-
 Two Highly Specific Mouse Monoclonal Antibodies to the Putative C-Telopeptide of Human Collagen XIα1, a Cancer Biomarker
Two Highly Specific Mouse Monoclonal Antibodies to the Putative C-Telopeptide of Human Collagen XIα1, a Cancer Biomarker -
 Developability Evaluation of Single-Domain Antibody-Chelator Conjugates for Diagnostic Radiotracers
Developability Evaluation of Single-Domain Antibody-Chelator Conjugates for Diagnostic Radiotracers -
 Dermatomyositis with Anti-MDA5 Autoantibodies After SARS-CoV-2 mRNA Vaccination Treated with Tofacitinib: Integrating Literature Evidence and a Novel Observation
Dermatomyositis with Anti-MDA5 Autoantibodies After SARS-CoV-2 mRNA Vaccination Treated with Tofacitinib: Integrating Literature Evidence and a Novel Observation -
 Discovery of Anti-SARS-CoV-2 XBB.1.5 and JN.1 Variant-Specific Monoclonal Single-Domain Antibodies from a Synthetic Library
Discovery of Anti-SARS-CoV-2 XBB.1.5 and JN.1 Variant-Specific Monoclonal Single-Domain Antibodies from a Synthetic Library -
 Evaluation of Anti-dsDNA Antibodies in Laboratory Practice: Management of Different Analytical Methods and Correlation with HEp-2 Immunofluorescence Patterns
Evaluation of Anti-dsDNA Antibodies in Laboratory Practice: Management of Different Analytical Methods and Correlation with HEp-2 Immunofluorescence Patterns
Journal Description
Antibodies
Antibodies
is an international, peer-reviewed, open access journal on immunoglobulins, published bimonthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: CiteScore - Q2 (Drug Discovery)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.5 days after submission; acceptance to publication is undertaken in 5.7 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
2.7 (2024);
5-Year Impact Factor:
4.7 (2024)
Latest Articles
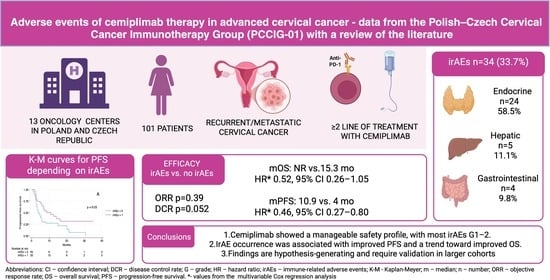
Immune-Related Adverse Events of Cemiplimab Therapy in Advanced Cervical Cancer—Data from the Polish–Czech Cervical Cancer Immunotherapy Group (PCCIG-01) with a Review of the Literature
Antibodies 2026, 15(3), 42; https://doi.org/10.3390/antib15030042 - 18 May 2026
Abstract
Background: Immunotherapy has become an integral part of systemic treatment for cervical cancer (CC). This study assessed the safety profile of cemiplimab and the association between immune-related adverse events (irAEs) and treatment outcomes in patients with persistent, recurrent or metastatic CC. Methods: This
[...] Read more.
Background: Immunotherapy has become an integral part of systemic treatment for cervical cancer (CC). This study assessed the safety profile of cemiplimab and the association between immune-related adverse events (irAEs) and treatment outcomes in patients with persistent, recurrent or metastatic CC. Methods: This ambispective, multicenter, real-world cohort study included 101 patients treated in 13 reference oncology centers as part of the PCCIG-01 study. We evaluated the frequency and severity of irAEs and their association with progression-free survival (PFS) and overall survival (OS). Survival outcomes were analyzed using the Kaplan–Meier method and Cox proportional hazards models, with p < 0.05 considered statistically significant. Results: After a median follow-up of 7.5 months, adverse events occurred in 45 patients (44.6%) and were mostly grade (G) 1–2. IrAEs were observed in 34 patients (33.7%). Endocrine toxicities predominated (n = 24, 58.5% of irAEs), followed by hepatic (n = 5, 12.2%) and gastrointestinal events (n = 4, 9.8%). G3 irAEs occurred in 8 patients (7.9%). Median PFS was 3.9 months (95% CI 2.9–5.6) in patients without irAEs and 10.9 months (95% CI 5.7–16.3) in those with irAEs (p = 0.03). Median OS was 15.3 months (95% CI 8.6–25.9) in patients without irAEs and was not reached in those with irAEs (95% CI 11.6-NR; p = 0.11). The development of irAEs was associated with a 54% reduction in the risk of progression (HR 0.46, 95% CI 0.27–0.80), with no statistically significant impact on OS. Conclusions: In exploratory analyses, the occurrence of irAEs was associated with improved PFS in cemiplimab-treated patients with persistent, recurrent or metastatic CC. Cemiplimab showed a manageable safety profile, with most toxicities being G1–G2.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►
Show Figures
Open AccessArticle
The Shifting Core: Antigenic Variability of the Influenza Virus Nucleoprotein Despite Evolutionary Conservation
by
Alexandra Rak, Veronika Muzurova, Svetlana Donina, Polina Prokopenko, Irina Isakova-Sivak and Larisa Rudenko
Antibodies 2026, 15(3), 41; https://doi.org/10.3390/antib15030041 - 15 May 2026
Abstract
Background. The highly mutable influenza virus causes severe annual infections worldwide and results in substantial socioeconomic losses. The spread of infection could be effectively controlled by cross-protective vaccines and universal diagnostic test systems based on the nucleoprotein (NP) as one of the most
[...] Read more.
Background. The highly mutable influenza virus causes severe annual infections worldwide and results in substantial socioeconomic losses. The spread of infection could be effectively controlled by cross-protective vaccines and universal diagnostic test systems based on the nucleoprotein (NP) as one of the most conserved viral antigens. However, NP also undergoes slow evolutionary changes, and little is known about the influence of these mutations on its antigenicity and immunogenicity. Methods. We expressed the full-length recombinant 6xHis-tagged NPs of ten evolutionary distant influenza A strains of different subtypes in E. coli BL21(DE3) cells and purified these proteins by immobilized metal affinity chromatography. The obtained antigens were identified by mass spectrometry and serological methods. NPs served as antigens for three immunizations of BALB/c mice (15 µg/animal at 14-day interval) and as capturing proteins in ELISA at 2 µg/mL, in order to study the effect of adaptive mutations on the antigenic and immunogenic properties of NPs. Results. A pronounced cross-reactivity of anti-NP antibodies induced in mice by immunization with different NPs was revealed. At the same time, we observed the differences in the humoral immunogenicity of NP, which are in line with the accumulation of evolutionarily driven NP mutations. In general, antibody affinity to heterologous NPs was reduced, indicating the differences in the specificity of anti-NP immunoglobulins, which may be caused by evolutionarily determined variability of immunogenic epitopes leading to the emergence of escape mutations. Conclusions. Overall, our results reflect the slightly evolving nature of the NP antigen, which influences the specificity spectrum of anti-NP antibodies and should be considered as a limitation for the development of NP-based cross-protective vaccines and test systems.
