Artemia spp. (Crustacea, Anostraca) in Crimea: New Molecular Genetic Results and New Questions without Answers
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Haplotypes | Species | GenBank Number | Geographical Locality | Reference |
|---|---|---|---|---|
| H1 | A. franciscana | ON872200 | Crimea (Aktashskoe) | this work |
| H2 | A. urmiana | ON872198 | Crimea (Aktashskoe) | this work |
| A. parthenogenetica | KF691333-337,343,345,357,358,359,361, | Iran | [6] | |
| A. parthenogenetica | KF691530-532 | Turkmenistan | [6] | |
| H3 | A. urmiana | ON872199, ON872202 | Crimea (Aktashskoe, Adzhjigol) | this work |
| A. parthenogenetica | KF691148-153,166-172,187-189,208-212,224-226,233-236,238,287-290, | China | [6] | |
| A. parthenogenetica | KF691338-342,344,346,348 | Iran | [6] | |
| A. parthenogenetica | KF691373-375 | Iraq | [6] | |
| A. parthenogenetica | KF691391-434 | Kazakhstan | [6] | |
| A. parthenogenetica | KF691442-448 | Pakistan | [6] | |
| A. parthenogenetica | KF691455,456,458-461,465,467-475,477,478,480,485-491,493,495-497 | Russia | [6] | |
| A. parthenogenetica | KF691534 | Turkmenistan | [6] | |
| A. parthenogenetica | KF691548-553,555 | Uzbekistan | [6] | |
| H4 | A. franciscana | ON872204 | Crimea (Adzhjigol) | this work |
| A. franciscana | KF691154-156,174-175,176,179,181,184,185,186,190,192,206,222,223,231,232,252,255,258,261,264,266,267,281,292,303,315, | China | [6] | |
| A. franciscana | KF691351,353,355, | Iran | [6] | |
| A. franciscana | KF691439-441 | Pakistan | [6] | |
| A. franciscana | KF691508 | Sri Lanka | [6] | |
| A. franciscana | KF691568 | Vietnam | [6] | |
| H5 | A. franciscana | ON872205 | Crimea (Adzhjigol) | this work |
| A. franciscana | KF691191,205,239,240,242-244,250,251,253,259,260,262,263,278,280,294,295,296,297,304,305,307,308,309-314, | China | [6] | |
| A. franciscana | KF691378-382 | Iraq | [6] | |
| A. franciscana | KF691449-454 | Portugal | [6] | |
| A. franciscana | KF691503-507 | Sri Lanka | [6] | |
| A. franciscana | KF691556-567 | Vietnam | [6] | |
| H6 | A. sinica | ON872201, ON872203 | Crimea (Adzhjigol) | this work |
| H7 | A. salina | ON872208 | Crimea (Sasyk-Sivash) | this work |
| H8 | A. salina | ON872209, ON872210, ON872211 | Crimea (Sasyk-Sivash) | this work |
| H9 | A. salina | ON872206 | Crimea (Sasyk-Sivash) | this work |
| H10 | A. salina | ON872207 | Crimea (Sasyk-Sivash) | this work |
| H11 | A. franciscana | DQ119645 | USA | [40] |
| H12 | A. sinica | DQ119650 | Mongolia | [40] |
| H13 | A. urmiana | JX512748,755,758,762,766,769,771,774,776,778,780,783,788,790,795,796,803,804,808 | Iran (Urmia) | [38] |
