Cyclic Block Copolymer Microchannel Fabrication and Sealing for Microfluidics Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Thermoplastic Substrates
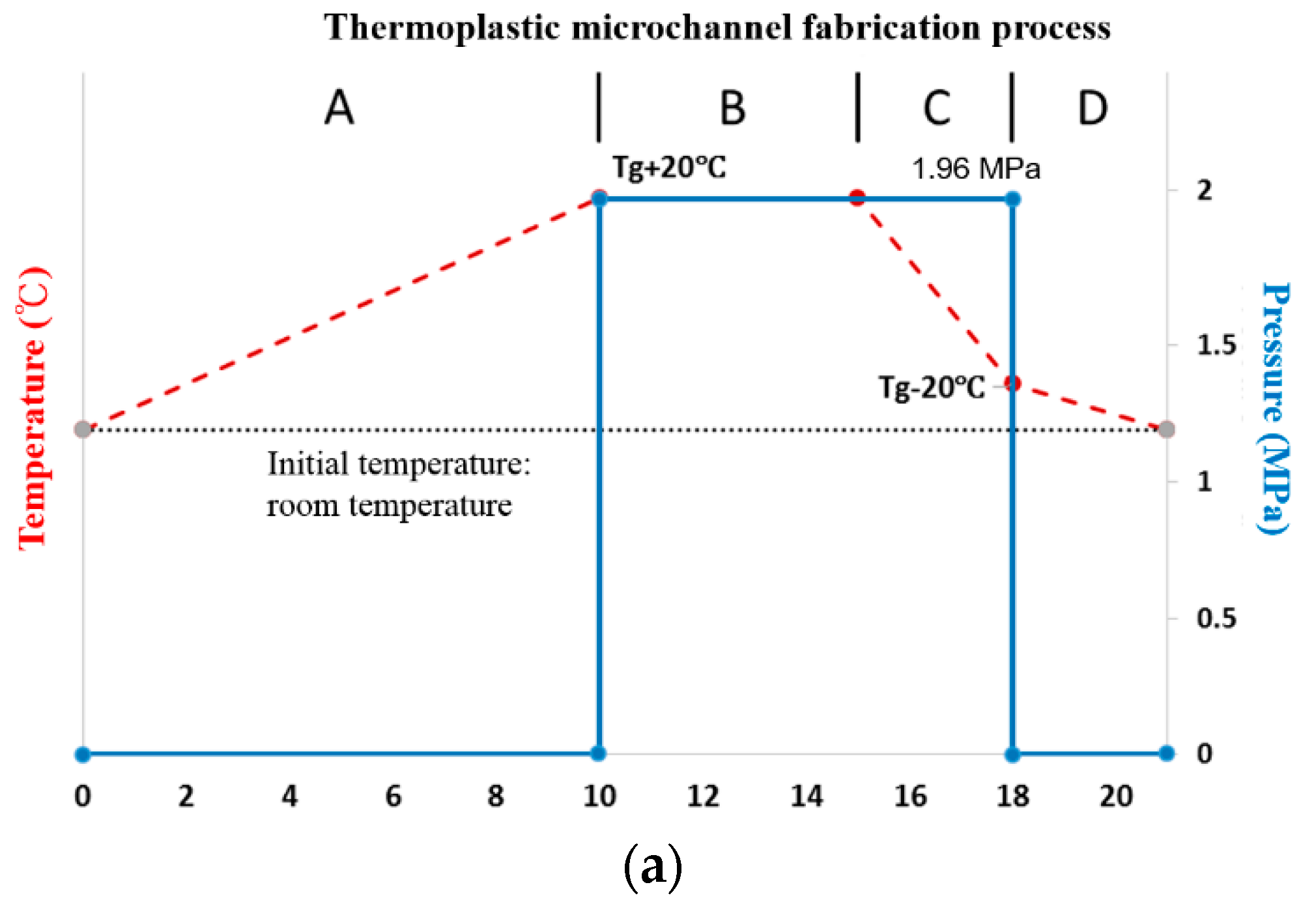
2.2. Microchannel Fabrication
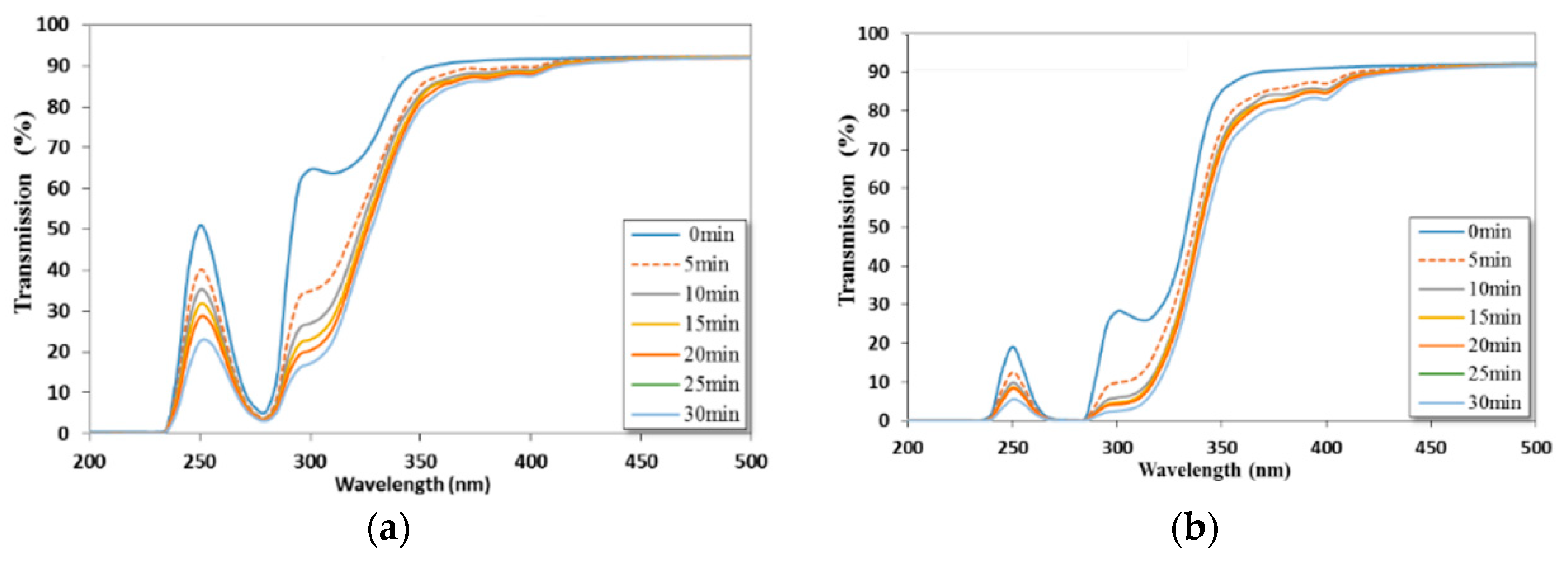
2.3. Water Contact Angle, and UV-VIS Transmissivity Measurements
3. Results and Discussion
3.1. Cyclic Block Copolymer
3.2. CBC Microchannel Fabrication Using the Hot Embossing Process
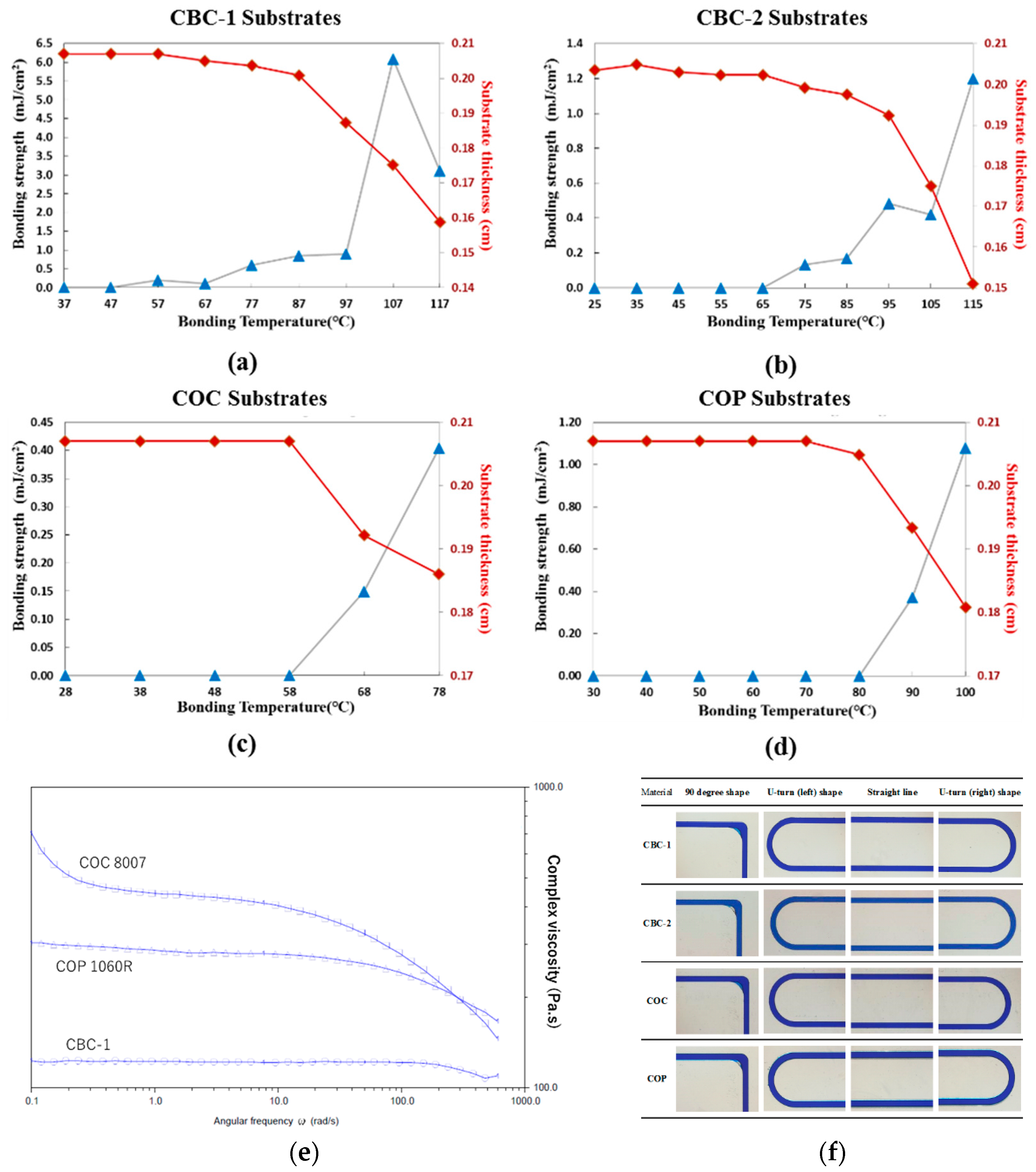
3.3. Bonding of the CBC Thermoplastic Microchannel
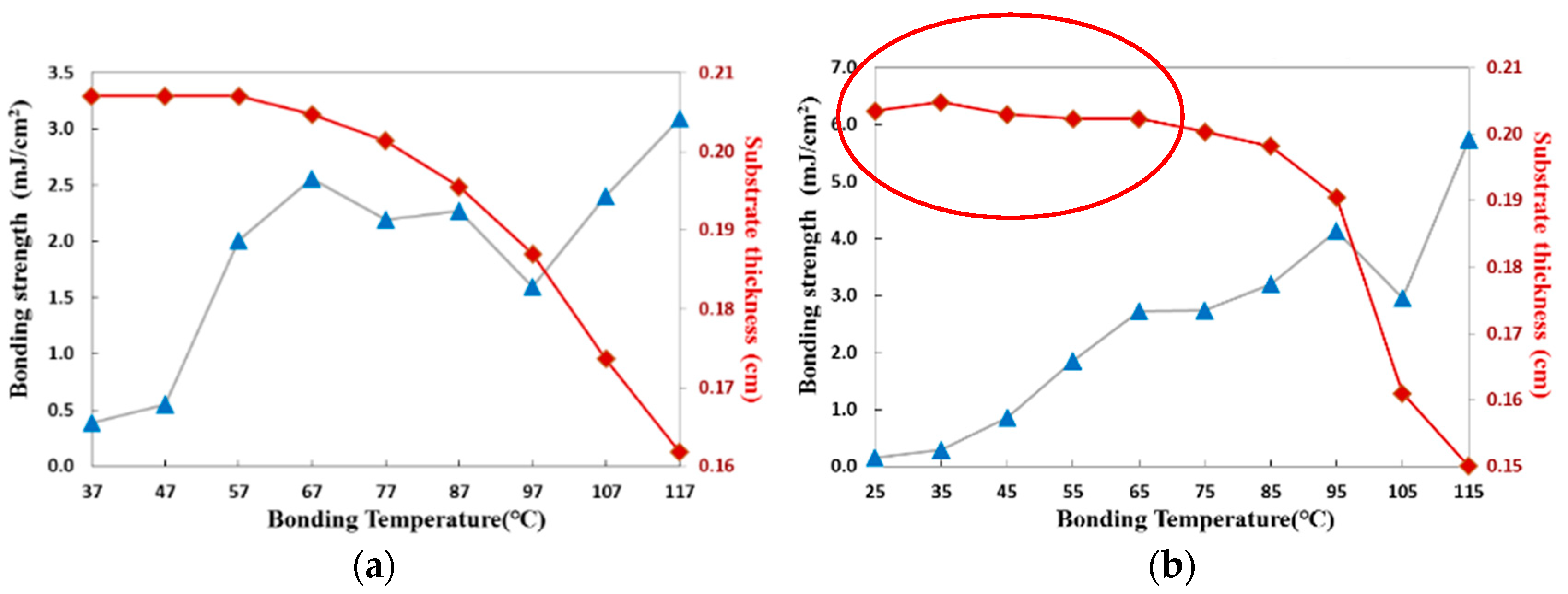
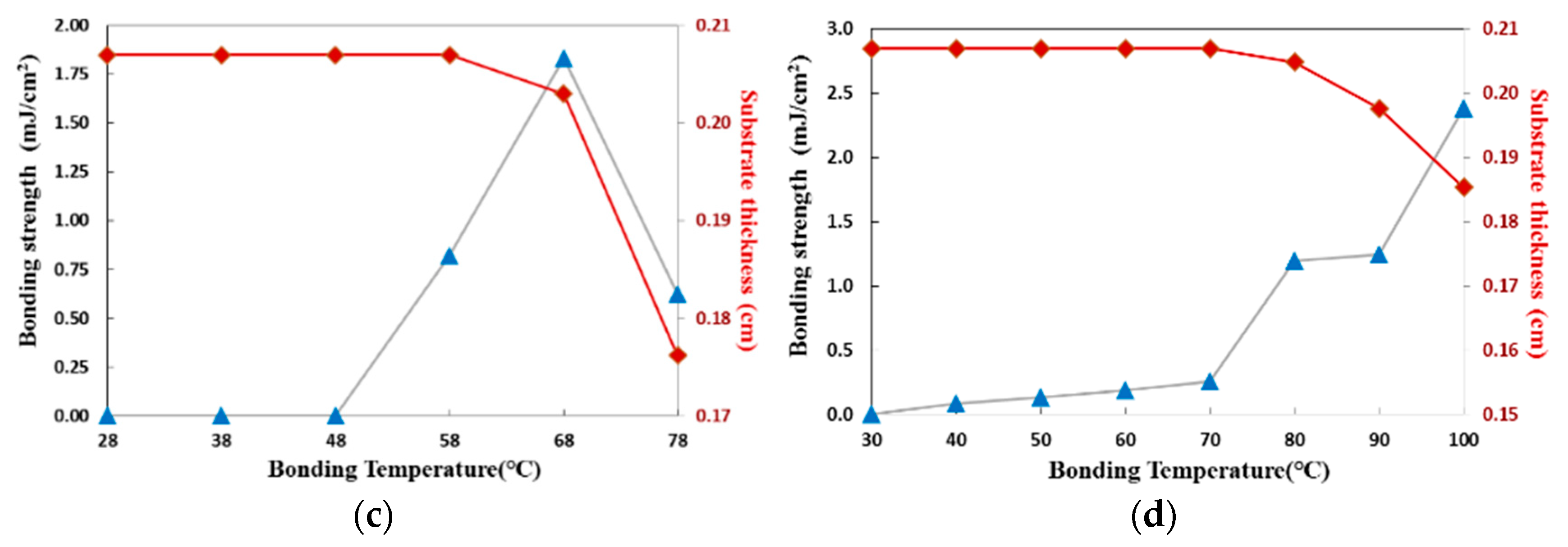
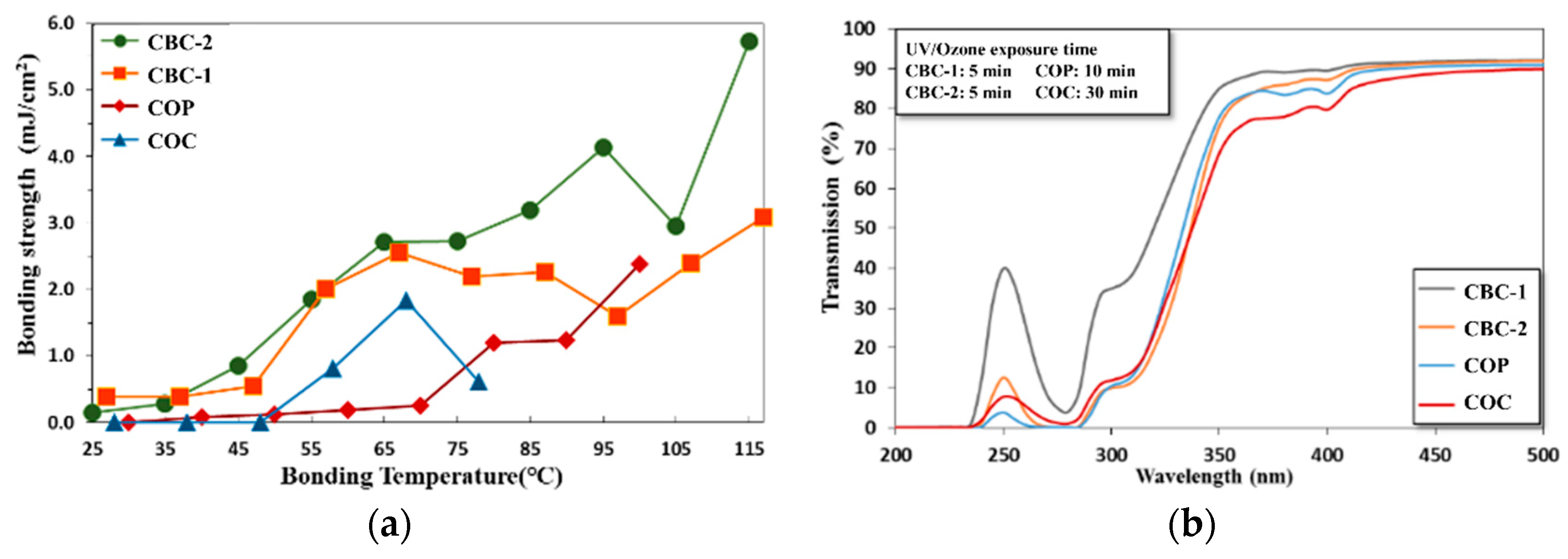
3.3.1. Thermal Fusion Bonding of the CBC Microchannel
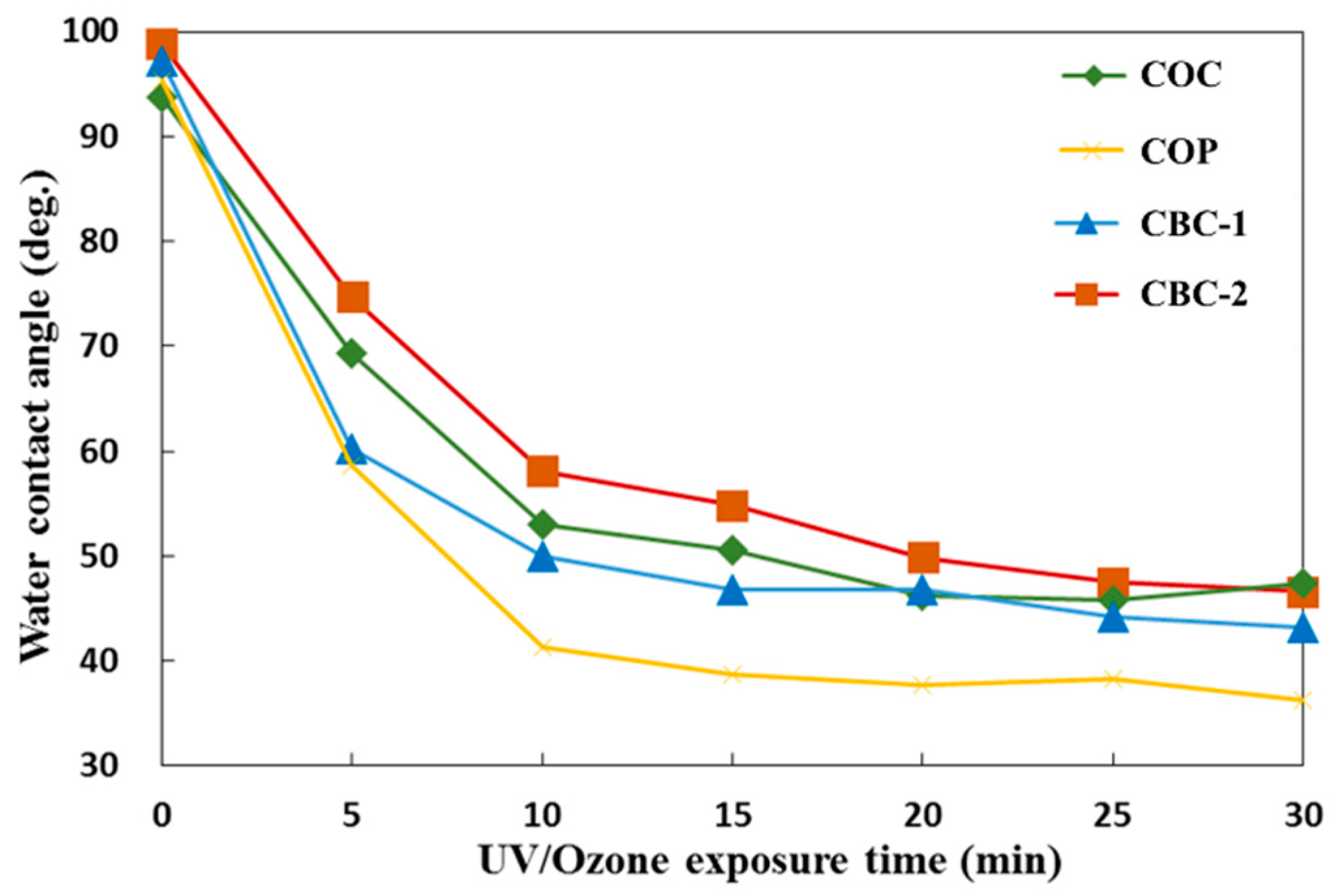
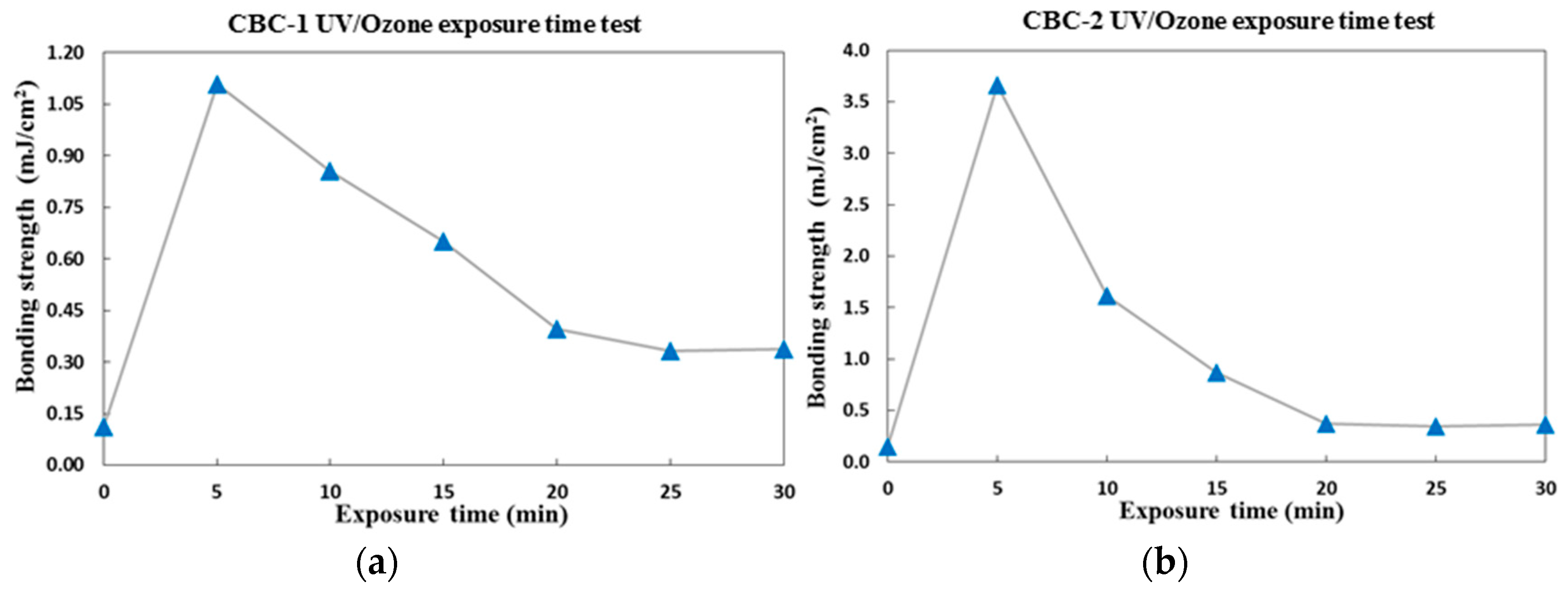
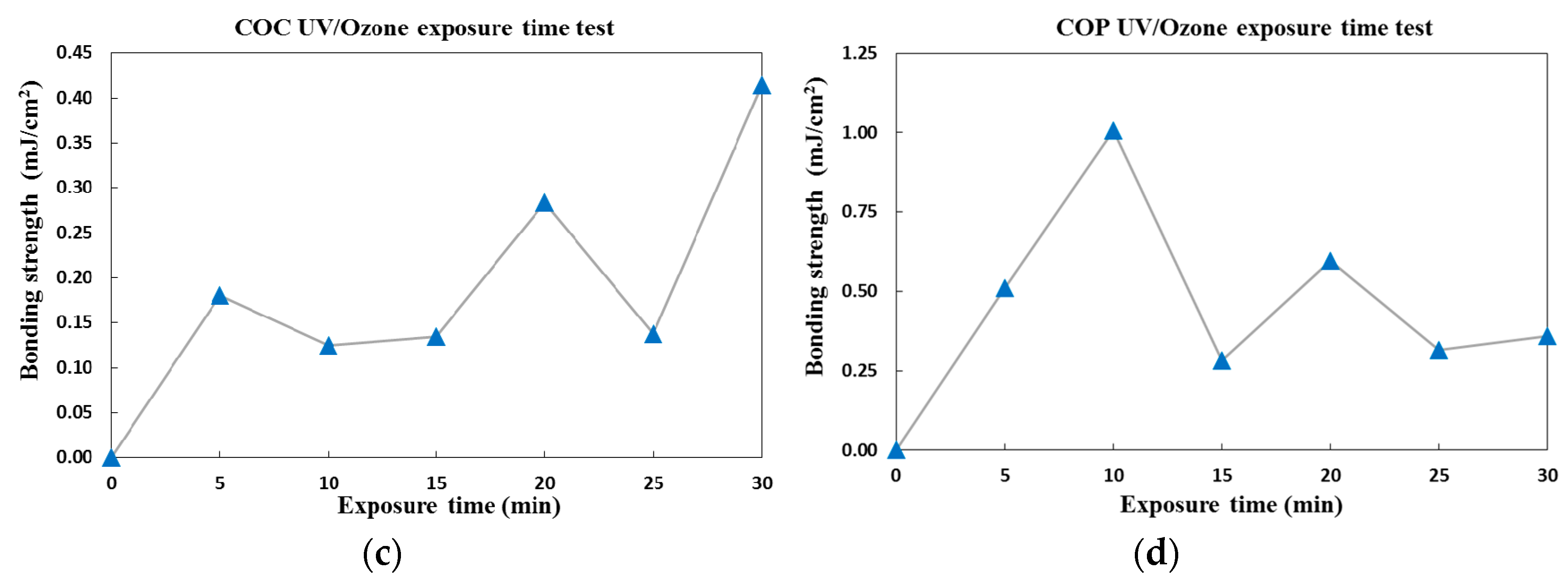
3.3.2. Surface Treatment Bonding of CBC Microchannels through UV/ozone Modification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sackmann, E.K.; Fulton, A.L.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Pandey, C.M.; Augustine, S.; Kumar, S.; Kumar, S.; Nara, S.; Srivastava, S.; Malhotra, B.D. Microfluidics based point-of-care diagnostics. Biotechnol. J. 