Abstract
In this study, sol–gel-synthesized nanoparticles were characterized by various physicochemical techniques, including scanning electron microscopy (SEM), X-ray powder diffraction (XRD), UV-Vis spectrophotometry, and thermogravimetric analysis (DTA/TG). The as-obtained powders were tested for their antimicrobial activity against the Gram-positive bacteria Staphylococcus aureus and Enterococcus faecalis, as well as the fungal strains Candida albicans and Saccharomyces cerevisiae. Additionally, the photocatalytic performance of the samples was evaluated under simulated solar light. The results are promising for possible environmental applications. The antimicrobial assessment also revealed notable effects, with varying degrees of growth inhibition observed across the tested microorganisms. The main approach in this study consists of the combination of physicochemical characterization with antibacterial and photocatalytic evaluations, resulting in promising multifunctional materials.
1. Introduction
The degradation of water quality has emerged as a serious environmental and public health concern because traditional treatment methods are often ineffective in eliminating complex contaminants. Wastewater from medical facilities and various commercial activities often contains hazardous combinations of microbial pathogens, including bacteria and fungi, as well as traces of pharmaceutical compounds. This combination of biological and chemical pollutants increases the risk of infectious diseases and contributes to the growing challenge of antimicrobial resistance [1]. These issues highlight the need for novel, multipurpose materials that can simultaneously address pharmaceutical pollutants and microbial contamination.
Photocatalysis has become a viable and sustainable method for environmental remediation, mainly in the removal of pathogenic microorganisms, pharmaceuticals, and organic pollutants from aquatic environments [2]. Semiconductor-based photocatalysts facilitate solar energy to initiate redox reactions, leading to the full removal of contaminants and mitigation of antimicrobial resistance risks.
The presence of pharmaceuticals and other organic contaminants poses a serious environmental and public health challenge, as conventional wastewater treatment processes often cannot remove these micropollutants efficiently and completely. Nanotechnology presents promising solutions for addressing this challenge. Pharmaceuticals such as β-lactam antibiotics (e.g., co-amoxiclav and amoxicillin) persist in aquatic environments and contribute to antibiotic resistance and ecosystem toxicity when insufficiently removed [3]. Conventional wastewater treatment methods, such as biological treatment, adsorption, membrane filtration, coagulation/flocculation, and advanced oxidation processes, vary in effectiveness and often require substantial infrastructure or produce secondary waste. In contrast, photocatalytic degradation has been demonstrated to be a promising alternative, utilizing semiconductor catalysts activated by light to produce reactive oxygen species capable of decomposing organic pollutants into harmless end products under ambient conditions [4]. Nanostructured photocatalysts such as TiO2, ZnO, and their heterojunctions have been extensively studied for their strong oxidative capacity, adjustable band gap properties, and stability under irradiation. For instance, TiO2/ZnO hybrid nanoparticles have achieved high removal efficiencies (~90%) in co-amoxiclav degradation, demonstrating the potential of nanophotocatalysts for treating antibiotic-contaminated wastewater [5]. Similarly, ZnO-TiO2 heterojunctions have shown high efficiency in the degradation of amoxicillin under UV–visible irradiation, with optimized conditions yielding a nearly full removal and favorable catalyst recyclability. These findings underline the efforts to enhance photocatalytic materials and processes for practical wastewater remediation [6]. Additionally, it has been reported that removal of tetracycline hydrochloride (TCH) is widely used and frequently detected in wastewater, where it contributes to the selective pressure driving resistance in bacterial species, including Staphylococcus aureus, Streptococcus pneumoniae, and Neisseria gonorrhoeae [7,8]. Sunlight-driven heterogeneous photocatalysis was used as an eco-friendly and cost-effective approach for degrading such pharmaceutical contaminants in aquatic environments.
Recent progress in the design and functionalization of nanoparticles (NPs) with combined photocatalytic and antimicrobial functions has created new opportunities for water treatment. Gram-positive pathogens such as Staphylococcus aureus and Enterococcus faecalis are clinically significant due to their involvement in soft-tissue and skin infections as well as their increasing resistance to multiple antibiotics [9,10]. Numerous studies have reported the strong antibacterial activity of metal-based nanoparticles, including ZnO, Ag, Au, and Pd, against Gram-positive bacteria, focusing on their potential as alternative antimicrobial agents [11,12].
Fungal pathogens are receiving increased attention due to their expanding role in infections. Candida albicans is one of the most common fungal species affecting humans and can cause both superficial and systemic disease [13]. Saccharomyces cerevisiae, typically considered non-pathogenic, has also been associated with various infections, particularly in immunocompromised individuals [14]. Metal-based nanoparticles such as Cu, Fe3O4/Ag, and MgO have exhibited notable antifungal activity, highlighting their potential as alternatives to conventional antimycotic agents [15,16,17].
Strontium titanate (SrTiO3) has attracted considerable interest as a photocatalyst for tetracycline degradation due to its chemical stability and favorable band structure [18,19]. However, studies examining its combined photocatalytic and antimicrobial properties, particularly when doped with transition metals such as Fe3+ remains limited. While Fe-modified SrTiO3 has shown antibacterial activity against Gram-negative bacteria [18,20], its efficacy against Gram-positive pathogens and clinically relevant fungi is still insufficiently explored. Jabbar et al. [21] have reported the potential of Fe incorporation in SrTiO3 for environmental applications for pollutant degradation and antimicrobial effect.
To the best of our knowledge, no prior studies have evaluated the activity of Fe-modified SrTiO3 against these specific bacterial strains, providing new insight into its potential as a multifunctional material for environmental and biomedical applications. Moreover, this work extends our previous findings on the antibacterial performance of 15% Fe-modified SrTiO3 against Escherichia coli and Pseudomonas aeruginosa [18], thereby broadening the understanding of its antimicrobial spectrum.
In this study, Fe3+-modified SrTiO3 nanoparticles were synthesized via the sol–gel method, and their sunlight-driven photocatalytic degradation of tetracycline was evaluated. In addition, the antimicrobial activity of Fe-modified SrTiO3 was investigated for the first time against the Gram-positive pathogens Staphylococcus aureus and Enterococcus faecalis, as well as the fungal species Candida albicans and Saccharomyces cerevisiae.
2. Materials and Methods
2.1. Materials
The following analytically pure chemicals were used in this study: C6H8O7·H2O (Merck & Co., Inc., Rahway, NJ, USA); C2H5OH (96%) (Sigma-Aldrich); NH3 (Sigma-Aldrich); Fe(NO3)3·9H2O (p.a., Sigma-Aldrich, Milwaukee, WI, USA); Sr(NO3)2·4H2O (p.a., Sigma Aldrich); and Ti(OBu)4 (reagent grade, 97%, Sigma-Aldrich, Milwaukee, WI, USA). The pharmaceutical compound tetracycline hydrochloride (Fisher BioReagents, Waltham, MA, USA) was employed as a model pollutant.
2.2. Sol–Gel Synthesis
In this study, pure SrTiO3 (FTO-0) and 15 mol% Fe-modified SrTiO3 (FTO-15) were synthesized using the sol–gel technique (Figure 1a,b). The synthesis procedure followed a previously established methodology [18]. Briefly, titanium butoxide was combined with ethanol and citric acid under vigorous stirring, while separate ethanolic solutions of Sr(NO3)2 and Fe(NO3)3 were prepared. These solutions were subsequently added dropwise to the titanium precursor mixture, after which the pH was adjusted to 5 using ammonia. Gelation occurred at room temperature, and the resulting gel was calcined at 1100 °C to achieve phase formation.

Figure 1.
Images of the gel samples of STO-0 (a) and FTO-15 (b).
2.3. Sample Characterization
Powder X-ray diffraction (XRD) patterns were recorded at room temperature using a Bruker D8 Advance diffractometer (Bruker, Berlin, Germany) equipped with Cu Kα radiation (λ = 1.54056 Å) and a LynxEye position-sensitive detector. The X-ray tube was operated at 40 kV and 40 mA. Data were collected over a 2θ range of 5.3–80° with a step size of 0.02°.
The morphology of both gels and the calcined samples was examined using a scanning electron microscope (SEM) JSM-5510 (JEOL, Tokyo, Japan) operated at an accelerating voltage of 10 kV. Prior to imaging, the samples were coated with a thin gold layer using a JFC-1200 fine coater (JEOL, Tokyo, Japan).
Thermal stability of the SrTiO3 (STO-0) and Fe-doped SrTiO3 (FTO-15) gels was assessed via differential thermal analysis (DTA) using a LABSYS™ EVO instrument (Setaram, Lyon, France). Measurements were performed in air with a heating rate of 10 K/min, employing a Pt-Pt/Rh thermocouple and Al2O3 as a reference. The temperature accuracy was ±5 °C, and the maximum temperature was set to 850 °C.
The optical absorption spectra of the powdered samples were recorded using a UV-Vis diffuse reflectance spectrophotometer (Evolution 300, Thermo Electron Corporation, Madison, WI, USA) in the wavelength range of 200–600 nm, using magnesium oxide as a reflectance standard. The band gap energies were determined using the Kubelka-Munk function [15] in combination with Tauc plots, based on the equation:
where F(R∞) is the Kubelka-Munk function, A is a constant, Eg is the band gap of the semiconductor, and n depends on the transition type. The band gap was obtained by extrapolating the linear portion of the Tauc plot to the x-axis, where it intersects the photon energy axis.
(ℎνF(R∞))n = A(ℎν − Eg),
A Varian Cary Eclipse Fluorescence UV-Vis spectrophotometer (Agilent Technologies, Santa Clara, CA, USA) with an excitation wavelength of 325 nm was used to detect the samples’ room temperature photoluminescence (PL).
