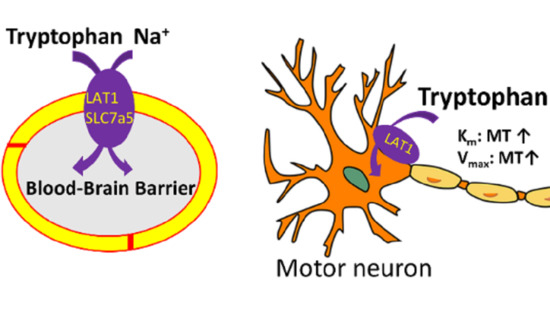
Pretreatment Effect of Inflammatory Stimuli and Characteristics of Tryptophan Transport on Brain Capillary Endothelial (TR-BBB) and Motor Neuron Like (NSC-34) Cell Lines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture
2.3. [3H]l-Tryptophan Uptake by NSC-34 and TR-BBB Cells
2.4. Transfection with rLAT1 and rCAT1 Small Interfering RNA (siRNA)
2.5. Pretreatment Effect of Pro-Inflammatory Cytokines on the Uptake of [3H]l-Tryptophan
2.6. Data Analysis
3. Results
3.1. Characteristics of [3H]l-Tryptophan Transport in TR-BBB Cell Lines
3.2. LAT1 and CAT1 siRNA Transfection Study in TR-BBB Cell Lines
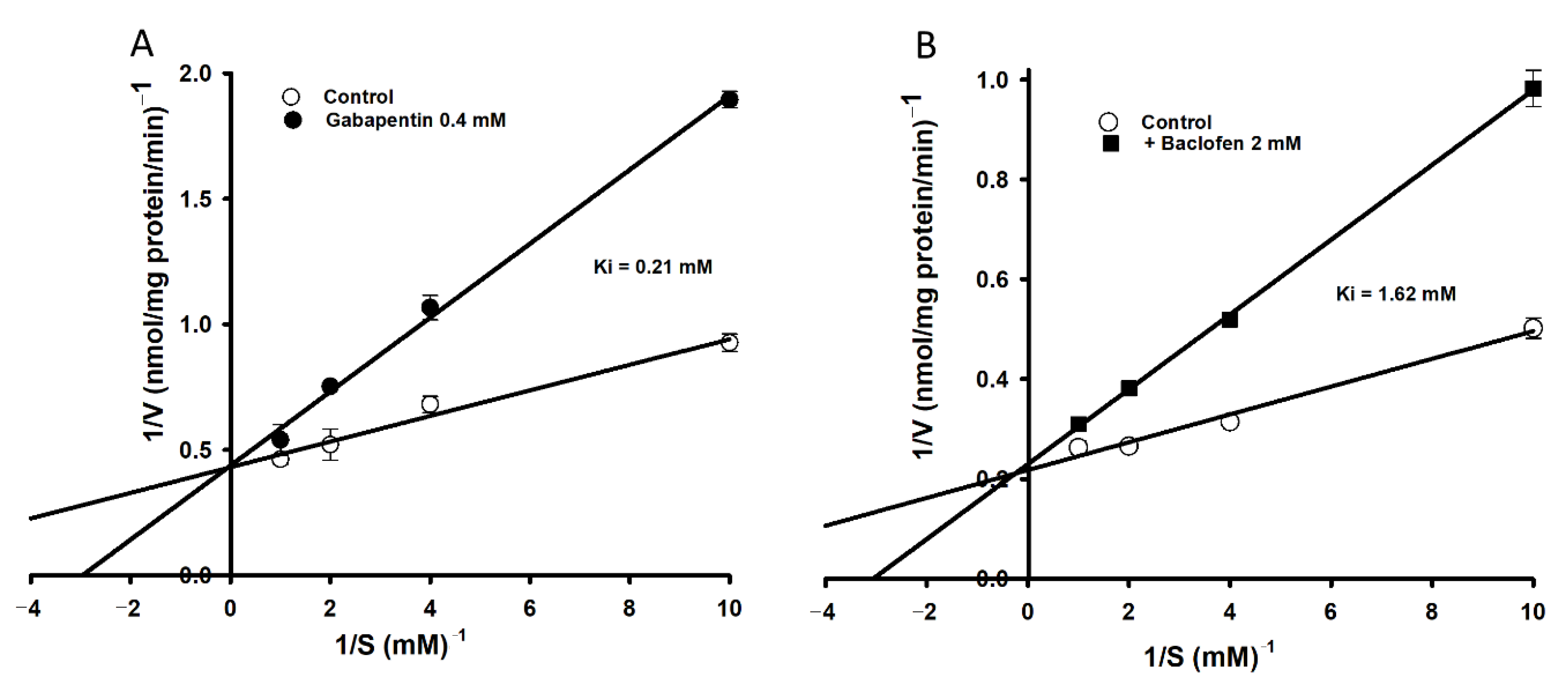
3.3. Effect of Pharmacological Drugs on the Uptake of [3H]l-Tryptophan by TR-BBB Cell Lines
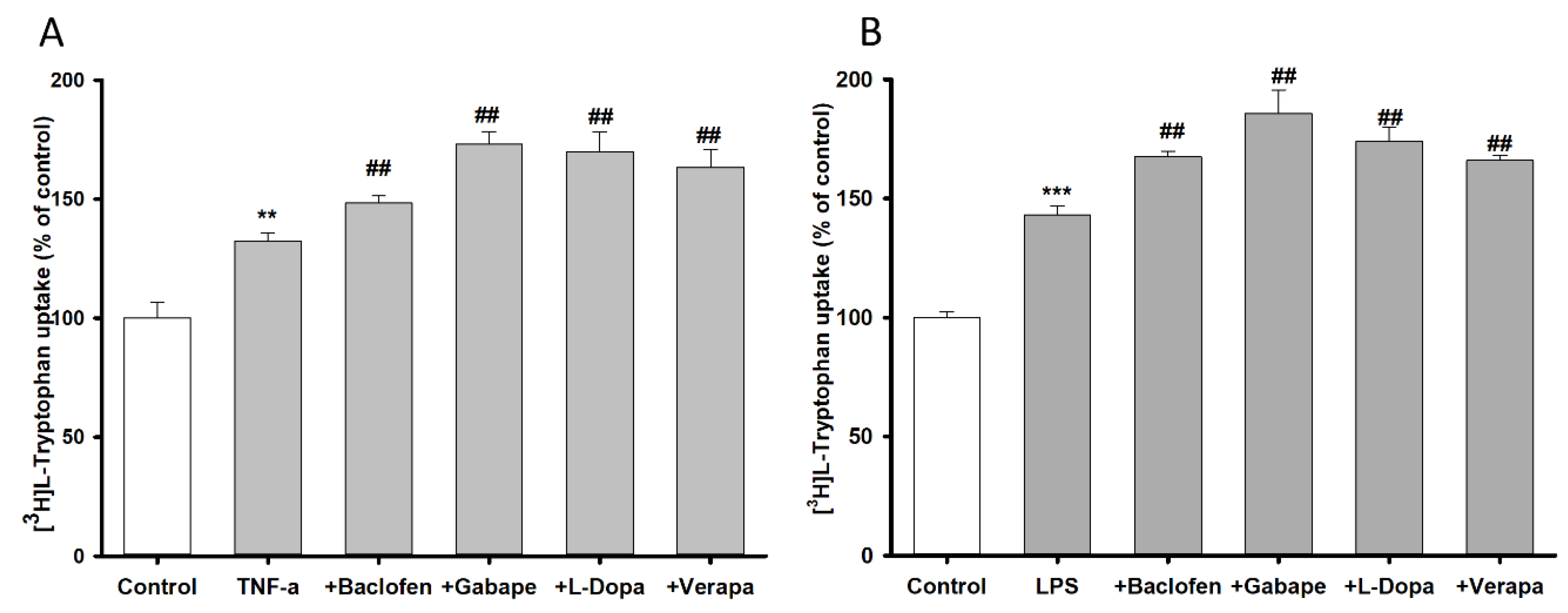
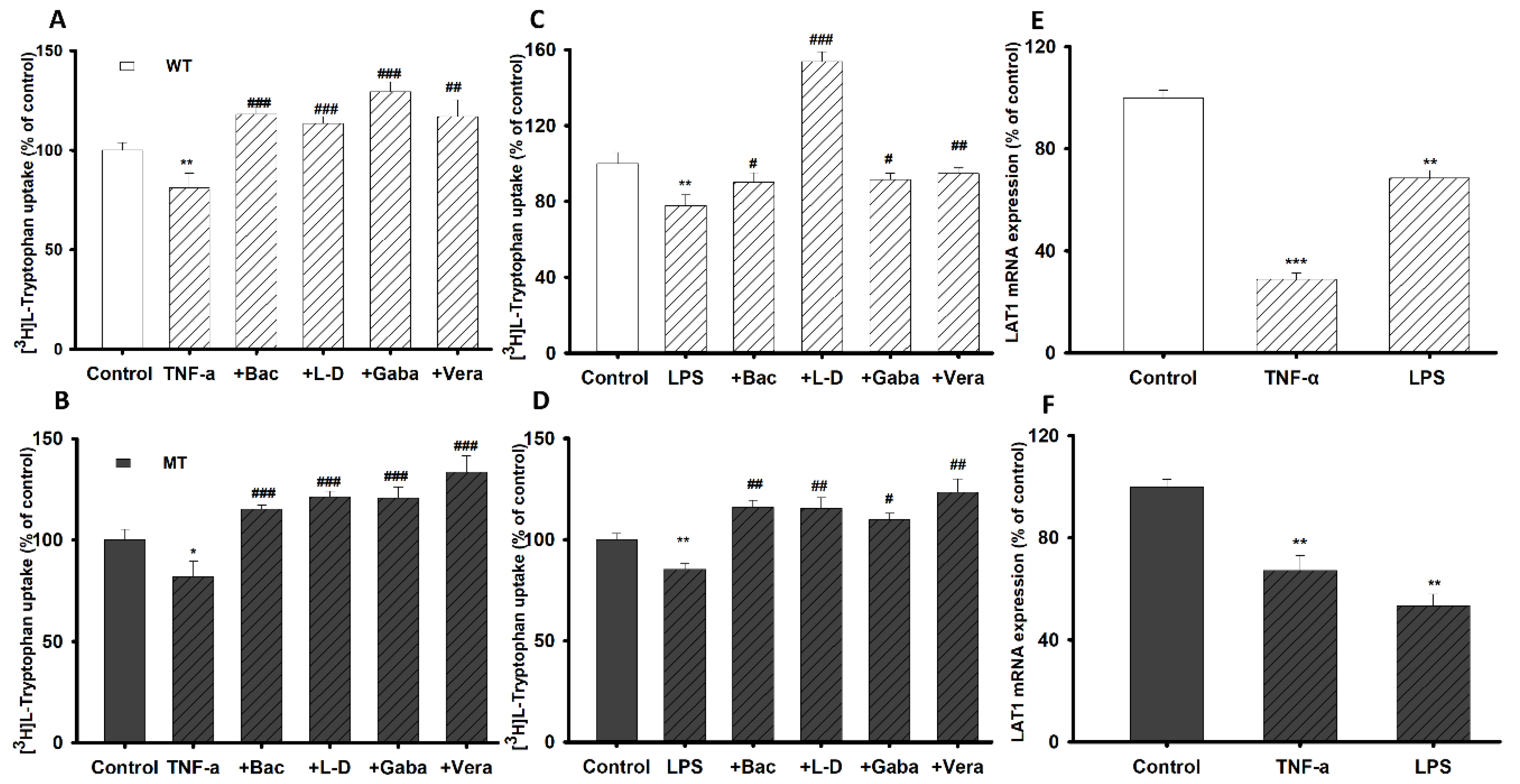
3.4. Pretreatment Effect of Inflammatory Stimuli on [3H]l-Tryptophan Uptake by TR-BBB Cell Lines
3.5. Changes in [3H]l-Tryptophan Uptake by NSC-34 Cell Lines.
3.6. Effect of LAT1 siRNA Transfection on NSC-34 Cell Lines
3.7. Pretreatment Effect of Inflammatory Stimuli on [3H]l-Tryptophan Uptake by NSC-34 Cell Lines
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALS | Amyotrophic lateral sclerosis |
| AD | Alzheimer’s disease |
| 5-HT | 5-Hydroxytryptamine; serotonin |
| BBB | Blood-brain barrier |
| BCH | 2-Aminobicyclo heptane-2-carboxylic acid |
| CAT1 | Cationic amino acid transporter 1 |
| CNS | Central nervous system |
| ECF | Extracellular fluid |
| IDO | Indoleamine 2, 3-dioxygenase |
| IF-γ | Interferon-γ |
| LNAA | Large neutral amino acids |
| LAT1 | l-type amino acid transporter 1 |
| MeAIB | α-Methylaminoisobutyric acid |
| NAA | Neutral amino acids |
| NMG | N-methyl d-glucamine |
| ND | Neurodegenerative disease |
| PD | Parkinson’s disease |
| RT-PCR | Real-time polymerase chain reaction |
| TDO | Tryptophan 2, 3-dioxygenase |
| TNF-α | Tumor necrosis factor α |
References
