Glycerophospholipid Profiles of Allomyrina dichotoma Larvae at Different Instars Based on Lipidomics and Transcriptomics Suggest a Promising Lipid Source
Simple Summary
Abstract
1. Introduction
2. Method
2.1. Rearing and Sampling of A. Dichotoma
2.2. Lipid Extraction from A. dichotoma Larvae
2.3. Untargeted Lipidomics for Lipid Analysis
2.4. Identification and Quantification of Glycerophospholipid Species
2.5. RNA Extraction and Transcriptomics Analysis
2.6. Statistical Analysis
3. Results
3.1. Overview of Glycerophospholipids in A. dichotoma Larvae
3.2. Dynamic Changes in Glycerophospholipid Content of A. dichotoma During Larval Development
3.3. Changes in Cardiolipin (CL) in A. dichotoma Larvae During Different Developmental Stages
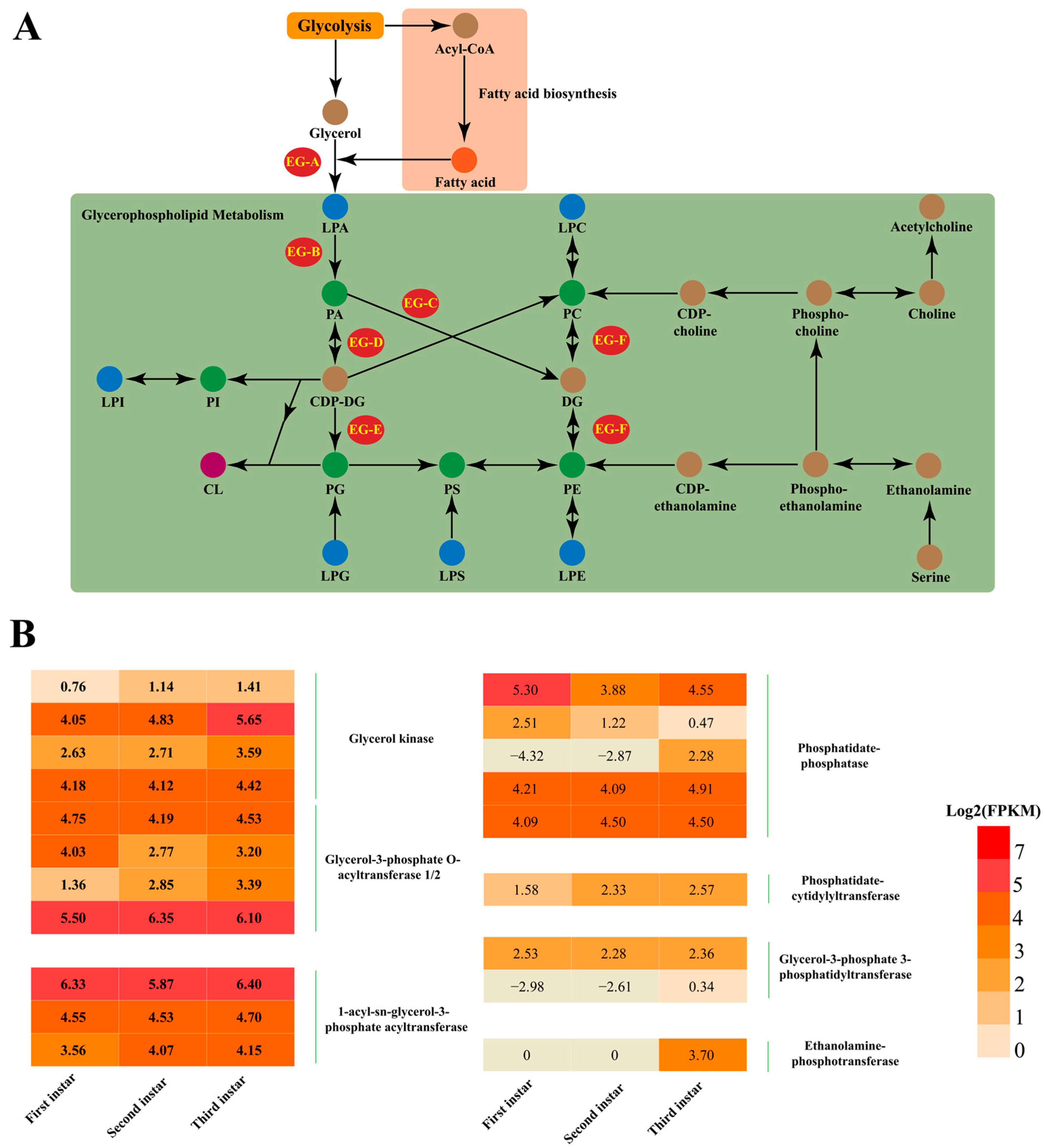
3.4. Analysis of the Biosynthesis Genes Involved in Metabolic Pathways of Glycerophospholipids
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hu, X.; Zhang, W.; Chi, X.; Wang, H.; Liu, Z.; Wang, Y.; Ma, L.; Xu, B. Non-targeted lipidomics and transcriptomics analysis reveal the molecular underpinnings of mandibular gland development in Apis mellifera ligustica. Dev. Biol. 2021, 479, 23–36. [Google Scholar] [CrossRef]
- Carvalho, M.; Sampaio, J.L.; Palm, W.; Brankatschk, M.; Eaton, S.; Shevchenko, A. Effects of diet and development on the Drosophila lipidome. Mol. Syst. Biol. 2012, 8, 600. [Google Scholar] [CrossRef]
- Grille, S.; Zaslawski, A.; Thiele, S.; Plat, J.; Warnecke, D. The functions of steryl glycosides come to those who wait: Recent advances in plants, fungi, bacteria and animals. Prog. Lipid Res. 2010, 49, 262–288. [Google Scholar] [CrossRef]
- Tzompa-Sosa, D.A.; Dewettinck, K.; Provijn, P.; Brouwers, J.F.; De Meulenaer, B.; Oonincx, D.G. Lipidome of cricket species used as food. Food Chem. 2021, 349, 129077. [Google Scholar] [CrossRef] [PubMed]
- van Huis, A. Potential of insects as food and feed in assuring food security. Annu. Rev. Entomol. 2013, 58, 563–583. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wang, G.; Shang, R.; Xu, Q.; Zhang, J.; Sun, R.; Li, L. Comparative lipid profile analysis of Hermetia illucens larvae fed food waste at different days of age using an LC-MS-based lipidomics approach. J. Insect Sci. 2021, 21, 17. [Google Scholar] [CrossRef] [PubMed]