2.4. Antimicrobial Testing
To evaluate the antimicrobial properties of the samples, two bacterial strains—Staphylococcus aureus ATCC 25923 and Enterococcus faecalis (ATCC 29212 (American Type Culture Collection, Manassas, VA, USA))—as well as two fungal strains—Candida albicans ATCC 10231 (American Type Culture Collection, Manassas, VA, USA) and Saccharomyces cerevisiae CCY 21-6-3- (Czech Collection of Yeasts, Institute of Chemistry, Slovak Academy of Sciences, Bratislava, Slovakia) were selected. The bacterial strains are medically significant representatives of Gram-positive pathogens, known for their involvement in hospital-acquired infections and varying resistance mechanisms. The fungal strains were included to assess the broader antimicrobial potential of the materials, as they represent common opportunistic yeasts of clinical relevance.
The antimicrobial activity of the nanopowders was assessed using both qualitative and quantitative methods. A spot test assay was performed at two-time intervals—after 3 h and 24 h of exposure—in order to evaluate both the immediate and prolonged effects of the materials on microbial growth.
For quantitative evaluation, the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) were determined using the broth microdilution method [22]. Microbial suspensions were adjusted to a turbidity equivalent to 0.5 on the McFarland scale, which was used as the final inoculum concentration in the microdilution wells, corresponding to approximately 1.5 × 108 CFU/mL for bacteria.
Mueller-Hinton broth was used for bacterial strains, while Sabouraud dextrose broth was used for fungal strains. The nanopowders were dispersed in the same media using an ultrasonic bath (Sonorex Bandelin, Berlin, Germany) at 35 kHz for 60 min at room temperature. Ultrasonication was applied to ensure homogeneous dispersion and to minimize potential microbial contamination. Serial twofold dilutions of the nanopowder dispersions were prepared to obtain the desired concentration range.
The assay was conducted in sterile 96-well microtiter plates, each well containing 100 µL of microbial suspension and 100 µL of nanopowder dispersion at various concentrations. Growth control wells containing microbial suspension without nanopowders and sterility control wells containing only culture medium were included. The plates were incubated under aerobic conditions at 35 ± 2 °C for 18–24 h. All MIC and MBC measurements were obtained from three parallel replicates, and the reported values represent the average of these replicates.
Post-incubation, the antimicrobial effect was quantified by performing tenfold serial dilutions of the treated cultures, followed by plating on Mueller-Hinton agar for bacterial strains and Sabouraud dextrose agar for fungal strains. Colony-forming units per milliliter (CFU/mL) were determined using the formula:
The MBC was defined as the lowest concentration of nanopowder that caused a ≥99.9% reduction in viable CFU/mL compared to the initial inoculum, indicating a bactericidal or fungicidal effect. The MIC was defined as the lowest concentration at which no visible microbial growth was observed, reflecting inhibition of microbial proliferation.
2.5. Photocatalytic Testing
The photocatalytic performance of the synthesized powdered catalysts was evaluated through the degradation of Tetracycline hydrochloride (TCH) under simulated sunlight irradiation (OSRAM Ultra-Vitalux 300 W, 230 V, e27), providing broad-spectrum irradiation including UV-A and visible light. A model solution of TCH (10 ppm, 200 mL) was prepared using ultra-pure water (18.2 MΩ·cm). The photocatalytic reaction was carried out in a batch-type slurry photoreactor (Lenz Laborglas, KL-100, Solana Beach, CA, USA), equipped with a circulating water bath (ArgoLab CB5-10, Modena, Italy) to maintain a constant reaction temperature of 25 ± 0.1 °C. Due to the high intensity of the 300 W lamp, the temperature was additionally monitored using a thermocouple to ensure thermal stability throughout the experiment. The depth of the slurry layer was adjusted to ~2 cm, and the light source was positioned 20 cm above the liquid surface.
Prior to illumination, 0.1 g of the photocatalyst was added to the solution, and the suspension was stirred magnetically for 30 min in the dark to reach adsorption–desorption equilibrium. Photodegradation was then initiated by switching on the light source, and the reaction was allowed to proceed for 60 min under continuous stirring. At regular intervals of 10 min, 3 mL aliquots were withdrawn, filtered through a 0.22 μm membrane syringe filter to remove the catalyst, and analyzed using a UV-Vis spectrophotometer (Thermo Scientific Evolution 300, Madison, WI, USA) by monitoring the absorbance at 355 nm, characteristic of TCH.
Photocatalytic activity was assessed by plotting C/C0 versus time, where C0 represents the TCH concentration at the end of the dark period. The reaction kinetics were analyzed using the Langmuir-Hinshelwood model, assuming pseudo-first-order kinetics (−ln(C/C0) vs. time), and the corresponding half-life (t1/2) values were calculated using the equation:
Additionally, the adsorption efficiency (AE) during the dark period was calculated using the following formula:
where C00 is the initial TCH concentration before the dark period, and C0 is the concentration after equilibrium was established.
3. Results & Discussion
3.1. XRD and DTA-TG of the Samples
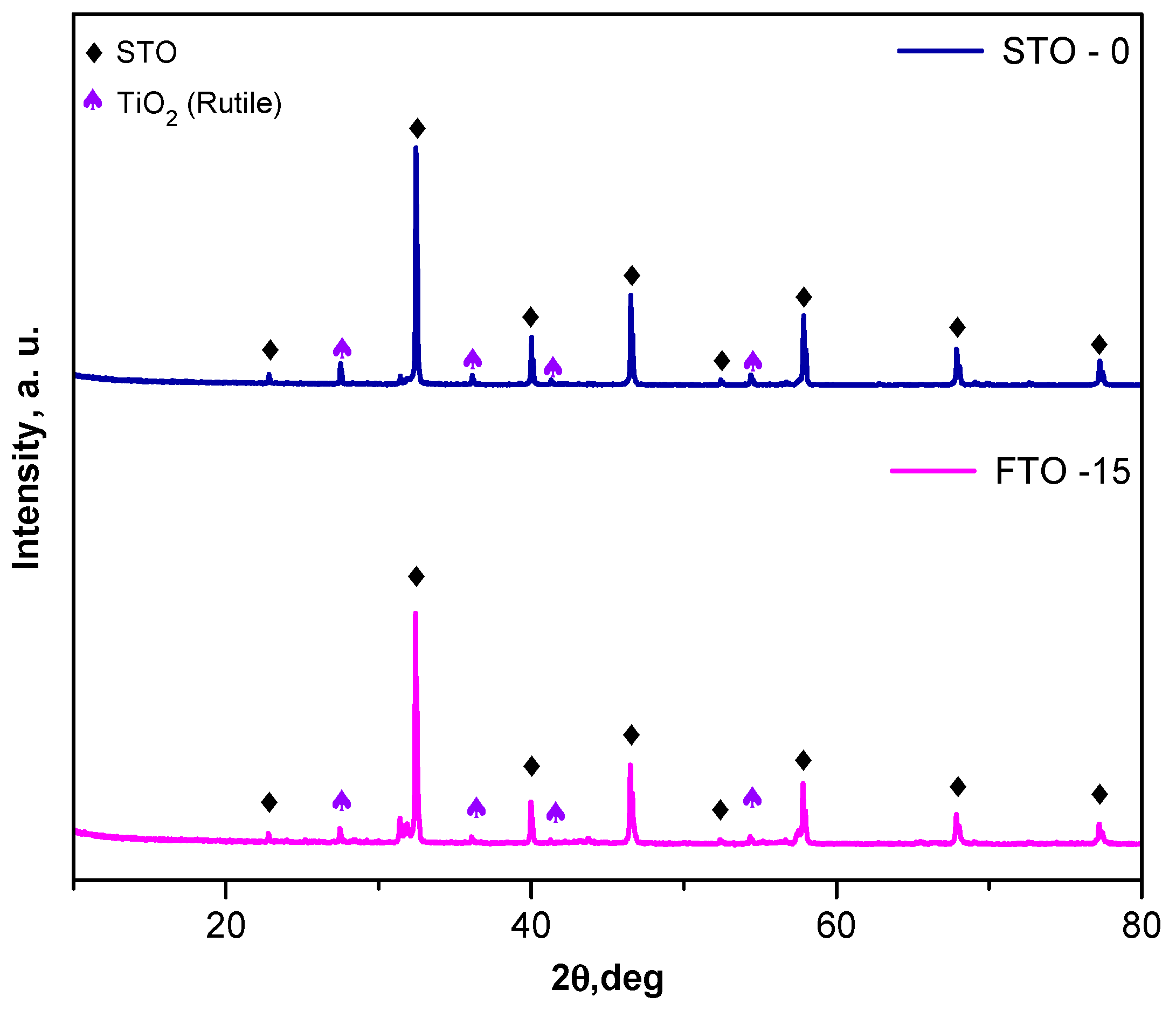
The crystal structures of the synthesized SrTiO3 (STO-0) and Fe-doped SrTiO3 (FTO-15) powders were examined by powder X-ray diffraction (XRD), and the corresponding diffractograms are presented in Figure 2. The diffraction peaks observed for both samples are consistent with the characteristic reflections of the cubic perovskite structure of SrTiO3 (ICDD 120 900-6864), confirming the successful formation of the target phase. The most prominent reflections appear at 2θ values of approximately 22.8°, 32.3°, 39.9°, 46.5°, 52.4°, 57.8°, 67.9°, and 76.8°, corresponding to the (100), (110), (111), (200), (210), (211), (220), and (310) planes, respectively.
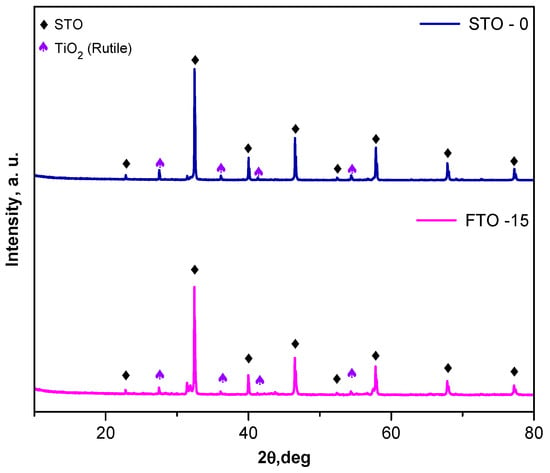
Figure 2.
XRD patterns of STO-0 and FTO-15.