| H14 | A. parthenogenetica | DQ426826 | Spain | [40] |
| H15 | A. salina | DQ426831,853,857 | Spain | [40] |
| H16 | A. salina | DQ426832, KF691502 | Spain | [40] |
| H17 | A. salina | DQ426833 | Spain | [40] |
| H18 | A. salina | DQ426834,856 | Spain | [40] |
| H19 | A. salina | DQ426836 | Spain | [40] |
| H20 | A. salina | DQ426837 | Spain | [40] |
| H21 | A. salina | DQ426841 | Spain | [40] |
| H22 | A. salina | DQ426845 | Spain | [40] |
| H23 | A. salina | DQ426846 | Spain | [40] |
| H24 | A. salina | DQ426847 | Spain | [40] |
| H25 | A. salina | DQ426848 | Spain | [40] |
| H26 | A. salina | DQ426858 | Spain | [40] |
| H27 | A. sinica | EF615592 | China | [40] |
| H28 | A. salina | EU543444 | Spain | [40] |
| H29 | A. salina | EU543445 | Spain | [40] |
| H30 | A. salina | EU543448 | Spain | [40] |
| H31 | A. salina | EU543452 | Morocco | [40] |
| H32 | A. salina | EU543453 | Morocco | [40] |
| H33 | A. salina | EU543456 | Tunisia | [40] |
| H34 | A. salina | EU543457 | Tunisia | [40] |
| H35 | A. salina | EU543467 | Algeria | [40] |
| H36 | A. salina | EU543468 | Algeria | [40] |
| H37 | A. salina | EU543470 | Egypt | [40] |
| H38 | A. salina | EU543480 | Italy | [40] |
| H39 | A. salina | EU543481 | Italy | [40] |
| H40 | A. salina | EU543485 | South Africa | [40] |
| H41 | A. salina | GU248381 | Italy | [41] |
| H42 | A. sinica | HM998990 | China | [42] |
| H43 | D. tenebrosa | HQ972028 | - | - |
| H44 | A. urmiana | JX512751 | Iran (Urmia) | [38] |
| H45 | A. urmiana | JX512756,764, | Iran (Urmia) | [38] |
| H46 | A. urmiana | JX512775,791,805 | Iran (Urmia) | [38] |
| H47 | A. urmiana | JX512777,801 | Iran (Urmia) | [38] |
| H48 | A. urmiana | JX512782 | Iran (Urmia) | [38] |
| H49 | A. urmiana | JX512792 | Iran (Urmia) | [38] |
| H50 | A. franciscana | KF691137-141 | Canada | [6] |
| H51 | A. franciscana | KF691143-147 | Cape Verde | [6] |
| H52 | A. parthenogenetica | KF691159,257 | China | [6] |
| H53 | A. franciscana | KF691160-165,173,177,178,180,182,207,227-230, 237,241,256,279,282-286,291,293 | China | [6] |
| A. franciscana | KF691328-332 | India | [6] | |
| A. franciscana | KF691347,349,354,356, | Iran | [6] | |
| A. franciscana | KF691376,377,381,383, | Iraq | [6] | |
| A. franciscana | KF691384-390 | Jamaica | [6] | |
| A. franciscana | KF691535,537,538,543,544,546 | USA | [6] | |
| H54 | A. parthenogenetica | KF691183 | China | [6] |
| A. parthenogenetica | KF691462 | Russia | [6] | |
| H55 | A. franciscana | KF691196,219 | China | [6] |
| H56 | A. parthenogenetica | KF691199-204,265,268, | China | [6] |
| H57 | A. sinica | KF691270,271 | China | [6] |