- Rabani, V.; Cheatsazan, H.; Davani, S. Proteomics and lipidomics of black soldier fly (Diptera: Stratiomyidae) and blow fly (Diptera: Calliphoridae) larvae. J. Insect Sci. 2019, 19, 29. [Google Scholar] [CrossRef]
- Li, C.; Li, X.; Huang, Q.; Zhuo, Y.; Xu, B.; Wang, Z. Changes in the phospholipid molecular species in water-boiled salted duck during processing based on shotgun lipidomics. Food Res. Int. 2020, 132, 109064. [Google Scholar] [CrossRef]
- Sagisaka, A.; Miyanoshita, A.; Ishibashi, J.; Yamakawa, M. Purification, characterization and gene expression of a glycine and proline-rich antibacterial protein family from larvae of a beetle, Allomyrina dichotoma. Insect Mol. Biol. 2001, 10, 293–302. [Google Scholar] [CrossRef]
- Pemberton, R.W. Insects and other arthropods used as drugs in Korean traditional medicine. J. Ethnopharmacol. 1999, 65, 207–216. [Google Scholar] [CrossRef]
- Guan, X.L.; Cestra, G.; Shui, G.; Kuhrs, A.; Schittenhelm, R.B.; Hafen, E.; van der Goot, F.G.; Robinett, C.C.; Gatti, M.; Gonzalez-Gaitan, M.; et al. Biochemical membrane lipidomics during Drosophila development. Dev. Cell 2013, 24, 98–111. [Google Scholar] [CrossRef]
- Masood, M.A.; Yuan, C.; Acharya, J.K.; Veenstra, T.D.; Blonder, J. Quantitation of ceramide phosphorylethanolamines containing saturated and unsaturated sphingoid base cores. Anal. Biochem. 2010, 400, 259–269. [Google Scholar] [CrossRef]
- Lau, M.J.; Nie, S.; Yang, Q.; Harshman, L.G.; Mao, C.; Williamson, N.A.; Hoffmann, A.A. Lipidomic profiling reveals concerted temporal patterns of functionally related lipids in Aedes aegypti females following blood feeding. Metabolites 2023, 13, 421. [Google Scholar] [CrossRef]
- Park, I.; Lee, W.; Yoo, Y.; Shin, H.; Oh, J.; Kim, H.; Kim, M.-A.; Hwang, J.S.; Bae, J.-S.; Na, M. Protective effect of tetrahydroquinolines from the edible insect Allomyrina dichotoma on LPS-induced vascular inflammatory responses. Int. J. Mol. Sci. 2020, 21, 3406. [Google Scholar] [CrossRef]
- Noh, J.H.; Yun, E.Y.; Park, H.; Jung, K.J.; Hwang, J.S.; Jeong, E.J.; Moon, K.-S. Subchronic oral dose toxicity of freeze-dried powder of Allomyrina dichotoma larvae. Toxicol. Res. 2015, 31, 69–75. [Google Scholar] [CrossRef]