For the Fe-doped sample (FTO-15), all principal diffraction peaks remain indexed to the perovskite STO-0 structure, confirming that the incorporation of Fe3+ ions into the lattice does not result in the emergence of any significant secondary phases. Minor additional peaks are present, particularly those marked with purple triangles, which correspond to the rutile phase of TiO2 (JCCDS 01-089-4920).
No diffraction peaks corresponding to iron oxides (e.g., Fe2O3 or Fe3O4) were observed, suggesting that Fe is successfully incorporated into the perovskite lattice. The retention of the cubic phase and the absence of significant peak broadening indicate good crystallinity of both STO-0 and FTO-15 powders. The average crystallite size of the samples was estimated using the Scherrer equation [23], based on the full width at half maximum (FWHM) of the (110) diffraction peak. The undoped SrTiO3 (STO-0) sample exhibited a crystallite size of approximately 30 nm, while the Fe-doped SrTiO3 (FTO-15) sample showed a reduced crystallite size of 20 nm. The decrease in crystallite size upon the introduction of iron may be attributed to the incorporation of Fe3+ ions into the Ti4+ lattice sites, which can introduce lattice strain and probably inhibit grain growth during calcination [24,25].
Overall, the XRD analysis confirms the formation of single-phase perovskite SrTiO3 for the undoped sample and the preservation of the perovskite structure upon Fe doping.
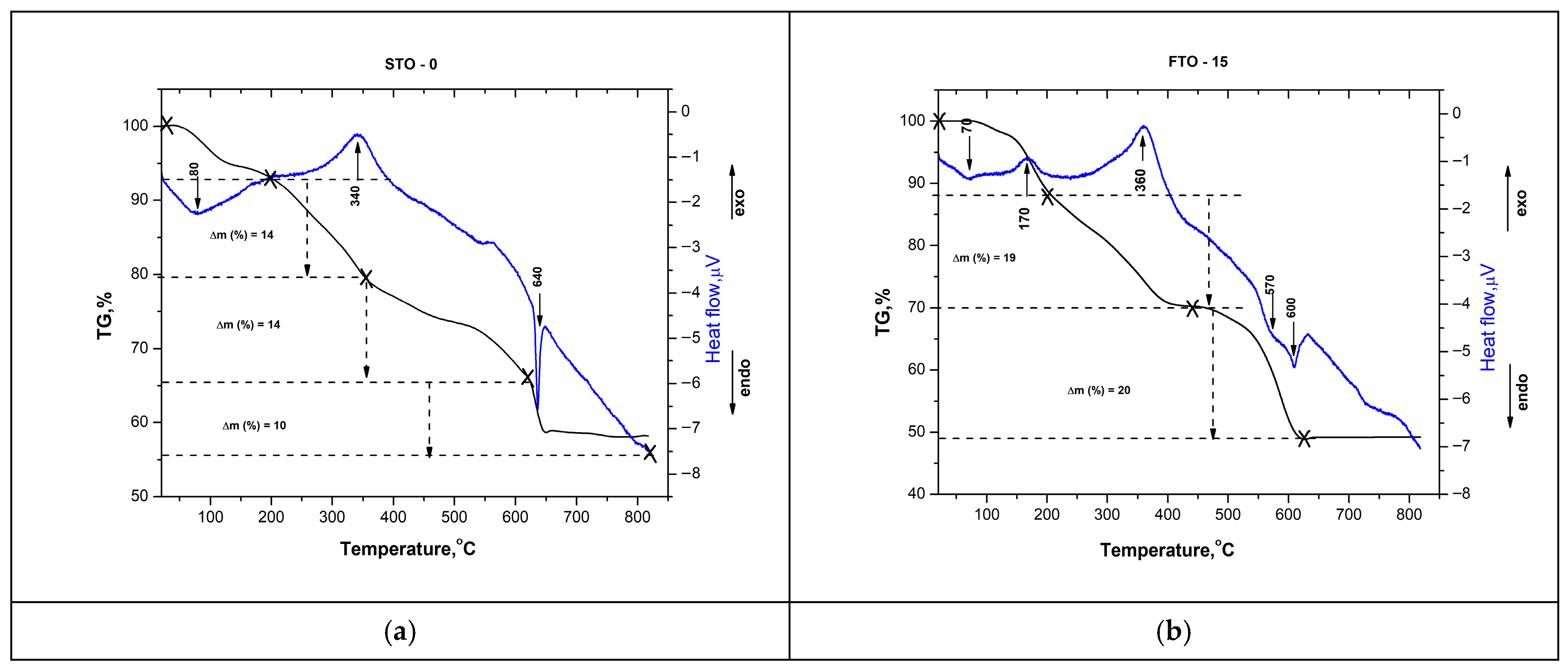
The thermal behavior of the as-prepared SrTiO3 and Fe-doped SrTiO3 (SrFe0.15Ti0.85O3) gels was investigated using differential thermal analysis (DTA) with thermogravimetric analysis (TG) (Figure 3a,b). The TG curve of the undoped SrTiO3 sample reveals several distinct mass loss steps, totaling approximately 50% in both samples. As shown in the figure, the first peak in the DTA curves of both samples is endothermic, centered at approximately 70–80 °C, and associated with a weight loss of about 15%. The observed effect could be associated with the evaporation of water molecules physically adsorbed onto the sample. A broad exothermic effect is observed near 170 °C, more pronounced in the FTO-15 sample (Figure 3b), and may be attributed to the onset of organic decomposition. This process is accompanied by an approximate 10% weight loss in both samples.
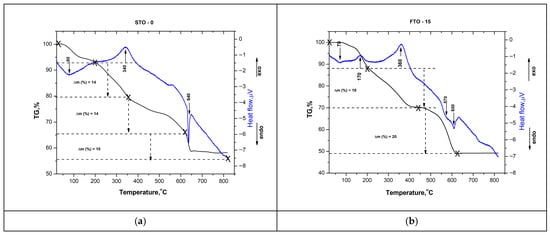
Figure 3.
DTA-TG curves of STO-0 (a) and FTO-15 (b).
The next step involves a more pronounced weight loss (~20%) occurring between 200 and 450 °C. It is characterized by a broad exothermic peak in the DTA curve, centered at approximately 340–360 °C. This stage is attributed to the stronger decomposition of residual organic compounds from the sol–gel process [26,27], accompanied by a mass loss of 14% for STO-0 and 19% for FTO-15. The final stage involves a weight loss of ~10% for STO-0 and ~20% for FTO-15 between 450 and 600 °C. A pronounced endothermic peak at approximately 600 °C signifies the crystallization of the SrTiO3 perovskite phase, consistent with previous reports of perovskite formation in the 600–700 °C range [28]. The Fe-doped sample exhibits comparable thermal behavior, though with a slightly higher total weight loss of ~51%.
3.2. SEM—Morphology of the Samples
The surface morphology of the calcined SrTiO3 (STO-0) and Fe-doped SrTiO3 (SrFe0.15Ti0.85O3, FTO-15) samples was investigated via scanning electron microscopy (SEM), and representative images at 1000× magnification are shown in Figure 4a,b. Both samples exhibit a strong tendency toward particle agglomeration, which is typical for materials synthesized by sol–gel methods due to the presence of residual hydroxyl groups and the capillary forces acting during the drying and calcination processes [29].

Figure 4.
SEM images of STO-0 (a) and FTO-15 (b).
The undoped SrTiO3 specimen (Figure 4a) exhibits compact aggregates consisting of irregularly shaped grains. The perovskite crystallites show partial sintering, with several domains exceeding 1 μm in diameter. Although larger crystals are present, the material maintains a porous and rough microstructure, potentially advantageous for surface-mediated processes such as photocatalysis.
In contrast, the Fe-doped SrTiO3 sample (Figure 4b) shows a more fragmented and disordered morphology. The particles are less compact, with enhanced surface irregularities and sharper edges compared to the undoped STO-0. This is consistent with previous reports indicating that Fe doping significantly influences the morphology of SrTiO3 by introducing lattice distortions and oxygen vacancies due to the substitution of Ti4+ with Fe3+ at the B-site [30].
