Oil-Based Drilling Cuttings from Shale Gas Wells Treated with CO2 Switchable Hydrophilic Solvents: Priority Pollutant Migration and Produced Wastewater Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extracting Petroleum Hydrocarbon from ODBCs with CO2 SHSs
2.2.1. Extracting Petroleum Hydrocarbon Procedure
2.2.2. Recovering CO2 SHSs Procedure
2.3. Analytical Methods
2.4. Characterizations
3. Results and Discussion
3.1. Morphology and Composition of Shale Gas OBDCs
3.2. Extracting Petroleum Hydrocarbon from shale gas ODBCs by Using CO2 SHSs
3.3. Priority Pollutant Migration and Produced Wastewater Assessment
3.3.1. FT-IR Analysis
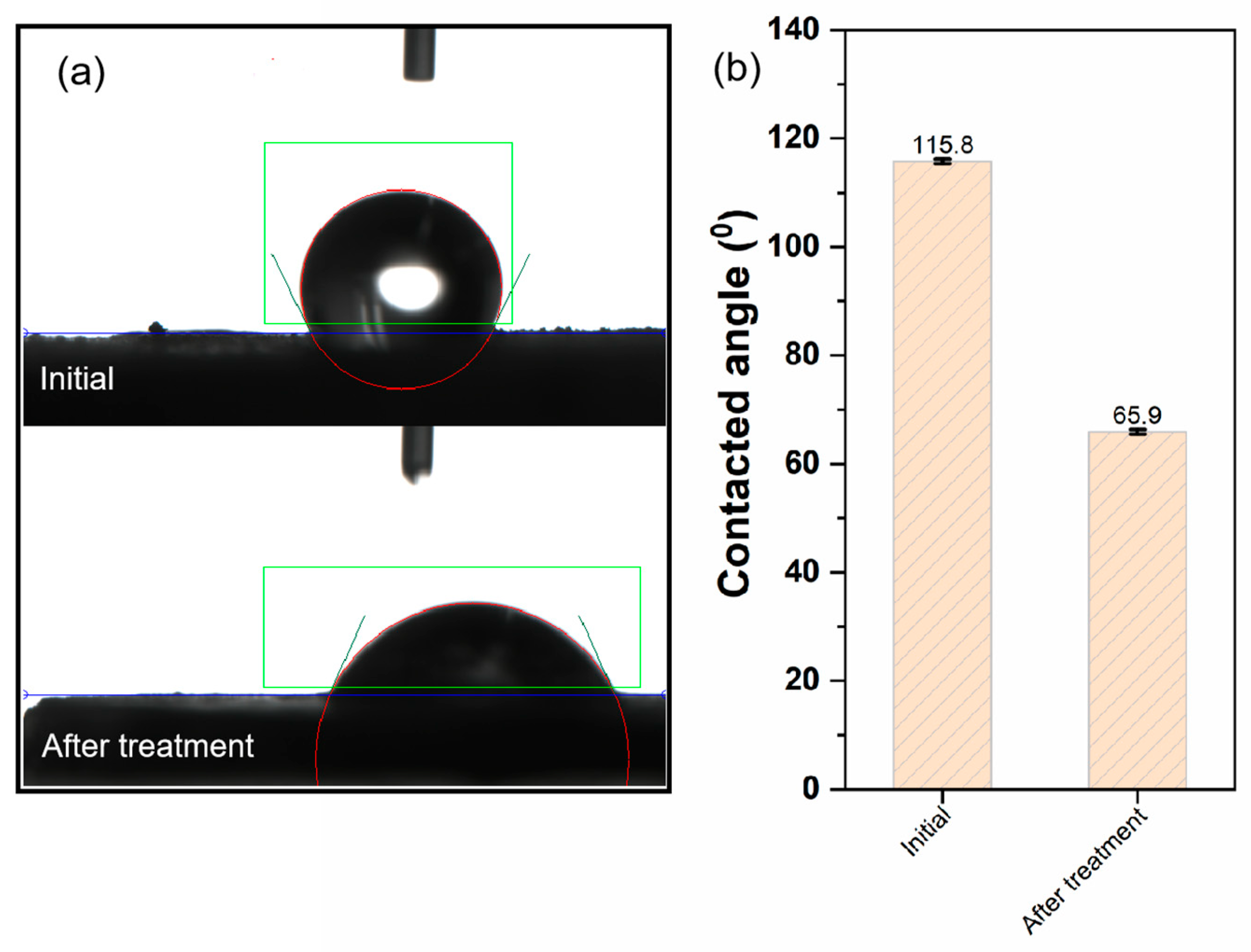
3.3.2. Contacted Angle Analysis
3.3.3. SEM Analysis
3.3.4. EDS Analysis
3.3.5. Produced Wastewater Analysis
3.4. Priority Pollutant Migration Mechanism from ODBCs to Produced Wastewater
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Milkov, A.V.; Faiz, M.; Etiope, G. Geochemistry of shale gases from around the world: Composition, origins, isotope reversals and rollovers, and implications for the exploration of shale plays. Org. Geochem. 2020, 143, 103997. [Google Scholar] [CrossRef]
- Naumenko-Dezes, M.; Wolfram Kloppmann, W.; Blessing, M.; Bondu, R.; Gaucher, E.C.; Mayer, B. Natural gas of radiolytic origin: An overlooked component of shale gas. Proc. Natl. Acad. Sci. USA 2022, 119, e2114720119. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.Y.; Hao, F.; Guo, X.S.; Yi, J.Z.; Shu, Z.G.; Bao, H.Y.; Zhu, X.Y. Effect of lithofacies on the pore system of over-mature Longmaxi shale in the Jiaoshiba area, Sichuan Basin, China. Mar. Pet. Geol. 2019, 109, 886–898. [Google Scholar] [CrossRef]
- Liu, Y.X. The thinking of shale gas development in China according to the US shale gas development experiments. In Proceedings of the 2nd International Conference on Energy, Environment and Sustainable Development (EESD 2012), Jilin, China, 27–29 February 2012. [Google Scholar]
- Iqbal, O.; Padmanabhan, E.; Mandal, A.; Dvorkin, J. Characterization of geochemical properties and factors controlling the pore structure development of shale gas reservoirs. J. Pet. Sci. Eng. 2021, 206, 109001. [Google Scholar] [CrossRef]
- Gao, S.; Dong, D.; Tao, K.; Guo, W.; Li, X.; Zhang, S. Experiences and lessons learned from China’s shale gas development: 2005–2019. J. Nat. Gas Sci. Eng. 2021, 85, 103648. [Google Scholar] [CrossRef]