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Kuo, J.S.; Chiu, D.T. Disposable microfluidic substrates: Transitioning from the research laboratory into the clinic. Lab Chip 2011, 11, 2656–2665. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.J.; Zhu, Z.X.; Ni, Z.H.; Xiang, N.; Yi, H. Inexpensive, rapid fabrication of polymer-film microfluidic autoregulatory valve for disposable microfluidics. Biomed. Microdevices 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Kim, J.; Jeon, C.-W.; Han, K.-H. A disposable microfluidic device with a reusable magnetophoretic functional substrate for isolation of circulating tumor cells. Lab Chip 2017, 17, 4113–4123. [Google Scholar] [CrossRef] [PubMed]
- Becker, H.; Gartner, C. Polymer microfabrication technologies for microfluidic systems. Anal. Bioanal. Chem. 2008, 390, 89–111. [Google Scholar] [CrossRef] [PubMed]
- Becker, H.; Locascio, L.E. Polymer microfluidic devices. Talanta 2002, 56, 267–287. [Google Scholar] [CrossRef]
- Qin, D.; Xia, Y.; Whitesides, G.M. Soft lithography for micro- and nanoscale patterning. Nat. Protocols 2010, 5, 491–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xia, Y.N.; Whitesides, G.M. Soft lithography. Angew. Chem. Int. Ed. 1998, 37, 550–575. [Google Scholar] [CrossRef]
- Eddings, M.A.; Johnson, M.A.; Gale, B.K. Determining the optimal pdms-pdms bonding technique for microfluidic devices. J. Micromech. Microeng. 2008, 18, 067001. [Google Scholar] [CrossRef]
- Velve-Casquillas, G.; Le Berre, M.; Piel, M.; Tran, P.T. Microfluidic tools for cell biological research. Nano Today 2010, 5, 28–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halldorsson, S.; Lucumi, E.; Gomez-Sjoberg, R.; Fleming, R.M.T. Advantages and challenges of microfluidic cell culture in polydimethylsiloxane devices. Biosens. Bioelectron. 2015, 63, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, R. When pdms isn‘t the best. Anal. Chem. 2007, 79, 3248–3253. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ganser, D.; Schneider, A.; Liu, R.; Grodzinski, P.; Kroutchinina, N. Microfabricated polycarbonate ce devices for DNA analysis. Anal. Chem. 2001, 73, 4196–4201. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.L.; Chen, X.; Yao, Z.; Chen, X.D.; Fu, B.D.; Zhang, L. Fabricated polycarbonate microchannel with different films using CO2 laser beam of two-pass for microfluidic chip. Microsyst. Technol. 2018, 24, 2325–2331. [Google Scholar] [CrossRef]
- Witek, M.A.; Hupert, M.L.; Park, D.S.W.; Fears, K.; Murphy, M.C.; Soper, S.A. 96-well polycarbonate-based microfluidic titer plate for high-throughput purification of DNA and rna. Anal. Chem. 2008, 80, 3483–3491. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.P.; Xu, T.; Chen, X.M.; Lin, S.; Cho, M.; Sun, D.; Yang, M.S. Effects of direct current electric fields on lung cancer cell electrotaxis in a pmma-based microfluidic device. Anal. Bioanal. Chem. 2017, 409, 2163–2178. [Google Scholar] [CrossRef] [PubMed]
- Liga, A.; Morton, J.A.S.; Kersaudy-Kerhoas, M. Safe and cost-effective rapid-prototyping of multilayer pmma microfluidic devices. Microfluid. Nanofluid. 2016, 20, 164. [Google Scholar] [CrossRef]
- Tan, H.Y.; Loke, W.K.; Nguyen, N.-T. A reliable method for bonding polydimethylsiloxane (pdms) to polymethylmethacrylate (pmma) and its application in micropumps. Sens. Actuators B Chem. 2010, 151, 133–139. [Google Scholar] [CrossRef]