3.3. UV-Vis Spectroscopy
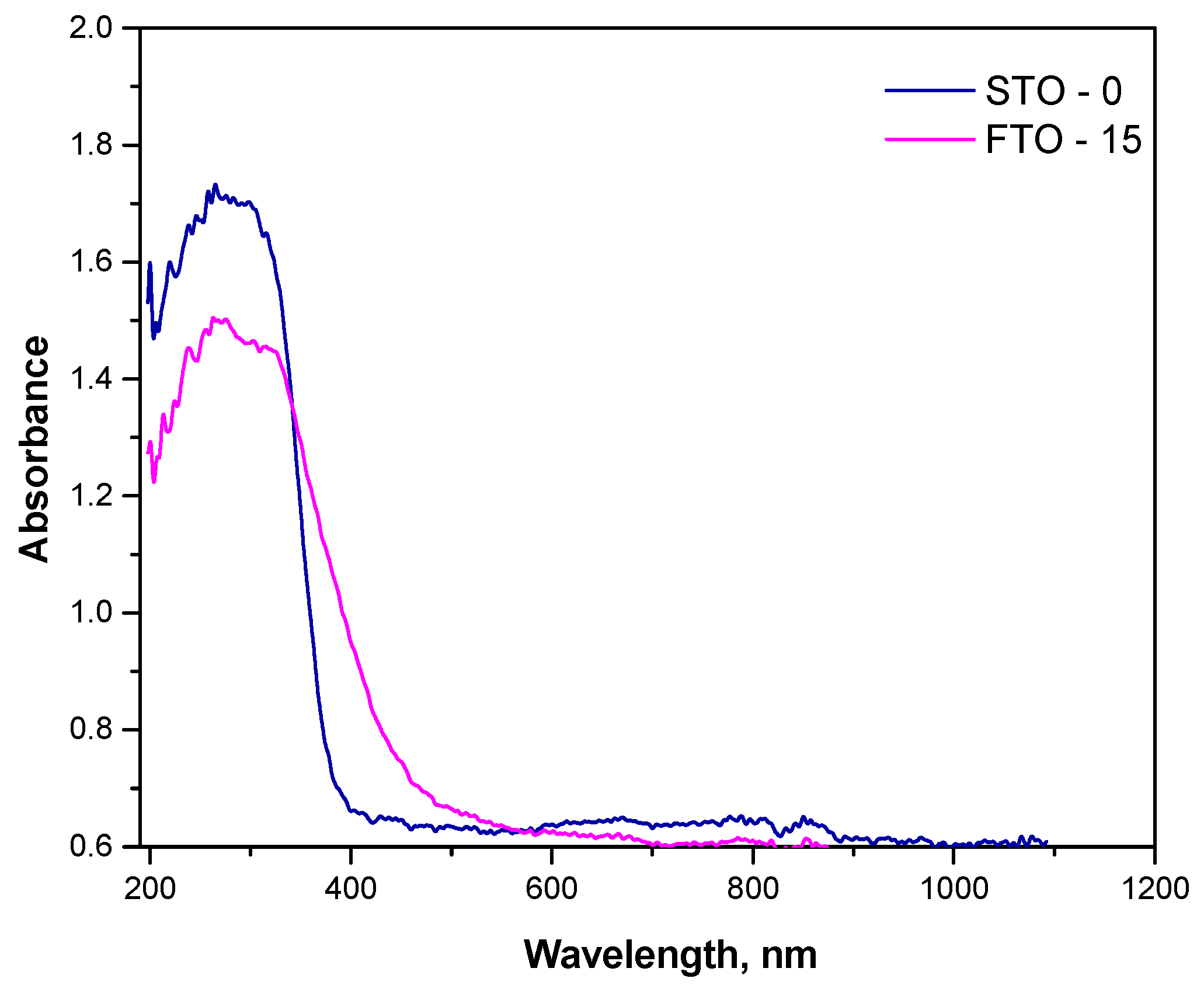
The optical absorption behavior of the synthesized SrTiO3 (STO-0) and Fe-doped SrTiO3 (FTO-15) powders was investigated using UV-Vis diffuse reflectance spectroscopy, and the spectra are presented in Figure 5. The corresponding plots used to determine the optical band gaps via the Kubelka-Munk function have been presented in our previous work [18]. Both samples exhibit strong absorption in the UV region, which is characteristic of perovskite-type titanates. However, significant differences in the absorption edges and intensities were observed between the two samples.
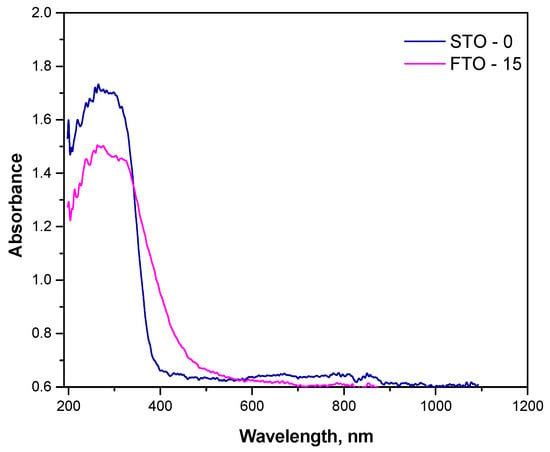
Figure 5.
Absorbance spectra of STO and FTO-15.
The undoped STO-0 sample shows a sharp absorption edge near 380 nm, consistent with its wide band gap energy of 3.28 eV. In contrast, the FTO-15 sample demonstrates a redshift of the absorption edge toward the visible region, extending up to ~450 nm. This shift indicates a narrowing of the band gap, calculated to be approximately 2.86 eV. The observed redshift and enhanced absorption in the visible region are attributed to the successful incorporation of Fe3+ ions into the Ti4+ sites of the perovskite lattice.
The reduction in band gap energy upon Fe substitution is beneficial for photocatalytic applications under visible light, as it allows more efficient utilization of the solar spectrum. However, excessive defect states may also lead to charge recombination if not properly balanced, which should be considered when correlating optical properties with photocatalytic performance [31,32].
These findings confirm that Fe doping effectively modifies the electronic structure of SrTiO3, improving its optical response in the visible range and supporting its theoretical potential as a visible-light-active photocatalyst.
3.4. Photoluminescence Spectroscopy
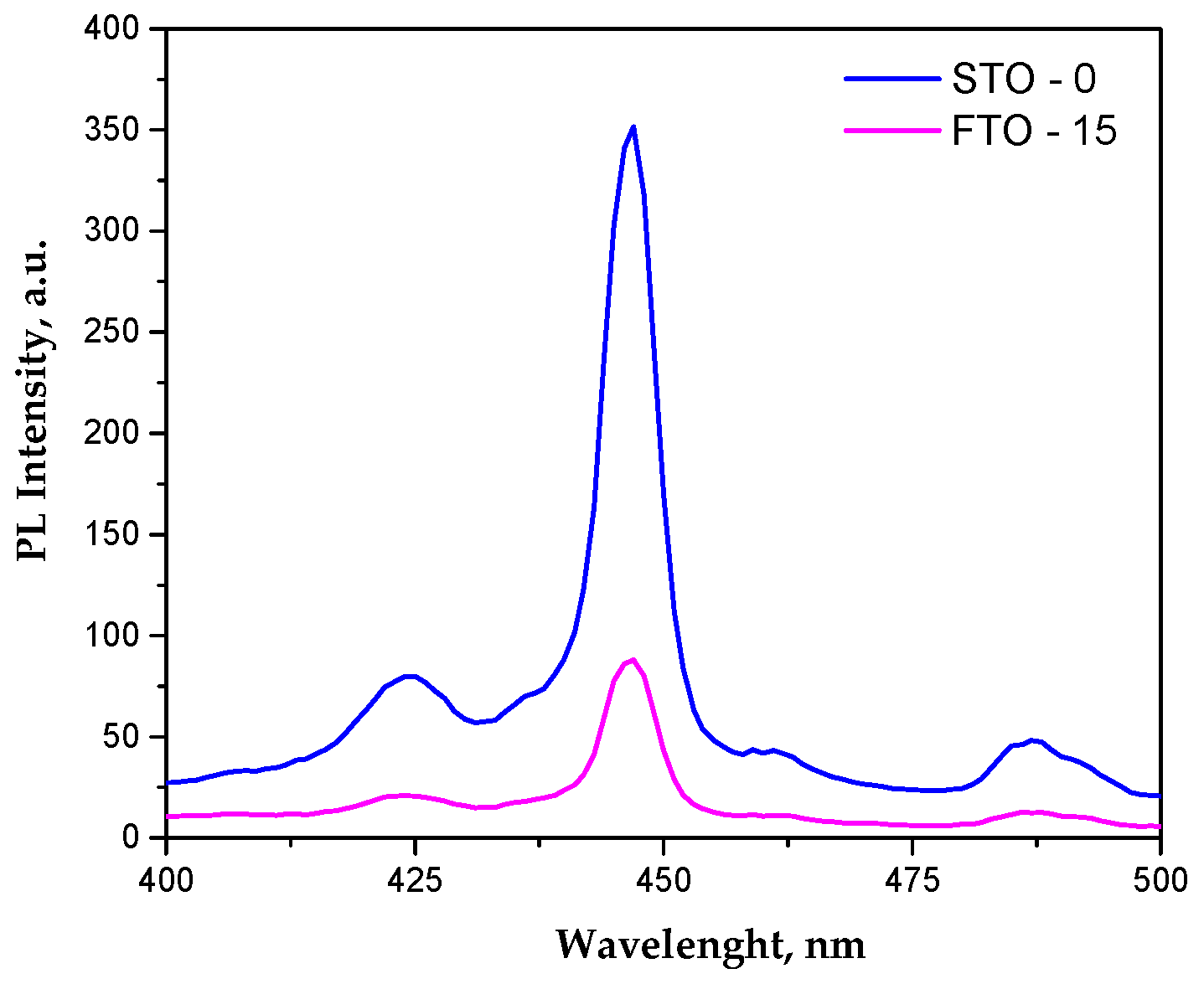
The undoped SrTiO3 sample exhibits a dominant emission band at ~445–450 nm (Figure 6), commonly associated with band-to-band or shallow defect-assisted recombination involving Ti3+-oxygen vacancy states [33], along with weaker emissions in the 420–430 nm and 480–490 nm regions attributed to deeper defect levels [34]. Upon Fe incorporation, the same emission features are preserved, while a pronounced decrease in PL intensity is observed without any significant peak shift, indicating that Fe doping does not alter the fundamental electronic transitions of SrTiO3 but strongly influences charge carrier recombination dynamics. The suppressed PL intensity for FTO-15 suggests the formation of Fe-related trap states and oxygen vacancies that favor non-radiative recombination pathways. Although reduced PL intensity is often linked to enhanced charge separation, an excessive density of defect states may act as recombination centers and limit charge carrier transport to the catalyst surface [35]. As a result, under simulated sunlight irradiation, the Fe-doped sample exhibits slightly lower photocatalytic efficiency than pristine SrTiO3, indicating that non-radiative losses outweigh the benefits of enhanced visible-light absorption.
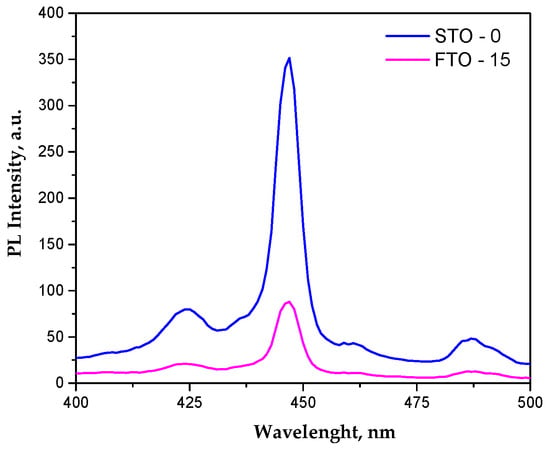
Figure 6.
Photoluminescence spectra of STO and FTO-15.
