1. Introduction
Given the environmental, social, and economic benefits of reprocessing mine waters, many treatment technologies have been developed to reuse water and recover resources such as metal ions [
1,
2,
3]. Ion exchange (IX) resins technology has emerged as a promising alternative given its high selectivity for metal removal, small concentrated output volume, high quality of treated water, and the great advantage that it is a reversible and reusable process [
4,
5,
6,
7]. Some challenges to be considered when using resins for mine water treatment are the hydraulic blocking of the material due to the presence of colloids in the water or their formation by the oxidation of iron, manganese, or other metals [
8]. The variation of pH in the process, depending on the type of exchange material, and the presence of calcium and sulfates in the water can develop secondary precipitations, which considerably reduce the capacity of the resin, modify the water flow inside the exchange column, and reduce the recovery process efficiency [
9]. However, these difficulties can be minimized with proper pretreatment of the water, monitoring of process parameters, and upward flow of the inlet solution [
4,
5,
6].
Although laboratory testing is vital for small-scale research, it involves sampling, analysis, and monitoring, becoming time consuming and costly [
10]. Therefore, once sufficient data have been obtained from experimental tests, it is necessary to visualize the system through an ion-exchange model, which would help to describe the exchange of ions onto resins and study its behavior and performance [
11]. Hydrogeochemical modeling helps to understand complex processes and is a powerful tool for assessing detailed geochemical reactions and transport processes [
12]. Since the early 1970s, many authors have developed different computer programs and databases for hydrogeochemical modeling [
13]. In this study, the software PhreeqC has been selected. This computer program has been evolving and increasing its capabilities over time, being able to model and simulate surface-controlled reactions such as IX and reactive mass transport in a 1D column [
14]. On the other hand, PhreeqC is a public domain software, which facilitates its use and expands its scope [
15].
There is a considerable amount of research on the modeling of reactive mass transport processes in natural environments, where the focus is the study of different phenomena such as solute transport, soil and freshwater contamination, and seawater intrusion [
13,
14,
16,
17,
18,
19]. However, surprisingly little information is available regarding the selective removal of metals from mine waters using manufactured resins [