Full article
(This article belongs to the Section Humoral Immunity)
Open AccessCase Report
Safety and Efficacy of Mosunetuzumab: Experience in the Hospital Cardinale Giovanni Panico
by
Giulio Turco, Donatella Tarantino, Antonietta Giuseppa Ferraro, Giuseppina Greco and Domenico Tricarico
Antibodies 2026, 15(3), 40; https://doi.org/10.3390/antib15030040 - 13 May 2026
Abstract
Background/Objective: Follicular lymphoma (FL) is one of the most common indolent B-cell non-Hodgkin lymphomas (NHL) and is characterized by recurrent relapses despite advances in therapy. Bispecific antibodies that redirect T lymphocytes toward malignant B cells represent a major innovation in the treatment of
[...] Read more.
Background/Objective: Follicular lymphoma (FL) is one of the most common indolent B-cell non-Hodgkin lymphomas (NHL) and is characterized by recurrent relapses despite advances in therapy. Bispecific antibodies that redirect T lymphocytes toward malignant B cells represent a major innovation in the treatment of relapsed or refractory disease. Mosunetuzumab is a CD20×CD3 bispecific antibody that induces T-cell mediated cytotoxicity against B-cell malignancies. In this manuscript, we describe the clinical experience with mosunetuzumab in three patients with relapsed or refractory FL treated at the Hospital Card. G. Panico, Tricase (LE). Methods: Clinical history, prior therapies, treatment responses, and safety outcomes are reported. Results: The cases illustrate the potential efficacy and manageable safety profile of mosunetuzumab in heavily pretreated FL patients. Conclusion: The effectiveness of this drug is confirmed in our center.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Figure 1
Open AccessArticle
Ca13Mab-17, a Novel Anti-Cadherin-13 Monoclonal Antibody for Versatile Applications
by
Kai Shimizu, Hiroyuki Suzuki, Mika K. Kaneko and Yukinari Kato
Antibodies 2026, 15(3), 39; https://doi.org/10.3390/antib15030039 - 11 May 2026
Abstract
Background/Objectives: Cadherin-13 (CDH13), part of the cadherin family, is attached to the plasma membrane through glycosylphosphatidylinositol. CDH13 plays essential roles in the development of the neurological and vascular systems and is a risk factor for neural and cardiovascular diseases. CDH13 is expressed on
[...] Read more.
Background/Objectives: Cadherin-13 (CDH13), part of the cadherin family, is attached to the plasma membrane through glycosylphosphatidylinositol. CDH13 plays essential roles in the development of the neurological and vascular systems and is a risk factor for neural and cardiovascular diseases. CDH13 is expressed on the plasma membrane in both mature and uncleaved precursor forms with the prodomain. Although several anti-CDH13 monoclonal antibodies (mAbs) are available for basic research, there have been no reports of anti-CDH13 mAbs that can detect both the mature form and the uncleaved precursor in flow cytometry. Methods: We developed novel anti-human CDH13 mAbs (named Ca13Mabs) using the mature form of CDH13-expressed cells as an antigen. Results: Among Ca13Mabs, a clone, Ca13Mab-17 (IgG2b, κ) specifically recognized the mature and uncleaved precursor CDH13-overexpressed Chinese hamster ovary-K1 (CHO/CDH13) cells with no detectable cross-reactivity toward 21 other cadherins by flow cytometry. Ca13Mab-17 also detected endogenous CDH13 in human glioblastoma (LN229 and U87MG) and lung mesothelioma (NCI-H2052) cell lines. The dissociation constant (KD) value of Ca13Mab-17 for LN229 was estimated at 4.1 × 10−8 M. Furthermore, Ca13Mab-17 detected both the mature and uncleaved precursor CDH13 in Western blotting. It also identified new blood vessels and glioblastoma cells by immunohistochemistry. Conclusions: Ca13Mab-17 is a versatile tool for detecting both mature and uncleaved precursor forms of CDH13 and has potential for tumor diagnosis and therapy.
Full article
(This article belongs to the Section Antibody Discovery and Engineering)
►▼
Show Figures

Figure 1
Open AccessArticle
A Recombinant Antibody Against Human DRP1 Serine 616 Phosphorylation Enables Detection of BRAFV600E-Associated Mitochondrial Division in Cancer
by
Shanon T. Nizard, Yiyang Chen, Madhavika N. Serasinghe, Ruben Fernandez-Rodriguez, Kamrin D. Shultz, Jesminara Khatun, Anthony Mendoza, Jesse D. Gelles, Juan F. Henao-Martinez, Ioana Abraham-Enachescu, Md Abdullah Al Noman, Stella G. Bayiokos, J. Andrew Duty, Shane Meehan, Mihaela Skobe and Jerry Edward Chipuk
Antibodies 2026, 15(2), 38; https://doi.org/10.3390/antib15020038 - 20 Apr 2026
Abstract
Background/Objectives: Mitochondria are dynamic organelles that continuously undergo balanced cycles of fusion and division to maintain optimal function. Mitochondrial division is mediated by Dynamin-Related Protein 1 (DRP1), a cytosolic large GTPase whose phosphorylation at serine 616 (DRP1-S616Ⓟ) promotes its translocation to the outer
[...] Read more.
