Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions
Abstract
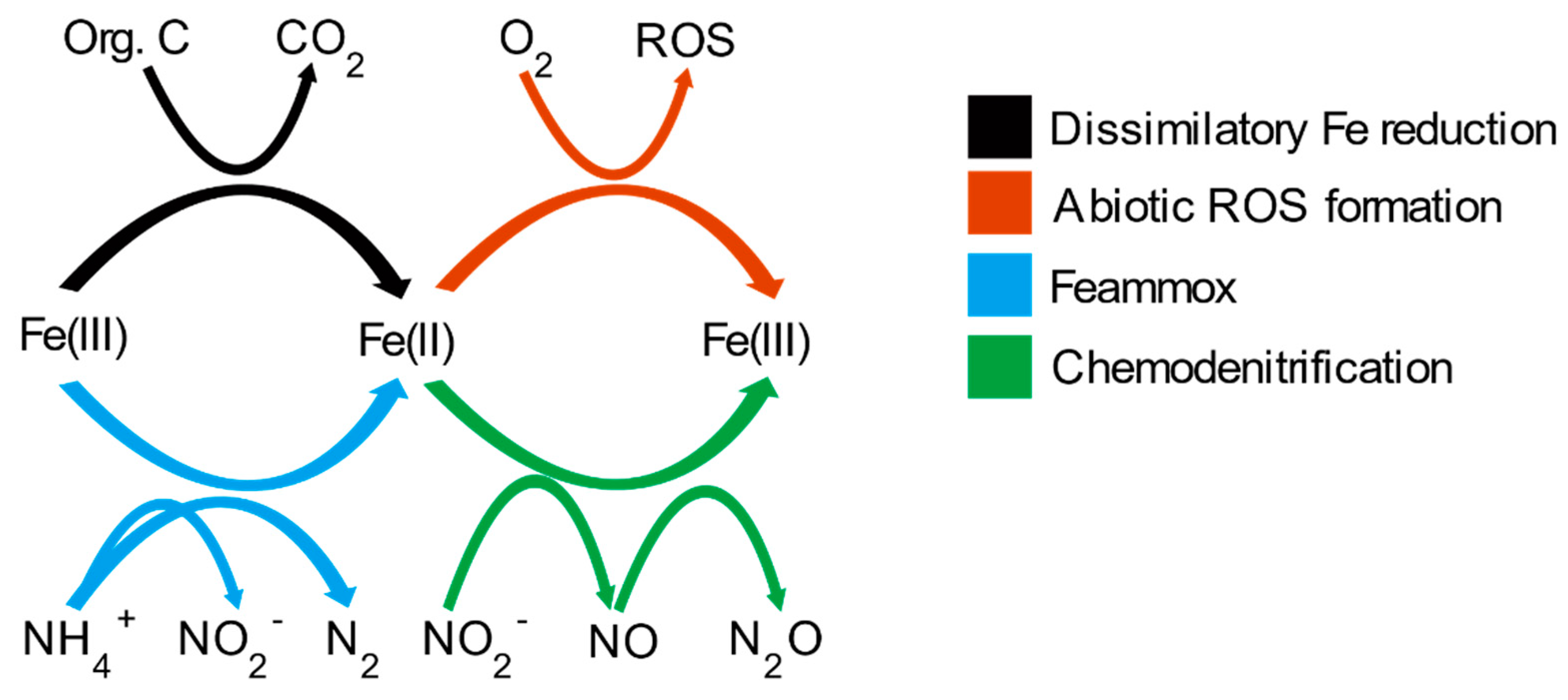
:1. Introduction
2. Methods
2.1. Study Site
2.2. Experimental Design
2.3. Soil Collection and Initial Characterization
2.4. Experimental Methods
2.5. Statistical Analyses
3. Results
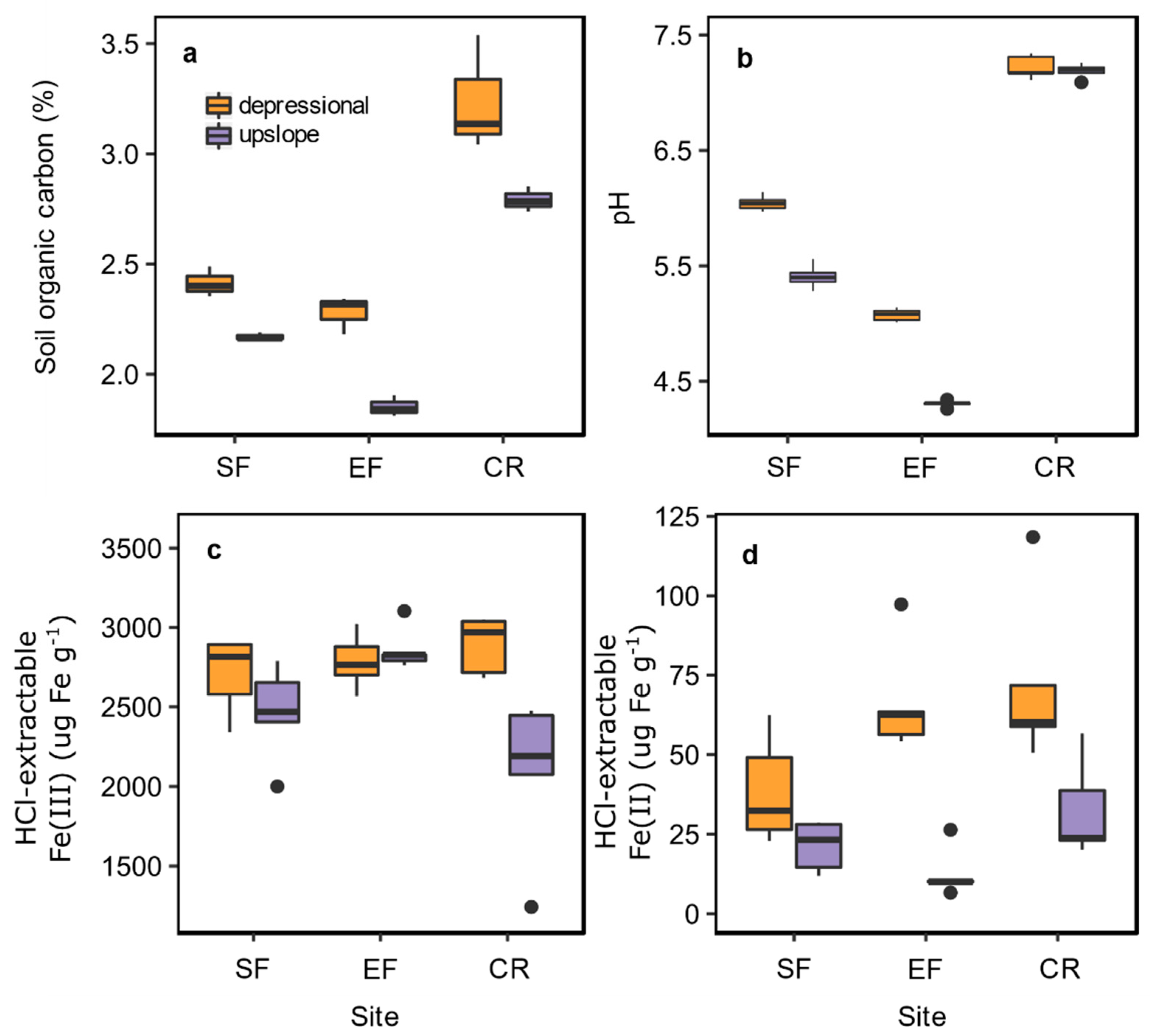
3.1. Background Soil Properties
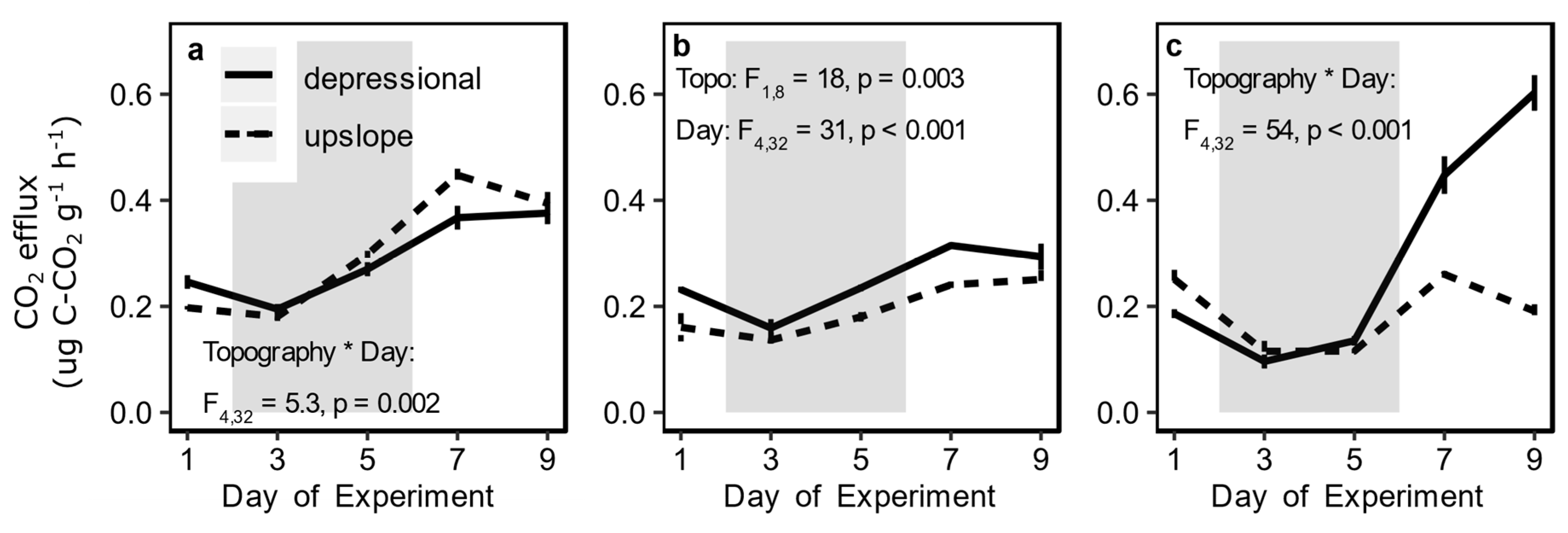
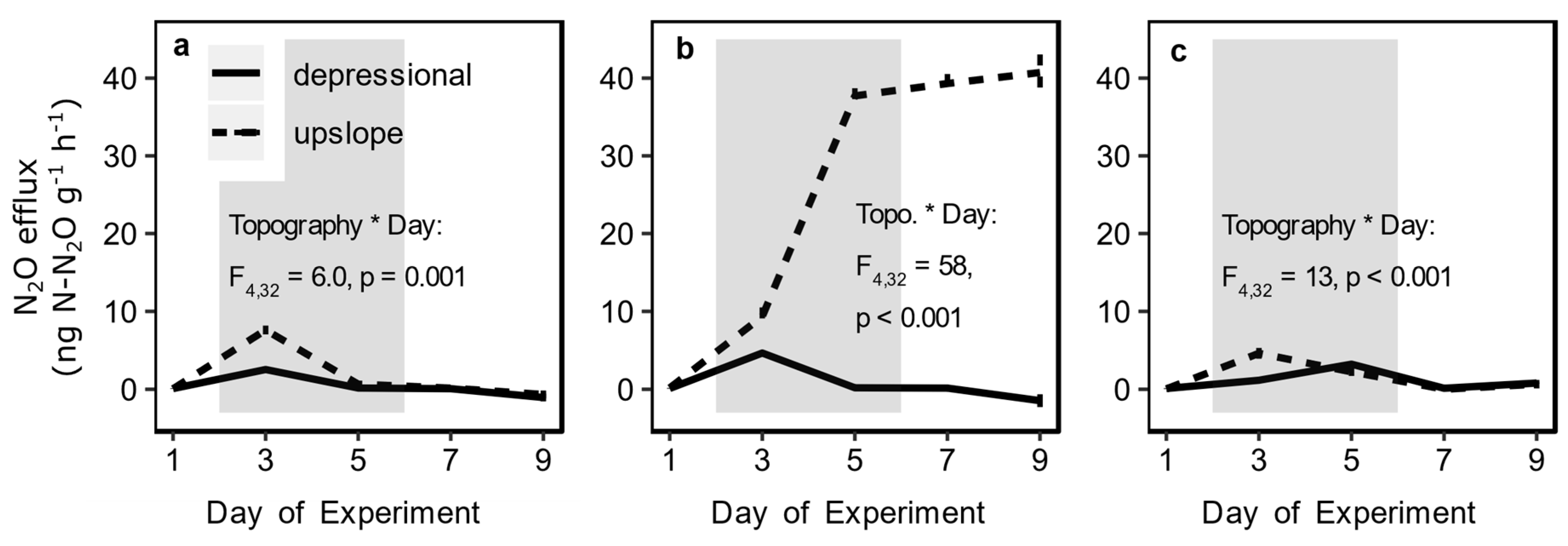
3.2. Redox Fluctuation Effects on Trace Gas Emissions
3.3. Amendment Effects on Trace Gas Emissions and Fe Reduction
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Min, S.K.; Zhang, X.; Zwiers, F.W.; Hegerl, G.C. Human contribution to more-intense precipitation extremes. Nature 2011, 470, 378–381. [Google Scholar] [CrossRef]
- Gleason, K. 2008 Midwestern, U.S. Floods; NOAA’s National Climatic Data Center: Ashville, NC, USA, 2008. [Google Scholar]
- USGCRP. Global Climate Change Impacts in the United States; Karl, T.R., Melillo, J.M., Peterson, T.C., Eds.; United States Global Change Research Program, Cambrige University Press: New York, NY, USA, 2009. [Google Scholar]
- Takai, Y.; Kamura, T. The mechanism of reduction in waterlogged paddy soil. Folia Microbiol. 1966, 11, 304–313. [Google Scholar] [CrossRef]
- Estop-Aragonés, C.; Knorr, K.H.; Blodau, C. Belowground in situ redox dynamics and methanogenesis recovery in a degraded fen during dry-wet cycles and flooding. Biogeosciences 2013, 10, 421–436. [Google Scholar] [CrossRef] [Green Version]