20]. Chelating resins have been developed for a broad range of applications using mine waters due to their high exchange capacity and remarkable performance at low pH values [
9,
21,
22]. In addition, weakly basic resins with a chelating bis-picolylamine functional group possess high selectivity for metal ions with economic potential such as copper, zinc, and nickel captured from acidic solutions [
23,
24].
Although the theoretical cation exchange capacity (CEC) is provided by the resin manufacturer, this value differs in practice, as it depends on the operating conditions of the system [
5,
25]. On the other hand, the selectivity of a resin can be defined by using exchange coefficients. These coefficients have been obtained from several experimental measurements of different soils and clay minerals, and their values are defined in existing geochemical software databases [
26]. Nevertheless, these default values are not always suitable for the modeling given their dependence on the characteristics of the resin and the mine water composition [
26,
27,
28,
29]. Furthermore, each ion of the inflow solution involved in the IX process requires the definition of its exchange coefficient. Otherwise, it will not be considered in the model [
15]. This is why the more complex the mine water composition, the more unknowns to estimate [
13]. This complexity is reflected in the limited literature on modeling IX processes using one, two, or up to three competing ions in the input solution [
2,
7,
11,
16,
17,
30]. Therefore, in order to address the study of more complex mine water matrices, this research contemplates a parallel work between laboratory and simulation to reduce simplifications and assumptions.
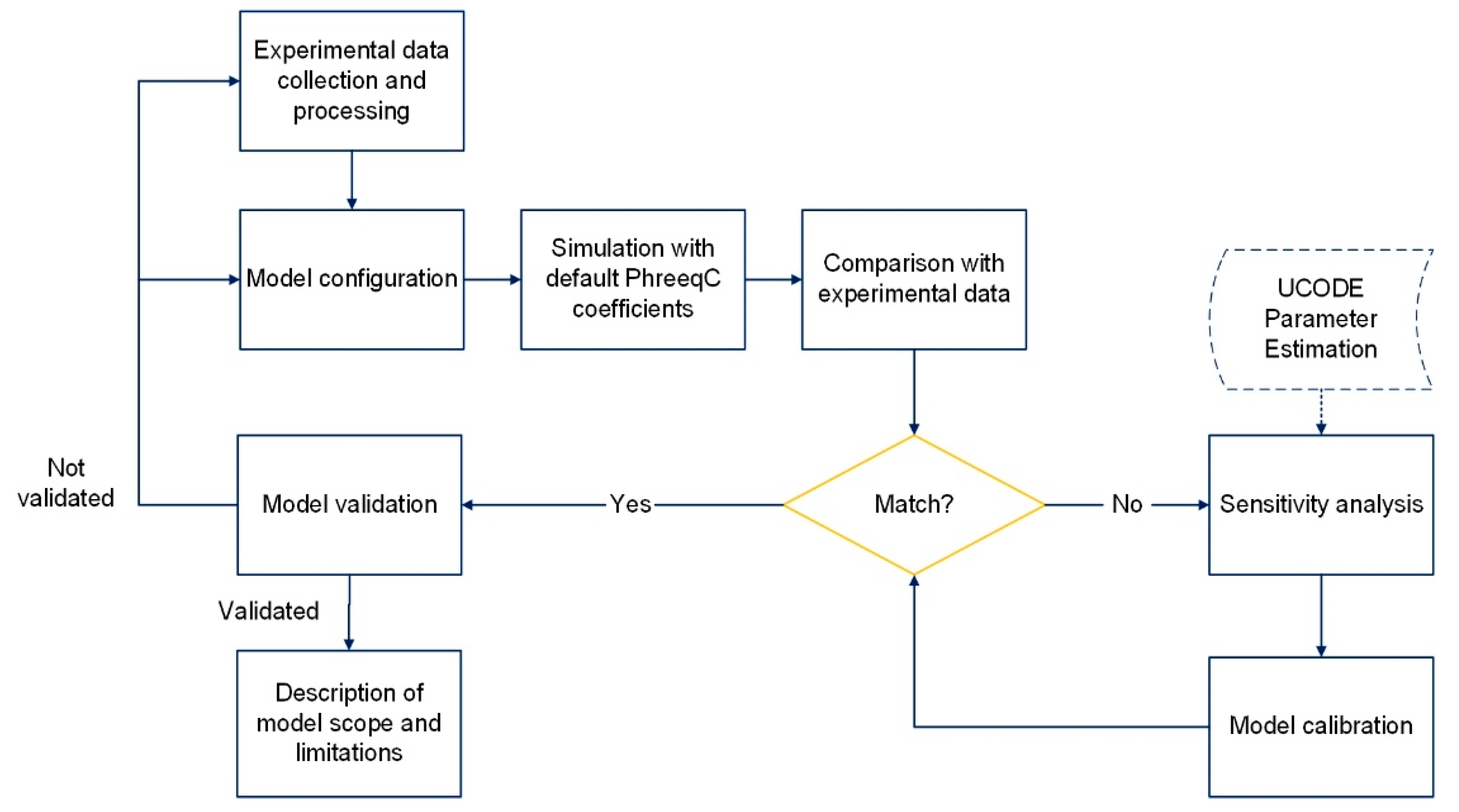
The outcomes of this project propose a well-organized methodology for a deeper assessment and support of IX column experiments using a model-based approach. Through this study, the main parameters that significantly influence the IX model have been identified and estimated for further extension and upgrading. In addition, as part of a simulation modeling, the developed model can be used to (i) predict the selective removal of metals from complex mine water matrices reducing the costs of laboratory tests and chemical analysis, (ii) speed up the study of cases over a long period of time, and (iii) upscale [
31]. Subsequently, it is expected to include the resin regeneration phase in the model to simulate the process of recovery of metal ions.
3. Results
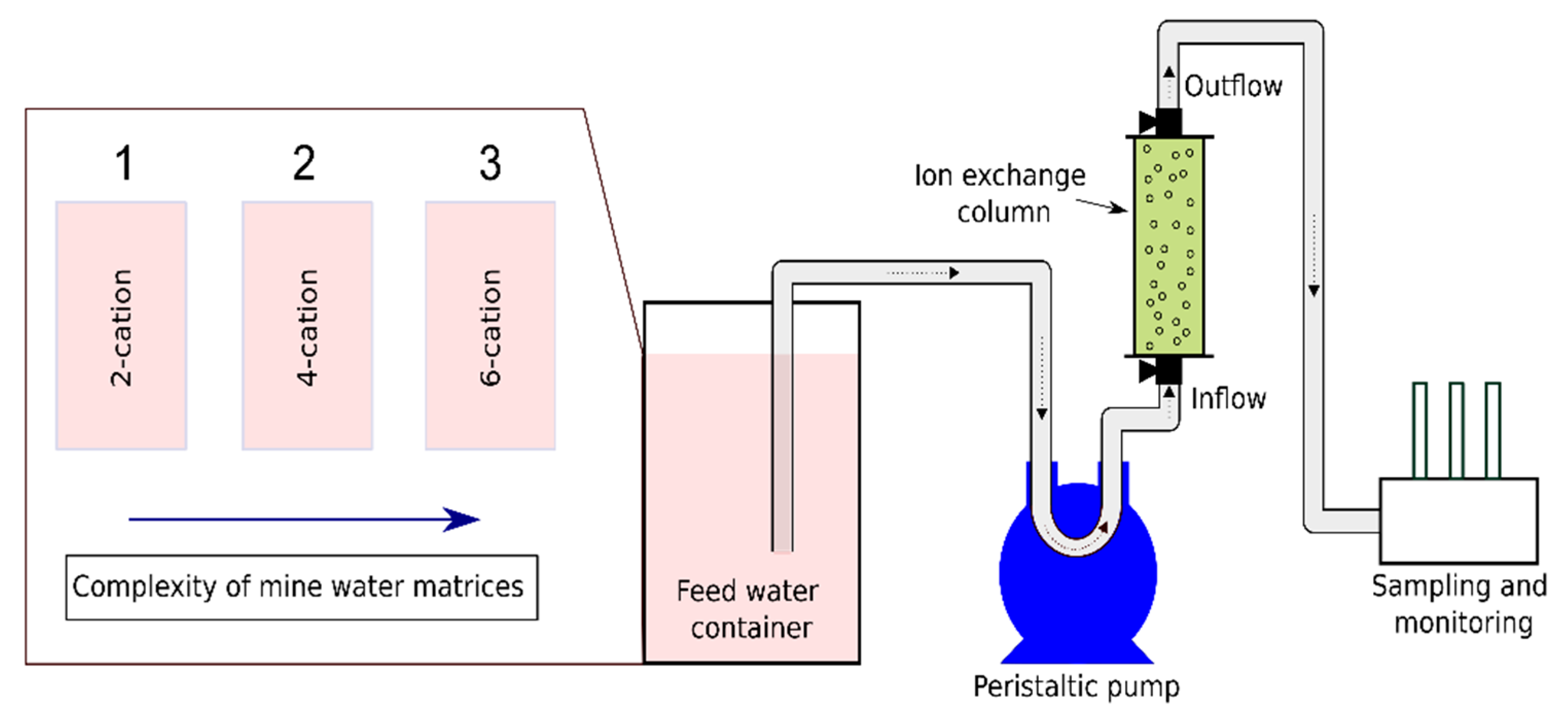
The experimental data obtained from two-cation (Scenario 1) and four-cation (Scenario 2) column tests at different operating conditions were employed to evaluate the prediction of BTCs for the selective metal removal from synthetic mine waters.