- Kim, J.; Yun, E.Y.; Park, S.W.; Goo, T.W.; Seo, M.; Kim, K. Allomyrina dichotoma larvae regulate food intake and body weight in high fat diet-induced obese mice through mTOR and Mapk signaling pathways. Nutrients 2016, 8, 100. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Kwak, M.K.; Bae, G.D.; Park, E.Y.; Baek, D.J.; Kim, C.Y.; Jang, S.-E.; Jun, H.-S.; Oh, Y.S. Allomyrina dichotoma larva extract attenuates free fatty acid-induced lipotoxicity in pancreatic beta cells. Nutr. Res. Pract. 2021, 15, 294–308. [Google Scholar] [CrossRef]
- Youn, K.; Kim, J.Y.; Yeo, H.; Yun, E.Y.; Hwang, J.S.; Jun, M. Fatty acid and volatile oil compositions of Allomyrina dichotoma larvae. Prev. Nutr. Food Sci. 2012, 17, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Shadyro, O.; Samovich, S.; Edimecheva, I. Free-radical and biochemical reactions involving polar part of glycerophospholipids. Free Radic. Biol. Med. 2019, 144, 6–15. [Google Scholar] [CrossRef]
- Musille, P.M.; Kohn, J.A.; Ortlund, E.A. Phospholipid-driven gene regulation. FEBS Lett. 2013, 587, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yang, F.; Zhao, M.; Zhang, M.; Liu, J.; Marchioni, E. Determination and comparison of phospholipid profiles in eggs from seven different species using UHPLC-ESI-Triple TOF-MS. Food Chem. 2021, 339, 127856. [Google Scholar] [CrossRef]
- Sun, T.; Wang, X.; Cong, P.; Xu, J.; Xue, C. Mass spectrometry-based lipidomics in food science and nutritional health: A comprehensive review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2530–2558. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Youn, K.; Yun, E.Y.; Hwang, J.S.; Ahn, M.R.; Jeong, W.S.; Jun, M. Effects of solvent fractions of Allomyrina dichotoma larvae through the inhibition of in vitro BACE1 and β-amyloid(25-35)-induced toxicity in rat pheochromocytoma PC12 cells. Entomol. Res. 2014, 44, 23–30. [Google Scholar] [CrossRef]
- Huang, Y.J.; Liu, X.; Huang, C.; Shi, X.J.; Zhao, Q.N.; Tian, Y.; Dai, L.; Zhang, J.; Man, Y.; Wang, S. Fatty oil content differences of Eupolyphaga sinensis products based on lipidomics. J. Binzhou Med. Univ. 2022, 45, 29–34. [Google Scholar] [CrossRef]
- Huang, X.; Li, W.Q.; Li, C.Y.; Zhang, X.D. Lipid group analysis of adult growth of Drosophila melanogaster. Chin. J. Biochem. Mol. Biol. 2018, 34, 982–989. [Google Scholar] [CrossRef]
- Wegener, J.; Krause, S.; Parafianczuk, V.; Chaniotakis, I.; Schiller, J.; Dannenberger, D.; Engel, K.M. Lipidomic specializations of honeybee (Apis mellifera) castes and ethotypes. J. Insect Physiol. 2022, 142, 104439. [Google Scholar] [CrossRef]
