Methods for Measuring and Estimating Methane Emission from Ruminants
Abstract
:Simple Summary
Abstract
1. Introduction
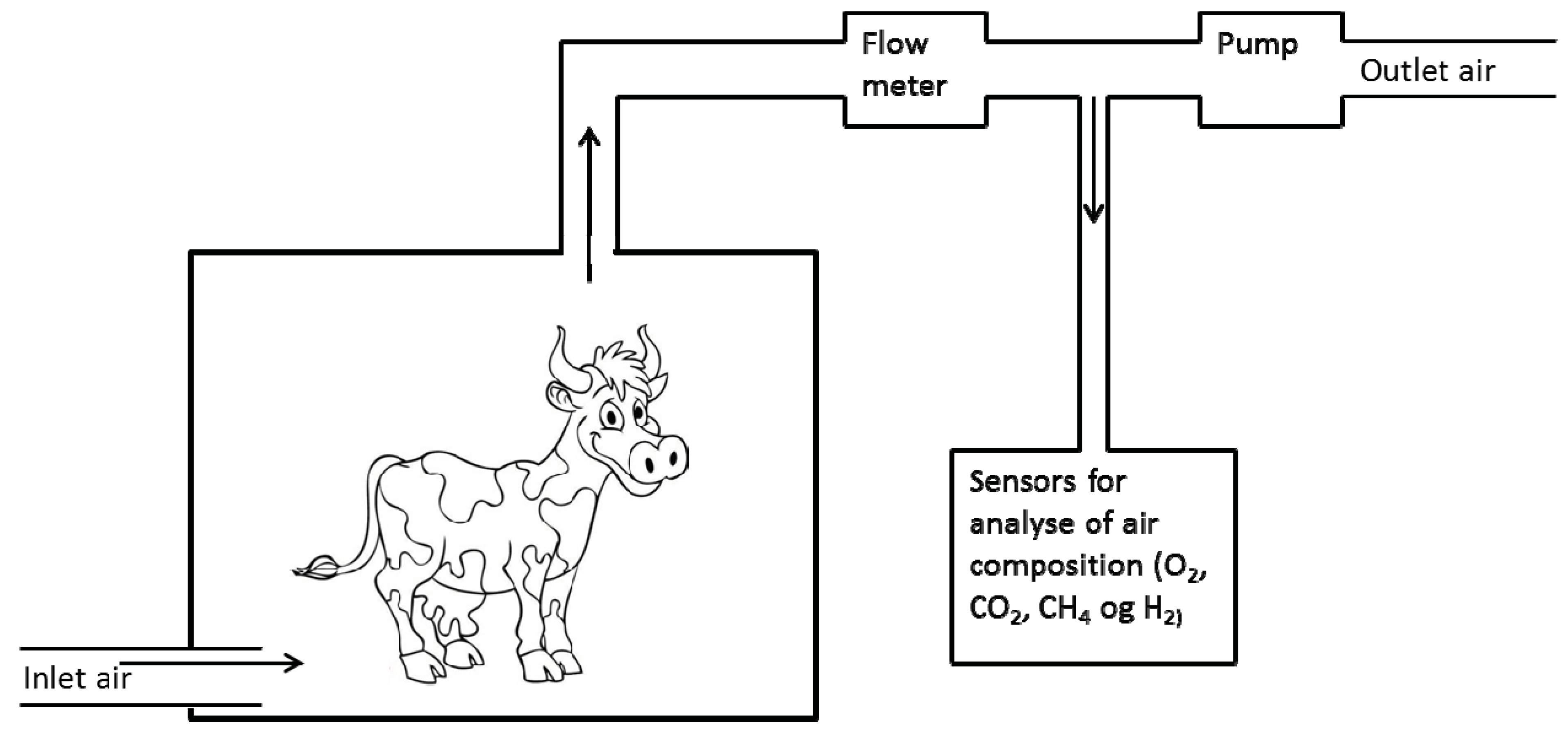
2. Measuring Methane by Means of Chambers

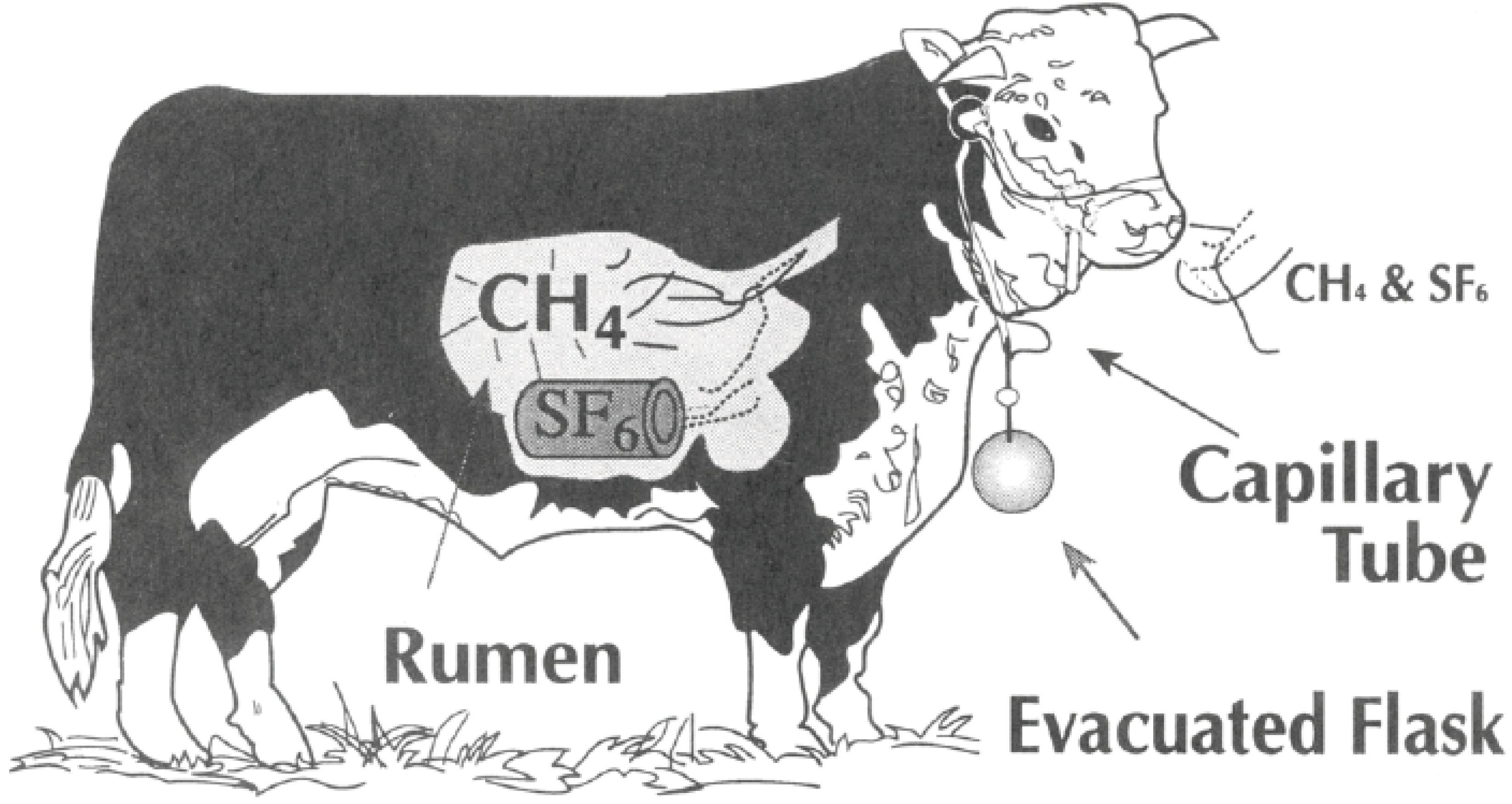
3. Measuring Methane with the SF6 Tracer Technique
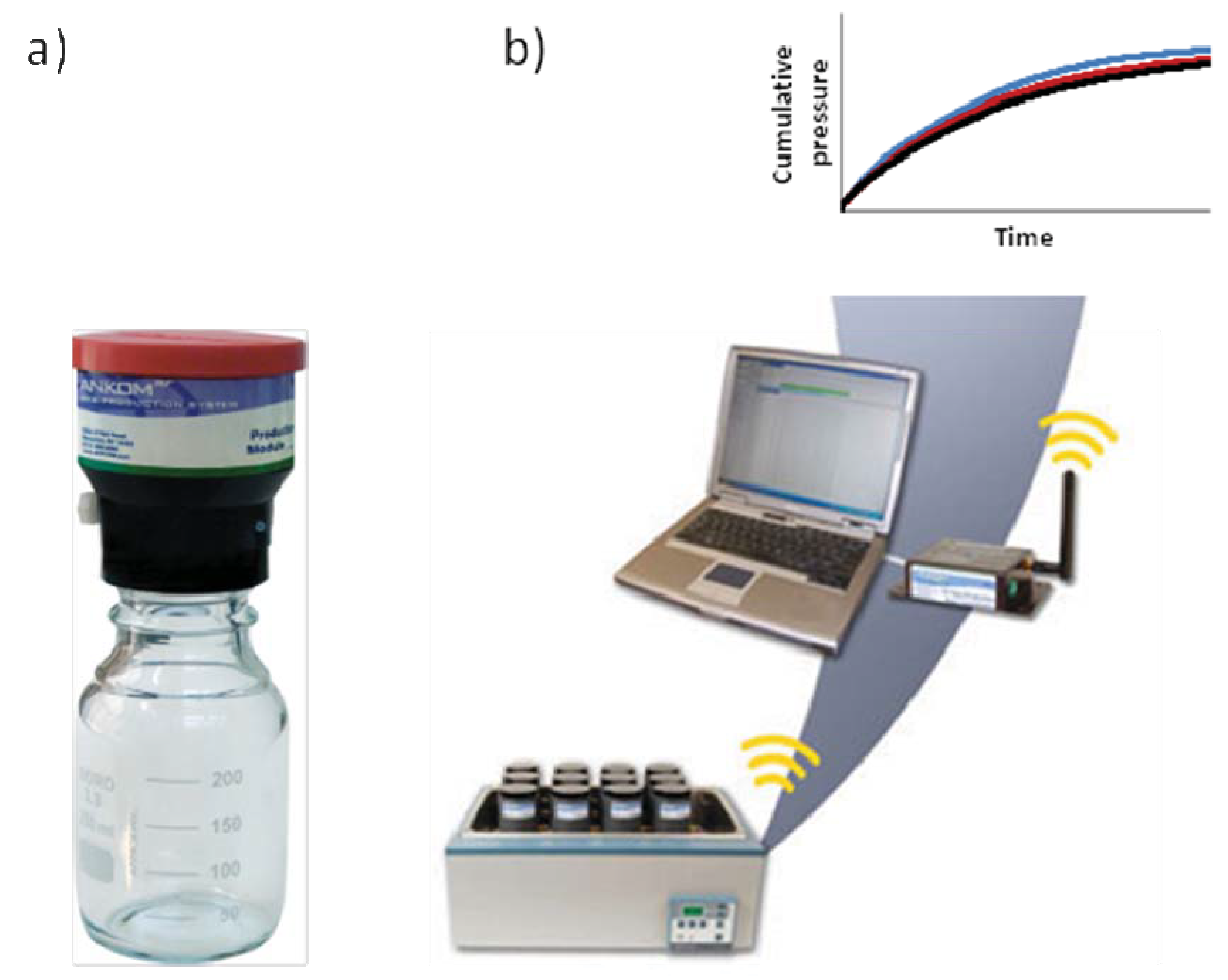
4. In Vitro Gas Production Technique for Methane Measurements
5. The CO2 Technique

6. Other Measuring Techniques
6.1. Methods Based on Whole Buildings or Areas
6.2. Combined Feeder and CH4 Analyzer
6.3. Proxy Methods
7. Models for Predicting Methane Production
7.1. IPCC
7.2. Methane Models
| Reference | Equation | R2 | N |
|---|---|---|---|
| IPCC [98] a | Methane (kg/dag) = GE (MJ/d) × Ym/55.65 | ||
| Yan et al. [103] b | Methane (L/d) = 47.8 × DMI − 0.76 × DMI2 − 41 (kg/d) | 0.75 | 315 |
| Yan et al. [103] bc | Methane (L/d) = 0.34 × BW (kg) + 19.7 × DMI (kg/d) + 12 | 0.77 | 315 |
| Kirchgessner et al. [104] d | Methane (g/d) = 63 + 79 × CF + 10 × NFE + 26 × CP – 212 × Cfat (kg/d) | 0.69 | 24 |
| Jentsch et al. [101] de | Methane (kJ/d) = 1.62 × d_CP − 0.38 × d_Cfat + 3.78 × d_CF + 1.49 × d_NFE +1142 (g/d) | 0.90 | 337 |
| Ellis et al. [21] | Methane (MJ/d) = 0.14 × forage (%) + 8.6 | 0.56 | 89 |
| Mills et al. [105] f | Methane (MJ/d) = 0.07 × ME (MJ/d) + 8.25 | 0.55 | 159 |
| Mills et al. [105] b | Methane (MJ/d) = 0.92 × DMI (kg/d) + 5.93 | 0.60 | 159 |
| Mills et al. [105] b | Methane (MJ/d) = 10.3 × forage (%) + 0.87 × DMI (kg/d) + 1.1 | 0.61 | 159 |
| Grainger et al. [26] b | Methane (g/d) = 18.5 × DMI (kg/d) − 9.5 | 0.56 | 16 |
