New Insights into the Role of Weak Electron–Phonon Coupling in Nanostructured ZnO Thin Films
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
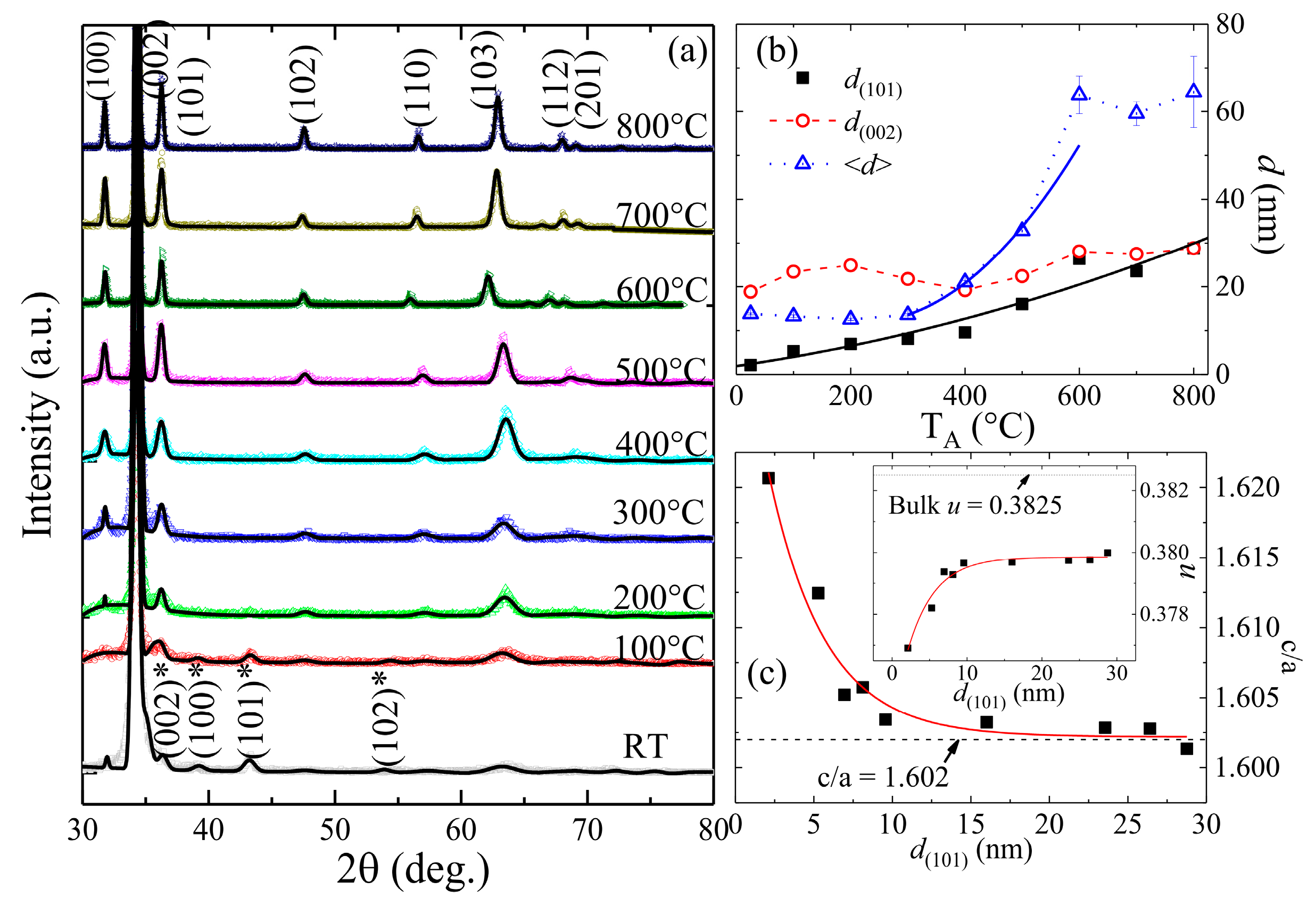
3.1. Morphology and Structural Characterization
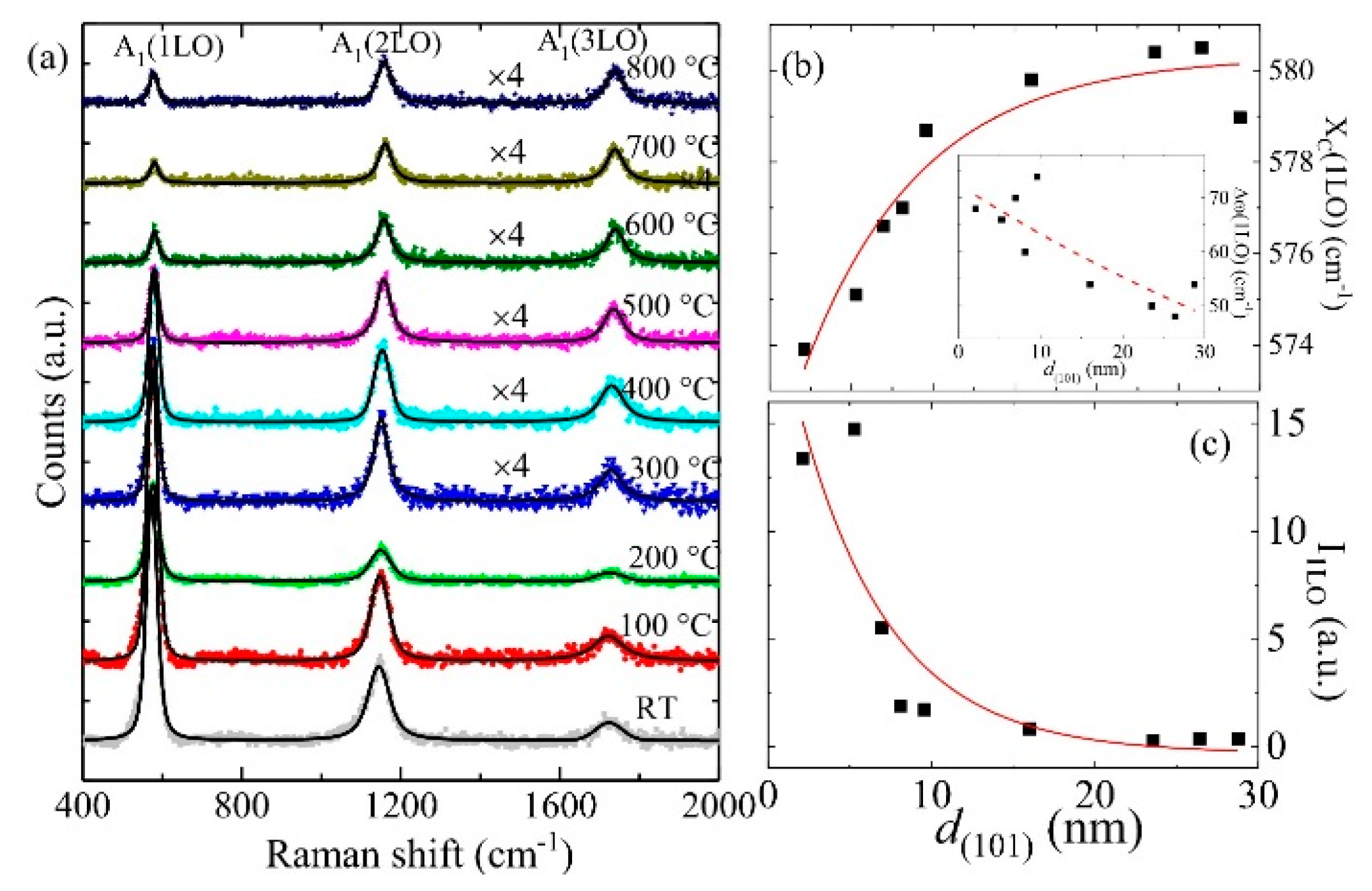
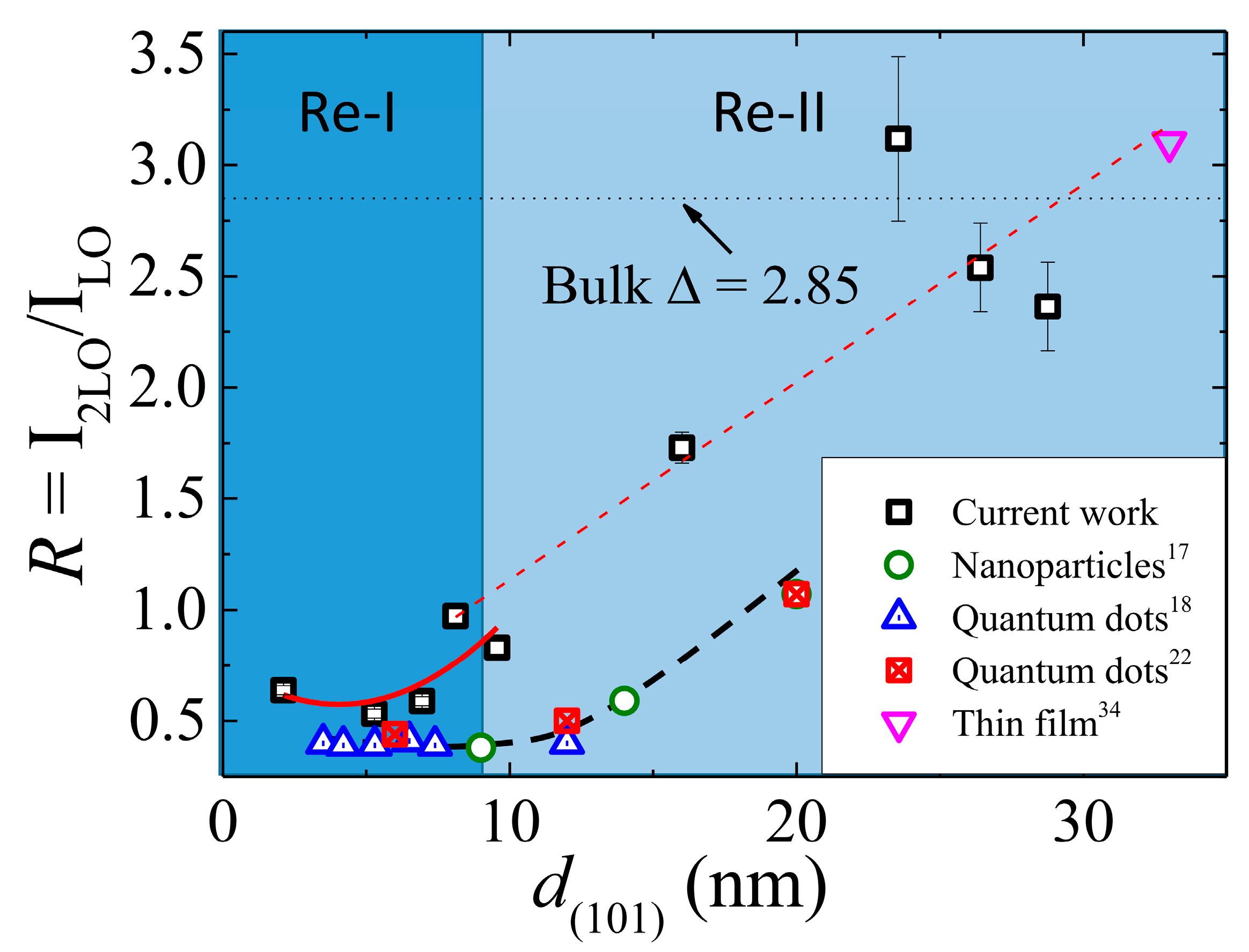
3.2. Electron–Phonon Coupling in ZnO Films
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Muchuweni, E.; Sathiaraj, T.S.; Nyakotyo, H. Effect of gallium doping on the structural, optical and electrical properties of zinc oxide thin films prepared by spray pyrolysis. Ceram. Int. 2016, 42, 10066–10070. [Google Scholar] [CrossRef]
- Muchuweni, E.; Sathiaraj, T.S.; Nyakotyo, H. Low temperature synthesis of radio frequency magnetron sputtered gallium and aluminium co-doped zinc oxide thin films for transparent electrode fabrication. Appl. Surf. Sci. 2016, 390, 570–577. [Google Scholar] [CrossRef]
- Lee, J.-H.; Ko, K.-H.; Park, B.-O. Electrical and optical properties of ZnO transparent conducting films by the sol–gel method. J. Cryst. Growth 2003, 247, 119–125. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, H.; Fu, Z.; Yang, B.; Zuo, J. Correlation between 577 cm−1 Raman scattering and green emission in ZnO ordered nanostructures. Appl. Phys. Lett. 2006, 88, 191909. [Google Scholar] [CrossRef]