- Friedman, M. Analysis, nutrition, and health benefits of tryptophan. Int. J. Tryptophan Res. 2018, 11, 1178646918802282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhutia, Y.D.; Babu, E.; Ganapathy, V. Interferon-γ induces a tryptophan-selective amino acid transporter in human colonic epithelial cells and mouse dendritic cells. Biochim. Biophys. Acta Biomembr. 2015, 1848, 453–462. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, T.A.; Nguyen, J.C.D.; Polglaze, K.E.; Bertrand, P.P. Influence of tryptophan and serotonin on mood and cognition with a possible role of the gut-brain axis. Nutrients 2016, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Keegan, M.R.; Winston, A.; Higgs, C.; Fuchs, D.; Boasso, A.; Nelson, M. Tryptophan metabolism and its relationship with central nervous system toxicity in people living with HIV switching from efavirenz to dolutegravir. J. Neurovirol. 2019, 25, 85–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seymour, R.L.; Ganapathy, V.; Mellor, A.L.; Munn, D.H. A high-affinity, tryptophan-selective amino acid transport system in human macrophages. J. Leukoc. Biol. 2006, 80, 1320–1327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mackay, G.M.; Forrest, C.M.; Stoy, N.; Christofides, J.; Egerton, M.; Stone, T.W.; Darlington, L.G. Tryptophan metabolism and oxidative stress in patients with chronic brain injury. Eur. J. Neurol. 2006, 13, 30–42. [Google Scholar] [CrossRef]
- Davies, N.W.S.; Guillemin, G.; Brew, B.J. Tryptophan, neurodegeneration and HIV-associated neurocognitive disorder. Int. J. Tryptophan Res. 2010, 3, 121–140. [Google Scholar] [CrossRef] [Green Version]
- Ruddick, J.P.; Evans, A.K.; Nutt, D.J.; Lightman, S.L.; Rook, G.A.W.; Lowry, C.A. Tryptophan metabolism in the central nervous system: Medical implications. Expert Rev. Mol. Med. 2006, 8, 1–27. [Google Scholar] [CrossRef]
- Davis, I.; Liu, A. What is the tryptophan kynurenine pathway and why is it important to neurotherapeutics? Expert Rev. Neurother. 2015, 15, 719–721. [Google Scholar] [CrossRef] [Green Version]