8. Comparison of Methods
| Method parameters | Chambers | SF6 technique | In vitro gas production | CO2 technique | IPCC | Other models |
|---|---|---|---|---|---|---|
| Prerequisites (except for instruments) | Access to rumen fluid | Information about CO2 production. Can be calculated [70]. | Information about e.g., number of animals, intake of gross energy | Model dependent, e.g., dry matter intake, nutrient composition | ||
| Aspects of feeding which can be investigated | ||||||
| Feeding level | Yes | Yes | No | Yes | No | Yes—some models |
| Physical form of the feed | Yes | Yes | No (all feed is ground) | Yes | No | No |
| Chemical composition of diet | Yes | Yes | Yes | Yes | No | Yes—some models |
| Supplementation of feed additives | Yes | Yes | Yes | Yes | No | No |
| Influence on animals | ||||||
| Fixation needed | Yes | No | * | Depends on aim | * | * |
| Animal needs to carry equipment | No | Yes | * | Depends on aim | * | * |
| Can be used in milking parlor or automatic milking | No | No | * | Yes | * | * |
| Method estimates | ||||||
| Individual animals | Yes | Yes | No | Yes | Yes | Yes |
| Within animal variation | Yes | Yes | No | Yes | No | No |
| Between animal variation | Yes | Yes | No | Yes | No | No |
| Daily variation | Yes | No | No | Yes | No | No |
| Time resolution 1 | A few minutes to hours | 8–24 h | Min. 6 h | Small intervals of a few minutes | * | * |
| Output format | ||||||
| Basic | l CH4/day/animal | l CH4/day/animal | l CH4/kg dry matter | l CH4/day/animal | l CH4/day/animal | l CH4/day/animal |