- Weber, K.A.; Achenbach, L.A.; Coates, J.D. Microorganisms pumping iron: Anaerobic microbial iron oxidation and reduction. Nat. Rev. Microbiol. 2006, 4, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Zhu-Barker, X.; Cavazos, A.R.; Ostrom, N.E.; Horwath, W.R.; Glass, J.B. The importance of abiotic reactions for nitrous oxide production. Biogeochemistry 2015, 126, 251–267. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J. Availability of ferric iron for microbial reduction in bottom sediments of the freshwater tidal potomac river. Appl. Environ. Microbiol. 1986, 52, 751–757. [Google Scholar] [PubMed]
- Burgin, A.J.; Yang, W.H.; Hamilton, S.K.; Silver, W.L. Beyond carbon and nitrogen: How the microbial energy economy couples elemental cycles in diverse ecosystems. Front. Ecol. Environ. 2011, 9, 44–52. [Google Scholar] [CrossRef]
- Dubinsky, E.A.; Silver, W.L.; Firestone, M.K. Tropical forest soil microbial communities couple iron and carbon biogeochemistry. Ecology 2010, 91, 2604–2612. [Google Scholar] [CrossRef]
- Suriyavirun, N.; Krichels, A.H.; Kent, A.D.; Yang, W.H. Microtopographic differences in soil properties and microbial community composition at the field scale. Soil. Biol. Biochem. 2019, 131, 71–80. [Google Scholar] [CrossRef]
- Barcellos, D.; Cyle, K.T.; Thompson, A. Faster redox fluctuations can lead to higher iron reduction rates in humid forest soils. Biogeochemistry 2018, 137, 367–378. [Google Scholar] [CrossRef]
- Ginn, B.; Meile, C.; Wilmoth, J.; Yang, Y.; Thompson, A. Rapid iron reduction rates are stimulated by high-amplitude redox fluctuations in a tropical forest soil. Environ. Sci. Technol. 2017, 51, 3250–3259. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.; Rancourt, D.G.; Chadwick, O.A.; Chorover, J. Iron solid-phase differentiation along a redox gradient in basaltic soils. Geochim. Cosmochim. Acta 2011, 75, 119–133. [Google Scholar] [CrossRef]