| H58 | A. sinica | KF691272 | China | [6] |
| H59 | A. sinica | KF691274,276,277,300,302 | China | [6] |
| H60 | A. sinica | KF691275,299 | China | [6] |
| H61 | A. franciscana | KF691306 | China | [6] |
| H62 | A. franciscana | KF691320,322 | Columbia | [6] |
| H63 | A. parthenogenetica | KF691360,367,369-372 | Iran | [6] |
| H64 | A. franciscana | KF691435,437,438 | Mexico | [6] |
| H65 | A. parthenogenetica | KF691457 | Russia | [6] |
| H66 | A. parthenogenetica | KF691464,466,476 | Russia | [6] |
| H67 | A. parthenogenetica | KF691479,482,484,492,494 | Russia | [6] |
| H68 | A. parthenogenetica | KF691481,483 | Russia | [6] |
| H69 | A. parthenogenetica | KF691520,522-525,528,529 | Turkey | [6] |
| H70 | A. franciscana | KF691536,540,542,545 | USA | [6] |
| A. franciscana | KJ863431,438,439,443,455,460,466,467,471,474,479,481,482,487,489 | - | [38] | |
| H71 | A. franciscana | KF691539 | Canada | [6] |
| A. franciscana | KJ863454 | USA | [38] | |
| H72 | A. urmiana | KF707695 | Iran | [43] |
| H73 | A. sinica | KF707886-889 | China | [43] |
| H74 | A. franciscana | KJ863484 | USA | [38] |
| H75 | A. parthenogenetica | KU053797-802 | Djarylpach | [39] |
| A. parthenogenetica | KU053803-807 | Sakskoye | [39] | |
| A. parthenogenetica | KU053808-818 | Dzharylhach | [39] | |
| H76 | A. parthenogenetica | KU053811,814 | Ukraine | [39] |
| H77 | A. sinica | LC195586 | Mongolia | [44] |
References
- Abatzopoulos, T.J.; Beardmore, J.; Clegg, J.S.; Sorgeloos, P. Artemia: Basic and Applied Biology; Springer Science & Business Media: Dordrecht, The Netherlands, 2013; 286p. [Google Scholar]
- Sainz-Escudero, L.; López-Estrada, E.K.; Rodríguez-Flores, P.C.; García-París, M. Settling taxonomic and nomenclatural problems in brine shrimps, Artemia (Crustacea: Branchiopoda: Anostraca), by integrating mitogenomics, marker discordances and nomenclature rules. PeerJ 2021, 9, e10865. [Google Scholar] [CrossRef] [PubMed]
- Gajardo, G.O.; Beardmore, J.A. Coadaptation: Lessons from the brine shrimp Artemia, “the aquatic Drosophila” (Crustacea; Anostraca). Rev. Chil. Hist. Nat. 2001, 74, 65–72. [Google Scholar] [CrossRef]
- Gajardo, G.M.; Beardmore, J.A. The brine shrimp Artemia: Adapted to critical life conditions. Front. Physiol. 2012, 3, 185. [Google Scholar] [CrossRef] [PubMed]
- Baxevanis, A.D.; Maniatsi, S.; Kouroupis, D.; Marathiotis, K.; Kappas, I.; Kaiser, H.; Abatzopoulos, T.J. Genetic identification of South African Artemia species: Invasion, replacement and co-occurrence. J. Mar. Biolog. Assoc. 2014, 94, 775–785. [Google Scholar] [CrossRef]