3.5. Antimicrobial Properties
Experiments were conducted to assess the efficacy of the STO-0 and FTO-15 samples’ antibacterial and antifungal activity. Suspensions containing different concentrations up to 100 mg/mL were created. Prior to additional testing, each suspension underwent ultrasonic treatment to guarantee appropriate dispersion and sample homogeneity. Both spot test and microdilution methods were employed; however, only the results from the latter, derived from the collected data, are presented here.
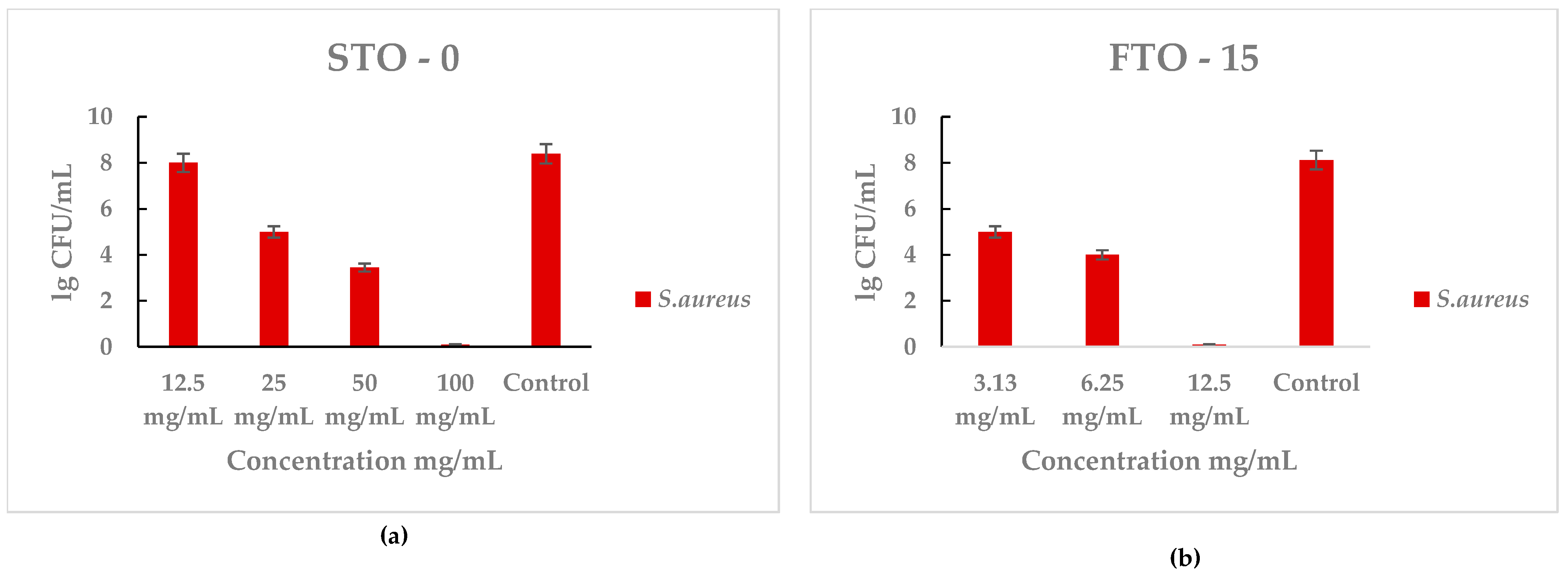
The antibacterial activity of STO-0 and FTO-15 against Staphylococcus aureus was evaluated by quantifying the viable bacterial counts (lg CFU mL−1) after incubation with different material concentrations (Figure 7a,b). A clear concentration-dependent reduction in bacterial viability was observed for both materials. All MIC and MBC values are summarized in Table 1.
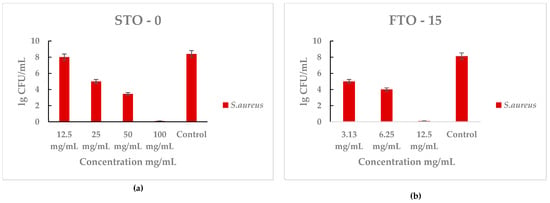
Figure 7.
Antimicrobial activity of STO-0 and FTO-15 materials against Staphylococcus aureus: (a) viable bacterial counts (lg 10 CFU mL−1) of S. aureus after exposure to STO-0 at different concentrations (12.5–100 mg mL−1), (b) viable bacterial counts (lg 10 CFU mL−1) of S. aureus after exposure to FTO-15 at different concentrations (3.13–12.5 mg mL−1).

Table 1.
Minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) values of the investigated perovskite materials against the tested bacterial and fungal strains, determined by the broth microdilution method. Lower MIC and MBC values indicate higher antimicrobial activity. MIC and MBC values reported as “X” indicate that no growth inhibition or bactericidal effect was observed at the highest tested concentration (100 mg/mL).
For STO-0 (Figure 7a), bacterial counts decreased markedly as the concentration increased from 12.5 mg/mL to 100 mg/mL, reaching bactericidal effect at the highest concentration. In contrast, untreated control samples showed high bacterial growth (~8 lg CFU mL−1). Similarly, FTO-15 (Figure 6) demonstrated strong antibacterial performance, with a significant decline in S. aureus viability even at lower concentrations (3.13–12.5 mg mL−1), achieving complete loss of cell viability at 12.5 mg mL−1.
These results indicate that both materials exhibit potent antibacterial effects against S. aureus, with FTO-15 showing higher efficacy at lower concentrations compared with STO-0.
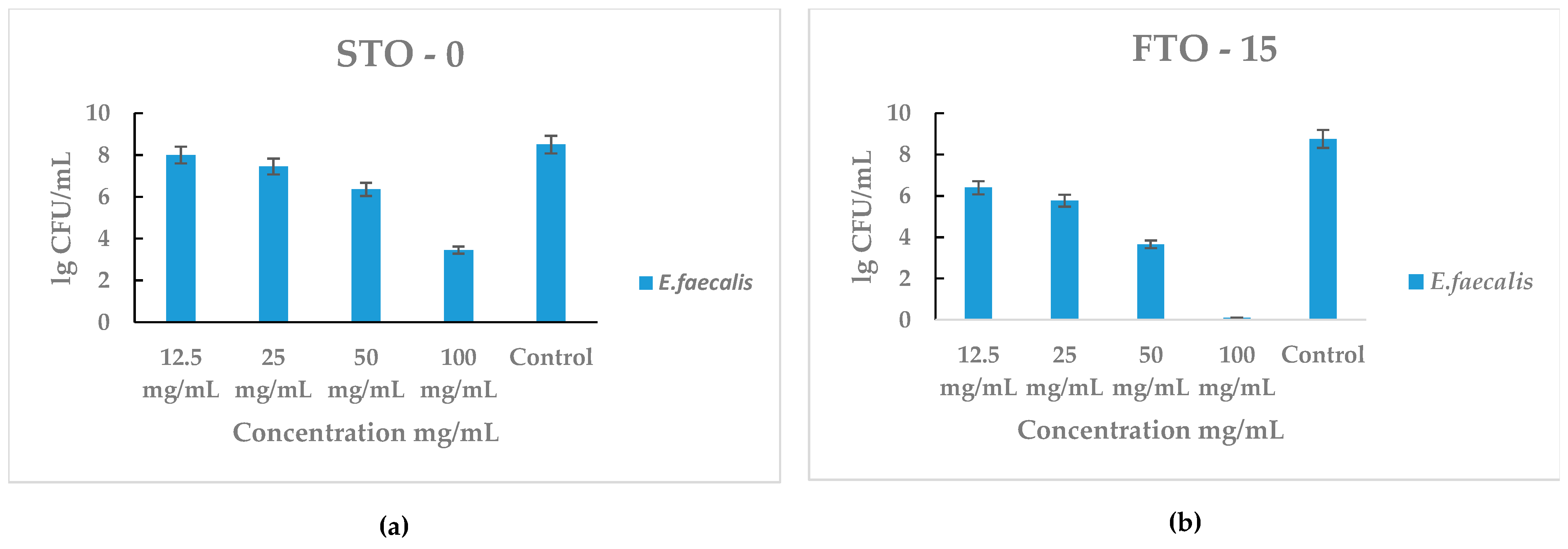
The antibacterial performance of STO-0 and FTO-15 against Enterococcus faecalis is presented in Figure 8a,b. Both materials exhibited a clear concentration-dependent antibacterial effect, with bacterial viability decreasing as the material concentration increased.
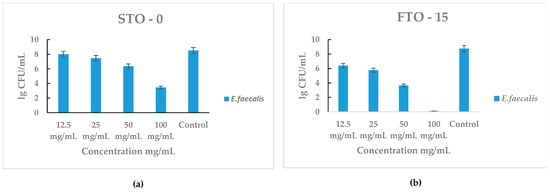
Figure 8.
Antimicrobial activity of STO-0 and FTO-15 materials against Enterococcus faecalis: (a) viable bacterial counts (lg CFU mL−1) of E. faecalis after exposure to STO-0 at various concentrations (12.5–100 mg mL−1), (b) viable bacterial counts (lg CFU mL−1) of E. faecalis after exposure to FTO-15 at concentrations ranging from 12.5 to 100 mg mL−1.
For STO-0 (Figure 8a), moderate inhibition of E. faecalis was observed at concentrations ≥ 50 mg mL−1, with a substantial reduction at 100 mg mL−1 compared with the control (~8 lg CFU mL−1). In contrast, FTO-15 (Figure 8b) demonstrated stronger antibacterial activity, showing a pronounced decline in bacterial counts even at lower concentrations. Complete loss of cell viability of E. faecalis was achieved at 100 mg mL−1, which corresponds to the minimum bactericidal concentration (MBC) for this material.
These findings indicate that FTO-15 possesses higher antibacterial efficacy than STO-0, consistent with the results obtained against S. aureus.