- Chassaing, P.-M.; Demangeot, F.; Paillard, V.; Zwick, A.; Combe, N.; Pagès, C.; Kahn, M.L.; Maisonnat, A.; Chaudret, B. Raman study of E2 and surface phonon in zinc oxide nanoparticles surrounded by organic molecules. Appl. Phys. Lett. 2007, 91, 053108. [Google Scholar] [CrossRef]
- Lu, J.G.; Ye, Z.Z.; Zhang, Y.Z.; Liang, Q.L.; Fujita, S.; Wang, Z.L. Self-assembled ZnO quantum dots with tunable optical properties. Appl. Phys. Lett. 2006, 89, 023122. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.P.; Xu, G.; Jin, P. Size dependence of electron-phonon coupling in ZnO nanowires. Phys. Rev. B 2004, 69, 113303. [Google Scholar] [CrossRef]
- Alivisatos, A.P.; Harris, T.D.; Carroll, P.J.; Steigerwald, M.L.; Brus, L.E. Electron–vibration coupling in semiconductor clusters studied by resonance Raman spectroscopy. J. Chem. Phys. 1989, 90, 3463–3468. [Google Scholar] [CrossRef]
- Schmitt-Rink, S.; Miller, D.A.B.; Chemla, D.S. Theory of the linear and nonlinear optical properties of semiconductor microcrystallites. Phys. Rev. B 1987, 35, 8113–8125. [Google Scholar] [CrossRef]
- Marini, J.C.; Stebe, B.; Kartheuser, E. Exciton-phonon interaction in CdSe and CuCl polar semiconductor nanospheres. Phys. Rev. B 1994, 50, 14302–14308. [Google Scholar] [CrossRef]
- Nomura, S.; Kobayashi, T. Exciton—LO-phonon couplings in spherical semiconductor microcrystallites. Phys. Rev. B 1992, 45, 1305–1316. [Google Scholar] [CrossRef]
- Shiang, J.J.; Risbud, S.H.; Alivisatos, A.P. Resonance Raman studies of the ground and lowest electronic excited state in CdS nanocrystals. J. Chem. Phys. 1993, 98, 8432–8442. [Google Scholar] [CrossRef]