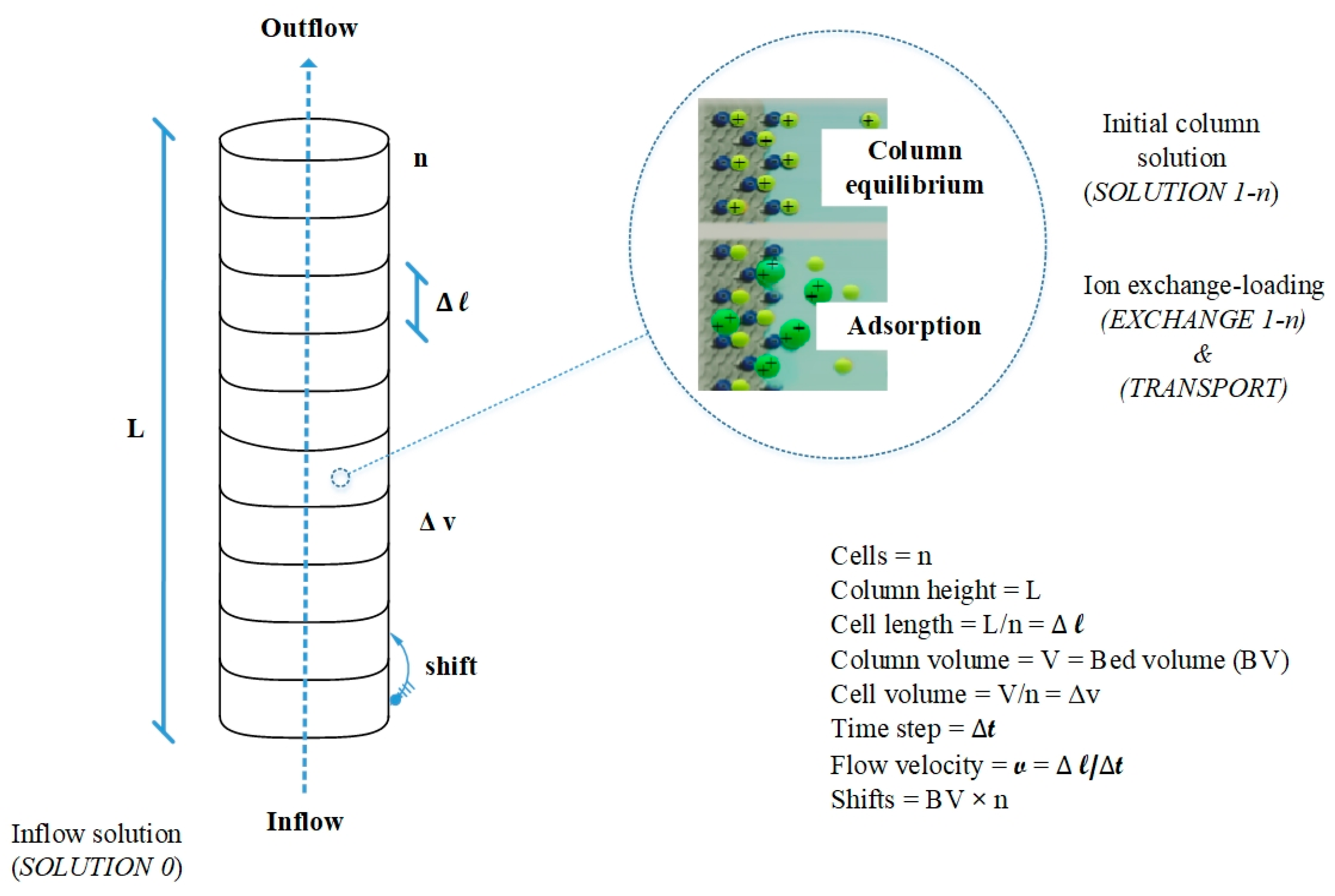
The PhreeqC code that couples both the Gaines–Thomas convention and the Advection–Reaction–Dispersion equation using a one-dimension flowline was employed to generate simulation results of fix-bed column tests. Additionally, the accuracy of the model has been evaluated by making comparisons between simulated and experimental data.
3.1. First Model Approach
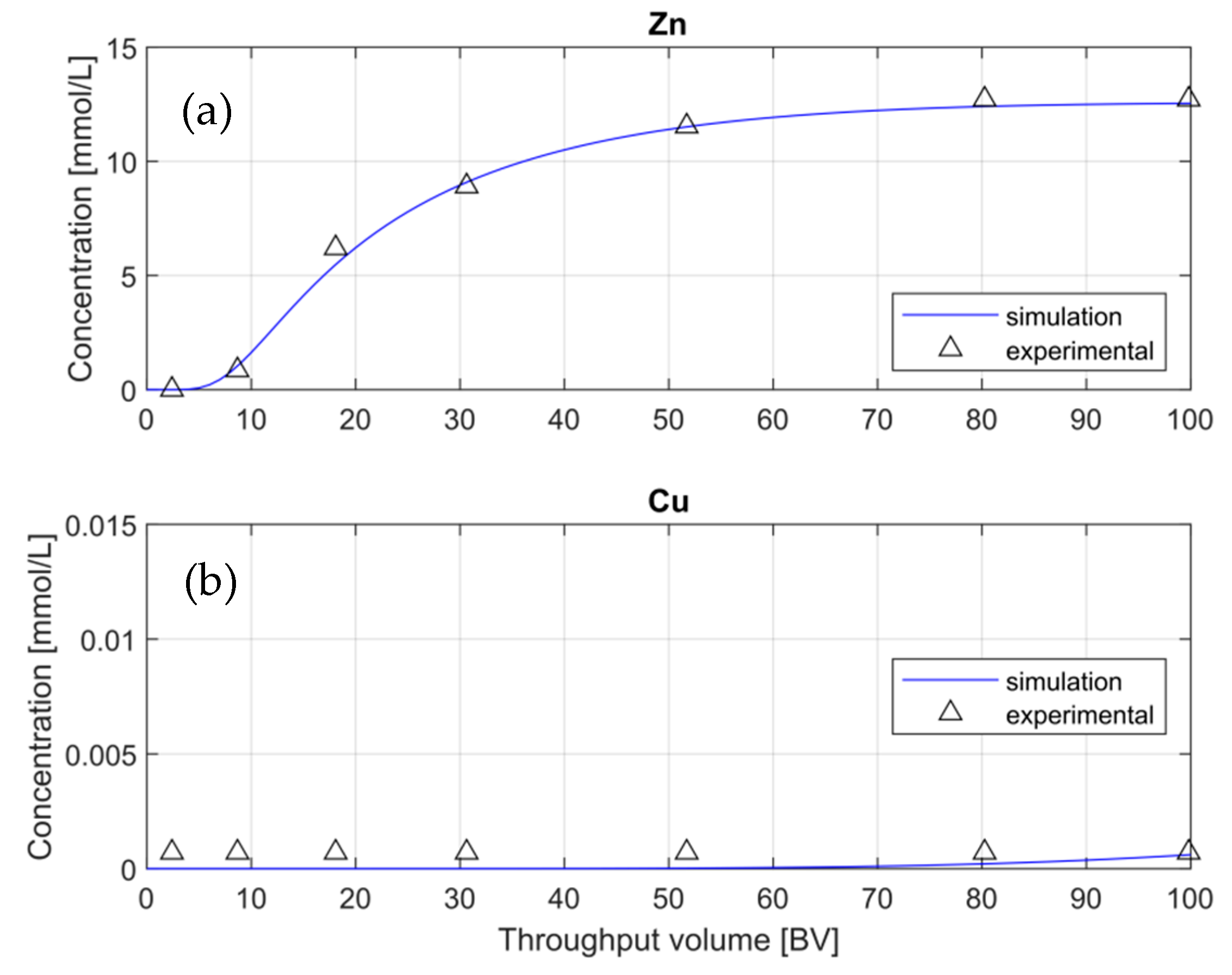
The first model approach was built using Scenario 1. For the first CEC approximation, a value of 48% of water retention was assumed, which is the lowest value from the theoretical rank provided by the manufacturer (
Table 2). For the first attempt, default values of exchange and activity coefficients from the phreeqc.dat database were used (see Table 6). Thus, it was not necessary to add extra data in the input file. The simulation and experimental BTCs for Zn
+2 and Cu
+2 are shown in
Figure 4.
As shown in
Figure 4a, the Zn-BTC simulation shows similar behavior to the experimental data, mainly at the breakthrough point (BTP) and at the exhaustion stage. On the other hand,
Figure 4b shows that the simulation of Cu-BTC does not fit with the experimental data along with the whole pattern. The experimental concentrations of copper were below the detection limit of the MP-AES and never reached the BTP during the loading phase due to its high affinity with the resin TP-220 and its very low concentration in the inflow solution. In any case,
Figure 4 shows that the simulated Zn
+2 and Cu
+2-ions have the BTP at nine and five-bed volumes, respectively.
3.2. Sensitivity Analysis of Parameters and Coefficients
A sensitivity analysis was performed for Scenario 1 in order to identify the main parameters and coefficients that govern the model. As mentioned above, the parameters identified as having the greatest impact on the IX process are CEC, selectivity coefficients, activity coefficients, pH, and flow rate. Although the pH and flow rate have a significant impact on the model, these parameters have values established during the experimental tests that will be taken for the input definition. Therefore, the behavior of the breakthrough curves at different CEC, exchange coefficients, and activity coefficients are shown in