| Relative to dry matter intake | Yes | Yes | Yes | Yes | Yes | Yes |
| Relative to digested organic matter | Yes | Yes | Yes | Yes | No | Yes, depends on model |
| Relative to digested NDF | Yes | Yes | Yes | Yes | No | Yes |
| Relative to milk yield | Yes | Yes | No | Yes | * | Yes |
| Relative to gross energy intake | Yes | Yes | No | Yes | * | Yes |
9. Conclusions
Acknowledgments
Conflict of Interest
References
- IPCC. Climate Change 2007: Mitigation of Climate Change. IPCC Fourth Assessment Report (AR4). 2007. Available online: http://www.ipcc.ch/publications_and_data/ar4/wg3/en/contents.html (accessed on 30 January 2012).
- IPCC. Climate change 2007: The Physical Science Basis. IPCC Fourth Assessment Report (AR4). 2007. Available online: http://www.ipcc.ch/publications_and_data/ar4/wg1/en/contents.html (accessed on 30 January 2012).
- Lassey, K.R. Livestock methane emission: From the individual grazing animal through national inventories to the global methane cycle. Agric. Forest Meteorol. 2007, 142, 120–132. [Google Scholar]
- Johnson, D.E.; Ferrell, C.L.; Jenkins, T.G. The history of energetic efficiency research: Where have we been and where are we going? J. Anim. Sci. 2003, 81, E27–E38. [Google Scholar]
- Mclean, J.A.; Tobin, G. Animal and Human Calorimetry; Cambridge University Press: Cambridge, UK, 1987. [Google Scholar]
- Wainman, F.W.; Blaxter, K.L. Closed-Circuit Respiration Apparatus for the Cow and Steer. In Proceedings of the 1st Symposium in Energy Metabolism, Principles, Methods and General Aspects, Copenhagen, Denmark, 15–19 September 1958; pp. 80–84.