- Dowhan, W.; Bogdanov, M. Lipid-dependent membrane protein topogenesis. Annu. Rev. Biochem. 2009, 78, 515–540. [Google Scholar] [CrossRef]
- Lessen, H.J.; Sapp, K.C.; Beaven, A.H.; Ashkar, R.; Sodt, A.J. Molecular mechanisms of spontaneous curvature and softening in complex lipid bilayer mixtures. Biophys. J. 2022, 121, 3188–3199. [Google Scholar] [CrossRef]
- Vance, J.E. Phosphatidylserine and phosphatidylethanolamine in mammalian cells: Two metabolically related aminophospholipids. J. Lipid Res. 2008, 49, 1377–1387. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Li, H.; Xiao, Y.; Harlina, P.W.; Geng, F. Quantitative lipidomic analysis of chicken egg yolk during its formation. J. Sci. Food Agric. 2023, 103, 3997–4005. [Google Scholar] [CrossRef]
- Hui, D.Y. Intestinal phospholipid and lysophospholipid metabolism in cardiometabolic disease. Curr. Opin. Lipidol. 2016, 27, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Wiedeman, A.M.; Barr, S.I.; Green, T.J.; Xu, Z.; Innis, S.M.; Kitts, D.D. Dietary choline intake: Current state of knowledge across the life cycle. Nutrients 2018, 10, 1513. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline. In Modern Nutrition in Health and Disease, 11th ed.; Ross, A.C., Caballero, B., Cousins, R.J., Tucker, K.L., Ziegler, T.R., Eds.; Lippincott Williams & Wilkins: Baltimore, MD, USA, 2014; pp. 416–426. [Google Scholar]
- Li, A.; Dewettinck, K.; Verheust, Y.; Van de Walle, D.; Raes, K.; Diehl, B.; Tzompa-Sosa, D.A. Edible insects as a novel source of lecithin: Extraction and lipid characterization of black soldier fly larvae and yellow mealworm. Food Chem. 2024, 452, 139391. [Google Scholar] [CrossRef]
- Cockcroft, S. Phosphatidylinositol transfer proteins: A requirement in signal transduction and vesicle traffic. BioEssays 1998, 20, 423–432. [Google Scholar] [CrossRef]
- Zheng, X.; Xin, Y.; Peng, Y.; Shan, J.; Zhang, N.; Wu, D.; Guo, J.; Huang, J.; Guan, W.; Shi, S.; et al. Lipidomic analyses reveal enhanced lipolysis in planthoppers feeding on resistant host plants. Sci. China Life Sci. 2021, 64, 1502–1521. [Google Scholar] [CrossRef]
- Wang, S.; De Souza, C.; Ramachandran, M.; Luo, Y.; Zhang, Y.; Yi, H.; Ma, Z.; Zhang, L.; Lin, K. Lipidomics insight on differences between human MFGM and dietary-derived lipids. Food Chem. 2023, 422, 136236. [Google Scholar] [CrossRef]