Background/Objectives: Mitochondria are dynamic organelles that continuously undergo balanced cycles of fusion and division to maintain optimal function. Mitochondrial division is mediated by Dynamin-Related Protein 1 (DRP1), a cytosolic large GTPase whose phosphorylation at serine 616 (DRP1-S616Ⓟ) promotes its translocation to the outer mitochondrial membrane and organelle division. Dysregulated mitochondrial division disrupts cellular homeostasis and contributes to disease pathogenesis, including cancer. Our prior work demonstrated that the oncogene-induced mitogen-activated protein kinase (MAPK) pathway constitutively phosphorylates DRP1 at serine 616, which is essential to cellular transformation and correlates with oncogene status in patient tissues. Similarly, DRP1-S616Ⓟ is subject to pharmacologic control by targeted therapies against oncogenic MAPK signaling. Methods: Building upon this foundation, we developed and characterized a recombinant murine monoclonal antibody (referred to as 3G11) with high specificity for human DRP1-S616Ⓟ, raised against a peptide derived from the human DRP1 sequence. Results: Using diverse experimental platforms, we demonstrate the robust utility of 3G11 to detect DRP1-S616Ⓟ in melanoma cell extracts and isolated organelles. Immunofluorescence revealed that pharmacologic inhibition of oncogenic MAPK signaling reduces DRP1-S616Ⓟ levels, which correlates with mitochondrial hyperfusion, while immunohistochemistry showed that elevated DRP1-S616Ⓟ expression in human tissues correlates with BRAFV600E disease. Conclusions: 3G11 is a new recombinant antibody for detecting DRP1-S616Ⓟ and supports studies of mitochondrial division in cancer. Together, these findings establish 3G11 as a specific, versatile, renewable, and cost-effective tool for studying mitochondrial division, with strong potential for clinical applications.
Full article
(This article belongs to the Section Antibody Discovery and Engineering)
►▼
Show Figures

Figure 1
Open AccessReview
Brain Delivery of Antibody-Derived Biologicals for Alzheimer’s Disease: An Updated Narrative Review
by
Rachita K. Sumbria and Ruben J. Boado
Antibodies 2026, 15(2), 37; https://doi.org/10.3390/antib15020037 - 17 Apr 2026
Abstract
Antibodies directed against β-amyloid (Aβ) have been developed for the treatment of Alzheimer’s disease (AD). However, the in vivo central efficacy is reduced by the poor penetration of antibodies across the blood–brain barrier (BBB). In addition, these antibodies have been associated with adverse
[...] Read more.
Antibodies directed against β-amyloid (Aβ) have been developed for the treatment of Alzheimer’s disease (AD). However, the in vivo central efficacy is reduced by the poor penetration of antibodies across the blood–brain barrier (BBB). In addition, these antibodies have been associated with adverse effects like amyloid-related imaging abnormalities. Thus, the development of new antibody-based therapies for AD with improved transport across the BBB may improve efficacy and reduce adverse effects. Antibodies targeting the BBB transferrin receptor (TfR) are able to cross the BBB through receptor-mediated transcytosis, producing a global distribution throughout the brain. Along the same line, bispecific antibodies directed to both the BBB TfR and Aβ showed enhanced brain uptake and pharmacological effects with diminished adverse side effects in experimental animal models of AD and in clinical trials. A generation of brain-penetrating fusion proteins targeting the BBB-TfR has been shown to represent novel treatments for AD, and this includes erythropoietin, tumor necrosis factor alpha inhibitors, neprilysin, somatostatin, oligonucleotides, and an antibody activating TREM2. The aim of this article is to review the progress made in the delivery of antibody-derived biologicals to the brain for AD, targeting the BBB-TfR.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Figure 1
Open AccessReview
Post-Chemotherapy Antibody-Based Continuation and Maintenance Strategies in HER2-Positive Metastatic Breast Cancer: A Translational Narrative Review
by
Katarzyna Pogoda, Karolina Lewińska, Paulina Kalman, Anna Bałata and Piotr J. Wysocki
Antibodies 2026, 15(2), 36; https://doi.org/10.3390/antib15020036 - 16 Apr 2026
Abstract
The treatment paradigm for HER2-positive metastatic breast cancer has evolved from continuous chemotherapy-based regimens to a model of finite chemotherapy induction followed by sustained antibody-driven disease control. The CLEOPATRA trial established dual HER2 blockade with trastuzumab and pertuzumab plus a taxane as the
[...] Read more.
The treatment paradigm for HER2-positive metastatic breast cancer has evolved from continuous chemotherapy-based regimens to a model of finite chemotherapy induction followed by sustained antibody-driven disease control. The CLEOPATRA trial established dual HER2 blockade with trastuzumab and pertuzumab plus a taxane as the biological and clinical anchor of this approach, demonstrating that chemotherapy is administered for a defined induction period, after which antibody maintains disease suppression. An increasing body of clinical evidence indicates that antibody-based regimens can be combined with targeted agents, including CDK4/6 inhibitors or HER2 tyrosine kinase inhibitors, to achieve durable disease control without the need for continuous chemotherapy. In the PATINA trial, the addition of palbociclib to trastuzumab, pertuzumab, and endocrine therapy was associated with a clinically meaningful improvement in progression-free survival in hormone receptor-positive, HER2-positive metastatic breast cancer. At the same time, quality of life was maintained despite higher rates of hematologic toxicity. More recently, HER2-CLIMB-05 demonstrated that the addition of tucatinib to dual HER2 antibody therapy significantly prolonged progression-free survival, supporting a model of sustained, multi-agent HER2 pathway suppression. The monarcHER trial provided biological proof of concept that antibody plus CDK4/6 inhibition can achieve disease control without chemotherapy in hormone receptor-positive, HER2-positive disease. Collectively, these advances support a translational framework in which antibody therapy serves as a central component of treatment strategies, with targeted partners selected according to tumor biology and prior therapy. This review summarizes the biological basis, clinical evidence, and future perspectives of antibody-driven maintenance in HER2-positive metastatic breast cancer.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Graphical abstract
Open AccessReview
IMGT® Nomenclature of Immunoglobulins (IG) or Antibodies and T Cell Receptors (TR): A Common Language for Immunoinformatics and Artificial Intelligence (AI)
by
Marie-Paule Lefranc and Gérard Lefranc
Antibodies 2026, 15(2), 35; https://doi.org/10.3390/antib15020035 - 15 Apr 2026
Abstract
The immunoglobulins (IG) or antibodies and the T cell receptors (TR) are the antigen receptors of the adaptive immune responses (AIR) of jawed vertebrates (Gnathostomata). IMGT®, the international ImMunoGeneTics information system®, was created in 1989 by Marie-Paule
[...] Read more.