- Krichels, A.; DeLucia, E.H.; Sanford, R.; Chee-Sanford, J.C.; Yang, W.H. Historical soil drainage mediates the response of soil greenhouse gas emissions to intense precipitation events. Biogeochemistry 2019, 142, 425–442. [Google Scholar] [CrossRef]
- Buettner, S.W.; Kramer, M.G.; Chadwick, O.A.; Thompson, A. Mobilization of colloidal carbon during iron reduction in basaltic soils. Geoderma 2014, 221–222, 139–145. [Google Scholar] [CrossRef]
- Coward, E.K.; Thompson, A.; Plante, A.F. Contrasting Fe speciation in two humid forest soils: Insight into organomineral associations in redox-active environments. Geochim. Cosmochim. Acta 2018, 238, 68–84. [Google Scholar] [CrossRef]
- Hall, S.J.; Berhe, A.A.; Thompson, A. Order from disorder: Do soil organic matter composition and turnover co-vary with iron phase crystallinity? Biogeochemistry 2018, 140, 93–110. [Google Scholar] [CrossRef]
- Grybos, M.; Davranche, M.; Gruau, G.; Petitjean, P.; Pédrot, M. Increasing pH drives organic matter solubilization from wetland soils under reducing conditions. Geoderma 2009, 154, 13–19. [Google Scholar] [CrossRef]
- Pan, W.; Kan, J.; Inamdar, S.; Chen, C.; Sparks, D. Dissimilatory microbial iron reduction release DOC (dissolved organic carbon) from carbon-ferrihydrite association. Soil. Biol. Biochem. 2016, 103, 232–240. [Google Scholar] [CrossRef]
- Nitzsche, K.N.; Kaiser, M.; Premke, K.; Gessler, A.; Ellerbrock, R.H.; Hoffman, C.; Kleeberg, A.; Kayler, Z.E. Organic matter distribution and retention along transects from hilltop to kettle hole within an agricultural landscape. Biogeochemistry 2017, 136, 47–70. [Google Scholar] [CrossRef]
- Yanai, J.; Sawamoto, T.; Oe, T.; Jusa, K.; Yamakawa, K.; Sakamoto, K.; Naganawa, T.; Inubushi, K.; Hatano, R.; Kosaki, T. Spatial variability of nitrous oxide emissions and their soil-related determining factors in an agricultural field. J. Environ. Qual. 2003, 32, 1965–1977. [Google Scholar] [CrossRef]
