbic acid and gallic acid were made fresh and diluted to (6.3, 4.8, 3.2, 2.4, 1.0) and (5, 3.5, 2.7, 1.4, 0.9) μg/mL concentrations, respectively, using phosphate buffer (75 mM, pH 7.4). The reaction mixture contained phosphate buffer (75 mM, pH 7.4), ABAP (40 mM), DCFH dye (13.26 μM) and the suitable amount of the pure antioxidant compound or sample extract. The dye was prehydrolyzed with 1 mM KOH to eradicate di-acetate before use and the reaction was carried out at 37 °C, in a total volume of 250 µL using a 96-well plate. Fluorescence generation was observed (excitation at 485 nm and emission at 538 nm) on a Fluoroskan Ascent fluorescent spectrophotometer (SoftMax systems, Molecular Devices, Sunnyvale, CA, USA). Data were analyzed using SoftMax Pro Software, Version 6.2 (SoftMax systems, Molecular Devices) running on a PC. The areas under the fluorescence reaction time kinetic curve (AUC) for both control and samples were included and used as the basis for the determination of peroxyl radical scavenging capacity (PSC) using equation:
where
SA is the AUC for the sample or standard dilution and
CA is the AUC for the control reaction. Compounds or extracts inhibiting the oxidation of DCFH produced lesser
SA and higher
PSC values. EC
50, the dose requisite to cause 50% inhibition (
PSC unit = 0.5) for each pure compound or sample extract, was used to assess antioxidant activity of different compounds or samples. Final values of hydro-PSC were expressed as μmol of vitamin C equivalent per 100 g of FW (μM vCe/100 g of FW), and data were reported as the mean ± SD of each triplicate.
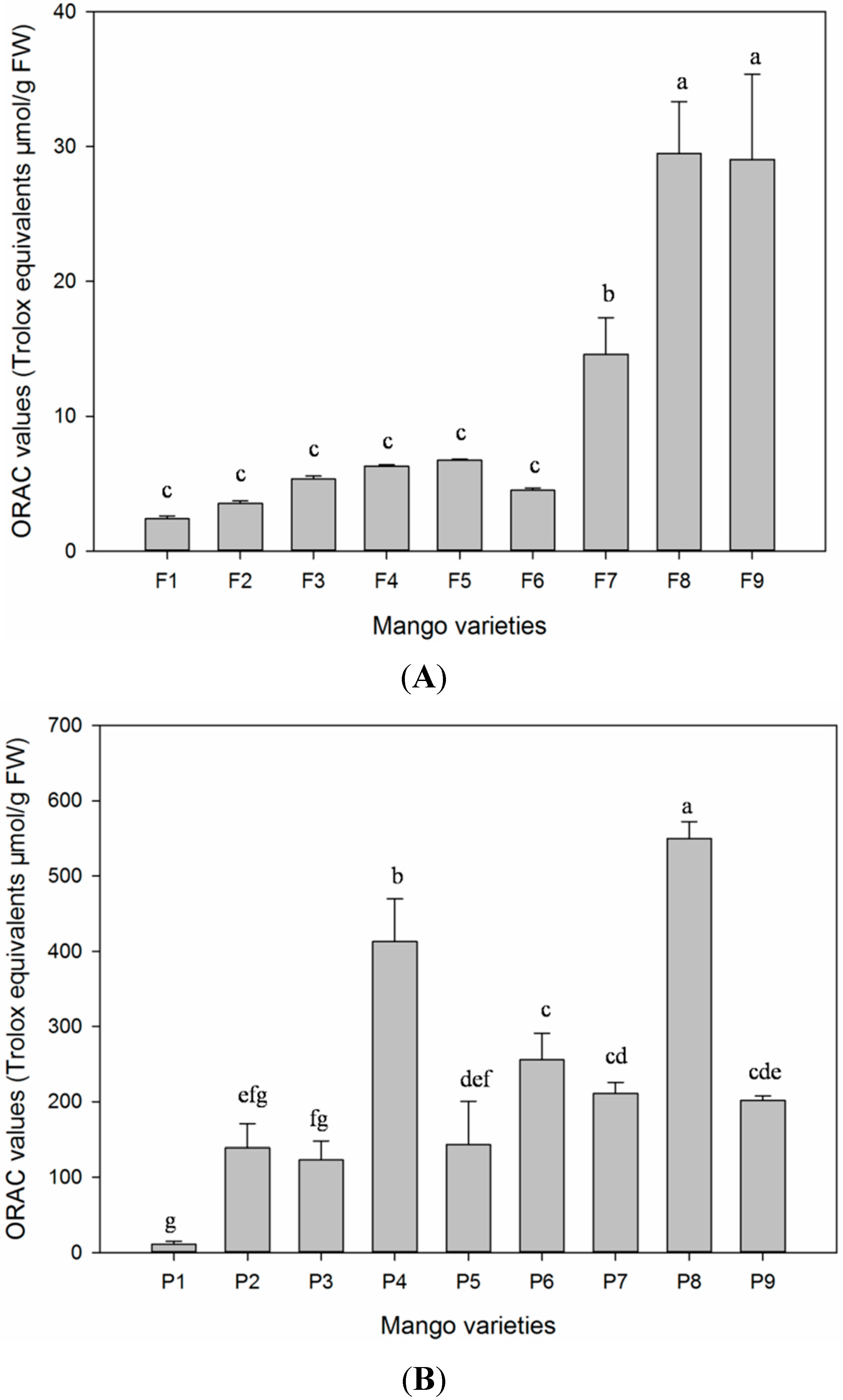
3.8.2. Oxygen Radical Scavenging Capacity Assay
The ORAC assay is a widely-used method to analyze the oxygen radical absorbance capacity of plant species extracts. This assay is based on free radical damage to a fluorescent probe through a change in its fluorescence intensity [
55]. In the typical ORAC assay, the fluorescent loss of probes as phycoerythrin or fluorescein is followed over time in the absence and presence of antioxidant [
41]. The oxygen radical absorbance capacity (ORAC) assay, as described by [
55], with modifications [
56], was conducted to measure the total antioxidant activity of the studied samples. Briefly, 20 μL of sample extracts in triplicate, diluted with 75 mM phosphate buffer (pH 7.4), were added in 96-well microplate, followed by the addition of 200 μL of fluorescein (0.96 μM), and incubated at 37 °C for 20 min. Outer wells were kept empty to avoid variation from inner wells. After incubation, 20 μL of freshly-prepared 119.4 mM AAPH in 75 mM phosphate buffer (pH 7.4) were added into each well, and the fluorescence intensity