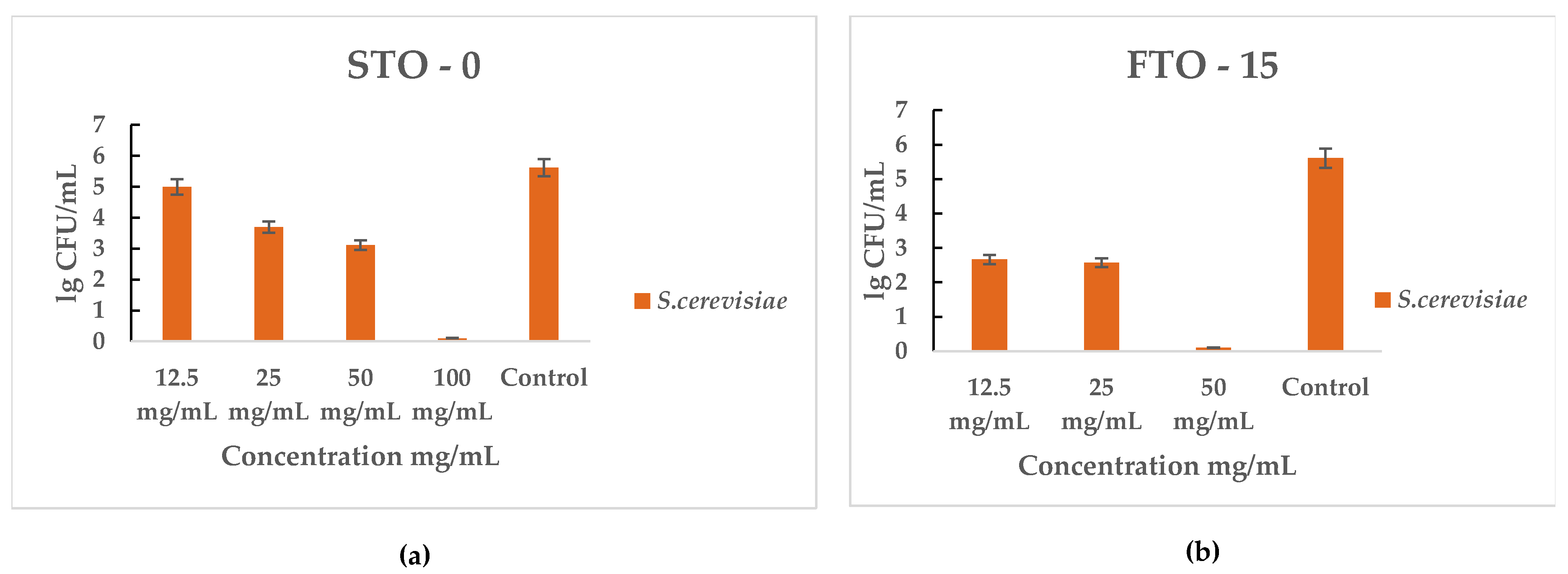
The antifungal activity of STO-0 and FTO-15 against Saccharomyces cerevisiae is illustrated in Figure 9a,b. Both materials exhibited a strong inhibitory effect on yeast growth in a concentration-dependent manner.
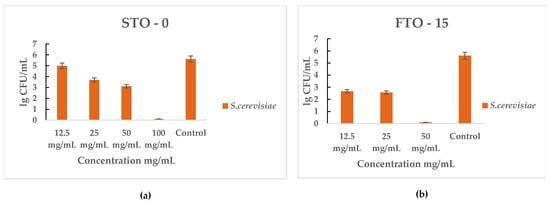
Figure 9.
Antimicrobial activity of STO-0 and FTO-15 materials against Saccharomyces cerevisiae: (a) viable cell counts (lg CFU mL−1) of S. cerevisiae after exposure to STO-0 at concentrations of 12.5–100 mg mL−1, (b) viable cell counts (lg CFU mL−1) of S. cerevisiae after exposure to FTO-15 at concentrations of 12.5–50 mg mL−1.
For STO-0 (Figure 9a), S. cerevisiae viability progressively decreased with increasing concentration, resulting in complete cell death observed at 100 mg mL−1. In the case of FTO-15 (Figure 9b), a pronounced reduction in viable cells was evident even at 25 mg mL−1, and complete eradication was achieved at 50 mg mL−1. Therefore, the minimum bactericidal concentration (MBC) for FTO-15 against S. cerevisiae was determined to be 50 mg mL−1, whereas for STO-0 it was 100 mg mL−1.
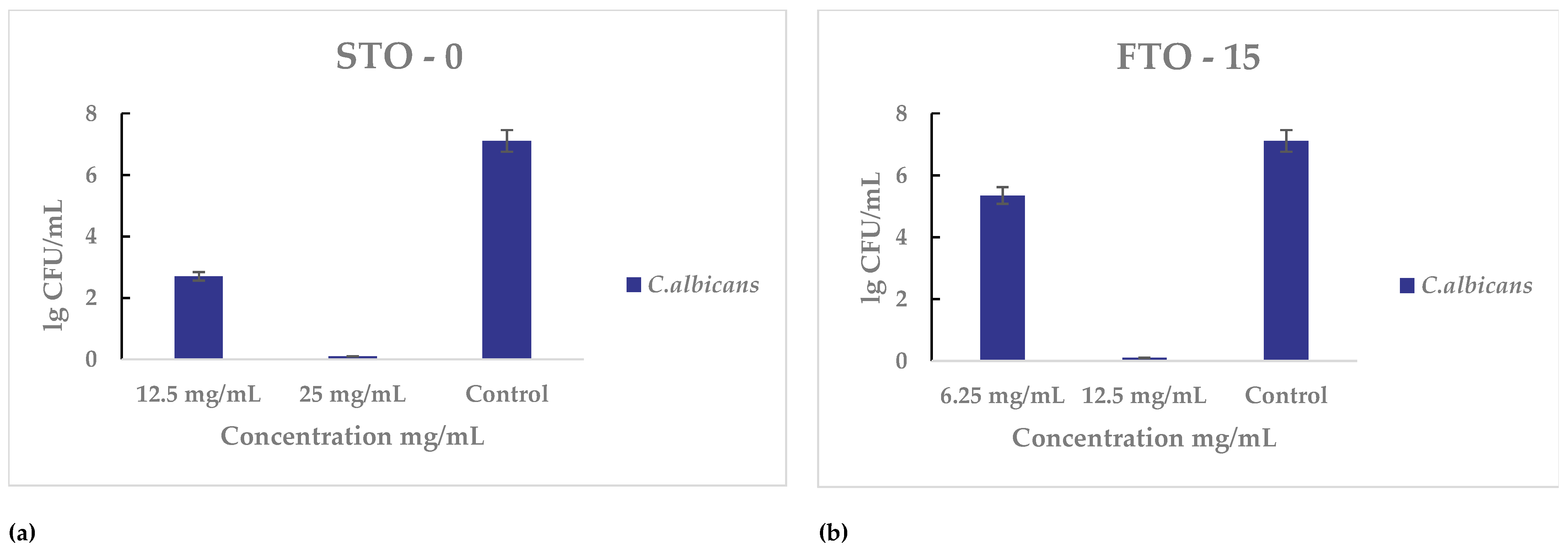
Both materials were also tested against C. albicans for their antifungal activity. For STO-0 (Figure 10a), inhibition was first noted at 12.5 mg mL−1, and complete cell death occurred at 25 mg mL−1. For FTO-15 partial growth inhibition was observed at 6.25 mg mL−1, and the minimum bactericidal concentration was determined to be 12.5 mg mL−1.
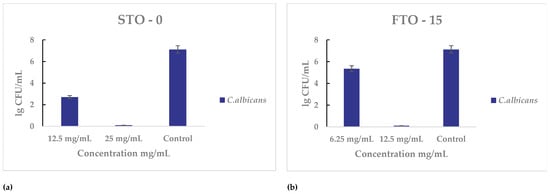
Figure 10.
Antimicrobial activity of STO-0 and FTO-15 materials against Candida albicans: (a) Viable cell counts (lg CFU mL−1) of C. albicans after exposure to STO-0 at concentrations of 12.5–25 mg mL−1, (b) Viable cell counts (lg CFU mL−1) of C. albicans after exposure to FTO-15 at concentrations of 6.25–12.5 mg mL−1.
The antimicrobial evaluation demonstrated that Fe3+-modified SrTiO3 (FTO) exhibited markedly enhanced activity compared to unmodified STO. Against Gram-positive bacteria, Staphylococcus aureus showed a reduction in the minimum bactericidal concentration (MBC) from 100 mg mL−1 for STO-0 to 12.5 mg mL−1 for FTO-15, while Enterococcus faecalis, which was only inhibited by STO-0 at 100 mg mL−1, was completely eradicated at the same concentration with FTO-15. Similarly, the antifungal assessment revealed that FTO-15 was more effective than STO-0: the MBC for Candida albicans decreased from 25 mg mL−1 to 12.5 mg mL−1, and for Saccharomyces cerevisiae, from 100 mg mL−1 to 50 mg mL−1. Overall, these results indicate that Fe3+ modification significantly enhances both antibacterial and antifungal efficacy, highlighting FTO-15 as a promising multifunctional material for biomedical and environmental applications (Table 1).
3.6. Photocatalytic Properties
The photocatalytic efficiency of the synthesized SrTiO3 (STO-0) and Fe-doped SrTiO3 (FTO-15) powders was evaluated for the degradation of tetracycline hydrochloride (TCH) under simulated solar irradiation. Prior to illumination, all suspensions underwent a 30 min dark equilibration period to establish adsorption–desorption equilibrium. The adsorption efficiencies were calculated to be around 22% for both samples, indicating a moderate affinity of both materials toward TCH molecules in the absence of light.
A control experiment for direct photolysis (TCH solution exposed to light without a catalyst) revealed a degradation efficiency of only 8% after 60 min of illumination, confirming that the removal of TCH in the experimental conditions is predominantly due to photocatalytic processes.