- Turner, D.A.; Chen, D.; Galbally, I.E.; Leuning, R.; Edis, R.B.; Kelly, K.; Phillips, F. Spatial variability of nitrous oxide emissions from an Australian irrigated dairy pasture. Plant Soil 2008, 309, 77–88. [Google Scholar] [CrossRef]
- Hall, S.J.; Silver, W.L. Iron oxidation stimulates organic matter decomposition in humid tropical forest soils. Glob. Chang. Biol. 2013, 19, 2804–2813. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.H.; Weber, K.; Silver, W.L. Nitrogen loss from soil through anaerobic ammonium oxidation coupled to iron reduction. Nat. Geosci. 2012, 5, 538–541. [Google Scholar] [CrossRef] [Green Version]
- Huang, S.; Jaffé, P.R. Characterization of incubation experiments and development of an enrichment culture capable of ammonium oxidation under iron-reducing conditions. Biogeosciences 2015, 12, 769–779. [Google Scholar] [CrossRef] [Green Version]
- Picardal, F. Abiotic and microbial interactions during anaerobic transformations of Fe(II) and NOx−. Front. Microbiol. 2012, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Carlson, H.K.; Clark, I.C.; Melnyk, R.A.; Coates, J.D. Toward a mechanistic understanding of anaerobic nitrate-dependent iron oxidation: Balancing electron uptake and detoxification. Front. Microbiol. 2012, 3, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Coby, A.J.; Picardal, F.; Shelobolina, E.; Xu, H.; Roden, E.D. Repeated anaerobic microbial redox cycling of iron. Appl. Environ. Microbiol. 2011, 77, 6036–6042. [Google Scholar] [CrossRef]
- Klueglein, N.; Zeitvogel, F.; Stierhof, Y.D.; Floetenmeyer, M.; Konhauser, K.O.; Kappler, A.; Obst, M. Potential role of nitrite for abiotic Fe(II) oxidation and cell encrustation during nitrate reduction by denitrifying bacteria. Appl. Environ. Microbiol. 2014, 80, 1051–1061. [Google Scholar] [CrossRef]