The immunoglobulins (IG) or antibodies and the T cell receptors (TR) are the antigen receptors of the adaptive immune responses (AIR) of jawed vertebrates (Gnathostomata). IMGT®, the international ImMunoGeneTics information system®, was created in 1989 by Marie-Paule Lefranc (Laboratoire d’ImmunoGénétique Moléculaire (LIGM), Université de Montpellier and CNRS) to deal with and to manage the huge diversity of IG or antibodies and TR. The founding of IMGT® marked the advent of immunoinformatics, a new science which emerged at the interface between immunogenetics and bioinformatics. For the first time, the IG and TR variable (V), diversity (D), joining (J) and constant (C) genes were officially recognized as ‘genes’, as were the conventional genes. The IMGT-ONTOLOGY CLASSIFICATION axiom and the concepts of classification have generated the IMGT nomenclature and the IMGT Scientific chart rules for assigning IMGT names to IG and TR genes and alleles of Homo sapiens and of any other jawed vertebrate species. The IMGT nomenclature is used for genes in locus, in sequences (genomic or rearranged, expressed or not) and in structures enabling comparative immunology, evolutionary immunogenetics, standardized analysis and comparison of IG and TR repertoires analysis in normal or pathologic situations. IMGT nomenclature is used in basic, veterinary, and medical research, in clinical applications (mutation analysis in leukemia and lymphoma), and in therapeutic antibody design, engineering and humanization. By providing consistent and high standard biocuration for the description of the IG and TR loci, genes and alleles, and for the analysis of the IG or antibody and TR-expressed rearranged sequences and proteins and structures, the IMGT nomenclature is the common language for immunoinformatics and artificial intelligence (AI).
Full article
(This article belongs to the Section Antibody Discovery and Engineering)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Development of a Human IgG1 Monoclonal Antibody Targeting Transferrin Receptor 1 for Antitumor Drug Delivery
by
Tingting Ji, Zhaoyun Zong, Ningyuan Gong, Minghui Yan and Shiyu Chen
Antibodies 2026, 15(2), 34; https://doi.org/10.3390/antib15020034 - 13 Apr 2026
Abstract
Background: Transferrin receptor protein 1 (TfR1) plays a central role in cellular iron uptake and is frequently overexpressed in malignant tumor cells, rendering it an attractive target for tumor-directed therapy and drug delivery. Methods: A fully human single-chain variable fragment (scFv)
[...] Read more.
Background: Transferrin receptor protein 1 (TfR1) plays a central role in cellular iron uptake and is frequently overexpressed in malignant tumor cells, rendering it an attractive target for tumor-directed therapy and drug delivery. Methods: A fully human single-chain variable fragment (scFv) antibody targeting TfR1, termed T8scFv, was isolated from a human scFv phage display library through three rounds of stringent biopanning and subsequently reformatted into a full-length IgG1 antibody (T8IgG1). Binding kinetics were characterized using Octet biolayer interferometry (BLI), while cellular binding and internalization were assessed by flow cytometry and immunofluorescence microscopy, respectively. T8IgG1 was further conjugated to DT3C, a recombinant truncated diphtheria toxin fusion protein, to evaluate its internalization-dependent cytotoxicity in vitro. Results: T8scFv exhibited nanomolar affinity for TfR1 (KD = 214 ± 1 nM), which was substantially enhanced following conversion to the IgG1 format (T8IgG1, KD = 18.5 ± 0.1 nM). T8IgG1 specifically recognized TfR1 on the surface of tumor cells and underwent efficient TfR1-mediated internalization. The T8IgG1-DT3C complex significantly reduced cell viability and induced apoptosis in K562 cells in vitro. Conclusions: These findings indicate that T8IgG1 is a moderate-affinity, internalizing anti-TfR1 antibody and highlight its potential as a promising candidate for TfR1-based targeted antitumor drug delivery systems.
Full article
(This article belongs to the Topic Antibody-Mediated Therapy and Other Emerging Therapies in Cancer Treatment)
►▼
Show Figures

Graphical abstract
Open AccessReview
Non-COVID-19 Vaccinations and the Induction of Autoantibodies in Pemphigus Diseases: A Review of the Speculative Issue and Our Clinical-Laboratory Experience
by
Maksymilian Markwitz, Natalia Welc, Klementyna Kępińska, Monika Bowszyc-Dmochowska and Marian Dmochowski
Antibodies 2026, 15(2), 33; https://doi.org/10.3390/antib15020033 - 10 Apr 2026
Abstract
Background: Pemphigus diseases are rare autoimmune blistering disorders mediated by pathogenic autoantibodies directed mainly against desmoglein 1 and desmoglein 3. Although most cases are considered idiopathic, external triggers that can disrupt immune tolerance have been described. Vaccination has been discussed as a
[...] Read more.
Background: Pemphigus diseases are rare autoimmune blistering disorders mediated by pathogenic autoantibodies directed mainly against desmoglein 1 and desmoglein 3. Although most cases are considered idiopathic, external triggers that can disrupt immune tolerance have been described. Vaccination has been discussed as a potential precipitating factor in autoimmune skin diseases. However, the relationship between vaccination and the induction of pemphigus-related autoantibodies has not been comprehensively summarized. Methods: We conducted a narrative review of all available studies published in the last 25 years identified through medical databases, excluding studies on COVID-19 vaccinations. Reports describing either new-onset pemphigus or exacerbation of preexisting pemphigus with a temporal association to vaccination were included. Clinical characteristics, vaccine type, latency period, direct immunofluorescence findings, and ELISA results for desmoglein autoantibodies were analyzed. In addition, we present our own clinical-laboratory experience illustrating this issue. Results: The current evidence consists predominantly of case reports and small case series. Published cases describe pemphigus vulgaris and pemphigus foliaceus occurring after vaccinations against influenza, hepatitis B, tetanus, diphtheria, pertussis, rabies, and other routinely administered immunizations. The latency period most often ranged from several days to a few weeks. Immunopathological findings were consistent with classical pemphigus diseases, including intercellular IgG deposits in the epidermis and circulating autoantibodies against desmoglein 1 and/or desmoglein 3. Our patient was a 78-year-old woman who developed cutaneous form of pemphigus vulgaris, diagnosed with direct immunofluorescence (DIF) and multiplex ELISA, 10 days after diphtheria–tetanus–pertussis vaccination. The patient had a positive family history of autoimmune blistering disease, namely mucous membrane pemphigoid. Conclusions: Based on the currently available evidence, a direct causal relationship between vaccination and pemphigus diseases cannot be established. Nevertheless, accumulated clinical and serological observations suggest that vaccination may act as a triggering factor in genetically or immunologically predisposed individuals, possibly by amplifying pre-existing subclinical autoreactive immune responses. Further population-based and mechanistic studies are required to clarify this association, while the overall benefits of vaccination remain substantial.