- Brown, D.; Cole, T.J.; Dauncey, M.J.; Marrs, R.W.; Murgatroyd, P.R. Analysis of gaseous exchange in open-circuit indirect calorimetry. Med. Biol. Eng. Comput. 1984, 22, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Derno, M.; Elsner, H.G.; Paetow, E.A.; Scholze, H.; Schweigel, M. Technical note: A new facility for continuous respiration measurements in lactating cows. J. Dairy Sci. 2009, 92, 2804–2808. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.H.; Koes, R.M. Construction and operation of an open-circuit indirect calorimetry system for small ruminants. J. Anim. Sci. 1988, 66, 1042–1047. [Google Scholar] [PubMed]
- Bryant, A.M.; Hughes, J.W.; Hutton, J.B.; Newth, R.P.; Parr, C.R.; Trigg, T.E.; Young, J. Calorimetric facilities for dairy cattle at Ruakua Animal Research Station. Proc. N. Z. Soc. Anim. Prod. 1977, 37, 158–162. [Google Scholar]
- Pinares-Patiño, C.S.; Lassey, K.R.; Martin, R.J.; Molano, G.; Fernandez, M.; MacLean, S.; Sandoval, E.; Luo, D.; Clark, H. Assessment of the sulphur hexafluoride (SF6) tracer technique using respiration chambers for estimation of methane emissions from sheep. Anim. Feed Sci. Technol. 2011, 166–167, 201–209. [Google Scholar]
- Yanez-Ruiz, D.R.; Hart, K.J.; Martin-Garcia, A.I.; Ramos, S.; Newbold, C.J. Diet composition at weaning affects the rumen microbial population and methane emissions by lambs. Aust. J. Exp. Agric. 2008, 48, 186–188. [Google Scholar] [CrossRef]
- Klein, L.; Wright, A.D.G. Construction and operation of open-circuit methane chambers for small ruminants. Aust. J. Exp. Agric. 2006, 46, 1257–1262. [Google Scholar] [CrossRef]
- Place, S.E.; Pan, Y.; Zhao, Y.; Mitloehner, F.M. Construction and operation of a ventilated hood system for measuring greenhouse gas and volatile organic compound emissions from cattle. Animals 2011, 1, 433–446. [Google Scholar] [CrossRef]
- Takahashi, J.; Chaudhry, A.S.; Beneke, R.G.; Young, B.A. An open-circuit hood system for gaseous exchange measurements in small ruminants. Small Rumin. Res. 1999, 32, 31–36. [Google Scholar] [CrossRef]
- Kelly, J.M.; Kerrigan, B.; Milligan, L.P.; McBride, B.W. Development of a mobile, open-circuit indirect calorimetry system. Can. J. Anim. Sci. 1994, 74, 65–71. [Google Scholar] [CrossRef]
- Murray, P.J.; Chadwick, D.C.; Newbold, C.J.; Lockyer, D.R. Measurement of Methane from Grazing Animals—The Tunnel Method. In Measuring Methane Production from Ruminants; Makkar, H.P.S., Vercoe, P.E., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 105–109. [Google Scholar]
- Murray, P.J.; Moss, A.; Lockyer, D.R.; Jarvis, S.C. A comparison of systems for measuring methane emissions from sheep. J. Agric. Sci. 1999, 133, 439–444. [Google Scholar] [CrossRef]
- Lockyer, D.R.; Jarvis, S.C. The measurement of methane losses from grazing animals. Environ. Pollut. 1995, 90, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.A.; Johnson, D.E. Methane emissions from cattle. J. Anim. Sci. 1995, 73, 2483–2492. [Google Scholar] [PubMed]
- Ellis, J.L.; Kebreab, E.; Odondo, N.E.; McBride, B.W.; Okine, E.K.; France, J. Prediction of methane production from dairy and beef cattle. J. Dairy Sci. 2007, 90, 3456–3467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.; Huyler, M.; Westberg, H.; Lamb, B.; Zimmerman, P. Measurement of methane emissions from ruminant livestock using a SF6 tracer technique. Environ. Sci. Technol. 1994, 28, 359–362. [Google Scholar] [CrossRef] [PubMed]
- Okelly, J.C.; Spiers, W.G. Effect of monensin on methane and heat productions of steers fed lucerne hay either ad-libitum or at the rate of 250 g/hour. Aust. J. Agric. Res. 1992, 43, 1789–1793. [Google Scholar] [CrossRef]
- Johannes, M.; Lund, P.; Weisbjerg, M.R.; Hellwing, A.L.F.; Hvelplund, T. The effect of different physical forms of rapeseed as fat supplement on rumen NDF digestion and methane emission in dairy cows. Adv. Anim. Biosci. 2011, 2, 516. [Google Scholar]
- Blaxter, K.L.; Clappert, J.L. Prediction of amount of methane produced by ruminants. Br. J. Nutr. 1965, 19, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Grainger, C.; Clarke, T.; Mcginn, S.M.; Auldist, M.J.; Beauchemin, K.A.; Hannah, M.C.; Waghorn, G.C.; Clark, H.; Eckard, R.J. Methane emissions from dairy cows measured using sulfur hexafluoride (SF6) and chamber techniques. J. Dairy Sci. 2007, 90, 2755–2766. [Google Scholar] [CrossRef] [PubMed]
- Blaxter, K.L. Techniques in energy metabolism studies and their limitations. Proc. Nutr. Soc. 1967, 26, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Thorbek, G. Studies on Protein and Energy Metabolism in Growing Calves; Landhusholdingsselskabets forlag: Copenhagen, Denmark, 1980. [Google Scholar]
- Zimmerman, P.R. System for Measuring Metabolic Gas Emissions from Animals. US Patent 5,265,618, 30 December 1993. [Google Scholar]
- Lassey, K.R.; Pinares-Patiño, C.S.; Martin, R.J.; Molano, G.; McMillan, A.M.S. Enteric methane emission rates determined by the SF6 tracer technique: Temporal patterns and averaging periods. Anim. Feed Sci. Technol. 2011, 166–167, 183–191. [Google Scholar] [CrossRef]