was measured immediately for 35 cycles every 4.5 min at an excitation of 485 nm and emission of 535 nm by the FilterMax F5 Multi-Mode Microplate Reader (Molecular Devices, Sunnyvale, CA, USA). Different concentrations of Trolox (range 6.25–50 μM) were used as a control. ORAC values were calculated by extrapolation on a calibration curve and expressed as the mean ± SD micromoles of Trolox equivalent per 100 g of fresh weigh (μM TE/100 g of FW) for three replicates.
3.9. Statistical Analysis
Statistical analyses were performed using SPSS software 13.0 (SPSS Inc., Chicago, IL, USA), and the dose effect was analyzed using Calcusyn software Version 2.0 (Biosoft, Cambridge, UK). Results were subjected to ANOVA, and differences among means were located using Tukey’s multiple comparison test. A p-value less than 0.05 (p < 0.05) was regarded as statistically significant. Basic statistical parameters and correlation coefficients among the measured variables were also calculated. All data were reported as the mean ± SD for three replicates.
4. Conclusions
The present study was focused on comparative assessment of phenolic content and in vitro antioxidant capacity in the pulp and peel of mango cultivars. Though phenolic compounds showed a significant contribution in the inhibition of free radicals, the antioxidant capacity of mango and other fruits is not only due to the content of phenolic acids. It may also be due to the presence of various bioactive compounds, such as carotenoids, vitamins and other polyphenolics phytochemicals present in the pulp and peel of mango, which were not identified in the present study. Our results showed that the Xiao Tainang and Aozhou cultivars contained maximum phenolic content and exhibited remarkable antioxidant capacity. Gallic acid and protocatechuic acid were predominant in the peel and pulp of the studied samples. Highly significant correlations (r = 0.997, 0.962, 0.922, etc.) were calculated between phenolic and antioxidant properties, particularly in the peel samples. The present study revealed that the antioxidant capacity of mango peel is due to the synergistic actions of phenolics and other bioactive compounds present in it. Therefore, it is suggest that mango peel may contribute to promoting human health as a functional food or a value-added ingredient. To our knowledge, this is the first report on the comparative assessment of phenolic compounds and antioxidant capacity determined by ORAC and hydro-PSC assays in the peel and pulp of mango cultivars in China, particularly in peel. However, additional studies are desirable to assess the bio-absorption, mechanism of action and associations between these compounds after consumption.