Full article
(This article belongs to the Section Humoral Immunity)
►▼
Show Figures

Figure 1
Open AccessCase Report
Monoclonal Antibodies in Pregnancy of Patients with Systemic Lupus Erythematosus: Friend or Foe? A Case Report of a Patient with Multiple Pregnancies
by
Chiara Orlandi, Angela Tincani, Micaela Fredi, Laura Andreoli, Francesca Crisafulli, Liala Moschetti, Cecilia Nalli, Maria Grazia Lazzaroni, Marco Taglietti, Matteo Filippini, Sonia Zatti, Laura Picciau, Franco Franceschini and Ilaria Cavazzana
Antibodies 2026, 15(2), 32; https://doi.org/10.3390/antib15020032 - 8 Apr 2026
Abstract
Systemic lupus erythematosus (SLE) is an autoimmune disease that predominantly affects women of childbearing age, and active disease during pregnancy is associated with increased maternal and fetal morbidity. Belimumab is an effective biologic therapy for active SLE; however, its use during pregnancy has
[...] Read more.
Systemic lupus erythematosus (SLE) is an autoimmune disease that predominantly affects women of childbearing age, and active disease during pregnancy is associated with increased maternal and fetal morbidity. Belimumab is an effective biologic therapy for active SLE; however, its use during pregnancy has long been limited by the scarcity of safety data. Recent evidence and updated international recommendations suggest that belimumab may be considered in selected cases when required to maintain maternal disease control. We report the case of a woman with SLE who experienced three consecutive pregnancies with live births between 2019 and 2024 while receiving belimumab, allowing an intra-individual comparison of different exposure strategies. During the first pregnancy, belimumab was discontinued at conception and was followed by a disease flare in late pregnancy and postpartum. In the second and third pregnancies, belimumab was continued until gestational week 20 following shared decision-making with the patient; nevertheless, disease flares occurred during the third trimester of both pregnancies. All pregnancies resulted in live births at term, with no congenital anomalies, placental insufficiency, or fetal growth restriction. One neonate from the third pregnancy developed early-onset neonatal sepsis and meningitis, which resolved completely after antibiotic treatment. All children are currently growing and developing normally. This case supports a risk-adapted approach to belimumab use during pregnancy. In selected women with SLE at high risk of disease reactivation, continuation of belimumab until mid-gestation may contribute to improved maternal disease control without evident adverse fetal outcomes.
Full article
(This article belongs to the Topic Antibody-Mediated Therapy and Other Emerging Therapies in Cancer Treatment)
►▼
Show Figures

Figure 1
Open AccessReview
The Possible Role of Antibodies in Alopecia: A Narrative Review
by
Julia Cieślawska, Mariola Pawlaczyk and Justyna Gornowicz-Porowska
Antibodies 2026, 15(2), 31; https://doi.org/10.3390/antib15020031 - 3 Apr 2026
Abstract
Human hair performs a number of important physiological and esthetic functions. Hair loss and alopecia are complex disorders which affect people all over the world. Hair loss can be an early manifestation of various autoimmunological disorders. Despite a growing interest of researchers in
[...] Read more.
Human hair performs a number of important physiological and esthetic functions. Hair loss and alopecia are complex disorders which affect people all over the world. Hair loss can be an early manifestation of various autoimmunological disorders. Despite a growing interest of researchers in the role of immune factors—especially autoantibodies—in the etiology of certain types of alopecia, their role in alopecia remains uncertain. Several potential autoantigens of follicular components, mainly derived from keratinocytes and melanocytes of the hair follicles, have been found to play a role in the development of alopecia areata. The list of autoantigens includes trichohyalin, keratin 16, fibroblast growth factor receptor 3, glycoprotein-100, melanoma-associated antigen recognized by T cells 1, dopachrome tautomerase/tyrosinase-related protein 2, tyrosinase, and tyrosine hydroxylase. This narrative review presents different aspects of immunopathogenesis of alopecia, from physiology (hair follicle immune privilege) to pathology (disruption of hair follicle immune privilege) and signaling pathways. Identification of key autoantigens could potentially pave the way for the development of new, effective, and more targeted immunotherapies for alopecia.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Figure 1
Open AccessArticle
SARS-CoV-2 Infection and COVID-19 Vaccine Antibody Responses in Two Canadian Cohorts of Persons Living with HIV
by
Sharon L. Walmsley, Leif Erik Lovblom, Bryan Boyachuk, Curtis Cooper, Valérie Martel-Laferrière, Mona Loutfy, Marie-Louise Vachon, Shariq Haider, Pamela Aldebes, Karen Colwill, Anne Claude Gingras, Freda Qi and Marina B. Klein
Antibodies 2026, 15(2), 30; https://doi.org/10.3390/antib15020030 - 3 Apr 2026
Abstract
Objectives: To determine the incidence and outcomes of SARS-CoV-2 infection and to evaluate seroconversion rates and quantify antibody responses to COVID-19 vaccines in two cohorts of persons living with HIV at a possible higher risk of poor outcomes (HCV coinfection and those over
[...] Read more.
Objectives: To determine the incidence and outcomes of SARS-CoV-2 infection and to evaluate seroconversion rates and quantify antibody responses to COVID-19 vaccines in two cohorts of persons living with HIV at a possible higher risk of poor outcomes (HCV coinfection and those over the age of 65 years). Methods: We included participants from two established cohorts of persons living with HIV, those who were older than 65 years of age, and those with hepatitis C (HCV) co-infection. Four hundred and seventy-one participants completed questionnaires on SARS-CoV-2 infection and COVID-19 vaccine doses and submitted peripheral blood specimens for measuring antibody levels to COVID-19 antigens, full-length spike trimer, its receptor binding domain (RBD), and nucleocapsid protein (N) at 6-month intervals up to three visits between February 2021 and December 2024. Logistic and ordinal logistic regression models evaluated predictors of seroconversion and antibody levels. Results: Overall, 51% of participants developed a SARS-CoV-2 infection, but it was mild, with only nine requiring hospital admission and no deaths. Overall, 99% of tested specimens had antibodies above threshold to either spike or RBD proteins. Specimens that did not and those with lower antibody levels had testing earlier in the pandemic, and were from participants with fewer vaccine doses, and did not have natural infection. Age, depression, comorbidity, HCV co-infection, current substance use, CD4 count, or HIV viral load were predictive of antibody level. Those with hybrid immunity had higher antibody responses. Conclusions: In cohorts of persons with HIV-HCV coinfection and those who are ageing, we observed high rates of seroconversion to COVID-19 antigens. Antibody levels were higher among those with more vaccine doses, hybrid immunity, and later in the pandemic waves. Although 51% developed a breakthrough infection, outcomes were mild with no deaths.