- Eimanifar, A.; Van Stappen, G.; Marden, B.; Wink, M. Artemia biodiversity in Asia with the focus on the phylogeography of the introduced American species Artemia franciscana Kellogg, 1906. Mol. Phylogenet. Evol. 2014, 79, 392–403. [Google Scholar] [CrossRef]
- Shadrin, N.; Yakovenko, V.; Anufriieva, E. Suppression of Artemia spp. (Crustacea, Anostraca) populations by predators in the Crimean hypersaline lakes: A review of the evidence. Int. Rev. Hydrobiol. 2019, 104, 5–13. [Google Scholar] [CrossRef]
- Sainz-Escudero, L.; López-Estrada, E.K.; Rodríguez-Flores, P.C.; García-París, M. Brine shrimps adrift: Historical species turnover in Western Mediterranean Artemia (Anostraca). Biol. Invasions 2022, 24, 2477–2498. [Google Scholar] [CrossRef]
- Green, A.J.; Sánchez, M.I.; Amat, F.; Figuerola, J.; Hontoria, F.; Ruiz, O.; Hortas, F. Dispersal of invasive and native brine shrimps Artemia (Anostraca) via waterbirds. Limnol. Oceanogr. 2005, 50, 737–742. [Google Scholar] [CrossRef]
- Muñoz, J.; Amat, F.; Green, A.J.; Figuerola, J.; Gomez, A. Bird migratory flyways influence the phylogeography of the invasive brine shrimp Artemia franciscana in its native American range. PeerJ 2013, 1, e200. [Google Scholar]
- Redón, S.; Gajardo, G.; Vasileva, G.P.; Sánchez, M.I.; Green, A.J. Explaining variation in abundance and species diversity of avian cestodes in brine shrimps in the Salar de Atacama and other Chilean wetlands. Water 2021, 13, 1742. [Google Scholar] [CrossRef]
- Van Stappen, G.; Sui, L.; Hoa, V.N.; Tamtin, M.; Nyonje, B.; de Medeiros Rocha, R.; Sorgeloos, P.; Gajardo, G. Review on integrated production of the brine shrimp Artemia in solar salt ponds. Rev. Aquac. 2020, 12, 1054–1071. [Google Scholar] [CrossRef]
- Nunes, B.S.; Carvalho, F.D.; Guilhermino, L.M.; Van Stappen, G. Use of the genus Artemia in ecotoxicity testing. Environ. Pollut. 2006, 144, 453–462. [Google Scholar] [CrossRef] [PubMed]
- El Fels, L.; Hafidi, M.; Ouhdouch, Y. Artemia salina as a new index for assessment of acute cytotoxicity during co-composting of sewage sludge and lignocellulose waste. Waste Manag. 2016, 50, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Lenormand, T.; Nougué, O.; Jabbour-Zahab, R.; Arnaud, F.; Dezileau, L.; Chevin, L.M.; Sánchez, M.I. Resurrection ecology in Artemia. Evol. Appl. 2018, 11, 76–87. [Google Scholar] [CrossRef]
- De Vos, S.; Rombauts, S.; Coussement, L.; Dermauw, W.; Vuylsteke, M.; Sorgeloos, P.; Clegg, J.S.; Nambu, Z.; Van Nieuwerburgh, F.; Norouzitallab, P.; et al. The genome of the extremophile Artemia provides insight into strategies to cope with extreme environments. BMC Genom. 2021, 22, 635. [Google Scholar] [CrossRef]