- Wirthgen, E.; Hoeflich, A.; Rebl, A.; Günther, J. Kynurenic acid: The Janus-faced role of an immunomodulatory tryptophan metabolite and its link to pathological conditions. Front. Immunol. 2018, 8, 1957–1969. [Google Scholar] [CrossRef] [Green Version]
- Campbell, B.M.; Charych, E.; Lee, A.W.; Möller, T. Kynurenines in CNS disease: Regulation by inflammatory cytokines. Front. Neurosci. 2014, 8, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, V.X.; Guillemin, G.J. Kynurenine pathway metabolites as biomarkers for amyotrophic lateral sclerosis. Front. Neurosci. 2019, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, K.; Baig, M.H.; Mushtaq, G.; Kamal, M.A.; Greig, N.H.; Choi, I. Commonalities in biological pathways, genetics, and cellular mechanism between Alzheimer disease and other neurodegenerative diseases: An in silico-updated overview. Curr. Alzheimer Res. 2017, 14, 1190–1197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vermeiren, Y.; Janssens, J.; Van Dam, D.; De Deyn, P.P. Serotonergic dysfunction in amyotrophic lateral sclerosis and Parkinson’s disease: Similar mechanisms, dissimilar outcomes. Front. Neurosci. 2018, 12, 185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banks, W.A. The Blood Brain Barrier. J. Neuroimmune Pharmacol. 2008, 21–38. [Google Scholar]
- Terasaki, T.; Hosoya, K.I. Conditionally immortalized cell lines as a new in vitro model for the study of barrier functions. Biol. Pharm. Bull. 2001, 24, 111–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richard, D.M.; Dawes, M.A.; Mathias, C.W.; Acheson, A.; Hill-Kapturczak, N.; Dougherty, D.M. L-Tryptophan: Basic metabolic functions, behavioral research and therapeutic indications. Int. J. Tryptophan Res. 2009, 2, 45–60. [Google Scholar] [CrossRef] [Green Version]
- Boado, R.J.; Li, J.Y.; Nagaya, M.; Zhang, C.; Pardridge, W.M. Selective expression of the large neutral amino acid transporter at the blood-brain barrier. Proc. Natl. Acad. Sci. USA 1999, 96, 12079–12084. [Google Scholar] [CrossRef] [Green Version]