Figure 5,
Figure 6 and
Figure 7.
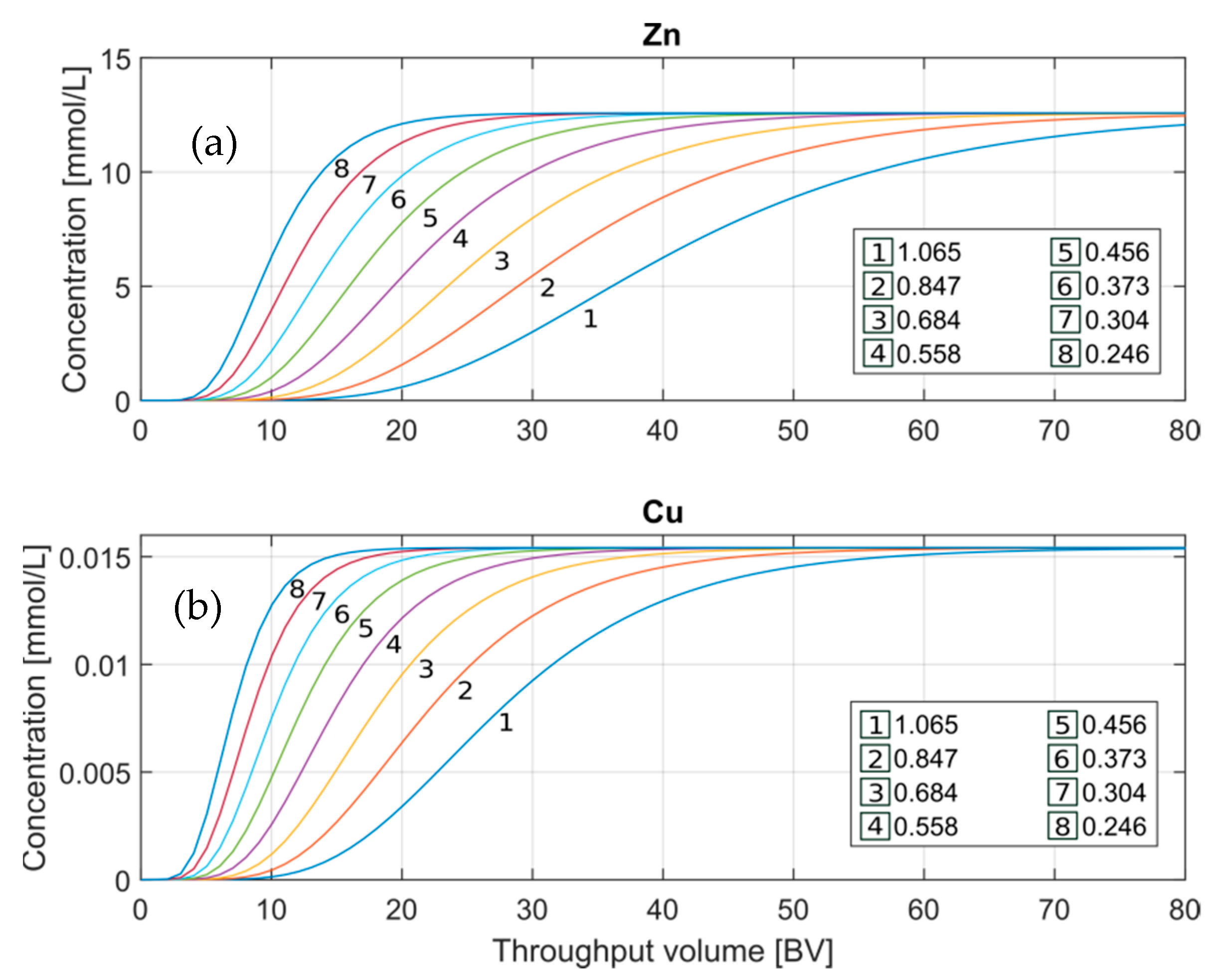
3.2.1. Cation Exchange Capacity
Since the theoretical capacity of the exchanger is not a defined parameter because it depends on the effective porosity, water retention of the resin, and operating conditions, it is necessary to evaluate it [
29]. For this purpose,
Figure 5 shows different CEC values in mol/L
w (mol per liter of water) that were set to evaluate their impact on the model. This range of values was estimated by defining different water retention percentages of the resin based on the results of the first model approach.
Due to the significant difference between the output concentrations of Zn
+2 and Cu
+2, separate graphs were plotted for a more detailed assessment of the trends. The behavior of Zn- and Cu-BTCs in
Figure 5 are affected following the same pattern. The higher the CEC, the later the BTP, observing a difference of 10-BV between the BTP calculated at the lowest (0.246) and highest (1.065) CEC. It can be seen in both cases a flatter slope for the highest CEC value selected. Due to the high impact of this parameter on the model, this variable will be part of the model calibration.
3.2.2. Exchange Coefficients
Scenario 1 is a two-cation system, and therefore, it is required to define two exchange coefficients, one for each ion. In order to evaluate the behavior of the BTCs at different exchange coefficient values, different configurations were stated as is described in
Table 4. Configuration A describes the values of the exchange coefficients defined by default in the PhreeqC database, and from these starting values, the other configurations were defined: first, increasing the values for the Cu ion and defining a constant value for the Zn ion (Configurations A–C); then, defining a constant value for the Cu ion and decreasing the value for the Zn ion (Configurations C–F). These configurations helped to evaluate the impact and interdependence of exchange coefficients on both metal ions.
Cu-BTCs show a high sensitivity for all the configurations due to its high affinity toward the resin [