- Shiang, J.J.; Wolters, R.H.; Heath, J.R. Theory of size-dependent resonance Raman intensities in InP nanocrystals. J. Chem. Phys. 1997, 106, 8981–8994. [Google Scholar] [CrossRef] [Green Version]
- Haiming, F.; Bingsuo, Z.; Yulong, L.; Sishen, X. Size effect on the electron–phonon coupling in CuO nanocrystals. Nanotechnology 2006, 17, 1099. [Google Scholar]
- Kelley, A.M. Electron-Phonon Coupling in CdSe Nanocrystals from an Atomistic Phonon Model. ACS Nano 2011, 5, 5254–5262. [Google Scholar] [CrossRef] [PubMed]
- Murphy-Armando, F.; Fagas, G.; Greer, J.C. Deformation Potentials and Electron-Phonon Coupling in Silicon Nanowires. Nano Lett. 2010, 10, 869–873. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-M.; Lin, K.-F.; Hsu, H.-C.; Lin, C.-J.; Lin, L.-J.; Hsieh, W.-F. Enhanced Resonant Raman Scattering and Electron−Phonon Coupling from Self-Assembled Secondary ZnO Nanoparticles. J. Phys. Chem. B 2005, 109, 18385–18390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, H.-M.; Lin, K.-F.; Hsu, H.-C.; Hsieh, W.-F. Size dependence of photoluminescence and resonant Raman scattering from ZnO quantum dots. Appl. Phys. Lett. 2006, 88, 261909. [Google Scholar] [CrossRef]
- Hsieh, W.F.; Cheng, H.M.; Lin, K.F.; Hsu, H.-C. Size dependence of band gap variation and electron-phonon coupling in ZnO Quantum Dots. In Proceedings of the Pacific Rim Conference on Lasers and Electro-Optics, Tokyo, Japan, July 2005; pp. 466–468. [Google Scholar]