- Scalise, M.; Galluccio, M.; Console, L.; Pochini, L.; Indiveri, C. The human SLC7A5 (LAT1): The intriguing histidine/large neutral amino acid transporter and its relevance to human health. Front. Chem. 2018, 6, 243. [Google Scholar] [CrossRef]
- Kärkkäinen, J.; Gynther, M.; Kokkola, T.; Petsalo, A.; Auriola, S.; Lahtela-Kakkonen, M.; Laine, K.; Rautio, J.; Huttunen, K.M. Structural properties for selective and efficient l-type amino acid transporter 1 (LAT1) mediated cellular uptake. Int. J. Pharm. 2018, 544, 91–99. [Google Scholar] [CrossRef] [Green Version]
- Huttunen, J.; Peltokangas, S.; Gynther, M.; Natunen, T.; Hiltunen, M.; Auriola, S.; Ruponen, M.; Vellonen, K.S.; Huttunen, K.M. L-type amino acid transporter 1 (LAT1/Lat1)-utilizing prodrugs can improve the delivery of drugs into neurons, astrocytes and microglia. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tina, E.; Prosén, S.; Lennholm, S.; Gasparyan, G.; Lindberg, M.; Göthlin Eremo, A. Expression profile of the amino acid transporters SLC7A5, SLC7A7, SLC7A8 and the enzyme TDO2 in basal cell carcinoma. Br. J. Dermatol. 2019, 180, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuki, S.; Yamaguchi, H.; Kang, Y.S.; Hori, S.; Terasaki, T. Reduction of L-type amino acid transporter 1 mRNA Expression in brain capillaries in a mouse model of Parkinson’s disease. Biol. Pharm. Bull. 2010, 33, 1250–1252. [Google Scholar] [CrossRef] [Green Version]
- Gyawali, A.; Gautam, S.; Hyeon, J.; Ryu, H. L-Citrulline level and transporter activity are altered in experimental models of amyotrophic lateral sclerosis. Mol. Neurobiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.K.; Kim, K.Y.; Lee, N.Y.; Kang, Y.S.; Hwang, Y.J.; Kim, Y.; Sung, J.J.; McKee, A.; Kowall, N.; Lee, J.R.H. Expression of taurine transporter (TauT) is modulated by heat shock factor 1 (HSF1) in motor neurons of ALS. Mol. Neurobiol. 2013, 47, 699–710. [Google Scholar] [CrossRef]
- Kang, Y.S.; Lee, K.E.; Lee, N.Y.; Terasaki, T. Donepezil, tacrine and -phenyl-n-tert-butyl nitrone (PBN) inhibit choline transport by conditionally immortalized rat. Arch. Pharm. Res. 2005, 28, 443–450. [Google Scholar] [CrossRef]