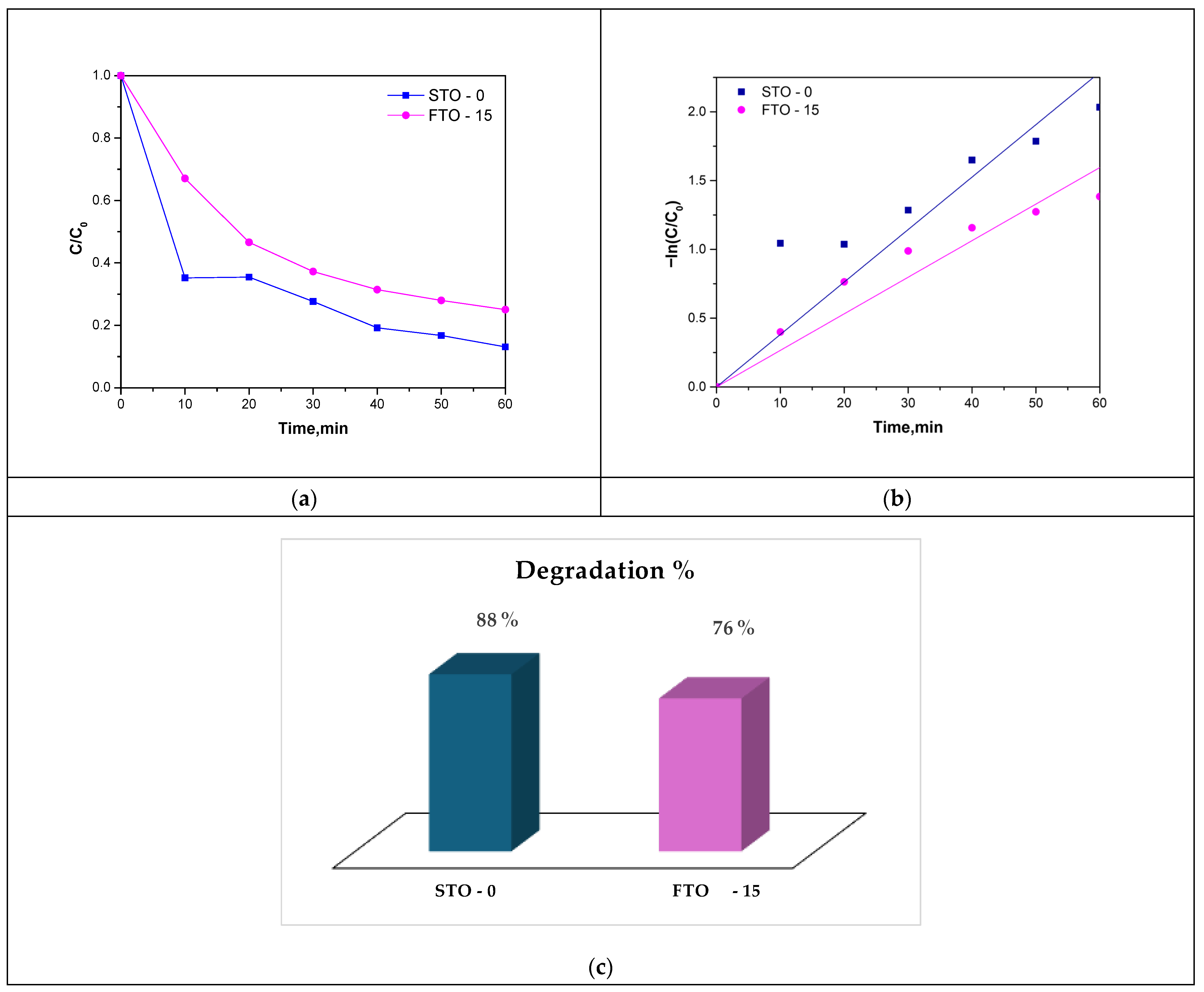
The photocatalytic degradation profiles of STO-0 and FTO-15 are shown in Figure 11a–c. Both catalysts demonstrated significant activity under solar simulation, with STO-0 achieving a final degradation efficiency of 88%, and FTO-15 reaching 76%. The C/C0 trend over time clearly illustrates that STO-0 degrades TCH more rapidly, particularly during the initial 30 min of irradiation (Figure 11a). These results are further visualized in the bar chart of Figure 11c.
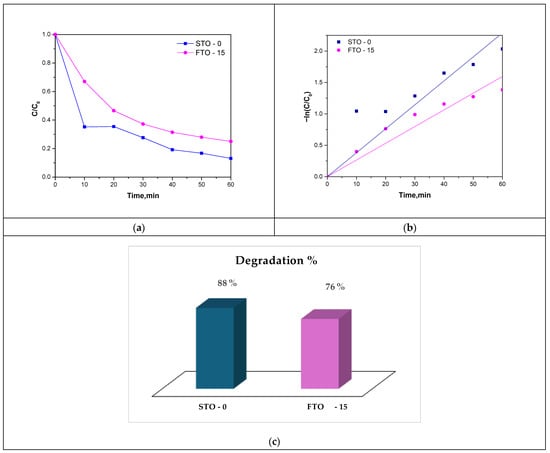
Figure 11.
Photocatalytic degradation curves of TC under sunlight illumination (a), Langmuir–Hinshelwood kinetic model of the reaction (b), and degradation % column chart (c).
The kinetics of the degradation process were evaluated using the Langmuir-Hinshelwood pseudo-first-order model. The plots of −ln(C/C0) vs. time (Figure 11b) showed linear behavior with high correlation coefficients (R2 = 0.9874 for STO-0 and 0.9704 for FTO-15), confirming that the degradation follows first-order kinetics. The calculated rate constants were k = 0.0351 min−1 for STO-0 and k = 0.0259 min−1 for FTO-15, consistent with the observed differences in photocatalytic performance with corresponding half-lives of 18.2 and 23.2 min (Table 2), respectively, further reflecting the faster degradation efficiency of STO-0.

Table 2.
Photocatalytic kinetic parameters.
Although Fe doping improves visible-light absorption by narrowing the band gap (Section 3.4), the lower photocatalytic performance of FTO-15 appears to be associated with defect-mediated recombination processes, as evidenced by the reduced PL intensity. The introduction of Fe-related states and oxygen vacancies possibly facilitates non-radiative charge recombination, which limits charge carrier availability for surface reactions and consequently suppresses ROS generation required for effective photocatalytic oxidation [18,31,32].
In summary, although FTO-15 exhibits enhanced visible-light absorption and improved adsorption capacity, its photocatalytic performance is slightly reduced, likely due to defect-induced non-radiative recombination, as indicated by the suppressed PL intensity. Sample STO-0 remains the superior photocatalyst under the tested conditions, achieving faster kinetics and higher degradation efficiency toward Tetracycline hydrochloride.
4. Conclusions
In this study, SrTiO3 (STO-0) and Fe-modified SrTiO3 (FTO-15) powders were successfully synthesized using the sol–gel method and subjected to comprehensive characterization. XRD analysis confirmed the formation of the perovskite structure. The DTA-TG analysis demonstrated the progressive removal of organic residues, followed by crystallization of the perovskite phase. UV-Vis diffuse reflectance spectroscopy revealed a red shift in the absorption edge, with optical band gaps of 3.28 eV for STO-0 and 2.86 eV for FTO-15, evidencing enhanced visible-light absorption upon Fe modification.
Photocatalytic experiments conducted under simulated solar irradiation demonstrated that both samples effectively degraded tetracycline, thereby confirming their potential as efficient photocatalysts. The antibacterial tests conducted against Staphylococcus aureus, Enterococcus faecalis, Candida albicans, and Saccharomyces cerevisiae revealed that Fe modification notably enhanced the antimicrobial activity. The FTO-15 sample exhibited the lowest minimum bactericidal concentration (MBC) of 12.5 mg/mL against S. aureus, compared to 50 mg/mL for the undoped STO-0, highlighting the significant improvement imparted by Fe doping. Overall, the findings demonstrate that Fe modification effectively tailors the optical and antimicrobial properties of SrTiO3.