- Kappas, I.; Baxevanis, A.D.; Maniatsi, S.; Abatzopoulos, T.J. Porous genomes and species integrity in the branchiopod Artemia. Mol. Phylogenet. Evol. 2009, 52, 192–204. [Google Scholar] [CrossRef]
- Asem, A.; Eimanifar, A.; Li, W.; Wang, P.Z.; Brooks, S.A.; Wink, M. Phylogeography and population genetic structure of an exotic invasive brine shrimp, Artemia Leach, 1819 (Crustacea: Anostraca), in Australia. Aust. J. Zool. 2019, 66, 307–316. [Google Scholar] [CrossRef]
- Thirunavukkarasu, S.; Karunasagaran, G.; Munuswamy, N. Morphometric and phylogenetic analysis of morphotypes in Artemia franciscana Kellogg, 1906 (Crustacea: Anostraca). Reg. Stud. Mar. Sci. 2022, 54, 102411. [Google Scholar] [CrossRef]
- Anufriieva, E.V.; Shadrin, N.V.; Shadrina, S.N. History of research on biodiversity in Crimean hypersaline waters. Arid Ecosyst. 2017, 7, 52–58. [Google Scholar] [CrossRef]
- Fedchenko, G.P. The deposited lump salt and salt lakes of the Caspian and Azov sea basins. Izv. Imper. O-va Lyubit. Estestvozn. Antropol. Etnogr. 1870, 5. (In Russian) [Google Scholar]
- Kulagin, N.M. Fauna of Crimean salt lakes. Izv. Imper. O-va Lyubit. Estestvozn. Antropol. Etnogr. 1888, 50, 430–444. (In Russian) [Google Scholar]
- Schmankewitsch, M.W.J. On the relations of Artemia salina and Artemia mühlhausenii, and on the genus Branchipus. J. Nat. Hist. 1876, 17, 256–258. [Google Scholar] [CrossRef]
- Gajewski, N. Uber die Variabilitat bei Artemia salina. Int. Rev. Hydrobiol. 1922, 10, 139–159. [Google Scholar] [CrossRef]
- Bond, R.M. Observations on Artemia “franciscana” Kellogg, especially on the relation of environment to morphology. Int. Rev. Hydrobiol. 1933, 28, 117–125. [Google Scholar] [CrossRef]
- Voronov, P.M. Reproduction of Artemia salina in saline waters of the Crimea. Zool. Zhurnal 1973, 52, 945–947. (In Russian) [Google Scholar]
- Mura, G.R.; Nagorskaya, L.I. Notes on the distribution of the genus Artemia in the former USSR countries (Russia and adjacent regions). J. Biol. Res. 2005, 4, 139–150. [Google Scholar]
- Shadrin, N.V.; Batogova, E.A.; Belmonte, D.; Moscatello, S.; Litvinchuk, L.F.; Shadrina, S.N. Artemia urmiana Gunther, 1890 (Anostraca, Artemiidae) in Lake Koyashskoe (Crimea, Black Sea) is the first find outside Lake Urmia (Iran). Mor. Ekol. Zhurn. 2008, 7, 30–31. (In Russian) [Google Scholar]
- Abatzopoulos, T.J.; Amat, F.; Baxevanis, A.D.; Belmonte, G.; Hontoria, F.; Maniatsi, S.; Moscatello, S.; Mura, G.; Shadrin, N.V. Updating geographic distribution of Artemia urmiana Günther, 1890 (Branchiopoda: Anostraca) in Europe: An integrated and interdisciplinary approach. Int. Rev. Hydrobiol. 2009, 94, 560–579. [Google Scholar] [CrossRef]