- Lee, K.E.; Kang, Y.S. Characteristics of L-citrulline transport through blood-brain barrier in the brain capillary endothelial cell line (TR-BBB Cells). J. Biomed. Sci. 2017, 24, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Gyawali, A.; Kang, Y.S. Blood-to-retina transport of imperatorin involves the carrier-mediated transporter system at the inner blood-retinal barrier. J. Pharm. Sci. 2019, 108, 1619–1626. [Google Scholar] [CrossRef]
- Kang, Y.S.; Ohtsuki, S.; Takanaga, H.; Tomi, M.; Hosoya, K.; Terasaki, T. Regulation of taurine transport at the blood-brain barrier by tumor necrosis factor-α, taurine and hypertonicity. J. Neurochem. 2002, 83, 1188–1195. [Google Scholar] [CrossRef]
- Gyawali, A.; Kang, Y.S. Transport alteration of 4-phenyl butyric acid mediated by a sodium- and proton-coupled monocarboxylic acid transporter system in ALS model cell lines (NSC-34) under inflammatory states. J. Pharm. Sci. 2020, 1–11. [Google Scholar] [CrossRef]
- Gyawali, A.; Kang, Y.S. The effect of drug pre-treatment on taurine transport at the inner blood-retinal barrier under variable conditions. Adv. Exp. Med. Biol. 2019, 1155, 959–975. [Google Scholar] [PubMed]
- Ravi, V.; Jessica, J.; Tommy, L.; Nikolaos, V. Tryptophan transport in human fibroblast cells- a functional characterization. Int. J. Tryptophan Res. 2011, 4, 19–27. [Google Scholar]
- del Amo, E.M.; Urtti, A.; Yliperttula, M. Pharmacokinetic role of L-type amino acid transporters LAT1 and LAT2. Eur. J. Pharm. Sci. 2008, 35, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Barar, J.; Rafi, M.A.; Pourseif, M.M.; Omidi, Y. Blood-brain barrier transport machineries and targeted therapy of brain diseases. BioImpacts 2016, 6, 225–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alles, S.R.A.; Gomez, K.; Moutal, A.; Khanna, R. Putative roles of SLC7A5 (LAT1) transporter in pain. Neurobiol. Pain 2020, 8, 100050. [Google Scholar] [CrossRef] [PubMed]