- Gao, D.; Xie, J.; Huang, S.; Wu, S.; Wu, P.; Huang, W. Research and application of evaluation methods for functional characteristics of oil-based drilling fluid in shale gas wells. Geofluids 2021, 2021, 8814032. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, F.; Cheng, X.; Huang, S.; Zhang, C.; Zhou, M.; Mei, K.; Zhang, L. Resource utilization from solid waste originated from oil-based shale drilling cutting during shale gas development. Chemosphere 2022, 298, 134318. [Google Scholar] [CrossRef]
- Ball, A.S.; Stewart, R.J.; Schliephake, K. A review of the current options for the treatment and safe disposal of drill cuttings. Waste Manag. Res. 2012, 30, 457–473. [Google Scholar] [CrossRef]
- Yang, H.; Diao, H.; Zhang, Y.; Xia, S. Treatment and novel resource-utilization methods for shale gas oil based drill cuttings—A review. J. Environ. Manag. 2022, 317, 115462. [Google Scholar] [CrossRef]
- Cao, T.T.; Song, Z.J.; Wang, S.B.; Cao, X.X.; Li, Y.; Xia, J. Characterizing the pore structure in the silurian and permian shales of the Sichuan Basin, China. Mar. Pet. Geol. 2015, 61, 140–150. [Google Scholar] [CrossRef]
- Cao, X.J.; Wang, M.G.; Kang, J.; Wang, S.H.; Liang, Y. Fracturing technologies of deep shale gas horizontal wells in the Weirong Block, southern Sichuan Basin. Nat. Gas Ind. B 2020, 7, 64–70. [Google Scholar] [CrossRef]
- Gou, Q.Y.; Xu, S.; Hao, F.; Shu, Z.; Zhang, Z.Y. Making sense of microfractures to the Longmaxi shale reservoir quality in the Jiaoshiba area, Sichuan Basin, China: Implications for the accumulation of shale gas. J. Nat. Gas Sci. Eng. 2021, 94, 104107. [Google Scholar] [CrossRef]
- Wei, Z.; Wang, Y.; Wang, G.; Sun, Z.; Xu, L. Pore characterization of organic-rich Late Permian Da-long Formation shale in the Sichuan Basin, southwestern China. Fuel 2018, 211, 507–516. [Google Scholar] [CrossRef]
- Gong, J.; Qiu, Z.; Zou, C.; Wang, H.; Shi, Z. An integrated assessment system for shale gas resources associated with graptolites and its application. Appl. Energy 2020, 262, 114524. [Google Scholar] [CrossRef]
- Jiang, G.; Yu, J.; Jiang, H.; Xu, B.; Tang, P.; Zhao, L.; Li, H.; Xiang, Q.; Hu, J. Physicochemical characteristics of oil-based cuttings from pretreatment in shale gas well sites. J. Environ. Sci. Health A 2020, 55, 1041–1049. [Google Scholar] [CrossRef]
- Hu, G.; Liu, H.; Chen, C.; Hou, H.; Li, J.; Hewage, K.; Sadiq, R. Low-temperature thermal desorption and secure landfill for oil-based drill cuttings management: Pollution control, human health risk, and probabilistic cost assessment. J. Hazard. Mater. 2021, 410, 124570. [Google Scholar] [CrossRef]