- Carlson, H.K.; Clark, I.C.; Blazewicz, S.J.; Iavarone, A.T.; Coates, J.D. Fe(II) oxidation is an innate capability of nitrate-reducing bacteria that involves abiotic and biotic reactions. J. Bacteriol. 2013, 195, 3260–3268. [Google Scholar] [CrossRef]
- Ding, L.; An, X.; Li, S.; Zhang, G.; Zhu, Y. Nitrogen loss through anaerobic ammonium oxidation coupled to iron reduction from paddy soils in a chronosequence. Environ. Sci. Technol. 2014, 48, 10641–10647. [Google Scholar] [CrossRef]
- Inois Cilllimate Network. Water and Atmospheric Resources Monitoring Program; Illinois State Water Survey: Champaign, IL, USA, 2017. [Google Scholar]
- Liptzin, D.; Silver, W.L. Effects of carbon additions on iron reduction and phosphorus availability in a humid tropical forest soil. Soil Biol. Biochem. 2009, 41, 1696–1702. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J.P. Organic-matter mineralization with reduction of ferric iron in anaerobic sediments. Appl. Environ. Microbiol. 1986, 51, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Chapuis-Lardy, L.; Wrage, N.; Metay, A.; Chottes, J.; Bernouxs, M. Soils, a sink for N2O? A review. Glob. Chang. Biol. 2007, 13, 1–17. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 2nd ed.; Sage: Thousand Oaks, CA, USA, 2011. [Google Scholar]
- Lenth, R.V. Least-Squares Means: The R Package lsmeans. J. Stat. Softw. 2016, 69, 1–33. [Google Scholar] [CrossRef]
- Pinheiro, J.; DebRoy, S.; Sarkar, D.; R Core Team. Nlme: Linear and Nonlinear Mixed Effects Models. 2017. Available online: https://CRAN.R-project.org/package=nlme (accessed on 10 March 2018).