- Bongiovanni, R.; Mchaourab, A.S.; McClellan, F.; Elsworth, J.; Double, M.; Jaskiw, G.E. Large neutral amino acids levels in primate cerebrospinal fluid do not confirm competitive transport under baseline conditions. Brain Res. 2016, 1648, 372–379. [Google Scholar] [CrossRef]
- Smith, Q.R. Transport of glutamate and other amino acids at the blood-brain barrier. J. Nutr. 2000, 130, 1016–1022. [Google Scholar] [CrossRef]
- Liu, Y.; Lo, Y.C.; Qian, L.; Crews, F.T.; Wilson, B.; Chen, H.L.; Wu, H.M.; Chen, S.H.; Wei, K.; Lu, R.B.; et al. Verapamil protects dopaminergic neuron damage through a novel anti-inflammatory mechanism by inhibition of microglial activation. Neuropharmacology 2011, 60, 373–380. [Google Scholar] [CrossRef] [Green Version]
- Haddad, F.; Sawalha, M.; Khawaja, Y.; Najjar, A.; Karaman, R. Dopamine and levodopa prodrugs for the treatment of Parkinson’s disease. Molecules 2018, 23, 40. [Google Scholar] [CrossRef] [Green Version]
- Abdel-Salam, O.M.; Sleem, A.A. Study of the analgesic, anti-inflammatory, and gastric effects of gabapentin. Drug Discov. Ther. 2009, 3, 18–26. [Google Scholar]
- Pilipenko, V.; Narbute, K.; Beitnere, U.; Rumaks, J.; Pupure, J.; Jansone, B.; Klusa, V. Very low doses of muscimol and baclofen ameliorate cognitive deficits and regulate protein expression in the brain of a rat model of streptozocin-induced Alzheimer’s disease. Eur. J. Pharmacol. 2018, 818, 381–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chataway, J.; De Angelis, F.; Connick, P.; Parker, R.A.; Plantone, D.; Doshi, A.; John, N.; Stutters, J.; MacManus, D.; Prados Carrasco, F.; et al. Efficacy of three neuroprotective drugs in secondary progressive multiple sclerosis (MS-SMART): A phase 2b, multiarm, double-blind, randomised placebo-controlled trial. Lancet Neurol. 2020, 19, 214–225. [Google Scholar] [CrossRef] [Green Version]
- Pardridge, W.M. Blood-brain barrier endogenous transporters as therapeutic targets: A new model for small molecule CNS drug discovery. Expert Opin. Ther. Targets 2015, 19, 1059–1072. [Google Scholar] [CrossRef] [PubMed]