21,
23]. This behavior is stated as
Figure 6b shows a distinct trend for each configuration. Analyzing the pattern illustrated in
Figure 6b, the following must be pointed out: (1) the higher the Cu coefficient, the flatter the slope and the later the Cu-BTC; (2) the higher the Zn coefficient, the sooner the Cu-BTP. It is observed that there is a difference of 10 BV approximately between the BTP at configuration A and F for the Cu
+2 ion.
On the other hand,
Figure 6a shows that Zn curve is only affected by its own exchange coefficient (configurations C to F) and follows the same trends as the Cu
+2 ion but to a minor degree. In this case, only a difference in the BTPs of 2 BV is visualized for all configurations.
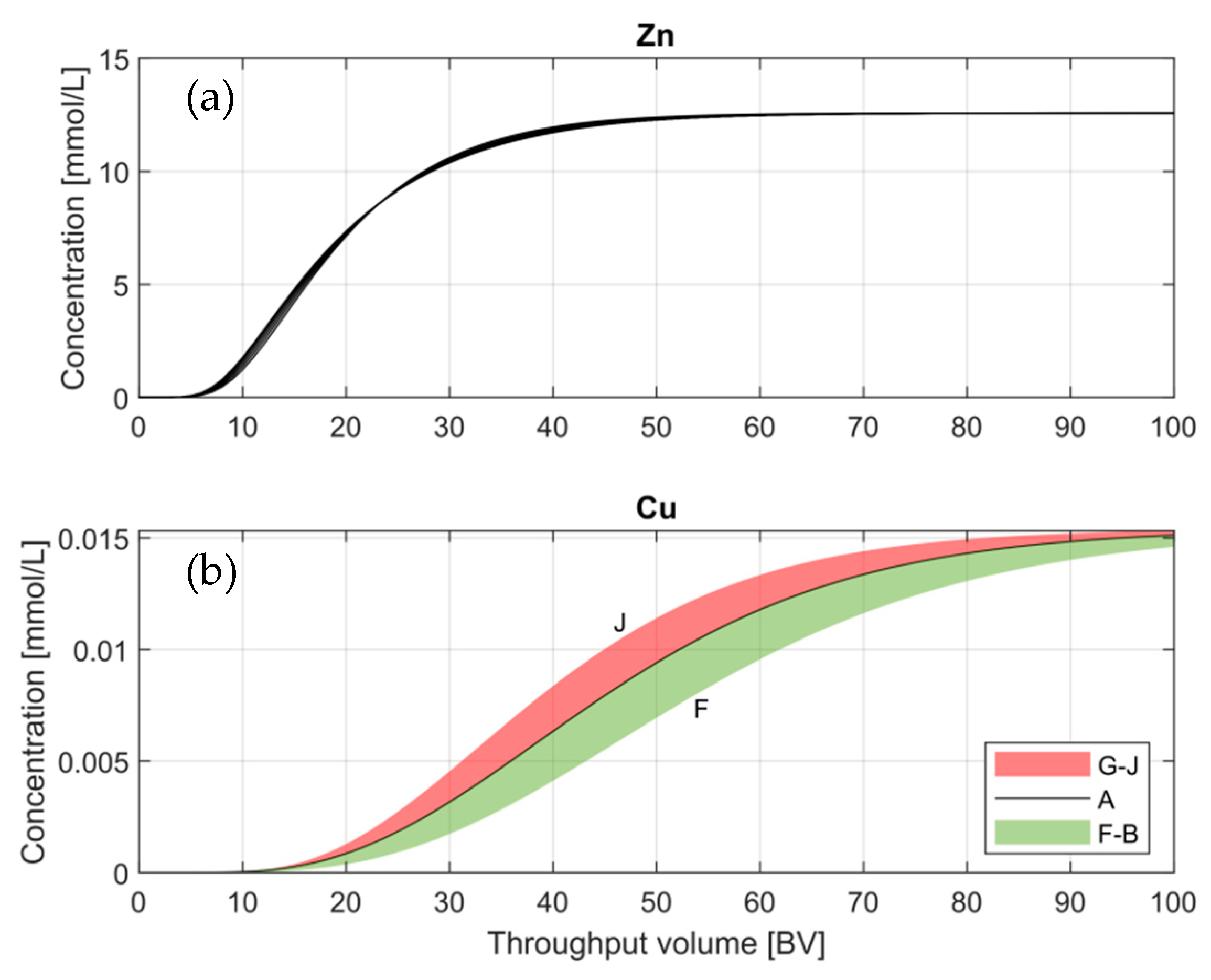
3.2.3. Ion Specific Parameters–Activity Coefficients
Although the parameters
a and
b are part of the database, an assessment of their behavior under variations will provide information in case further adjustment is required. For example, the default ion-specific parameters
b has a value of 0.0 for Cu
+2 and Zn
+2 ions, while the parameter
a presents a different value for each element. Therefore, in order to reduce the number of unknowns, a value of zero will be defined for parameter
b for all simulations, while parameter
a will be evaluated by setting different values, as shown in
Table 5.
Starting with default values,
Table 5 describes the different configurations applied to evaluate the impact of activity coefficients.
Figure 6 shows the Zn- and Cu-BTCs under configurations A to J.
Figure 7b was plotted using areas (G-J), (F-B) due to the slight variations between one BTC and the other. The trends of the BTCs constitute the areas that curiously fit in the different configurations defined in
Table 5 for Cu
+2. The behavior shows that at a constant
a-parameter for Zn
+2, the trend of Cu curves follows: the lower the value of Cu-
a, the flatter the slope of the curve. Moreover, at a constant
a parameter for Cu
+2, the lower the Zn-
a, the steeper the slope of the curve. During all the proposed configurations, variations in the slope of the curves can be observed; however, the breakthrough and exhaustion points remain without significant modifications.