Full article
(This article belongs to the Special Issue Antiviral Antibody Immune Responses in the Context of Vaccination and Infection)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Development of Bispecific Antibody Targeting Human IL-17A and IL-6
by
Beata Pamuła, Martyna Banach, Marta Mikońska, Karolina Korytkowska, Krzysztof Lacek, Oliwia Śniadała, Małgorzata Marczak, Krzysztof Flis, Aleksandra Sowińska, Damian Kołakowski, Jerzy Pieczykolan, Beata Zygmunt, Maciej Wieczorek and Olga Abramczyk
Antibodies 2026, 15(2), 29; https://doi.org/10.3390/antib15020029 - 30 Mar 2026
Abstract
Background/Objectives: Antibodies are a rapidly expanding field in drug discovery, but their monospecificity limits therapeutic applications, particularly in complex inflammatory diseases. Multispecific therapeutics, which combine variable regions targeting two or more antigens, offer potential advantages such as enhanced efficacy, broader target modulation,
[...] Read more.
Background/Objectives: Antibodies are a rapidly expanding field in drug discovery, but their monospecificity limits therapeutic applications, particularly in complex inflammatory diseases. Multispecific therapeutics, which combine variable regions targeting two or more antigens, offer potential advantages such as enhanced efficacy, broader target modulation, and reduced side effects. This study aimed to identify and characterize bispecific, VHH-based antibodies simultaneously targeting IL-6 and IL-17A—two key cytokines involved in autoimmune and chronic inflammatory conditions. Methods: A phage display screening was conducted using llama-derived VHH libraries to select binders against human IL-6 and IL-17A. Binding affinities of individual VHHs and assembled bispecific constructs were assessed using Bio-Layer Interferometry (BLI). Functional activity was evaluated using reporter cell lines responsive to IL-6 and IL-17A signaling. Biophysical and quality assessments of selected VHHs and bispecific antibodies were performed using the Uncle screening platform and LabChip capillary electrophoresis. Results: Several high-affinity VHH binders were identified for both IL-6 and IL-17A, and incorporated into bispecific antibody formats. The bispecific candidates exhibited simultaneous inhibition of both cytokine pathways in functional reporter assays. Biophysical characterization confirmed good stability and purity profiles for selected molecules. Conclusions: This study demonstrates the feasibility of generating stable, functional bispecific VHH-based antibodies targeting IL-6 and IL-17A. These constructs show potential as therapeutic agents for treating autoimmune and chronic inflammatory diseases by modulating multiple signaling pathways simultaneously.
Full article
(This article belongs to the Section Antibody Discovery and Engineering)
►▼
Show Figures

Figure 1
Open AccessReview
Dynamics of 1,3-β-D-Glucan in Invasive Candidiasis: A Narrative Review of Microbiological Aspects and Diagnostic Implications
by
Maddalena Calvo, Marta Caccamo, Dalila Maria Cammarata and Laura Trovato
Antibodies 2026, 15(2), 28; https://doi.org/10.3390/antib15020028 - 27 Mar 2026
Abstract
►▼
Show Figures
Invasive candidiasis (IC) remains a significant cause of morbidity and mortality among critically ill, hematologic, and neonatal patients worldwide. Rapid and accurate diagnosis is essential to guide timely antifungal therapy and improve outcomes. Among available diagnostic tools, 1,3-β-D-glucan (BDG), a polysaccharide component of
[...] Read more.
Invasive candidiasis (IC) remains a significant cause of morbidity and mortality among critically ill, hematologic, and neonatal patients worldwide. Rapid and accurate diagnosis is essential to guide timely antifungal therapy and improve outcomes. Among available diagnostic tools, 1,3-β-D-glucan (BDG), a polysaccharide component of the fungal cell wall, has emerged as a key biomarker. BDG assays allow for early detection of probable IC, often preceding positive blood cultures, and offer prognostic information based on serial measurements. Species-specific differences in Candida cell wall composition influence BDG release and diagnostic sensitivity. Candida albicans generally correlates with high BDG levels, whereas Nakaseomyces glabrata, Candida parapsilosis, and Candida auris exhibit variable or lower glucan exposure, limiting assay sensitivity. BDG performance is affected by patient-specific factors, such as prior surgery, transfusions, or coexisting bacterial infections, which may lead to false-positive results. Molecular techniques, including PCR-based assays, provide complementary diagnostic accuracy and species identification, and their combination with BDG testing enhances sensitivity up to 90%. Serial BDG monitoring supports risk stratification and treatment response assessment, with persistent elevations predicting worse outcomes. In neonatal and pediatric populations, optimal cut-off values remain under investigation, highlighting the need for integration with clinical and microbiological data. Overall, BDG represents a valuable adjunct in a multimodal diagnostic workflow, providing both diagnostic and prognostic insights in invasive candidiasis management.
Full article

Figure 1
Open AccessArticle
The Effect of FcRn Binding on Ocular Disposition of Monoclonal Antibodies
by
Sanika Naware, Saurav Kulkarni, Sahil Salvi, Dhvani Patel and Dhaval K. Shah
Antibodies 2026, 15(2), 27; https://doi.org/10.3390/antib15020027 - 25 Mar 2026
Abstract
Background/Objectives: The neonatal Fc receptor (FcRn) plays a crucial role in extending the systemic half-life of monoclonal antibodies (mAbs), but its influence on ocular distribution remains incompletely understood. This study investigated the impact of FcRn on the ocular disposition of mAbs following
[...] Read more.