- Denef, K.; Six, J.; Bossuyt, H.; Frey, A.D.; Elliott, E.T.; Merckx, R.; Paustian, K. Influence of dry-wet cycles on the interrelationship between aggregate, particulate organic matter, and microbial community dynamics. Soil. Biol. Biochem. 2001, 33, 1599–1611. [Google Scholar] [CrossRef]
- Six, J.; Bossuyt, H.; Degryze, S.; Denef, K. A history of research on the link between (micro)aggregates, soil biota, and soil organic matter dynamics. Soil Tillage Res. 2004, 79, 7–31. [Google Scholar] [CrossRef]
- De-Campos, A.B.; Mamedov, A.I.; Huang, C. Short-term reducing conditions decrease soil aggregation. Soil Sci. Soc. Am. J. 2009, 73, 550–559. [Google Scholar] [CrossRef]
- Blazewicz, S.J.; Schwartz, E.; Firestone, M.K. Growth and death of bacteria and fungi underlie rainfall-induced carbon dioxide pulses from seasonally dried soil. Ecology 2014, 95, 1162–1172. [Google Scholar] [CrossRef]
- Linn, D.M.; Doran, J.W. Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tillied and nontilled soils. Soil. Sci. Soc. Am. J. 1984, 48, 1267–1272. [Google Scholar] [CrossRef]
- Davidson, E.A.; Samanta, S.; Caramori, S.S.; Savage, K. The Dual Arrhenius and Michaelis-Menten kinetics model for decomposition of soil organic matter at hourly to seasonal time scales. Glob. Chang. Biol. 2012, 18, 371–384. [Google Scholar] [CrossRef]