The Zn ion once again shows no dependence under Cu-
a variations and no significant changes for different Zn-
a values. There can be clearly observed in
Figure 7a a thick BTC where the impact of the different configurations cannot be visualized due to the low sensitivity.
3.3. Estimation of CEC and Exchange Coefficients
After identifying the key parameters through sensitivity analysis, two of them, CEC and exchange coefficients, were selected for the model calibration based on their high impact on the model. The calibration consists of comparing model and experimental results by adjusting the key parameters until the simulation results match the experimental data [
28].
Calibration was performed manually by trial and error. As this is complicated when many parameters and variables are involved, the calibration was performed in two stages: first, a calibration of CEC using default exchange coefficients; then, the calibration of exchange coefficients using the estimated CEC.
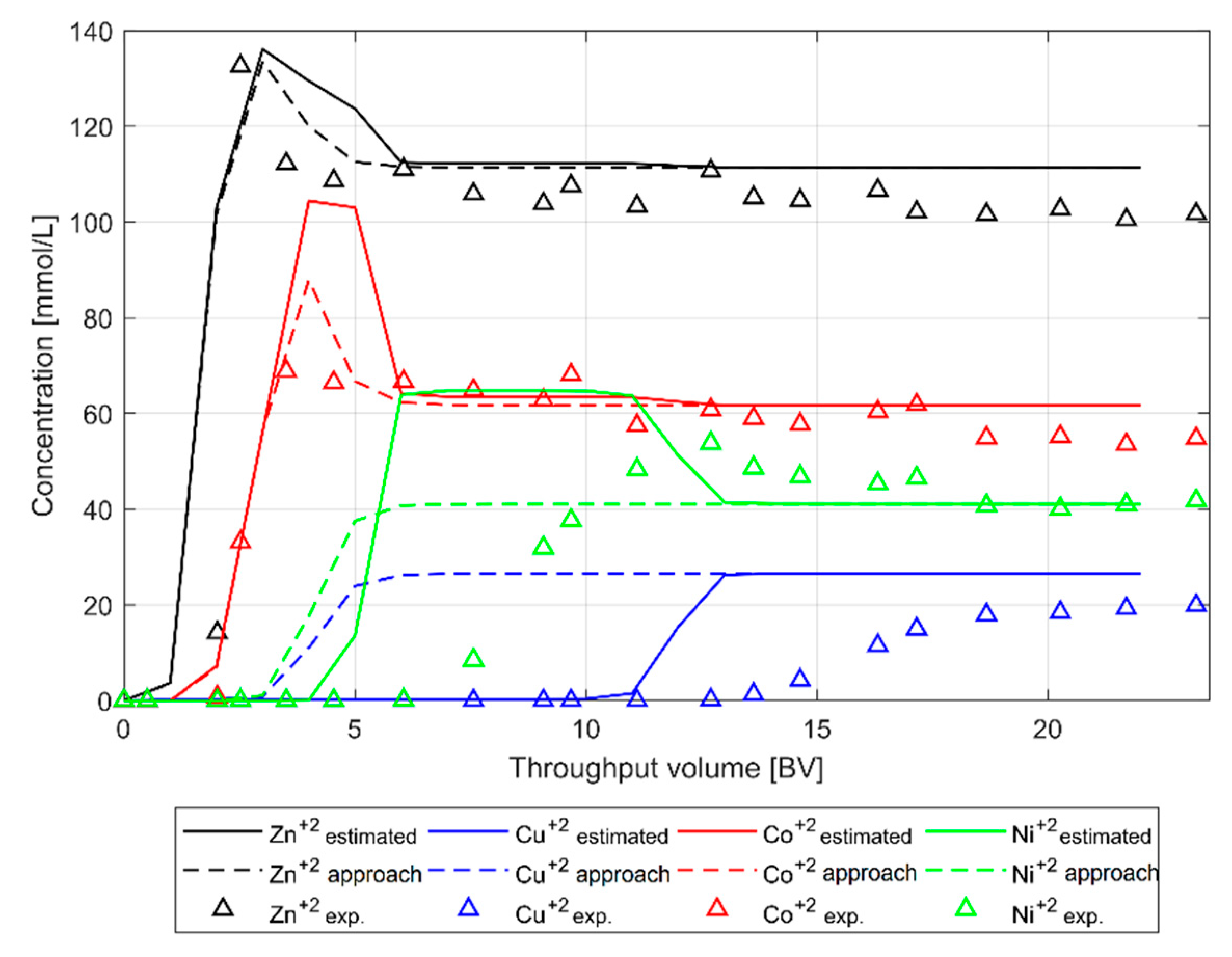
The first calibration was performed using Scenario 1 in order to estimate the CEC value and the coefficients for zinc and copper ions. Then, a second calibration was conducted for Scenario 2 using the estimated values from the first calibration and estimating the values for the new metal ions nickel and cobalt. Results can be observed in
Figure 8 and
Figure 9 where a good approximation for both scenarios is visible.
Figure 8a shows a complete match between the simulation and the experimental data of the Zn-BTC. Then, the Zn-exchange coefficient value is fixed to 0.55. On the other hand, in the case of the Cu
+2 ion, as shown in
Figure 8b, the experimental data points to compare with are set as 7e
−4 [mmol/L]. This value was defined as the limit of detection (LOD) of the MP-AES for the quantification of the Cu
+2 ion concentration. Then, a value for the Cu-exchange coefficient must be at least 1.5 to meet this upper limit condition. A lower value would surpass this limit, and a higher one would flatten out the curve. It is important to take in mind that a variation of the Cu-exchange coefficient does not affect the behavior of the Zn curves, as it was shown in