Author Contributions
Conceptualization, S.P., K.I. and A.B.-N.; methodology, A.B.-N. and I.I.; investigation, S.P., K.I. and I.I.; writing—original draft preparation, S.P., K.I. and A.B.-N.; writing—photocatalytic properties, S.P.; writing—antibacterial properties, K.I. and I.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research is financed by the European Union-Next Generation EU, through the National Recovery and Resilience Plan of the Republic of Bulgaria, project SUMMIT BG-RRP-2.004-0008-C01 contract 70-123-661/16.04.2024 Green synthesis on nanoparticles and study on the antimicrobial and cytotoxic effect.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Acknowledgments
The authors are thankful to the support by European Regional Development Fund under “Research Innovation and Digitization for Smart Transformation” program 2021–2027 under the Project BG16RFPR002-1.014-0006 National Centre of Excellence Mechatronics and Clean Technologies. Research equipment of distributed research infrastructure INFRAMAT, supported by the Bulgarian Ministry of Education and Science was used. The author S.P. would like to express her gratitude to the Laboratory of Chemistry of Rare-Earth elements for the valuable discussion on the photocatalytic properties.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Kao, P.H.-N.; Ch’ng, J.-H.; Chong, K.K.L.; Stocks, C.J.; Wong, S.L.; Kline, K.A. Enterococcus Faecalis Suppresses Staphylococcus Aureus -Induced NETosis and Promotes Bacterial Survival in Polymicrobial Infections. FEMS Microbes 2023, 4, xtad019. [Google Scholar] [CrossRef]
- Ahmad, K.; Ghatak, H.R.; Ahuja, S.M. A review on photocatalytic remediation of environmental pollutants and H2 production through water splitting: A sustainable approach. Environ. Technol. Innov. 2020, 19, 100893. [Google Scholar] [CrossRef]
- Lázaro-Mass, S.; Quintana, P.; Gómez-Cornelio, S.; Hernández-Bolio, G.; Herrera-Candelario, L.; López-González, R.; Fuentes, A.F.; De la Rosa-García, S.; Ruiz-Gómez, M. Efficiency of CaZn2(OH)6·2H2O and ZnO nanoparticles in photocatalytic degradation of amoxicillin after multiple cycles. Chemosphere 2024, 367, 143577. [Google Scholar] [CrossRef]
- Srivastava, S.K. Recent advances in removal of pharmaceutical pollutants in wastewater using metal oxides and carbonaceous materials as photocatalysts: A review. RSC Appl. Interfaces 2024, 1, 340–429. [Google Scholar] [CrossRef]
- Heris, S.Z.; Khaniani, P.B.; Mousavi, S.B. Photocatalytic degradation of co-amoxiclav using hybrid TiO2/ZnO nanoparticles: Experimental and optimization. J. Water Process Eng. 2025, 70, 107040. [Google Scholar] [CrossRef]
- Charafi, S.; Janani, F.Z.; Elhalil, A.; Abdennouri, M.; Sadiq, M.; Barka, N. Optimization of photocatalytic degradation of amoxicillin by ZnO-TiO2 heterojunction under UV-Visible irradiation. Clean. Chem. Eng. 2025, 11, 100183. [Google Scholar] [CrossRef]
- Grossman, T.H. Tetracycline Antibiotics and Resistance. Cold Spring Harb. Perspect. Med. 2016, 6, a025387. [Google Scholar] [CrossRef]
- Morse, S.A.; Johnson, S.R.; Biddle, J.W.; Roberts, M.C. High-Level Tetracycline Resistance in Neisseria Gonorrhoeae Is Result of Acquisition of Streptococcal tetM Determinant. Antimicrob. Agents. Chemother. 1986, 30, 664–670. [Google Scholar] [CrossRef]
- Hao, Y.; Wang, Y.; Zhang, L.; Liu, F.; Jin, Y.; Long, J.; Chen, S.; Duan, G.; Yang, H. Advances in Antibacterial Activity of Zinc Oxide Nanoparticles against Staphylococcus Aureus (Review). Biomed. Rep. 2024, 21, 161. [Google Scholar] [CrossRef] [PubMed]
- Abdeta, A.; Beyene, D.; Negeri, A.A. Antimicrobial Resistance Patterns of Staphylococcus Aureus and Enterococcus Species at the Ethiopian Public Health Institute, Ethiopia: A Five-Year Retrospective Analysis. Infect. Drug Resist. 2023, 16, 6155–6166. [Google Scholar] [CrossRef]
- Roszczenko, P.; Szewczyk, O.K.; Czarnomysy, R.; Bielawski, K.; Bielawska, A. Biosynthesized Gold, Silver, Palladium, Platinum, Copper, and Other Transition Metal Nanoparticles. Pharmaceutics 2022, 14, 2286. [Google Scholar] [CrossRef]
- Wilson, D. A Tale of Two Yeasts: Saccharomyces Cerevisiae as a Therapeutic against Candidiasis. Virulence 2017, 8, 15–17. [Google Scholar] [CrossRef]
- Pérez-Torrado, R.; Querol, A. Opportunistic Strains of Saccharomyces Cerevisiae: A Potential Risk Sold in Food Products. Front. Microbiol. 2016, 6, 1522. [Google Scholar] [CrossRef]
- Martínez, A.; Apip, C.; Meléndrez, M.F.; Domínguez, M.; Sánchez-Sanhueza, G.; Marzialetti, T.; Catalán, A. Dual Antifungal Activity against Candida Albicans of Copper Metallic Nanostructures and Hierarchical Copper Oxide Marigold-like Nanostructures Grown in Situ in the Culture Medium. J. Appl. Microbiol. 2021, 130, 1883–1892. [Google Scholar] [CrossRef] [PubMed]
- Prucek, R.; Tuček, J.; Kilianová, M.; Panáček, A.; Kvítek, L.; Filip, J.; Kolář, M.; Tománková, K.; Zbořil, R. The Targeted Antibacterial and Antifungal Properties of Magnetic Nanocomposite of Iron Oxide and Silver Nanoparticles. Biomaterials 2011, 32, 4704–4713. [Google Scholar] [CrossRef] [PubMed]
- De La Rosa-García, S.C.; Martínez-Torres, P.; Gómez-Cornelio, S.; Corral-Aguado, M.A.; Quintana, P.; Gómez-Ortíz, N.M. Antifungal Activity of ZnO and MgO Nanomaterials and Their Mixtures against Colletotrichum Gloeosporioides Strains from Tropical Fruit. J. Nanomater. 2018, 2018, 3498527. [Google Scholar] [CrossRef]
- Amangelsin, Y.; Semenova, Y.; Dadar, M.; Aljofan, M.; Bjørklund, G. The Impact of Tetracycline Pollution on the Aquatic Environment and Removal Strategies. Antibiotics 2023, 12, 440. [Google Scholar] [CrossRef]
- Petrova, S.; Ivanova, K.; Ivanova, I.; Bachvarova-Nedelcheva, A. Optical, Photocatalytic, and Antibacterial Properties of Sol-Gel Derived Fe Doped SrTiO3 Powders. Water 2025, 17, 2072. [Google Scholar] [CrossRef]
- Sharma, N.; Hernadi, K. The Emerging Career of Strontium Titanates in Photocatalytic Applications: A Review. Catalysts 2022, 12, 1619. [Google Scholar] [CrossRef]
- Bajpai, P. Optical Properties of Paper. In Biermann’s Handbook of Pulp and Paper; Elsevier: Amsterdam, The Netherlands, 2018; pp. 237–271. [Google Scholar]
- Jabbar, Z.H.; Ebrahim, S.E. Recent advances in nano-semiconductors photocatalysis for degrading organic contaminants and microbial disinfection in wastewater: A comprehensive review. Environ. Nanotechnol. Monit. Manag. 2022, 17, 100666. [Google Scholar] [CrossRef]
- Jorgensen, J.H.; Ferraro, M.J. Antimicrobial Susceptibility Testing: A Review of General Principles and Contemporary Practices. Clin. Infect. Dis. 2009, 49, 1749–1755. [Google Scholar] [CrossRef]
- Patterson, A.L. The Scherrer Formula for X-Ray Particle Size Determination. Phys. Rev. 1939, 56, 978–982. [Google Scholar] [CrossRef]
- Hasan, T.; Saha, A.; Khan, M.N.I.; Rashid, R.; Basith, M.A.; Bashar, M.S.; Ahmed, I. Structural, Electrical, and Magnetic Properties of Ce and Fe Doped SrTiO3. AIP Adv. 2022, 12, 095003. [Google Scholar] [CrossRef]
- Wang, K.; De Souza, R.A.; Peng, X.-L.; Merkle, R.; Rheinheimer, W.; Albe, K.; Xu, B.-X. A Defect-Chemistry-Informed Phase-Field Model of Grain Growth in Oxide Ceramics: Application to Fe-Doped SrTiO3. arXiv 2024, arXiv:2407.17650. [Google Scholar]
- Schoofs, B.; Van De Vyver, D.; Vermeir, P.; Schaubroeck, J.; Hoste, S.; Herman, G.; Van Driessche, I. Characterisation of the Sol-Gel Process in the Superconducting NdBa2Cu3O7−y System. J. Mater. Chem. 2007, 17, 1714–1724. [Google Scholar] [CrossRef]
- Fynn, D.; Billing, C.; Billing, D.G. Thermal Decomposition of Sol-Gel Synthesized Bismuth Citrate. J. Therm. Anal. Calorim. 2025, 150, 9039–9052. [Google Scholar] [CrossRef]
- Malghe, Y.S. Nanosized SrTiO3 Powder from Oxalate Precursor Microwave Aided Synthesis and Thermal Characterization. J. Therm. Anal. Calorim. 2010, 102, 831–836. [Google Scholar] [CrossRef]
- Ehrhardt, C.; Fettkenhauer, C.; Glenneberg, J.; Münchgesang, W.; Leipner, H.S.; Wagner, G.; Diestelhorst, M.; Pientschke, C.; Beige, H.; Ebbinghaus, S.G. Enhanced Dielectric Properties of Sol-Gel-BaTiO3/P(VDF-HFP) Composite Films without Surface Functionalization. RSC Adv. 2014, 4, 40321–40329. [Google Scholar] [CrossRef]
- Shafique, H.; Kashif, M.; Zahid, M.; Yaseen, M.; Iqbal, J.; Aldaghfag, S.A.; Neffati, R. Magnetic and Optical Characteristics of Fe Doped SrTiO3 Perovskite Compound: A First Principle Study. Chalcogenide Lett. 2021, 18, 589–599. [Google Scholar] [CrossRef]
- Koshi, N.A.; Murthy, D.H.K.; Chakraborty, S.; Lee, S.-C.; Bhattacharjee, S. Probing Photoexcited Charge Carrier Trapping and Defect Formation in Synergistic Doping of SrTiO3. ACS Appl. Energy Mater. 2022, 5, 1159–1168. [Google Scholar] [CrossRef]
- Kubacki, J.; Kajewski, D.; Goraus, J.; Szot, K.; Koehl, A.; Lenser, C.; Dittmann, R.; Szade, J. Impact of Fe Doping on the Electronic Structure of SrTiO3 Thin Films Determined by Resonant Photoemission. J. Chem. Phys. 2018, 148, 154702. [Google Scholar]
- Rubano, A.; Paparo, D.; Radović, M.; Sambri, A.; Granozio, F.M.; Scotti Di Uccio, U.; Marrucci, L. Time-Resolved Photoluminescence of n-Doped SrTiO3. Appl. Phys. Lett. 2008, 92, 021102. [Google Scholar] [CrossRef]
- Liu, C.M.; Zu, X.T.; Zhou, W.L. Photoluminescence of Nitrogen Doped SrTiO3. J. Phys. D Appl. Phys. 2007, 40, 7318–7322. [Google Scholar] [CrossRef]
- Wu, P.; Zhou, T.; Tong, Z.; Xi, F.; Lu, J.; Li, X.; Ma, W.; Li, S.; Yang, X. Balancing Charge Carrier Density and Exciton Recombination in Defective G-C3N4 for Efficient Photocatalytic Hydrogen Evolution. Prog. Nat. Sci. Mater. Int. 2025, 35, 238–244. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.