- O’Kane, R.L.; Viña, J.R.; Simpson, I.; Zaragozá, R.; Mokashi, A.; Hawkins, R.A. Cationic amino acid transport across the blood-brain barrier is mediated exclusively by system Y+. Am. J. Physiol. Endocrinol. Metab. 2006, 291, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vumma, R.; Johansson, J.; Venizelos, N. Proinflammatory cytokines and oxidative stress decrease the transport of dopamine precursor tyrosine in human fibroblasts. Neuropsychobiology 2018, 75, 178–184. [Google Scholar] [CrossRef]
- Clark, I.A.; Vissel, B. Excess cerebral TNF causing glutamate excitotoxicity rationalizes treatment of neurodegenerative diseases and neurogenic pain by anti-TNF agents. J. Neuroinflammation 2016, 13, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Batista, C.R.A.; Gomes, G.F.; Candelario-Jalil, E.; Fiebich, B.L.; de Oliveira, A.C.P. Lipopolysaccharide-induced neuroinflammation as a bridge to understand neurodegeneration. Int. J. Mol. Sci. 2019, 20, 2293. [Google Scholar] [CrossRef] [Green Version]
- Zoccolella, S.; Santamato, A.; Lamberti, P. Current and emerging treatments for amyotrophic lateral sclerosis. Neuropsychiatr. Dis. Treat. 2009, 5, 577–595. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Chen, S.; Lu, K.; Wang, F.; Deng, J.; Xu, Z.; Wang, X.; Zhou, Q.; Le, W.; Zhao, Y. Verapamil ameliorates motor neuron degeneration and improves lifespan in the SOD1G93A mouse model of Als by enhancing autophagic flux. Aging Dis. 2019, 10, 1159–1173. [Google Scholar] [CrossRef] [Green Version]


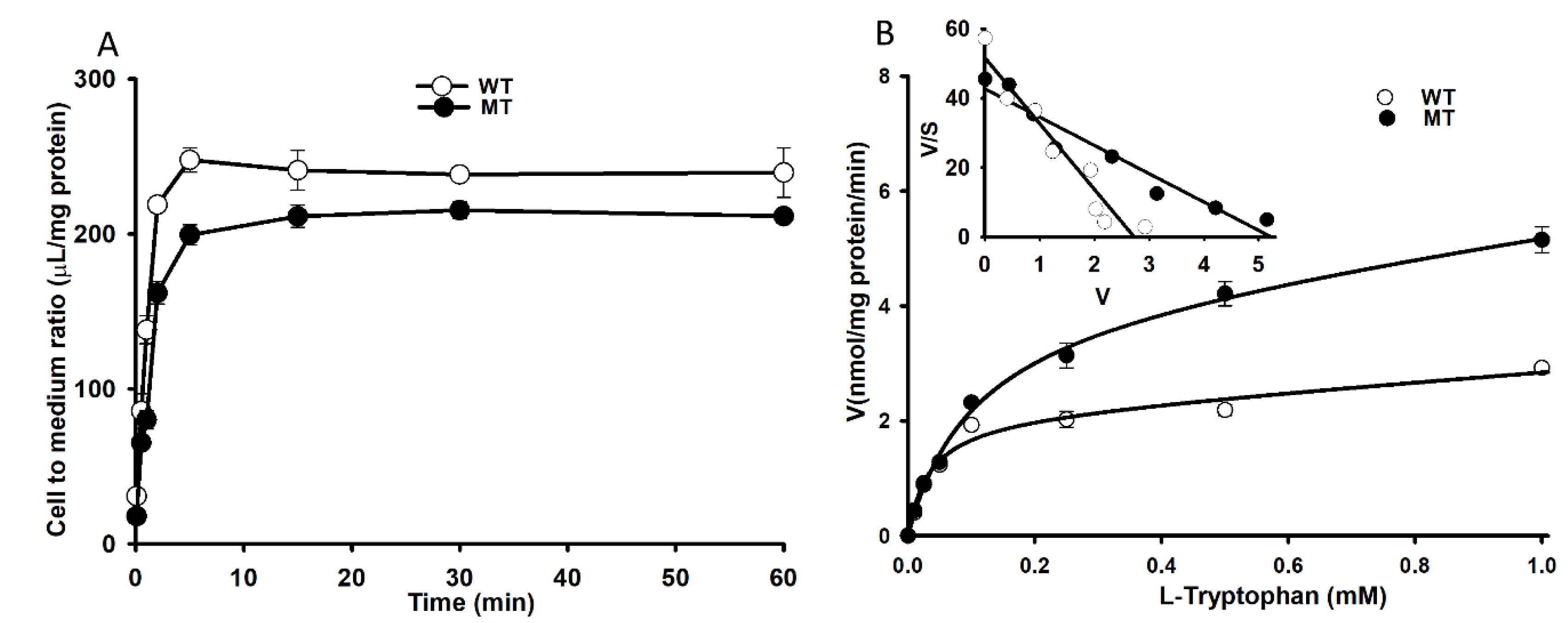

| Cell lines | WT (NSC-34 hSOD1WT) | MT (NSC-34 hSOD1G93A) | TR-BBB |
|---|---|---|---|
| Km (µM) | 33.1 ± 1.2 | 101 ± 2 | 121 ± 11 |
| Vmax (nmol/mg protein/min) | 2.10 ± 0.27 | 4.07 ± 0.42 | 4.69 ± 0.20 |
| Kd (µL/mg protein/min) | 0.805 | 1.46 | 3.66 |
| Vmax/Km (µL/mg protein/min) | 0.063 | 0.041 | 0.704 |
| Compounds | Concentration | Relative Uptake |
|---|---|---|
| mM | (% of Control) | |
| Control | 100 ± 4 | |
| + BCH | 2 | 21.0 ± 1.3 *** |
| + d-Methionine | 2 | 30.8 ± 3.4 *** |