- Lassey, K.R.; Ulyatt, M.J.; Martin, R.J.; Walker, C.F.; Shelton, I.D. Methane emissions measured directly from grazing livestock in New Zealand. Atmos. Environ. 1997, 31, 2905–2914. [Google Scholar] [CrossRef]
- Mcginn, S.M.; Chung, Y.H.; Beauchemin, K.A.; Iwaasa, A.D.; Grainger, C. Use of corn distillers’ dried grains to reduce enteric methane loss from beef cattle. Can. J. Anim. Sci. 2009, 89, 409–413. [Google Scholar] [CrossRef]
- McCaughey, W.P.; Wittenberg, K.; Corrigan, D. Methane production by steers on pasture. Can. J. Anim. Sci. 1997, 77, 519–524. [Google Scholar] [CrossRef]
- Grainger, C.; Williams, R.; Clarke, T.; Wright, A.D.G.; Eckard, R.J. Supplementation with whole cottonseed causes long-term reduction of methane emissions from lactating dairy cows offered a forage and cereal grain diet. J. Dairy Sci. 2010, 93, 2612–2619. [Google Scholar] [CrossRef]
- Goopy, J.P.; Hegarty, R.S. Repeatability of methane production in cattle fed concentrate and forage diets. J. Anim. Feed Sci. 2004, 13, 75–78. [Google Scholar]
- Tekippe, J.A.; Hristov, A.N.; Heyler, K.S.; Cassidy, T.W.; Zheljazkov, V.D.; Ferreira, J.F.S.; Karnati, S.K.; Varga, G.A. Rumen fermentation and production effects of Origanum vulgare L. leaves in lactating dairy cows. J. Dairy Sci. 2011, 94, 5065–5079. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Wredle, E.; Borjesson, G.; Danielsson, R.; Iwaasa, A.D.; Sporndly, E.; Bertilsson, J. Enteric methane emissions from dairy cows fed different proportions of highly digestible grass silage. Acta Agric. Scand. A Anim. 2011, 61, 128–136. [Google Scholar]
- Nes, S.K.; Garmo, T.; Chaves, A.V.; Harstad, O.M.; Iwaasa, A.D.; Krizsan, S.J.; Beauchemin, K.A.; McAllister, T.A.; Norell, L.; Thuen, E.; Vedres, D.; Volden, H. Effects of Neutral Detergent Fiber (aNDF) Digestibility of Grass Silage on Enteric Methane Emissions from Dairy Cows. In Proceedings of Inteternational Conference Greenhouse Gases in Animal Agriculture, Banff, AB, Canada, 3 October 2010; p. 110.
- Lester, D.; Greenberg, L.A. The toxicity of sulfur hexafluoride. Arch. Ind. Hyg. Occup. Med. 1950, 2, 348–349. [Google Scholar] [PubMed]
- Johnson, K.A.; Huyler, M.; Pierce, C.S.; Westberg, H.; Lamb, B.; Zimmerman, P. The use of SF6 as an inert gas tracer for use in methane measurements. J. Anim. Sci. 1992, 70, 302. [Google Scholar]
- Johnson, K.A.; Westberg, H.H.; Michal, J.J.; Cossalman, M.W. The SF6 Tracer Technique: Methane Measurement from Ruminants. In Measuring Methane Production from Ruminants; Makkar, H.P.S., Vercoe, P.E., Eds.; Springer: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Cécile, M.; Koolaard, J.; Rochette, Y.; Clark, H.; Jouany, J.P.; Pinares-Patiño, C.S. Effect of release rate of SF6 Tracer on Methane and Carbon Dioxide Emission Estimates Based on Ruminal and Breath Gas Samples. In Proceedings of 4th International Conference on Greenhouse Gases Animal Agriculture, Banff, AB, Canada, 3 October 2010; p. 85.
- Coates, T.; Farr, B.; Beauchemin, K.A.; Mcginn, S.M. Can the SF6 Tracer Gas Technique be Used to Accurately Measure Methane Production from Ruminally Cannulated Cattle. In Proceedings of 4th International Conference on Greenhouse Gases Animal Agriculture, Banff, AB, Canada, 3 October 2010; p. 86.
- Pinares-Patiño, C.S. Methane Emission from Forage-Fed Sheep, a Study of Variation Between Animals. Ph.D. Thesis, Animal and Biomedical Sciences, College of Sciences, Massey University, Palmerston, New Zealand, 2000. [Google Scholar]
- Pinares-Patiño, C.S.; Clark, H. Reliability of the sulfur hexafluoride tracer technique for methane emission measurement from individual animals: An overview. Aust. J. Exp. Agric. 2008, 48, 223–229. [Google Scholar] [CrossRef]
- Ulyatt, M.J.; Baker, S.K.; McCrabb, G.J.; Lassey, K.R. Accuracy of SF6 tracer technology and alternatives for field measurements. Aust. J. Agric. Res. 1999, 50, 1329–1334. [Google Scholar] [CrossRef]
- Lassey, K.R.; Walker, C.F.; McMillan, A.M.S.; Ulyatt, M.J. On the performance of SF6 permeation tubes used in determining methane emission from grazing livestock. Chemosphere Glob. Chang. Sci. 2001, 3, 367–376. [Google Scholar] [CrossRef]
- Vlaming, J.B. Qantifying Variation in Estimated Methane Emission from Ruminants Using the SF6 Tracer Technique. Ph.D. Thesis, Massey Univesity, Palmerton North, New Zealand, 2007. [Google Scholar]
- Pinares-Patiño, C.S.; Machmuller, A.; Molano, G.; Smith, A.; Vlaming, J.B.; Clark, H. The SF6 tracer technique for measurements of methane emission from cattle—Effect of tracer permeation rate. Can. J. Anim. Sci. 2008, 88, 309–320. [Google Scholar] [CrossRef]
- Williams, S.R.O.; Moate, P.J.; Hannah, M.C.; Ribaux, B.E.; Wales, W.J.; Eckard, R.J. Background matters with the SF6 tracer method for estimating enteric methane emissions from dairy cows: A critical evaluation of the SF6 procedure. Anim. Feed Sci. Technol. 2011, 170, 265–276. [Google Scholar] [CrossRef]
- Johnson, K.A.; Huyler, M.T.; Westberg, H.H.; Lamb, B.K.; Zimmerman, P. Measurements of Methane Emissions from Ruminant Livestock Using a Sulfur Hexafluoride Tracer Technique. In Proceedings of 13th Symposium on Energy Metabolism of Farm Animals Aguilera, Mojácar, Spain, 18–24 September 1994; pp. 335–338.