- Athenstaedt, K. Phosphatidic acid biosynthesis in the model organism yeast Saccharomyces cerevisiae—A survey. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2021, 1866, 158907. [Google Scholar] [CrossRef]
- Henneberry, A.L.; McMaster, C.R. Cloning and expression of a human choline/ethanolaminephosphotransferase: Synthesis of phosphatidylcholine and phosphatidylethanolamine. Biochem. J. 1999, 339, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Horibata, Y.; Hirabayashi, Y. Identification and characterization of human ethanolaminephosphotransferase1. J. Lipid Res. 2007, 48, 503–508. [Google Scholar] [CrossRef] [PubMed]
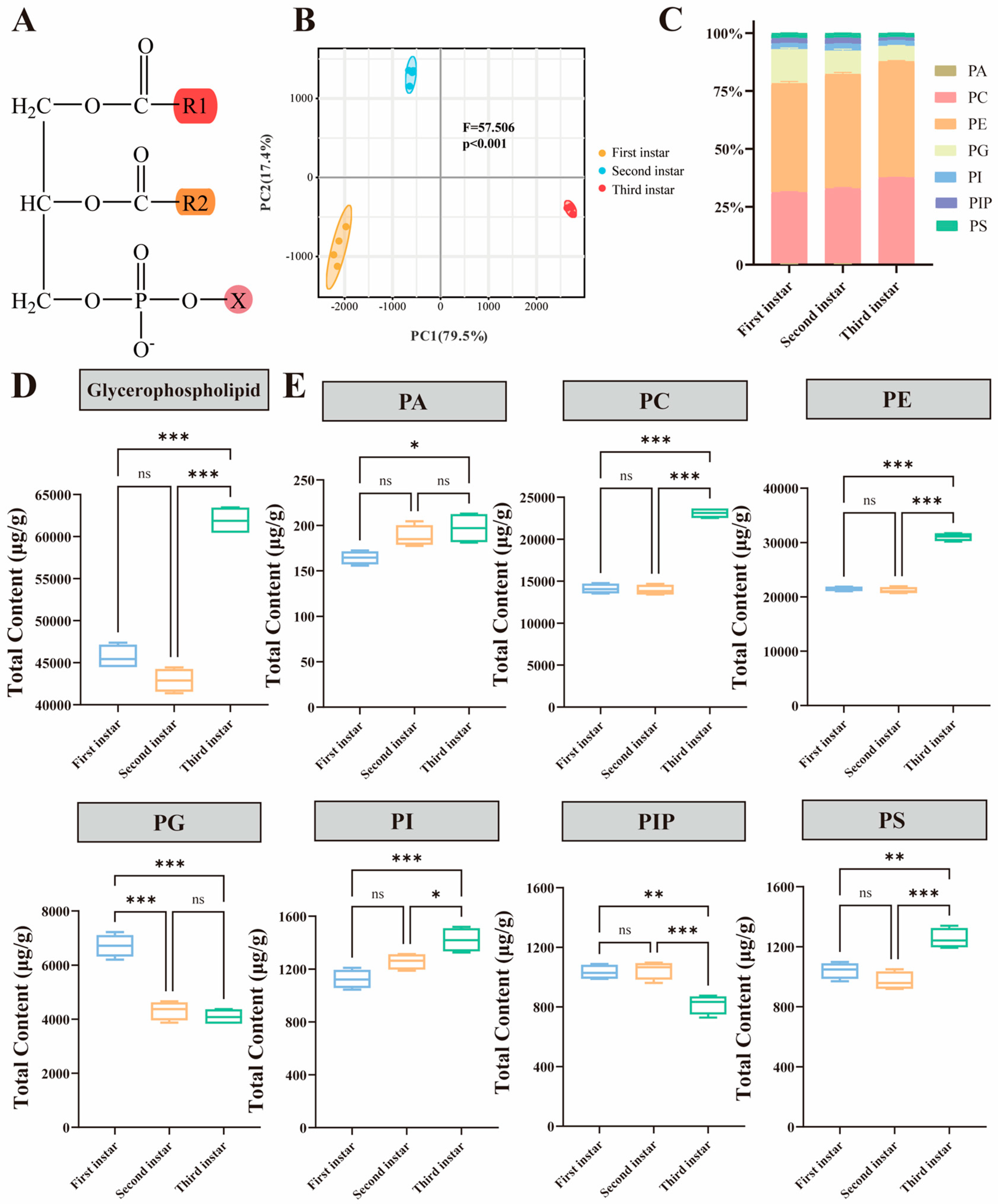

| Class | Number | Content (ug/g) | Percent (%) |
|---|---|---|---|
| CL | 72 | 4384.64 | 2.83 |
| PA | 14 | 549.39 | 0.35 |
| PC | 328 | 51,155.36 | 33.03 |
| PE | 223 | 73,696.16 | 47.58 |
| PG | 84 | 15,125.26 | 9.77 |
| PI | 57 | 3802.01 | 2.45 |
| PIP | 21 | 2899.63 | 1.87 |
| PS | 34 | 3267.48 | 2.11 |
| Total | 833 | 15,4879.93 | 100.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, K.; Liu, J.-N.; Wang, C.-X.; Wang, T.; Pan, Y.; Wang, S.; Xi, J.-H. Glycerophospholipid Profiles of Allomyrina dichotoma Larvae at Different Instars Based on Lipidomics and Transcriptomics Suggest a Promising Lipid Source. Insects 2025, 16, 1220. https://doi.org/10.3390/insects16121220
Fang K, Liu J-N, Wang C-X, Wang T, Pan Y, Wang S, Xi J-H. Glycerophospholipid Profiles of Allomyrina dichotoma Larvae at Different Instars Based on Lipidomics and Transcriptomics Suggest a Promising Lipid Source. Insects. 2025; 16(12):1220. https://doi.org/10.3390/insects16121220
Chicago/Turabian StyleFang, Kui, Jia-Nan Liu, Cong-Xu Wang, Tao Wang, Yu Pan, Shang Wang, and Jing-Hui Xi. 2025. "Glycerophospholipid Profiles of Allomyrina dichotoma Larvae at Different Instars Based on Lipidomics and Transcriptomics Suggest a Promising Lipid Source" Insects 16, no. 12: 1220. https://doi.org/10.3390/insects16121220
APA StyleFang, K., Liu, J.-N., Wang, C.-X., Wang, T., Pan, Y., Wang, S., & Xi, J.-H. (2025). Glycerophospholipid Profiles of Allomyrina dichotoma Larvae at Different Instars Based on Lipidomics and Transcriptomics Suggest a Promising Lipid Source. Insects, 16(12), 1220. https://doi.org/10.3390/insects16121220