Background/Objectives: The neonatal Fc receptor (FcRn) plays a crucial role in extending the systemic half-life of monoclonal antibodies (mAbs), but its influence on ocular distribution remains incompletely understood. This study investigated the impact of FcRn on the ocular disposition of mAbs following systemic administration in rabbits. Methods: New Zealand White rabbits received a single intravenous dose (1 mg/kg) of either wild-type trastuzumab (TS-WT) or its FcRn non-binding variant (IHH). Plasma and ocular tissues (retina, iris–ciliary body, vitreous humor, aqueous humor, cornea, conjunctiva, and tears) were collected at terminal time points up to 336 h for TS-WT and 168 h for IHH. Antibody concentrations were quantified using a validated sandwich ELISA. Pharmacokinetic parameters and antibody biodistribution coefficients (ABC) were calculated to assess the FcRn-mediated effects on ocular distribution. Results: TS-WT demonstrated 2-fold higher systemic exposure compared to IHH. The iris–ciliary body exhibited the highest absolute exposure for both antibodies, with TS-WT showing significantly higher accumulation (ABC0–168h: 14.95% vs. 8.89%). Retinal distribution remained comparable between antibodies (5.96% vs. 5.51%). Both antibodies were detectable in tears, with ABC value of ~4% reported for TS-WT. TS-WT also demonstrated markedly increased distribution in vitreous humor and tear fluid (3.5- and 5.5-fold higher ABC values, respectively) compared to IHH. The cornea (5.76% vs. 5.57%) and conjunctiva (7.71% vs. 7.21%) showed comparable relative distribution between TS-WT and IHH, while aqueous humor showed minimal differences (0.44% vs. 0.52%). Conclusions: This investigation reveals distinct tissue-specific patterns of FcRn-mediated mAb distribution within the eye. FcRn binding significantly enhanced antibody distribution in ocular tissues, such as the iris–ciliary body, and tears, with less pronounced effects on the retina, cornea, conjunctiva and aqueous humor. These findings provide mechanistic insights for optimizing mAb-based therapeutics for ocular disease and understanding the ocular toxicity of mAb-based therapeutics, such as antibody–drug conjugates.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Graphical abstract
Open AccessArticle
The Multi-Attribute Method (MAM), An Advanced LC-MS Approach for Protein A Resin Performance and Lifecycle Evaluation
by
Jingming Zhang, Matthew Larsen, Timothy Blanc, Babita S. Parekh and Ming-Ching Hsieh
Antibodies 2026, 15(2), 26; https://doi.org/10.3390/antib15020026 - 23 Mar 2026
Abstract
Background: Protein A resins are indispensable for monoclonal antibody (mAb) production, yet their condition and performance are traditionally assessed using indirect or qualitative methods. In this study, the multi-attribute method (MAM), previously applied to therapeutic protein characterization, is systematically adapted for the first
[...] Read more.
Background: Protein A resins are indispensable for monoclonal antibody (mAb) production, yet their condition and performance are traditionally assessed using indirect or qualitative methods. In this study, the multi-attribute method (MAM), previously applied to therapeutic protein characterization, is systematically adapted for the first time as a unified liquid chromatography–mass spectrometry (LC-MS) platform for Protein A resin analysis. Method: Four Cytiva Protein A resins, MabSelect™, MabSelect SuRe™, MabSelect SuRe™ LX, and MabSelect™ PrismA, were evaluated by MAM for resin identity, Protein A ligand integrity, fouling by impurities, and cleaning performance. Results: MAM enables resin-specific peptide fingerprinting and quantitative monitoring of Protein A ligand post-translational modifications (PTMs), including deamidation, isomerization, and fragmentation induced by repeated clean-in-place (CIP) cycles. Comparative analysis of virgin and used resins revealed ligand degradation and fouling despite engineered alkaline stability, with MabSelect™ showing the greatest susceptibility. Importantly, residual monoclonal antibodies (mAbs) and host cell proteins (HCPs) were directly detected and quantified from the resin matrix, providing a molecular-level assessment of resin cleaning effectiveness not achievable with conventional approaches. Conclusions: This work establishes MAM as a novel, sensitive, and comprehensive strategy for Protein A resin lifecycle management, delivering actionable insight for resin selection, cleaning optimization, and downstream process development.
Full article
(This article belongs to the Section Antibody-Based Therapeutics)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Immunogenetic Architecture of Chronic Lymphocytic Leukemia at Early Stage: Insights from the O-CLL1 Cohort
by
Davide Bagnara, Andrea Nicola Mazzarello, Monica Colombo, Ennio Nano, Niccolò Cardente, Fabiana Ferrero, Nadia Bertola, Vanessa Cossu, Fabio Ghiotto, Adalberto Ibatici, Emanuele Angelucci, Antonino Neri, Massimo Gentile, Fortunato Morabito, Manlio Ferrarini, Giovanna Cutrona and Franco Fais
Antibodies 2026, 15(2), 25; https://doi.org/10.3390/antib15020025 - 18 Mar 2026
Abstract
Background/Objectives: The immunoglobulin heavy-chain variable (IGHV) gene repertoire represents a characteristic feature of chronic lymphocytic leukemia (CLL), although its configuration is not well defined at the early disease stages. The IGHV repertoire of a cohort of early CLL patients was analyzed and compared
[...] Read more.
Background/Objectives: The immunoglobulin heavy-chain variable (IGHV) gene repertoire represents a characteristic feature of chronic lymphocytic leukemia (CLL), although its configuration is not well defined at the early disease stages. The IGHV repertoire of a cohort of early CLL patients was analyzed and compared to that of a “real-world” reference cohort. Methods: Patients from the O-CLL1 observational protocol, which enrolled only Binet stage A cases within twelve months from diagnosis, were studied. IGHV/IGHJ rearrangements were sequenced and annotated following ERIC recommendations, and stereotyped subsets were assigned using ARResT/AssignSubsets. The repertoire features were compared with the dataset of a real-world cohort of patients with heterogeneous staging (CTR cohort) and with published early-diagnosis series. Results: IGHV and IGHJ gene distributions and HCDR3-length profiles in O-CLL1 closely mirrored those of CTR, indicating that the BcR IG repertoire at diagnosis is already defined rather than being selected during disease progression. Mutated IGHV (M-CLL) predominated, with a frequency of stereotyped BcR IG comparable to that of other early-diagnosis cohorts. However, within this conserved framework, subset #4 was over-represented among M-CLL from O-CLL without an increased overall IGHV4-34 gene usage, suggestive of a selective expansion rather than a recombinational bias. Subset #4 cases retained canonical HCDR3 motifs and showed time-to-first-treatment like other M-CLL, likely reflecting the younger age structure of O-CLL1. Conclusions: Early-diagnosis CLL displays a biased IGHV repertoire with stereotyped configurations characteristic of CLL, including subsets that are rare in the normal B-cell repertoire. These findings support a central role for antigen-driven selection in shaping CLL evolution.