- Goulding, L. Spotlights: Safety work and working safe, rule violations, managing drill cuttings, and more. ACS Chem. Health Saf. 2022, 29, 3–4. [Google Scholar] [CrossRef]
- Miao, W.; Li, X.; Wang, Y.; Lv, Y. Pyrolysis characteristics of oilfield sludge and the comparison of kinetic analysis with two representative methods. J. Pet. Sci. Eng. 2019, 182, 106309. [Google Scholar] [CrossRef]
- Abnisa, F.; Alaba, P.A. Recovery of liquid fuel from fossil-based solid wastes via pyrolysis technique: A review. J. Environ. Chem. Eng. 2021, 9, 106593. [Google Scholar] [CrossRef]
- Fei, Z.; Yanxia, L.; Zhongliang, L.; Yongzhi, T. Flow and heat transfer characteristics of oil-based drilling cuttings in a screw-driving spiral heat exchanger. Appl. Therm. Eng. 2020, 181, 115881. [Google Scholar] [CrossRef]
- Alves, G.M.; Junior, I.P. Microwave remediation of oil-contaminated drill cuttings—A review. J. Pet. Sci. Eng. 2021, 207, 109137. [Google Scholar] [CrossRef]
- Hou, Y.; Qi, S.; You, H.; Huang, Z.; Niu, Q. The study on pyrolysis of oil-based drilling cuttings by microwave and electric heating. J. Environ. Manag. 2018, 228, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhou, J.; Chen, Z.; Chen, H.; Chen, Q.; He, C.; Liu, X.; Xu, Y. A laboratory evaluation of superheated steam extraction process for decontamination of oil-based drill cuttings. J. Environ. Chem. Eng. 2018, 6, 6691–6699. [Google Scholar] [CrossRef]
- Chen, Z.; Li, D.; Tong, K.; Chen, Z.; Chen, H.; Chen, Q.; Xu, Y. Static decontamination of oil-based drill cuttings with pressurized hot water using response surface methodology. Environ. Sci. Pollut. Res. 2019, 26, 7216–7227. [Google Scholar] [CrossRef]
- Liu, H.; Li, J.; Zhao, M.; Li, Y.; Chen, Y. Remediation of oil-based drill cuttings using low-temperature thermal desorption: Performance and kinetics modeling. Chemosphere 2019, 235, 1081–1088. [Google Scholar] [CrossRef]
- Khanpour, R.; Sheikhi-Kouhsar, M.R.; Esmaeilzadeh, F.; Mowla, D. Removal of contaminants from polluted drilling mud using supercritical carbon dioxide extraction. J. Supercrit. Fluids 2014, 88, 1–7. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, Z.; Yin, F.; Wang, G.; Chen, H.; He, C.; Xu, Y. Supercritical water oxidation of oil-based drill cuttings. J. Hazard. Mater. 2017, 332, 205–213. [Google Scholar] [CrossRef]
- Poyai, T.; Getwech, C.; Dhanasin, P.; Punyapalakul, P.; Painmanakul, P.; Chawaloesphonsiya, N. Solvent-based washing as a treatment alternative for onshore petroleum drill cuttings in Thailand. Sci. Total Environ. 2020, 718, 137384. [Google Scholar] [CrossRef]