- Huang, W.; Hall, S.J. Elevated moisture stimulates carbon loss from mineral soils by releasing protected organic matter. Nat. Commun. 2017, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- LaRowe, D.E.; Van Cappellen, P. Degradation of natural organic matter: A thermodynamic analysis. Geochim. Cosmochim. Acta. 2011, 75, 2030–2042. [Google Scholar] [CrossRef]
- Stuckey, J.W.; Schaefer, M.V.; Kocar, B.D.; Benner, S.G.; Fendorf, S. Arsenic release metabolically limited to permanently water-saturated soil in Mekong Delta. Nat. Geosci. 2016, 9, 70–76. [Google Scholar] [CrossRef]
- Henderson, R.; Kabengi, N.; Mantripragada, N.; Cabrera, M.; Hassan, S.; Thompson, A. Anoxia-induced release of colloid- and nanoparticle-bound phosphorus in grassland soils. Environ. Sci. Technol. 2012, 46, 11727–11734. [Google Scholar] [CrossRef] [PubMed]
- Coby, A.J.; Picardal, F.W. Inhibition of NO3− and NO2− reduction by microbial Fe( III ) reduction: Evidence of a reaction between NO2− and cell surface-bound Fe2+. Appl. Environ. Microbiol. 2005, 71, 5267–5274. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.J.; Reyes, L.; Huang, W.; Homyak, P.M. Wet Spots as Hotspots: Moisture Responses of Nitric and Nitrous Oxide Emissions From Poorly Drained Agricultural Soils. J. Geophys. Res. Biogeosci. 2018, 123, 3589–3602. [Google Scholar] [CrossRef] [Green Version]
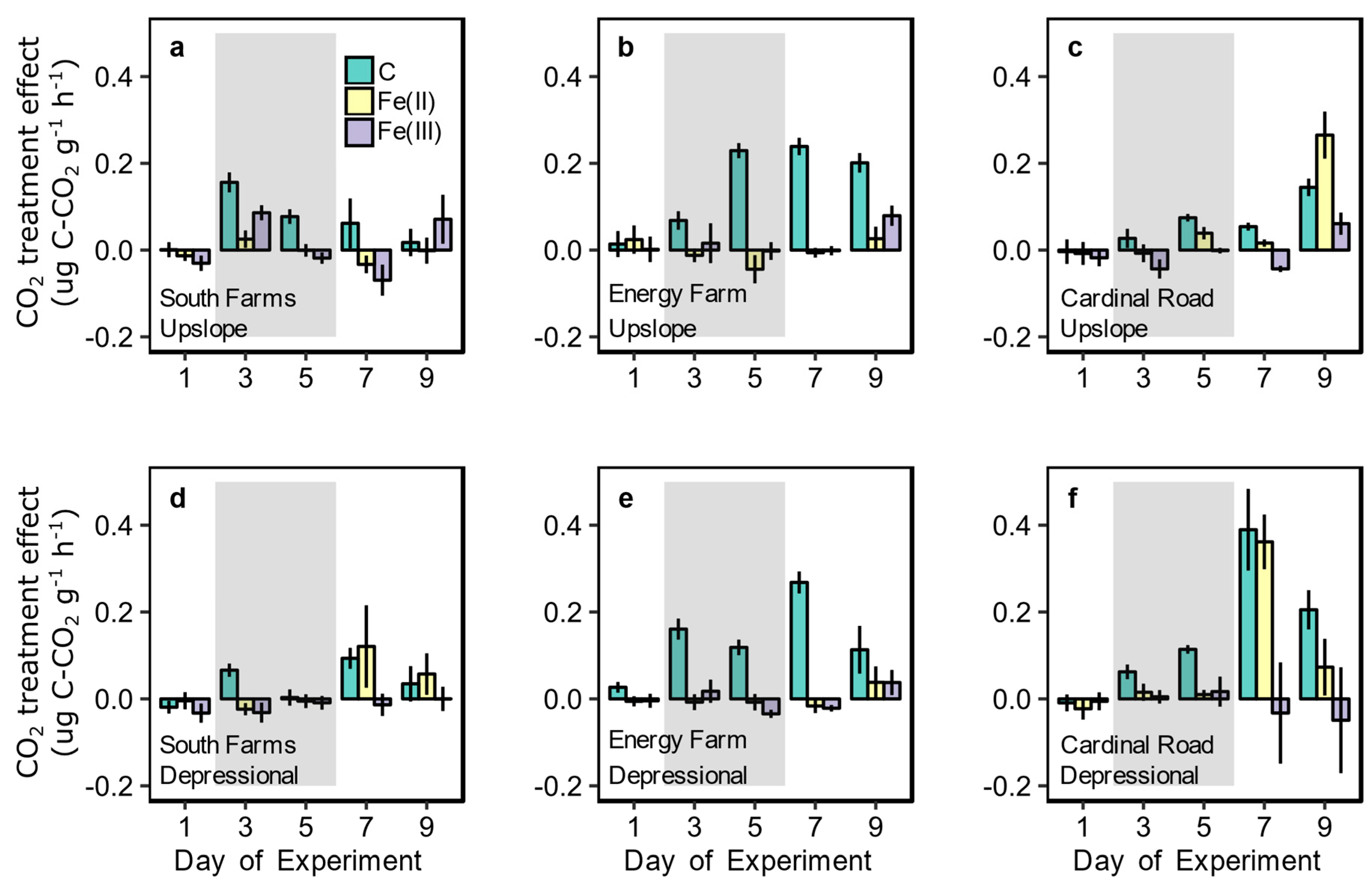
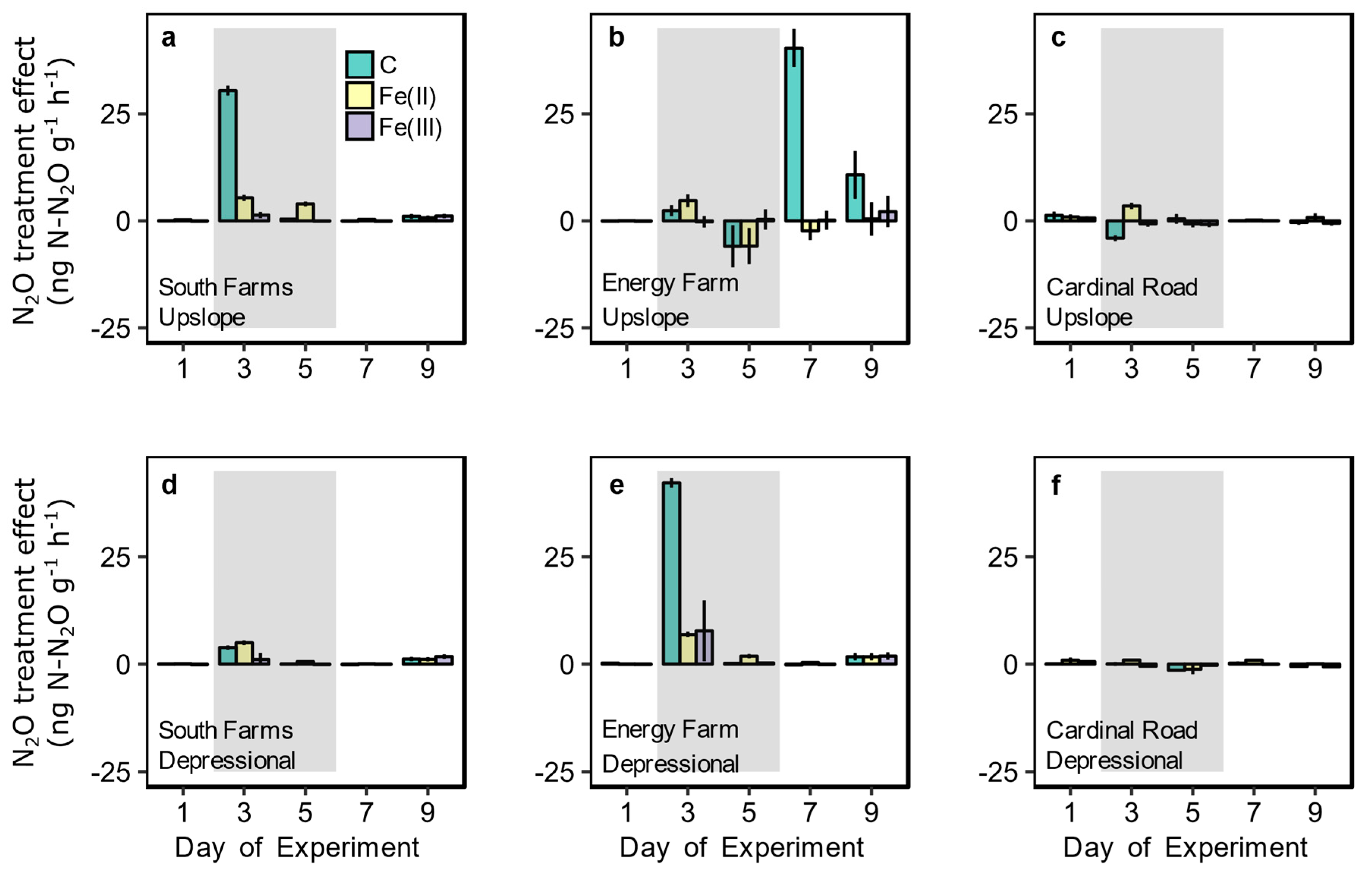
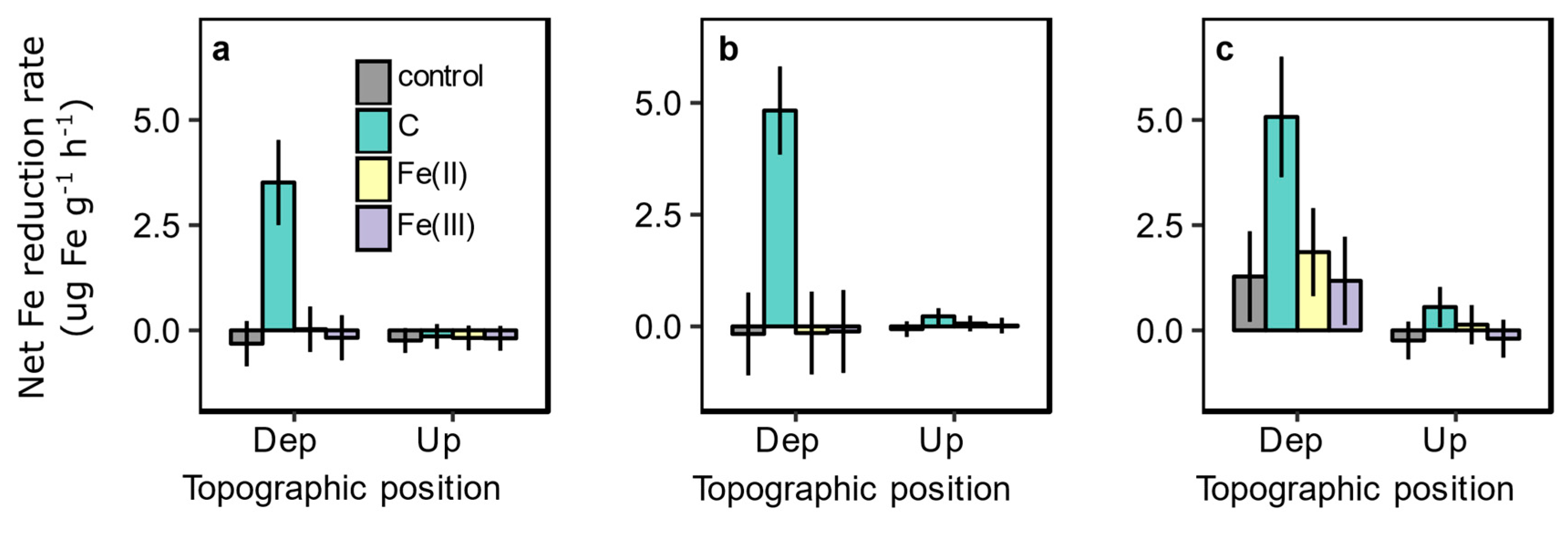
| Site | Drainage Class | Degrees of Freedom (n,d) | F Statistic | p Value |
|---|---|---|---|---|