Full article
(This article belongs to the Section Antibody-Based Diagnostics)
►▼
Show Figures

Graphical abstract
Open AccessReview
Dermatomyositis with Anti-MDA5 Autoantibodies After SARS-CoV-2 mRNA Vaccination Treated with Tofacitinib: Integrating Literature Evidence and a Novel Observation
by
Maurizio Benucci, Elisa Cioffi, Francesca Li Gobbi, Emanuele Antonio Maria Cassarà, Riccardo Terenzi, Edda Russo, Valentina Grossi, Barbara Lari, Maria Infantino and Mariangela Manfredi
Antibodies 2026, 15(2), 24; https://doi.org/10.3390/antib15020024 - 9 Mar 2026
Abstract
COVID-19 mRNA vaccines activate type I interferon pathways and in genetically or immunologically predisposed individuals may trigger autoimmune responses, including autoantibodies against melanoma differentiation-associated protein 5 (MDA5). Although cases of dermatomyositis (DM), particularly anti-MDA5-positive DM, have been increasingly reported after SARS-CoV-2 vaccination, its
[...] Read more.
COVID-19 mRNA vaccines activate type I interferon pathways and in genetically or immunologically predisposed individuals may trigger autoimmune responses, including autoantibodies against melanoma differentiation-associated protein 5 (MDA5). Although cases of dermatomyositis (DM), particularly anti-MDA5-positive DM, have been increasingly reported after SARS-CoV-2 vaccination, its clinical spectrum and management remain incompletely defined. We conducted a narrative review of the literature on post-vaccination dermatomyositis, focusing on clinical features, autoantibody profiles, therapeutic approaches, and outcomes. The review was enriched by the inclusion of a new case: a 60-year-old woman who developed anti-MDA5-positive dermatomyositis two weeks after receiving her fourth dose of the BNT162b2 (Pfizer/BioNTech) vaccine. She presented predominantly with cutaneous and articular manifestations in the absence of interstitial lung disease. Treatment with oral prednisone, intravenous alprostadil, and the Janus kinase inhibitor tofacitinib resulted in marked clinical improvement. This case, together with the literature review, illustrates both typical and atypical presentations of vaccine-associated anti-MDA5 DM, highlights diagnostic challenges without lung involvement, and suggests JAK inhibition as a potential therapeutic option, contributing to a more comprehensive understanding of post-vaccination dermatomyositis.
Full article
(This article belongs to the Section Humoral Immunity)
►▼
Show Figures

Figure 1
Open AccessArticle
Evaluation of Anti-dsDNA Antibodies in Laboratory Practice: Management of Different Analytical Methods and Correlation with HEp-2 Immunofluorescence Patterns
by
Massimo Papale, Carmela Paolillo, Loredana Iafelice, Tiziana Trivisano, Giuseppe Stefano Netti, Elena Ranieri and Gaetano Corso
Antibodies 2026, 15(2), 23; https://doi.org/10.3390/antib15020023 - 5 Mar 2026
Cited by 1
Abstract
Background: Anti-double-stranded DNA (anti-dsDNA) antibodies are a key serological marker for systemic lupus erythematosus (SLE) and are commonly assessed in conjunction with anti-nuclear antibody (ANA) testing by indirect immunofluorescence (IIF) on HEp-2 cells. However, their detection is influenced both by the heterogeneity of
[...] Read more.
Background: Anti-double-stranded DNA (anti-dsDNA) antibodies are a key serological marker for systemic lupus erythematosus (SLE) and are commonly assessed in conjunction with anti-nuclear antibody (ANA) testing by indirect immunofluorescence (IIF) on HEp-2 cells. However, their detection is influenced both by the heterogeneity of the autoimmune response and by the characteristics of the analytical method employed, thereby complicating diagnostic interpretation. Methods: In this retrospective single-center study, 3090 consecutive patients undergoing anti-dsDNA analysis were screened, and 138 positive individuals, with anti-dsDNA levels ≥ 15 IU/mL by fluoroenzyme immunoassay (FEIA), were included in the study. A control group of 29 anti-dsDNA-negative patients was also analyzed. Anti-dsDNA-positive patients were stratified by antibody level (low, mild, high), and the results were correlated with HEp-2 IIF titers and fluorescence patterns. Furthermore, in a subset of 30 positive patients, anti-dsDNA antibodies were evaluated using immunoblotting (IB) and the Crithidia luciliae indirect immunofluorescence test (CLIFT). Statistical analyses assessed associations and concordance among methods. Results: Higher anti-dsDNA levels were generally associated with higher HEp-2 IIF titers. However, a considerable percentage (35%) of patients with positive anti-dsDNA were negative by HEp-2 IIF. Notably, high anti-dsDNA levels were detected in 19% of HEp-2 IIF-negative patients (titer < 1:80), 18% of mildly HEp-2 IIF-positive patients (titer 1:80–1:160), and 25% of HEp-2 IIF-positive patients (titer > 1:320). In the subset of 30 positive patients, FEIA analysis showed high concordance with the immunoblot in both IIF-positive (81%) and -negative (100%) patients, while CLIFT demonstrated lower agreement with both FEIA and IB independently of the IIF. Conclusions: Our findings indicate that anti-dsDNA antibody detection may occur independently of HEp-2 IIF positivity and that FEIA, especially when confirmed by immunoblot, represents a reliable approach for anti-dsDNA assessment. The observed results in this study likely reflect differences in epitope recognition and assay sensitivity among methods, suggesting the use of a multi-step diagnostic strategy in the serological evaluation of SLE.
Full article
(This article belongs to the Section Antibody-Based Diagnostics)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Antibodies Home
- Aims & Scope
- Editorial Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Cancers, CIMB, Current Oncology, Sci. Pharm., Antibodies, IJMS, IJTM
Antibody-Mediated Therapy and Other Emerging Therapies in Cancer Treatment
Topic Editors: Won Sup Lee, Yaewon Yang, Seil GoDeadline: 31 July 2026

Conferences
Special Issues
Special Issue in
Antibodies
Antiphospholipid Antibodies: Beyond Biomarkers
Guest Editors: Md Asiful Islam, Przemysław KotylaDeadline: 25 May 2026
Special Issue in
Antibodies
Antibody-Mediated Rejection in Kidney Transplantation
Guest Editors: Kazuhiro Iwadoh, Hiroto EgawaDeadline: 25 June 2026
Special Issue in
Antibodies
A Festschrift Celebrating Dr. Dimiter Stanchev Dimitrov: Antibodies, Innovation, and Impact on Infectious Disease and Cancer Research
Guest Editors: Ponraj Prabakaran, Tianlei Ying, Wei LiDeadline: 30 June 2026
Special Issue in
Antibodies
Antibody and Autoantibody Specificities in Autoimmunity
Guest Editors: Loredana Frasca, E. Helen KempDeadline: 30 June 2026
Topical Collections
Topical Collection in
Antibodies
Computational Antibody and Antigen Design
Collection Editor: Buyong Ma