- Belmonte, G.; Moscatello, S.; Batogova, E.A.; Pavlovskaya, T.; Shadrin, N.V.; Litvinchuk, L.F. Fauna of hypersaline lakes of the Crimea (Ukraine). Thalass. Salentina 2012, 34, 11–24. [Google Scholar]
- Shadrin, N.; Anufriieva, E.; Galagovets, E. Distribution and historical biogeography of Artemia Leach, 1819 (Crustacea: Anostraca) in Ukraine. Int. J. Artemia Biol. 2012, 2, 30–42. [Google Scholar]
- Shadrin, N.V.; Anufriieva, E.V.; Amat, F.; Eremin, O.Y. Dormant stages of crustaceans as a mechanism of propagation in the extreme and unpredictable environment in the Crimean hypersaline lakes. Chin. J. Oceanol. Limnol. 2015, 33, 1362–1367. [Google Scholar] [CrossRef]
- Shadrin, N.V.; Anufriieva, E.V. Size polymorphism and fluctuating asymmetry of Artemia (Branchiopoda: Anostraca) populations from the Crimea. J. Sib. Fed. Univ. Biol. 2017, 10, 114–126. [Google Scholar] [CrossRef]
- Anufriieva, E.; Shadrin, N. The long-term changes in plankton composition: Is Bay Sivash transforming back into one of the world’s largest habitats of Artemia sp. (Crustacea, Anostraca)? Aquac. Res. 2020, 51, 341–350. [Google Scholar] [CrossRef]
- Anufriieva, E.; Hołyńska, M.; Shadrin, N. Current invasions of Asian cyclopid species (Copepoda: Cyclopidae) in Crimea, with taxonomical and zoogeographical remarks on the hypersaline and freshwater fauna. Ann. Zool. 2014, 64, 109–130. [Google Scholar] [CrossRef]
- Golubkov, S.M.; Shadrin, N.V.; Golubkov, M.S.; Balushkina, E.V.; Litvinchuk, L.F. Food chains and their dynamics in ecosystems of shallow lakes with different water salinities. Russ. J. Ecol. 2018, 49, 442–448. [Google Scholar] [CrossRef]
- Geller, J.; Meyer, C.; Parker, M.; Hawk, H. Redesign of PCR primers for mitochondrial cytochrome c oxidase subunit I for marine invertebrates and application in all-taxa biotic surveys. Mol. Ecol. Resour. 2013, 13, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Eimanifar, A.; Wink, M. Fine-scale population genetic structure in Artemia urmiana (Günther, 1890) based on mtDNA sequences and ISSR genomic fingerprinting. Org. Divers. Evol. 2013, 13, 531–543. [Google Scholar] [CrossRef]
- Eimanifar, A.; Asem, A.; Djamali, M.; Wink, M. A note on the biogeographical origin of the brine shrimp Artemia urmiana Gunther, 1899 from Urmia Lake, Iran. Zootaxa 2016, 4097, 294–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñoz, J.; Gómez, A.; Green, A.J.; Figuerola, J.; Amat, F.; Rico, C. Phylogeography and local endemism of the native Mediterranean brine shrimp Artemia salina (Branchiopoda: Anostraca). Mol. Ecol. 2008, 17, 3160–3177. [Google Scholar] [CrossRef]