- Azouz, A.B.; Murphy, S.; Karazi, S.; Vázquez, M.; Brabazon, D. Fast fabrication process of microfluidic devices based on cyclic olefin copolymer. Mater. Manuf. Process. 2014, 29, 93–99. [Google Scholar] [CrossRef] [Green Version]
- Jena, R.K.; Yue, C.Y.; Lam, Y.C. Micro fabrication of cyclic olefin copolymer (COC) based microfluidic devices. Microsyst. Technol. 2011, 18, 159–166. [Google Scholar] [CrossRef]
- Zhang, J.; Das, C.; Fan, Z.H. Dynamic coating for protein separation in cyclic olefin copolymer microfluidic devices. Microfluid. Nanofluid. 2007, 5, 327–335. [Google Scholar] [CrossRef]
- Steigert, J.; Haeberle, S.; Brenner, T.; Müller, C.; Steinert, C.P.; Koltay, P.; Gottschlich, N.; Reinecke, H.; Rühe, J.; Zengerle, R.; et al. Rapid prototyping of microfluidic chips in COC. J. Micromech. Microeng. 2007, 17, 333–341. [Google Scholar] [CrossRef]
- Pentecost, A.M.; Martin, R.S. Fabrication and characterization of all-polystyrene microfluidic devices with integrated electrodes and tubing. Anal. Methods 2015, 7, 2968–2976. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Goral, V.N.; DeRosa, M.E.; Huang, T.J.; Yuen, P.K. A polystyrene-based microfluidic device with three-dimensional interconnected microporous walls for perfusion cell culture. Biomicrofluidics 2014, 8, 046505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, A.S.; Anderson, K.B.; Halpin, S.T.; Kirkpatrick, D.C.; Spence, D.M.; Martin, R.S. Integration of multiple components in polystyrene-based microfluidic devices part I: Fabrication and characterization. Analyst 2013, 138, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Li, J.M.; Liu, C.; Qiao, H.C.; Zhu, L.Y.; Chen, G.; Dai, X.D. Hot embossing/bonding of a poly(ethylene terephthalate) (PET) microfluidic chip. J. Micromech. Microeng. 2008, 18, 015008. [Google Scholar] [CrossRef]
- Hu, Z.L.; Chen, X.Y. Fabrication of polyethylene terephthalate microfluidic chip using CO2 laser system. Int. Polym. Proc. 2018, 33, 106–109. [Google Scholar] [CrossRef]
- Tsao, C.W. Polymer microfluidics: Simple, low-cost fabrication process bridging academic lab research to commercialized production. Micromachines 2016, 7, 225. [Google Scholar] [CrossRef]
- Nunes, P.S.; Ohlsson, P.D.; Ordeig, O.; Kutter, J.P. Cyclic olefin polymers: Emerging materials for lab-on-a-chip applications. Microfluid. Nanofluid. 2010, 9, 145–161. [Google Scholar] [CrossRef]
- Becker, H.; Gartner, C. Polymer microfabrication methods for microfluidic analytical applications. Electrophoresis 2000, 21, 12–26. [Google Scholar] [CrossRef]
- Tsao, C.W.; DeVoe, D.L. Bonding of thermoplastic polymer microfluidics. Microfluid. Nanofluid. 2009, 6, 1–16. [Google Scholar] [CrossRef]
- Temiz, Y.; Lovchik, R.D.; Kaigala, G.V.; Delamarche, E. Lab-on-a-chip devices: How to close and plug the lab? Microelectron. Eng. 2015, 132, 156–175. [Google Scholar] [CrossRef]
- Maszara, W.P.; Goetz, G.; Caviglia, A.; McKitterick, J.B. Bonding of silicon wafers for silicon-on-insulator. J. Appl. Phys. 1988, 64, 4943. [Google Scholar] [CrossRef]
- Tsao, C.W.; Hromada, L.; Liu, J.; Kumar, P.; DeVoe, D.L. Low temperature bonding of pmma and coc microfluidic substrates using uv/ozone surface treatment. Lab Chip 2007, 7, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, H.; Mizuno, J.; Shoji, S. Studies on low-temperature direct bonding of vuv, vuv/o3 and o2 plasma pretreated cyclo-olefin polymer. Sens. Actuators A Phys. 2011, 165, 124–131. [Google Scholar] [CrossRef]