- Murray, R.M.; Bryant, A.M.; Leng, R.A. Rates of production of methane in rumen and large-intestine of sheep. Br. J. Nutr. 1976, 36, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mcginn, S.M.; Beauchemin, K.A.; Iwaasa, A.D.; McAllister, T.A. Assessment of the sulfur hexafluoride (SF6) tracer technique for measuring enteric methane emissions from cattle. J. Environ. Qual. 2006, 35, 1686–1691. [Google Scholar] [CrossRef] [PubMed]
- Pinares-Patiño, C.S.; D’Hour, P.; Jouany, J.P.; Martin, C. Effects of stocking rate on methane and carbon dioxide emissions from grazing cattle. Agric. Ecosyst. Environ. 2007, 121, 30–46. [Google Scholar] [CrossRef]
- Boadi, D.A.; Wittenberg, K.M.; Kennedy, A.D. Validation of the sulphur hexafluoride (SF6) tracer gas technique for measurement of methane and carbon dioxide production by cattle. Can. J. Anim. Sci. 2002, 82, 125–131. [Google Scholar] [CrossRef]
- Pinares-Patiño, C.S.; Holmes, C.W.; Lassey, K.R.; Ulyatt, M.J. Measurement of methane emission from sheep by the sulphur hexafluoride tracer technique and by the calorimetric chamber: Failure and success. Animal 2008, 2, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Fredeen, A.H.; Cooper, S.; Tate, M.; Main, M.; Yuill, J. Comparison of whole body and breath- sampling methods of measuring methane emission in dairy cows. Can. J. Anim. Sci. 2004, 84, 774. [Google Scholar]
- Rymer, C.; Huntington, J.A.; Williams, B.A.; Givens, D.I. In vitro cumulative gas production techniques: History, methodological considerations and challenges. Anim. Feed Sci. Technol. 2005, 123–124, 9–30. [Google Scholar] [CrossRef]
- Pellikaan, W.F.; Hendriks, W.H.; Uwimana, G.; Bongers, L.J.G.M.; Becker, P.M.; Cone, J.W. A novel method to determine simultaneously methane production during in vitro gas production using fully automated equipment. Anim. Feed Sci. Technol. 2011, 168, 196–205. [Google Scholar] [CrossRef]
- Navarro-Villa, A.; O’Brien, M.; Lopez, S.; Boland, T.M.; O’Kiely, P. Modifications of a gas production technique for assessing in vitro rumen methane production from feedstuffs. Anim. Feed Sci. Technol. 2011, 166–167, 163–174. [Google Scholar] [CrossRef]
- Blümmel, M.; Ørskov, E.R. Comparison of in vitro gas production and nylon bag degradability of roughages in predicting feed intake in cattle. Anim. Feed Sci. Technol. 1993, 40, 109–119. [Google Scholar] [CrossRef]
- Bhatta, R.; Tajima, K.; Takusari, N.; Higuchi, K.; Enishi, O.; Kurihara, M. Comparison of sulfur hexafluoride tracer technique, rumen simulation technique and in vitro gas production techniques for methane production from ruminant feeds. Int. Congr. Ser. 2006, 1293, 58–61. [Google Scholar] [CrossRef]
- Cornou, C.; Hindrichsen, I.K.; Worgan, H.; Bakewell, E.; Yáñez Ruiz, D.R.; Abecia, L.; Tagliapietra, F.; Cattani, M.; Storm, I.M.L.D.; Ritz, C.; Hansen, H.H. A ring test of a wireless in vitro gas production system. Anim. Prod. Sci. 2012. submitted. [Google Scholar]
- Mould, F.L.; Kliem, K.E.; Morgan, R.; Mauricio, R.M. In vitro microbial inoculum: A review of its function and properties. Anim. Feed Sci. Technol. 2005, 123–124, 31–50. [Google Scholar] [CrossRef]
- Bhatta, R.; Enishi, O.; Takusari, N.; Higuchi, K.; Nonaka, I.; Kurihara, M. Diet effects on methane production by goats and a comparison between measurement methodologies. J. Agric. Sci. 2008, 146, 705–715. [Google Scholar] [CrossRef]
- Klevenhusen, F.; Zeitz, J.O.; Duval, S.; Kreuzer, M.; Soliva, C.R. Garlic oil and its principal component diallyl disulfide fail to mitigate methane, but improve digestibility in sheep. Anim. Feed Sci. Technol. 2011, 166–167, 356–363. [Google Scholar] [CrossRef]
- Patra, A.K.; Saxena, J. A new perspective on the use of plant secondary metabolites to inhibit methanogenesis in the rumen. Phytochemistry 2010, 71, 1198–1222. [Google Scholar] [CrossRef] [PubMed]
- Williams, Y.J.; Popovski, S.; Rea, S.M.; Skillman, L.C.; Toovey, A.F.; Northwood, K.S.; Wright, A.D. A vaccine against rumen methanogens can alter the composition of archaeal populations. Appl. Environ. Microbiol. 2009, 75, 1860–1866. [Google Scholar] [CrossRef] [PubMed]
- Madsen, J.; Bjerg, B.S.; Hvelplund, T.; Weisbjerg, M.R.; Lund, P. Methane and carbondioxide ration in excreted air for quantification of the methane prodction from ruminants. Livest. Sci. 2010, 129, 223–227. [Google Scholar] [CrossRef]
- Pedersen, S.; Sällvik, K. 4th Report from Working Group on Climatization in Animal Houses—Heat and Moisture Production at Animal and House Level. 2002. Available online: http://www.cigr.org/documents/CIGR_4TH_WORK_GR.pdf (accessed on 30 January 2012).