- Maniatsi, S.; Kappas, I.; Baxevanis, A.D.; Farmaki, T.; Abatzopoulos, T.J. Sharp phylogeographic breaks and patterns of genealogical concordance in the brine shrimp Artemia franciscana. Int. J. Mol. Sci. 2009, 10, 5455–5470. [Google Scholar] [CrossRef]
- Maniatsi, S.; Baxevanis, A.D.; Kappas, I.; Deligiannidis, P.; Triantafyllidis, A.; Papakostas, S.; Bougiouklis, D.; Abatzopoulos, T.J. Is polyploidy a persevering accident or an adaptive evolutionary pattern? The case of the brine shrimp Artemia. Mol. Phylogenet. Evol. 2011, 58, 353–364. [Google Scholar] [CrossRef]
- Maccari, M.; Amat, F.; Gomez, A. Origin and genetic diversity of diploid parthenogenetic Artemia in Eurasia. PLoS ONE 2013, 8, e83348. [Google Scholar]
- Naganawa, H.; Mura, G. Two new cryptic species of Artemia (Branchiopoda, Anostraca) from Mongolia and the possibility of invasion and disturbance by the aquaculture industry in East Asia. Crustaceana 2017, 90, 1679–1698. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Ruebhart, D.R.; Cock, I.E.; Shaw, G.R. Invasive character of the brine shrimp Artemia franciscana Kellogg 1906 (Branchiopoda: Anostraca) and its potential impact on Australian inland hypersaline waters. Mar. Freshw. Res. 2008, 59, 587–595. [Google Scholar] [CrossRef]
- Vikas, P.A.; Sajeshkumar, N.K.; Thomas, P.C.; Chakraborty, K.; Vijayan, K.K. Aquaculture related invasion of the exotic Artemia franciscana and displacement of the autochthonous Artemia populations from the hypersaline habitats of India. Hydrobiologia 2012, 684, 129–142. [Google Scholar] [CrossRef]
- Scalone, R.; Rabet, N. Presence of Artemia franciscana (Branchiopoda, Anostraca) in France: Morphological, genetic, and biometric evidence. Aquat. Invasions 2013, 8, 67–76. [Google Scholar] [CrossRef]
- Khomenko, S.V.; Shadrin, N.V. Iranian endemic Artemia urmiana in hypersaline Lake Koyashskoe (Crimea, Ukraine): A preliminary discussion of introduction by birds. Branta. Trans. Azov Black Sea Ornithol. Station 2009, 12, 81–91. [Google Scholar]
- Muñoz, J.; Gómez, A.; Figuerola, J.; Amat, F.; Rico, C.; Green, A.J. Colonization and dispersal patterns of the invasive American brine shrimp Artemia franciscana (Branchiopoda: Anostraca) in the Mediterranean region. Hydrobiologia 2014, 726, 25–41. [Google Scholar] [CrossRef]
- Kappas, I.; Abatzopoulos, T.J.; Van Hoa, N.; Sorgeloos, P.; Beardmore, J.A. Genetic and reproductive differentiation of Artemia franciscana in a new environment. Mar. Biol. 2004, 146, 103–117. [Google Scholar] [CrossRef]