| South Farms | upslope | 9,47 | 3.2 | 0.004 |
| South Farms | depressional | 9,48 | 2.1 | <0.05 |
| Energy Farm | upslope | 9,48 | 4.6 | <0.001 |
| Energy Farm | depressional | 9,46 | 3.8 | 0.001 |
| Cardinal Road | upslope | 9,45 | 9.2 | <0.001 |
| Cardinal Road | depressional | 9,47 | 3.5 | 0.002 |
| Site | Drainage Class | Degrees of Freedom (n,d) | F Statistic | p value |
|---|---|---|---|---|
| South Farms | upslope | 9,47 | 46 | <0.001 |
| South Farms | depressional | 9,48 | 6.9 | <0.001 |
| Energy Farm | upslope | 9,48 | 7.6 | <0.001 |
| Energy Farm | depressional | 9,46 | 8.7 | <0.001 |
| Cardinal Road | upslope | 9,45 | 7.1 | <0.001 |
| Cardinal Road | depressional | 9,47 | 3.1 | 0.006 |
| Site | Degrees of Freedom (n,d) | F Statistic | p Value |
|---|---|---|---|
| South Farms | 3,32 | 17 | <0.001 |
| Energy Farm | 3,32 | 172 | <0.001 |
| Cardinal Road | 3,32 | 6.1 | 0.002 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krichels, A.H.; Sipic, E.; Yang, W.H. Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions. Soil Syst. 2019, 3, 60. https://doi.org/10.3390/soilsystems3030060
Krichels AH, Sipic E, Yang WH. Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions. Soil Systems. 2019; 3(3):60. https://doi.org/10.3390/soilsystems3030060
Chicago/Turabian StyleKrichels, Alexander H., Emina Sipic, and Wendy H. Yang. 2019. "Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions" Soil Systems 3, no. 3: 60. https://doi.org/10.3390/soilsystems3030060