- Yan, P.; Lu, M.; Guan, Y.; Zhang, W.; Zhang, Z. Remediation of oil-based drill cuttings through a biosurfactant-based washing followed by a biodegradation treatment. Bioresour. Technol. 2011, 102, 10252–10259. [Google Scholar] [CrossRef]
- Huang, X.; Jiang, G.; Deng, Z. Oil extraction from oil-contaminated drill cuttings using a recyclable single-phase O/W microemulsion. Tenside Surfact. Det. 2015, 52, 454–463. [Google Scholar] [CrossRef]
- Feng, Q.; Zhong, M.-J.; Liu, H.; Liu, X.-J.; Peng, Z.-G. Preparation, investigation and characterization of microemulsion used in cleaning waste liquid and drilling cuttings. Energy Sources Part A Recover. Util. Environ. Eff. 2020, 42, 919–929. [Google Scholar] [CrossRef]
- Dantas, T.N.D.; da Silva, D.N.N.; Neto, A.A.D.; Cabral, T.J.D.; dos Anjos, A.S.D. Treatment of drill cuttings using microemulsion. J. Pet. Explor. Prod. Technol. 2020, 10, 1243–1251. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Jiao, Y.; Luo, Q.; Hu, W.; Fang, S.; Tang, C.; Liu, Q. Treatment of oil-based drill cuttings by hydrophobic deep eutectic solvents. Can. J. Chem. Eng. 2021, 100, 1747–1754. [Google Scholar] [CrossRef]
- Phan, L.; Andreatta, J.R.; Horvey, L.K.; Edie, C.F.; Luco, A.L.; Mirchandani, A.; Darensbourg, D.J.; Jessop, P.G. Switchable-polarity solvents prepared with a single liquid component. J. Org. Chem. 2008, 73, 127–132. [Google Scholar] [CrossRef]
- Phan, L.; Brown, H.; White, J.; Hodgson, A.; Jessop, P.G. Soybean oil extraction and separation using switchable or expanded solvents. Green Chem. 2009, 11, 53–59. [Google Scholar] [CrossRef]
- Jessop, P.G.; Phan, L.; Carrier, A.; Robinson, S.; Dürr, C.J.; Harjani, J.R. A solvent having switchable hydrophilicity. Green Chem. 2010, 12, 809. [Google Scholar] [CrossRef]
- Jessop, P.G.; Kozycz, L.; Rahami, Z.G.; Schoenmakers, D.; Boyd, A.R.; Wechsler, D.; Holland, A.M. Tertiary amine solvents having switchable hydrophilicity. Green Chem. 2011, 13, 619. [Google Scholar] [CrossRef]
- Jessop, P.G.; Vanderveen, J.R.; Durelle, J. Design and evaluation of switchable-hydrophilicity solvents. Green Chem. 2014, 16, 1187–1197. [Google Scholar]
- Holland, A.; Wechsler, D.; Patel, A.; Molloy, B.M.; Boyd, A.R.; Jessop, P.G. Separation of bitumen from oil sands using a switchable hydrophilicity solvent. Can. J. Chem. 2012, 90, 805–810. [Google Scholar] [CrossRef]