| Bonding pressure (kg/cm2) | 30 |
| Bonding time (min) | 20 |
| CBC-1 * Bonding temperature (°C) | 27/37/47/57/67/77/87/97/107/117 |
| CBC-2 Bonding temperature (°C) | 25/35/45/55/65/75/85/95/105/115 |
| COC * Bonding temperature (°C) | 28/38/48/58/68/78 |
| COP * Bonding temperature (°C) | 30/40/50/60/70/80/90/100 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yen, C.-Y.; Chang, M.-C.O.; Shih, Z.-F.; Lien, Y.-H.; Tsao, C.-W. Cyclic Block Copolymer Microchannel Fabrication and Sealing for Microfluidics Applications. Inventions 2018, 3, 49. https://doi.org/10.3390/inventions3030049
Yen C-Y, Chang M-CO, Shih Z-F, Lien Y-H, Tsao C-W. Cyclic Block Copolymer Microchannel Fabrication and Sealing for Microfluidics Applications. Inventions. 2018; 3(3):49. https://doi.org/10.3390/inventions3030049
Chicago/Turabian StyleYen, Chia-Yi, Moh-Ching O. Chang, Zong-Fu Shih, Yi-Hsing Lien, and Chia-Wen Tsao. 2018. "Cyclic Block Copolymer Microchannel Fabrication and Sealing for Microfluidics Applications" Inventions 3, no. 3: 49. https://doi.org/10.3390/inventions3030049