- Chwalibog, A. Husdyrernæring, Bestemmelse af Næringsværdi og Næringsbehov; Faculty of Life Sciences, University of Copenhagen, DSR forlag.: Copenhagen, Denmark, 1991. [Google Scholar]
- Elia, M.; Livesey, G. Theory and validity of indirect calorimetry during net lipid synthesis. Am. J. Clin. Nutr. 1988, 47, 591–607. [Google Scholar] [PubMed]
- Pedersen, S.; Blanes-Vidal, V.; Jørgensen, H.; Chwalibog, A.; Haeussermann, A.; Heetkamp, M.J.W.; Aarnink, A.J.A. Carbon dioxide production in animal houses: A litterature review. Agric. Eng. Int. CIGR E J. 2008, X, BC 08 008. [Google Scholar]
- Bjerg, B.; Zhang, G.; Madsen, J.; Rom, H.B. Methane emission from naturally ventilated livestock buildings can easily be determined from gas concentration measurements. Environ. Monit. Assess. 2012. [Google Scholar] [CrossRef]
- Lassen, J.; Løvendahl, P.; Madsen, J. Experiences with large scale breath measurements in dairy cattle in order to select for lower methane production. J. Dairy Sci. 2012, 95, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Madsen, J.; Bertelsen, M.F. Methane production by Bennet’s wallabies (Macropus rufogriseus). J. Anim. Sci. 2012. accepted. [Google Scholar]
- Teye, F.K.; Alkkiomaki, E.; Simojoki, A.; Pastell, M.; Ahokas, J. Instrumentation, measurement and performance of three air quality measurement systems for dairy buildings. Appl. Eng. Agric. 2009, 25, 247–256. [Google Scholar] [CrossRef]
- Harper, L.A.; Denmead, O.T.; Flesch, T.K. Micrometeorological techniques for measurement of enteric greenhouse gas emissions. Anim. Feed Sci. Technol. 2011, 166–167, 227–239. [Google Scholar] [CrossRef]
- Denmead, O.T. Approaches to measuring fluxes of methane and nitrous oxide between landscapes and the atmosphere. Plant Soil 2008, 309, 5–24. [Google Scholar] [CrossRef]
- Griffith, D.W.T.; Bryant, G.R.; Hsu, D.; Reisinger, A.R. Methane emissions from free-ranging cattle: Comparison of tracer and integrated horizontal flux techniques. J. Environ. Qual. 2008, 37, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Kaharabata, S.K.; Schuepp, P.H. Estimating methane emissions from dairy cattle housed in a barn and feedlot using an atmospheric tracer. Environ. Sci. Technol. 2000, 34, 3296–3302. [Google Scholar] [CrossRef]
- Marik, T.; Levin, I. A new tracer experiment to estimate the methane emissions from a dairy cow shed using sulfur hexafluoride (SF6). Glob. Biogeochem. Cycles 1996, 10, 413–418. [Google Scholar] [CrossRef]
- Demmers, T.G.M.; Burgess, L.R.; Short, J.L.; Philips, V.R.; Clark, J.A.; Wathes, C.M. Ammonia emissions from two mechanically ventilated UK livestock buildings. Atmos. Environ. 1999, 33, 217–227. [Google Scholar] [CrossRef]
- Laubach, J.; Kelliher, F.M. Measuring methane emission rates of a dairy cow herd by two micrometeorological techniques. Agric. Forest Meteorol. 2004, 125, 279–303. [Google Scholar] [CrossRef]
- Harper, L.A.; Denmead, O.T.; Freney, J.R.; Byers, F.M. Direct measurements of methane emissions from grazing and feedlot cattle. J. Anim. Sci. 1999, 77, 1392–1401. [Google Scholar]
- Judd, M.J.; Kellier, F.M.; Ulyatt, M.J.; Lassey, K.R.; Tate, K.R.; Shelton, D.; Harvey, M.J.; Walker, C.F. Net methane emissions from grazing sheep. Glob. Chang. Biol. 1999, 5, 647–657. [Google Scholar] [CrossRef]
- Dengel, S.; Levy, P.E.; Grace, J.; Jones, S.K.; Skiba, U.M. Methane emissions from sheep pasture, measured with an open-path eddy covariance system. Glob. Chang. Biol. 2011, 17, 3524–3533. [Google Scholar] [CrossRef]
- Baum, K.A.; Ham, J.M.; Brunsell, N.A.; Coyne, P.I. Surface boundary layer of cattle feedlots: Implications for air emissions measurement. Agric. Forest Meteorol. 2008, 148, 1882–1893. [Google Scholar] [CrossRef]
- Laubach, J.; Kelliher, F.A. Measuring methane emission rates of a dairy cow herd (II): Results from a backward-Lagrangian stochastic model. Agric. Forest Meteorol. 2005, 129, 137–150. [Google Scholar] [CrossRef]
- Flesch, T.K.; Wilson, J.D.; Yee, E. Backward-time lagrangian stochastic dispersion models and their application to estimate gaseous emissions. J. Appl. Meteorol. 1995, 34, 1320–1332. [Google Scholar] [CrossRef]
- Flesch, T.K.; Wilson, J.D.; Harper, L.A. Deducing ground-to-air emissions from observed trace gas concentrations: A field trial with wind disturbance. J. Appl. Meteorol. 2005, 44, 475–484. [Google Scholar] [CrossRef]