- Samorì, C.; Torri, C.; Samorì, G.; Fabbri, D.; Galletti, P.; Guerrini, F.; Pistocchi, R.; Tagliavini, E. Extraction of hydrocarbons from microalga Botryococcus braunii with switchable solvents. Bioresour. Technol. 2010, 101, 3274–3279. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, C.; Zhao, J.; Li, X.; Lu, H. Extracting and recovering diesel from oil-based drill cuttings using switchable hydrophilic solven. Chem. Eng. Res. Des. 2017, 128, 27–36. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association: Washington, DC, USA; American Water Works Association: Denver, CO, USA; Water Environment Federation: Alexandria, VA, USA, 1998. [Google Scholar]
- Lauzurique, Y.; Espinoza, L.C.; Huiliñir, C.; García, V.; Salazar, R. Anodic oxidation of industrial winery wastewater using different anodes. Water 2022, 12, 95. [Google Scholar] [CrossRef]











| Samples | pH | Conductivity (µS/cm) | COD (mg/L) | BOD5 (mg/L) | Ammonia-N (mg/L) | Total Nitrogen (mg/L) | TOC (mg/L) | Petroleum (mg/L) |
|---|---|---|---|---|---|---|---|---|
| DI water | 6.8 | 0.75 | ND | ND | ND | 0.19 | 1.00 | ND |
| Produced water | 8.9 | 341.00 | 561.00 | 238.00 | ND | 40.60 | 309.00 | 0.27 |
| Samples | K (mg/L) | Na (mg/L) | Ca (mg/L) | Ba (mg/L) | Cu (mg/L) | Fe (mg/L) | Sr (mg/L) | Cl− (mg/L) | NO3− (mg/L) | SO42− (mg/L) | TDS (mg/L) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| DI water | ND | ND | ND | ND | ND | ND | ND | 5.36 | 0.096 | 0.984 | ND |
| Produced water | 6.44 | 12.60 | 28.80 | 0.28 | 0.01 | 0.01 | 0.16 | 145 | 0.802 | 9.830 | 264 |
| Samples | Phenol (mg/L) | Naphthalene (mg/L) | Phenanthrene (mg/L) | Anthracene (mg/L) |
|---|---|---|---|---|
| DI water | ND | ND | ND | ND |
| Produced water | 0.0031 | 0.0000129 | 0.000059 | 0.000058 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, J.; Zhao, G.; Zhang, Y.; Wu, Z.; Tang, Q. Oil-Based Drilling Cuttings from Shale Gas Wells Treated with CO2 Switchable Hydrophilic Solvents: Priority Pollutant Migration and Produced Wastewater Assessment. Water 2022, 14, 3433. https://doi.org/10.3390/w14213433
Huang J, Zhao G, Zhang Y, Wu Z, Tang Q. Oil-Based Drilling Cuttings from Shale Gas Wells Treated with CO2 Switchable Hydrophilic Solvents: Priority Pollutant Migration and Produced Wastewater Assessment. Water. 2022; 14(21):3433. https://doi.org/10.3390/w14213433
Chicago/Turabian StyleHuang, Junhan, Guoguang Zhao, Yibo Zhang, Zheng Wu, and Quanwu Tang. 2022. "Oil-Based Drilling Cuttings from Shale Gas Wells Treated with CO2 Switchable Hydrophilic Solvents: Priority Pollutant Migration and Produced Wastewater Assessment" Water 14, no. 21: 3433. https://doi.org/10.3390/w14213433