- Lin, K.-F.; Cheng, H.-M.; Hsu, H.-C.; Hsieh, W.-F. Band gap engineering and spatial confinement of optical phonon in ZnO quantum dots. Appl. Phys. Lett. 2006, 88, 263117. [Google Scholar] [CrossRef]
- Ojha, A.K.; Srivastava, M.; Kumar, S.; Hassanein, R.; Singh, J.; Singh, M.K.; Materny, A. Influence of crystal size on the electron-phonon coupling in ZnO nanocrystals investigated by Raman spectroscopy. Vib. Spectrosc. 2014, 72, 90–96. [Google Scholar] [CrossRef]
- Ray, S.C.; Low, Y.; Tsai, H.M.; Pao, C.W.; Chiou, J.W.; Yang, S.C.; Chien, F.Z.; Pong, W.F.; Tsai, M.-H.; Lin, K.F.; et al. Size dependence of the electronic structures and electron-phonon coupling in ZnO quantum dots. Appl. Phys. Lett. 2007, 91, 262101. [Google Scholar] [CrossRef]
- Gaikwad, S.S.; Gandhi, A.C.; Pandit, S.D.; Pant, J.; Chan, T.-S.; Cheng, C.-L.; Ma, Y.-R.; Wu, S.Y. Oxygen induced strained ZnO nanoparticles: An investigation of Raman scattering and visible photoluminescence. J. Mater. Chem. C 2014, 2, 7264–7274. [Google Scholar] [CrossRef]
- Ismail, A.; Abdullah, M.J. The structural and optical properties of ZnO thin films prepared at different RF sputtering power. J. King Saud Univ. Sci. 2013, 25, 209–215. [Google Scholar] [CrossRef]
- Husna, J.; Aliyu, M.M.; Islam, M.A.; Chelvanathan, P.; Hamzah, N.R.; Hossain, M.S.; Karim, M.R.; Amin, N. Influence of Annealing Temperature on the Properties of ZnO Thin Films Grown by Sputtering. Energy Procedia 2012, 25, 55–61. [Google Scholar] [CrossRef]