- Ogello, E.O.; Nyonje, B.M.; Van Stappen, G. Genetic differentiation of Artemia franciscana (Kellogg, 1906) in Kenyan coastal saltworks. Int. J. Adv. Res. 2014, 2, 1154–1164. [Google Scholar]
- Lee, J.; Cho, B.C.; Park, J.S. Transcriptomic analysis of brine shrimp Artemia franciscana across a wide range of salinities. Mar. Genom. 2022, 61, 100919. [Google Scholar] [CrossRef] [PubMed]
- Shadrin, N.; Anufriieva, E. Review of the biogeography of Artemia Leach, 1819 (Crustacea: Anostraca) in Russia. Int. J. Artemia Biol. 2012, 2, 51–61. [Google Scholar]
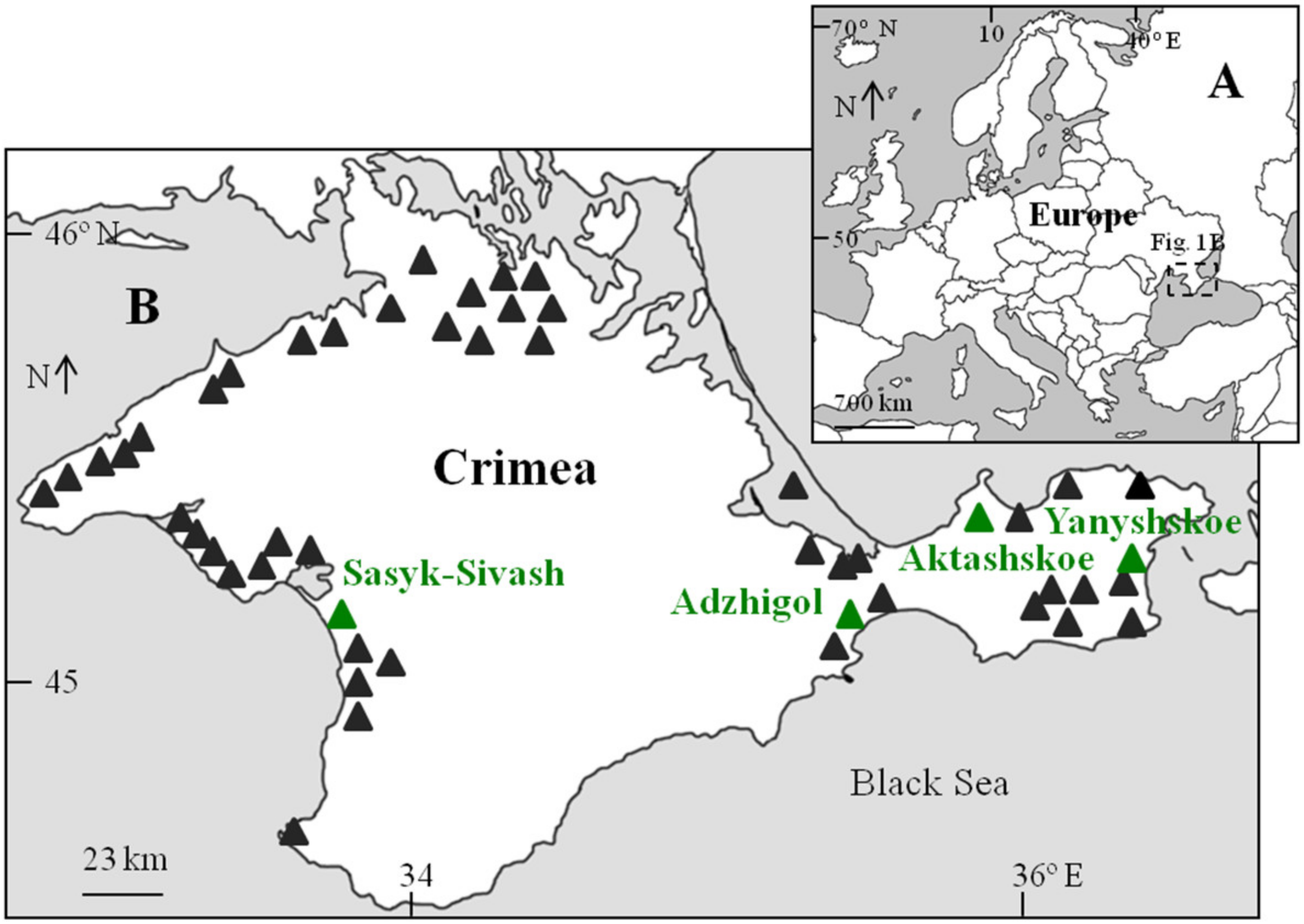
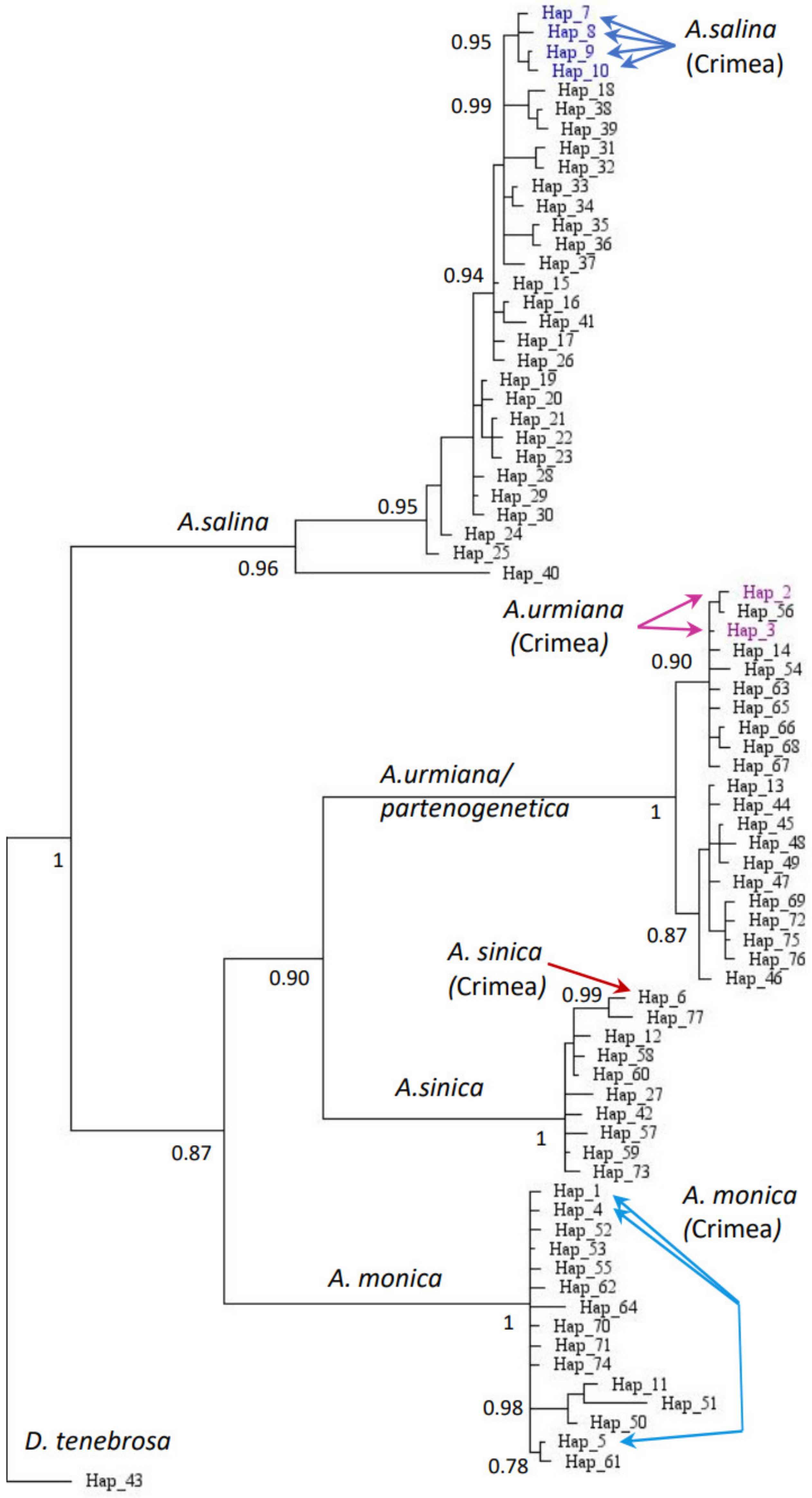
| Lake | Coordinates | Area, km2 | Date of Sampling | Salinity (during Sampling), g L−1 | Temperature, °C | Number of Analyzed Individuals (Female/Male) | Total Artemia Abundance, Ind. m−3 |
|---|---|---|---|---|---|---|---|
| Aktashskoe | 45°22′31″ N 35°49′45″ E | 26.8 | 1 July 2021 | 173 | 28.5 | A. urmiana—2 f A. monica—1 m | 34,820 |
| Adzhjigol | 45°06′32″ N 35°27′58″ E | 0.6 | 1 July 2021 | 50 | 34.5 | A. monica—2 m A. sinica—2 f A. urmiana—1 f | 902,960 |
| Sasyk-Sivash | 45°09′21″ N 33°31′09″ E | 75.3 | 3 July 2021 | 245 | 36.0 | A. salina—4 m/2 f | 220 |
| Species | Abbreviation/Haplotypes | Lake | GenBank Number |
|---|---|---|---|
| A. monica (=A. franciscana) | 3M2/H1 | Aktashskoe | ON872200 |
| A. urmiana | 3F2/H2 | Aktashskoe | ON872198 |
| A. urmiana | 3F3/H3; 4F2/H3 | Aktashskoe, Adzhigol | ON872199 ON872202 |
| A. monica (=A. franciscana) | 4M1/H4 | Adzhigol | ON872204 |
| A. monica (=A. franciscana) | 4M2/H5 | Adzhigol | ON872205 |
| A. sinica | 4F1/H6; 4F3/H6 | Adzhigol | ON872201 ON872203 |
| A. salina | 5M1/H7 | Sasyk-Sivash | ON872208 |
| A. salina | 5M2/H8,5M3/H8,5M5/H8 | Sasyk-Sivash | ON872209 ON872210 ON872211 |
| A. salina | 5F1/H9 | Sasyk-Sivash | ON872206 |
| A. salina | 5F2/H9 | Sasyk-Sivash | ON872207 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lantushenko, A.; Meger, Y.; Gadzhi, A.; Anufriieva, E.; Shadrin, N. Artemia spp. (Crustacea, Anostraca) in Crimea: New Molecular Genetic Results and New Questions without Answers. Water 2022, 14, 2617. https://doi.org/10.3390/w14172617
Lantushenko A, Meger Y, Gadzhi A, Anufriieva E, Shadrin N. Artemia spp. (Crustacea, Anostraca) in Crimea: New Molecular Genetic Results and New Questions without Answers. Water. 2022; 14(17):2617. https://doi.org/10.3390/w14172617
Chicago/Turabian StyleLantushenko, Anastasia, Yakov Meger, Alexandr Gadzhi, Elena Anufriieva, and Nickolai Shadrin. 2022. "Artemia spp. (Crustacea, Anostraca) in Crimea: New Molecular Genetic Results and New Questions without Answers" Water 14, no. 17: 2617. https://doi.org/10.3390/w14172617