| + Methylmaleimide | 2 | 60.4 ± 1.2 *** |
| + Homoarginine | 2 | 88.4 ± 3.4 |
| + MeAIB | 2 | 96.8 ± 2.7 |
| + Harmaline | 2 | 108 ± 5 |
| Compounds | Concentration | Relative Uptake |
|---|---|---|
| mM | (% of Control) | |
| Control | 100 ± 4 | |
| + Tryptophan | 2 | 7.27 ± 0.9 *** |
| + Phenylalanine | 2 | 8.68 ± 0.3 *** |
| + Tyrosine | 2 | 14.1 ± 1.0 *** |
| + Methionine | 2 | 10.3 ± 7.4 *** |
| + Glutamine | 2 | 19.4 ± 1.2 *** |
| + Citrulline | 2 | 27.3 ± 1.4 *** |
| + Valine | 2 | 38.4 ± 1.8 *** |
| + Cysteine | 2 | 41.3 ± 1.3 *** |
| + Lysine | 2 | 53.1 ± 4.1 *** |
| + Serine | 2 | 58.6 ± 1.5 *** |
| + Aspartic acid | 2 | 90.2 ± 3.4 |
| + Glutamic acid | 2 | 97.4 ± 3.8 |
| + Taurine | 2 | 102 ± 5 |
| + Choline | 2 | 110 ± 4 |
| Drugs | Concentration | Relative Uptake |
|---|---|---|
| mM | (% of Control) | |
| Control | 100 ± 4 | |
| + Verapamil | 0.5 | 15.0 ± 0.9 *** |
| + l-Dopa | 0.5 | 18.6 ± 1.6 *** |
| + Gabapentin | 0.5 | 46.0 ± 0.5 *** |
| + Baclofen | 2 | 73.1 ± 5.3 ** |
| + Quinidine | 0.5 | 78.5 ± 5 ** |
| + Dopamine | 2 | 94.4 ± 3.3 |
| + Donepezil | 2 | 97.1 ± 7.6 |
| + Riluzole | 0.5 | 102 ± 8 |
| + Amiloride | 2 | 111 ± 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gyawali, A.; Kang, Y.-S. Pretreatment Effect of Inflammatory Stimuli and Characteristics of Tryptophan Transport on Brain Capillary Endothelial (TR-BBB) and Motor Neuron Like (NSC-34) Cell Lines. Biomedicines 2021, 9, 9. https://doi.org/10.3390/biomedicines9010009
Gyawali A, Kang Y-S. Pretreatment Effect of Inflammatory Stimuli and Characteristics of Tryptophan Transport on Brain Capillary Endothelial (TR-BBB) and Motor Neuron Like (NSC-34) Cell Lines. Biomedicines. 2021; 9(1):9. https://doi.org/10.3390/biomedicines9010009
Chicago/Turabian StyleGyawali, Asmita, and Young-Sook Kang. 2021. "Pretreatment Effect of Inflammatory Stimuli and Characteristics of Tryptophan Transport on Brain Capillary Endothelial (TR-BBB) and Motor Neuron Like (NSC-34) Cell Lines" Biomedicines 9, no. 1: 9. https://doi.org/10.3390/biomedicines9010009