- Gardeniers, J.G.E.; Rittersma, Z.M.; Burger, G.J. Preferred orientation and piezoelectricity in sputtered ZnO films. J. Appl. Phys. 1998, 83, 7844–7854. [Google Scholar] [CrossRef]
- Gandhi, A.C.; Pant, J.; Pandit, S.D.; Dalimbkar, S.K.; Chan, T.-S.; Cheng, C.-L.; Ma, Y.-R.; Wu, S.Y. Short-Range Magnon Excitation in NiO Nanoparticles. J. Phys. Chem. C 2013, 117, 18666–18674. [Google Scholar] [CrossRef]
- Wojdyr, M. Fityk: A General-purpose Peak Fitting Program. J. Appl. Cryst. 2010, 43, 1126–1128. [Google Scholar] [CrossRef]
- Lupan, O.; Chow, L.; Chai, G.; Heinrich, H. Fabrication and characterization of Zn–ZnO core–shell microspheres from nanorods. Chem. Phys. Lett. 2008, 465, 249–253. [Google Scholar] [CrossRef]
- Ellmer, K. Magnetron sputtering of transparent conductive zinc oxide: Relation between the sputtering parameters and the electronic properties. J. Phys. D Appl. Phys. 2000, 33, R17–R32. [Google Scholar] [CrossRef]
- Hong, R.; Qi, H.; Huang, J.; He, H.; Fan, Z.; Shao, J. Influence of Oxygen Partial Pressure on the Structure and Photoluminescence of Direct Current Reactive Magnetron Sputtering ZnO Thin Films. Thin Solid Films 2005, 473, 58–62. [Google Scholar] [CrossRef]
- Von Dreele, R.B.; Larson, A.C. General Structure Analysis System (GSAS), Los Alamos National Laboratory Report LAUR 86–748; Los Alamos National Laboratory: Los Alamos, NM, USA, 2000; p. 221.