Section 3.2.2. Then, the Cu coefficient could be adjusted further, without changing the trend of Zn-BTC.
For the second calibration applying Scenario 2, the Zn-coefficient was not adjusted, since the value of 0.55 was estimated in the first calibration. In the case of the Cu-exchange coefficient from Scenario 1, the value of 1.5 was the minimum starting value for Scenario 2. Therefore, there are three variables for the second model calibration: Co-, Ni-, and Cu-exchange coefficients. Although the Zn ion was not adjusted in this scenario, its trend has to be monitored as well.
The results of the second calibration are shown in
Figure 9, where is clearly seen that the calculated BTCs follow the selectivity order of adsorption determined experimentally (Cu
+2 > Ni
+2 > Co
+2 > Zn
+2) presented in
Table 2. There are no default coefficients for Co
+2 and Ni
+2 ions in the database. Thus, these coefficients were fit between the most (Cu
+2) and the least (Zn
+2) affine ions, as is described in
Table 6 (Second approach).
In order to show the significant variation that exists in the model when modifying the exchange coefficients, and the possibility to manipulate them,
Figure 9 shows the BTCs before and after the calibration of Scenario 2. As a result of the interdependence between ions, the manipulation of one coefficient affects all competing ions in the system. This condition is observed in the estimated Zn and Co curves where the values of their coefficients were kept constant but, nevertheless, they deviate from the experimental data in the range 3 > BV > 5. However, for these two ions, Zn
+2 and Co
+2, a good calibration of the model is observed. In the case of the Cu
+2 and Ni
+2 ions, a considerable approximation of the BTCs with the experimental data was achieved. While it is possible to further increase the values of these coefficients to achieve better results, this manipulation would decalibrate the already calibrated ions.
Table 6 summarizes the various default and estimated CEC and exchange coefficients over the calibrations performed.