- Tomkins, N.W.; Mcginn, S.M.; Turner, D.A.; Charmley, E. Comparison of open-circuit respiration chambers with a micrometeorological method for determining methane emissions from beef cattle grazing a tropical pasture. Anim. Feed Sci. Technol. 2011, 166–167, 240–247. [Google Scholar] [CrossRef]
- Penning, P. Herbage Intake Handbook; British Grassland Society: Warwickshire, UK, 2004. [Google Scholar]
- Chilliard, Y.; Martin, C.; Rouel, J.; Doreau, M. Milk fatty acids in dairy cows fed whole crude linseed, extruded linseed, or linseed oil, and their relationship with methane output. J. Dairy Sci. 2009, 92, 5199–5211. [Google Scholar] [CrossRef] [PubMed]
- Vlaeminck, B.; Fievez, V.; Cabrita, A.R.J.; Fonseca, A.J.M.; Dewhurst, R.J. Factors affecting odd- and branched-chain fatty acids in milk: A review. Anim. Feed Sci. Technol. 2006, 131, 389–417. [Google Scholar] [CrossRef]
- Dijkstra, J.; van Zijderveld, S.M.; Apajalahti, J.A.; Bannink, A.; Gerrits, W.J.J.; Newbold, J.R.; Perdok, H.B.; Berends, H. Relationships between methane production and milk fatty acid profiles in dairy cattle. Anim. Feed Sci. Technol. 2011, 166–167, 590–595. [Google Scholar] [CrossRef]
- Montoya, J.C.; Bhagwat, A.M.; Peiren, N.; De Campeneere, S.; De Baets, B.; Fievez, V. Relationships between odd- and branched-chain fatty acid profiles in milk and calculated enteric methane proportion for lactating dairy cattle. Anim. Feed Sci. Technol. 2011, 166–167, 596–602. [Google Scholar] [CrossRef]
- IPCC. IPCC Guidelines for National Greenhouse Gas Inventories. 2006. Available online: http://www.ipcc-nggip.iges.or.jp/public/2006gl/vol4.html (accessed on 30 January 2012).
- Danfær, A. Methane Emissions from Dairy Cows. In Arbejdsrapport nr. 11 fra Miljøstyrelsen; Olesen, J.E., Jørgensen, H., Danfær, A., Mikkelsen, M.H., Asman, W.A.H., Pedersen, S.O., Eds.; Miljøstyrelsen: Copenhagen, Denmark, 2005. [Google Scholar]
- NERI. Denmark’s National Inventory Report. 2011. Available online: http://unfccc.int/national_reports/annex_i_ghg_inventories/national_inventories_submissions/items/5888.php (accessed on 30 January 2012).
- Jentsch, W.; Schweigel, M.; Weissbach, F.; Scholze, H.; Pitroff, W.; Derno, M. Methane production in cattle calculated by the nutrient composition of the diet. Arch. Anim. Nutr. 2007, 61, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Volden, H.; Larsen, M. Digestion and Metabolism in the Gastrointestinal Tract. In NorFor—The Nordic Feed Evaluation System; EAAP Publication No. 130; Volden, H., Ed.; Academic Publishers: Wageningen, The Netherlands, 2011. [Google Scholar]
- Yan, T.; Mayne, C.S.; Porter, M.G. Effects of dietary and animal factors on methane production in dairy cows offered grass silage based diets. Int. Congr. Ser. 2006, 1293, 123–126. [Google Scholar] [CrossRef]
- Kirchgessner; Windisch, W.; Müller, H.L. Nutritional Factors for the Quantification of Methane Production. In Proceedings of the 8th International Symposium on Ruminant Physiology, Willingen, Hesse, Germany, 25–30 September 1994; pp. 333–348.
- Mills, J.A.N.; Kebreab, E.; Yates, C.M.; Crompton, L.A.; Cammell, S.B.; Dahnoa, M.S.; Agnew, R.E.; France, J. Alternative approaches to predicting methane emissions from dairy cows. J. Anim. Sci. 2003, 81, 3141–3150. [Google Scholar] [PubMed]
- Kristensen, T.; Mogensen, L.; Weisbjerg, M.R.; Lund, P.; Aaes, O. Variation i udskillelsen af metan fra kvæg. KvægInfo. 2009. Available online: http://www.landbrugsinfo.dk/kvaeg (accessed on 12 March 2012).
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Storm, I.M.L.D.; Hellwing, A.L.F.; Nielsen, N.I.; Madsen, J. Methods for Measuring and Estimating Methane Emission from Ruminants. Animals 2012, 2, 160-183. https://doi.org/10.3390/ani2020160
Storm IMLD, Hellwing ALF, Nielsen NI, Madsen J. Methods for Measuring and Estimating Methane Emission from Ruminants. Animals. 2012; 2(2):160-183. https://doi.org/10.3390/ani2020160
Chicago/Turabian StyleStorm, Ida M. L. D., Anne Louise F. Hellwing, Nicolaj I. Nielsen, and Jørgen Madsen. 2012. "Methods for Measuring and Estimating Methane Emission from Ruminants" Animals 2, no. 2: 160-183. https://doi.org/10.3390/ani2020160