- Huang, K.; Rhys, A. Theory of light absorption and non-radiative transitions in F-centres. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1950, 204, 406–423. [Google Scholar] [CrossRef]
- Zhang, X.T.; Liu, Y.C.; Zhi, Z.Z.; Zhang, J.Y.; Lu, Y.M.; Shen, D.Z.; Xu, W.; Zhong, G.Z.; Fan, X.W.; Kong, X.G. Resonant Raman scattering and photoluminescence from high-quality nanocrystalline ZnO thin films prepared by thermal oxidation of ZnS thin films. J. Phys. D Appl. Phys. 2001, 34, 3430. [Google Scholar] [CrossRef]
- Hsieh, P.T.; Chen, Y.C.; Wang, C.M.; Tsai, Y.Z.; Hu, C.C. Structural and photoluminescence characteristics of ZnO films by room temperature sputtering and rapid thermal annealing process. Appl. Phys. A Mater. Sci. Process. 2006, 84, 345–349. [Google Scholar] [CrossRef]
- Deng, B.; da Rosa, A.L.; Frauenheim, T.; Xiao, J.P.; Shi, X.Q.; Zhang, R.Q.; Van Hove, M.A. Oxygen vacancy diffusion in bare ZnO nanowires. Nanoscale 2014, 6, 11882–11886. [Google Scholar] [CrossRef] [PubMed]
- Kelley, A.M. Electron-Phonon Coupling in CdSe Nanocrystals. J. Phys. Chem. Lett. 2010, 1, 1296–1300. [Google Scholar] [CrossRef]

| TA (°C) | Zn | ZnO | wRp | Rp | χ2 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| a = b (Å) | c (Å) | a = b (Å) | c (Å) | u | ε (%) | σ (GPa) | ||||
| 25 (RT) | 2.6531(11) | 5.1315(16) | 3.2358(11) | 5.2442(3) | 0.3769 | 0.71 | −1.66 | 0.2638 | 0.1538 | 6.132 |
| 100 | 2.6569(13) | 5.0304(24) | 3.2462(8) | 5.2343(7) | 0.3782 | 0.52 | −1.22 | 0.3403 | 0.2317 | 5.741 |
| 200 | 3.2514(8) | 5.2191(2) | 0.3794 | 0.23 | −0.54 | 0.2639 | 0.1794 | 4.989 | ||
| 300 | 3.2495(7) | 5.2179(2) | 0.3793 | 0.21 | −0.49 | 0.2492 | 0.1597 | 5.448 | ||
| 400 | 3.2525(6) | 5.2151(5) | 0.3797 | 0.16 | −0.36 | 0.3169 | 0.2233 | 4.235 | ||
| 500 | 3.2449(7) | 5.2023(12) | 0.3797 | −0.09 | 0.21 | 0.3092 | 0.204 | 3.878 | ||
| 600 | 3.2485(5) | 5.2066(9) | 0.3798 | −0.01 | 0.02 | 0.3743 | 0.2436 | 3.426 | ||
| 700 | 3.2509(2) | 5.2106(2) | 0.3797 | 0.07 | −0.16 | 0.3084 | 0.1907 | 4.043 | ||
| 800 | 3.2488(4) | 5.2024(6) | 0.3800 | −0.09 | 0.21 | 0.3515 | 0.2318 | 3.3 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gandhi, A.C.; Yeoh, W.-S.; Wu, M.-A.; Liao, C.-H.; Chiu, D.-Y.; Yeh, W.-L.; Huang, Y.-L. New Insights into the Role of Weak Electron–Phonon Coupling in Nanostructured ZnO Thin Films. Nanomaterials 2018, 8, 632. https://doi.org/10.3390/nano8080632
Gandhi AC, Yeoh W-S, Wu M-A, Liao C-H, Chiu D-Y, Yeh W-L, Huang Y-L. New Insights into the Role of Weak Electron–Phonon Coupling in Nanostructured ZnO Thin Films. Nanomaterials. 2018; 8(8):632. https://doi.org/10.3390/nano8080632
Chicago/Turabian StyleGandhi, Ashish C., Wei-Shan Yeoh, Ming-An Wu, Ching-Hao Liao, Dai-Yao Chiu, Wei-Li Yeh, and Yue-Lin Huang. 2018. "New Insights into the Role of Weak Electron–Phonon Coupling in Nanostructured ZnO Thin Films" Nanomaterials 8, no. 8: 632. https://doi.org/10.3390/nano8080632