3.4. Model Predictability for a Complex Mine Water Matrix
For the validation of the model, Scenario 3 with a six-cation composition inlet solution as described in
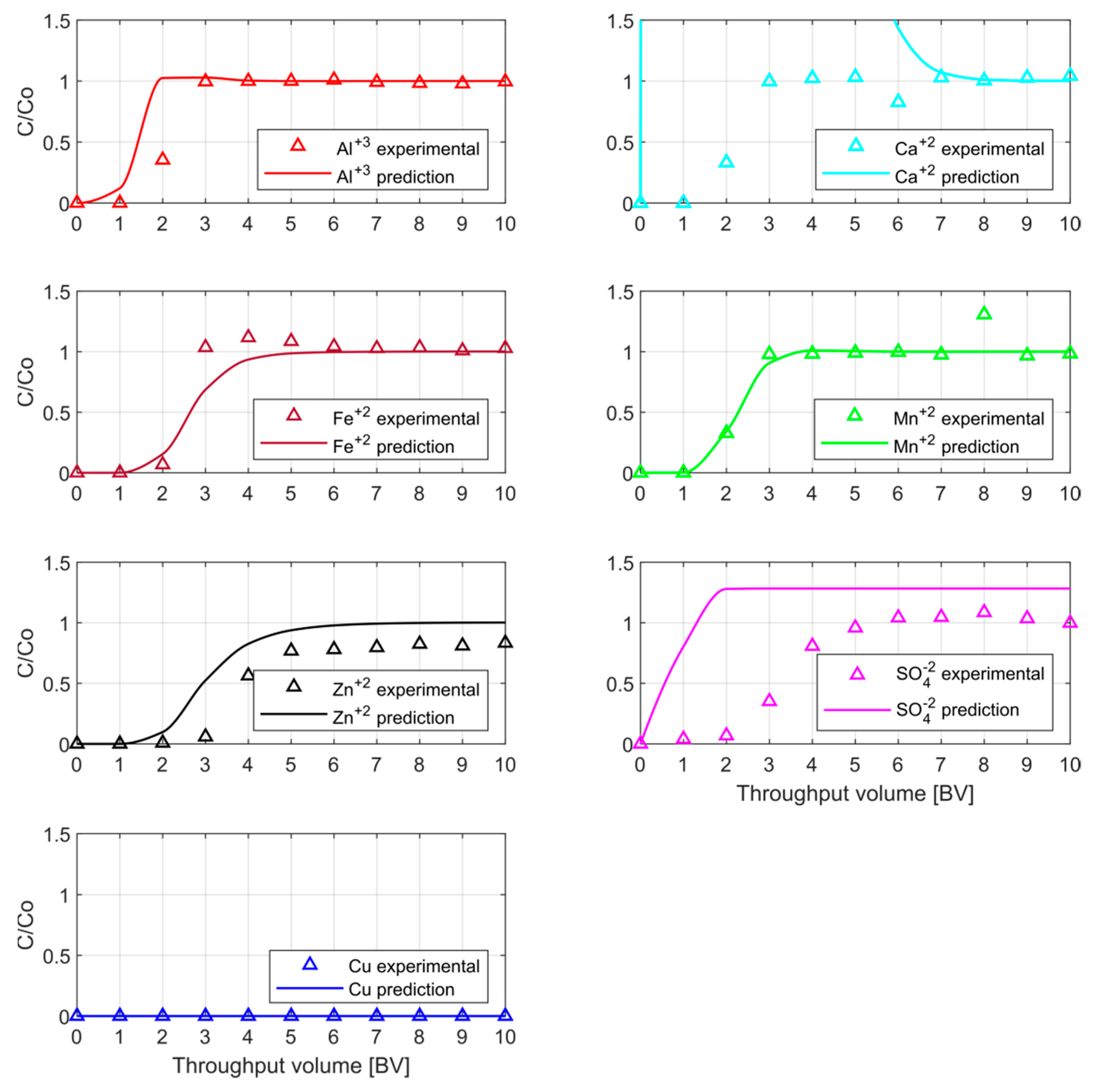
Table 1 was predicted. Once the BTCs were obtained, the column was set up to obtain the experimental results. As mentioned previously, the comparison of simulation and experimental BTCs’ position and shape is used to evaluate the accuracy of the model predictability.
The behavior of the BTCs shows a very low resin affinity of all ions but Cu
+2. That is why the BTPs of the cations were reached before the 5-BV was injected. In order to assess in more detail the predicted and experimental BTC,
Figure 10 shows a closer view of each ion separately.
In this second part of the research, not only metal ions but also sulfate and calcium ions were evaluated in order to make a deeper evaluation of the model under more real conditions. From
Figure 10, it can be observed that for the only anion considered, SO
4−2, there is no match between predicted and experimental data. The predicted curve has a sharp increase in concentration from the beginning of the loading phase, while the experimental one has a sigmoidal-shaped curve with a BTP at one BV. On the other hand, the alkali earth metal, Ca
+2 that was part of the inflow solution, matches only from the 7-BV onwards, having an overestimated concentration in the predicted curve. Experimental BTCs also show a sigmoidal-shaped behavior until they reach maximum concentration at 3-BV.
In the case of Al+3, the shapes of predicted and experimental curves are similar. The predicted BTP is slightly earlier than the experimental one, but there is a complete match from 3-BV onwards. The predicted Mn+2 -BTC overlaps the experimental one, showing a good approximation. It can be seen as an auspicious outcome for Fe+2, matching both curves along with the pattern. The metal ion Zn+2 shows a similar sigmoidal shape between predictions and experimental BTCs. Nevertheless, the predicted BTP takes place 1-BV earlier than the experimental one. Finally, for Cu+2, the most resin-affine ion, the graph shows no presence of this cation in the outflow solution during the experimental loading, and this characteristic was also predicted.
5. Conclusions
A 1D reactive transport model based on PhreeqC code has been developed to simulate the selective removal of metals from acidic mine waters by chelating bis-picolylamine resins (Lewatit® MonoPlus TP-220). The BTCs obtained through simulations helped to identify and study the three key parameters that can govern the adsorption model: CEC, exchange coefficients, and activity coefficients. The model was used to simulate systems with two-cation, four-cation, and six-cation inflow compositions. This configuration of scenarios helped gradually evaluate and upgrade the model while estimating values for CEC and exchange coefficients. The significant percentage difference determined between the theoretical and estimated values confirms, firstly, that the default exchange coefficients require adjustment when using synthetic resins. In addition, the theoretical CEC of the exchanger should be experimentally verified according to the characteristics of each case. For the particularities and boundary conditions proposed in this study, the estimated values are considered stochastic variables.
This study took a novel approach to model IX processes with complex mine water matrices. We obtained good correlations and were able to improve the results through iterative calibrations.
This first approach presents deviations in some components of the proposed systems and does not take into account other possible reactions that may be part of the process. However, these deficiencies can be overcome with further iterative calibration and post-use resin studies. Due to the great potential of the process and synthetic resins, this methodology is proposed, as it allows results to be obtained in very short periods of time and at low or no cost.
To improve the knowledge, we want to strengthen this research with microscopic analysis of the exchanger material to evaluate other types of reactions that may occur and affect the IX process and incorporate the UCODE program to estimate more parameters that can further boost the model’s scope. Once the upgraded model is established, it could be used to model IX processes for all kind of real scenarios.