In Vivo Evidence for Voltage-Gated Sodium Channel Expression in Carcinomas and Potentiation of Metastasis
Abstract
:1. Introduction
2. mRNA Level Studies
3. Protein Expression
4. Tissue Electrolytes
5. In Vivo Animal Tests
6. Clinical Studies
7. Conclusions and Future Perspective
Funding
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Djamgoz, M.B.A.; Coombes, R.C.; Schwab, A. Ion transport and cancer: From initiation to metastasis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130092. [Google Scholar] [CrossRef] [PubMed]
- Lastraioli, E.; Iorio, J.; Arcangeli, A. Ion channel expression as promising cancer biomarker. Biochim. et Biophys. Acta 2015, 1848, 2685–2702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prevarskaya, N.; Skryma, R.; Shuba, Y. Ion channels in cancer: Are cancer hallmarks oncochannelopathies? Physiol. Rev. 2018, 98, 559–621. [Google Scholar] [CrossRef] [PubMed]
- Tuszynski, J.; Tilli, T.M.; Levin, M. Ion channel and neurotransmitter modulators as electroceutical approaches to the control of cancer. Curr. Pharm. Des. 2017, 23, 4827–4841. [Google Scholar] [CrossRef] [PubMed]
- Grimes, J.A.; Fraser, S.P.; Stephens, G.J.; Downing, J.E.; Laniado, M.E.; Foster, C.S.; Abel, P.D.; Djamgoz, M.B.A. Differential expression of voltage-activated Na+ currents in two prostatic tumour cell lines: Contribution to invasiveness in vitro. FEBS Lett. 1995, 369, 290–294. [Google Scholar] [CrossRef]
- Laniado, M.E.; Lalani, E.-N.; Fraser, S.P.; Grimes, J.A.; Bhangal, G.; Djamgoz, M.B.A.; Abel, P.D. Expression and functional analysis of voltage-activated Na+ channels in human prostate cancer cell lines and their contribution to invasion in vitro. Am. J. Pathol. 1997, 150, 1213–1221. [Google Scholar]
- Fraser, S.P.; Diss, J.K.J.; Chioni, A.M.; Mycielska, M.; Pan, H.; Yamaci, R.F.; Pani, F.; Siwy, Z.; Krasowska, M.; Grzywna, Z.; et al. Voltage-gated sodium channel expression and potentiation of human breast cancer metastasis. Clin. Cancer Res. 2005, 11, 5381–5389. [Google Scholar] [CrossRef]
- Brackenbury, W.J.; Chioni, A.M.; Diss, J.K.J.; Djamgoz, M.B.A. The neonatal splice variant of Nav1.5 potentiates in vitro invasive behaviour of MDA-MB-231 human breast cancer cells. Breast Cancer Res. Treat. 2007, 101, 149–160. [Google Scholar] [CrossRef]
- Nelson, M.; Yang, M.; Millican-Slater, R.; Brackenbury, W.J. Nav1.5 regulates breast tumor growth and metastatic dissemination in vivo. Oncotarget 2015, 6, 32914–32929. [Google Scholar] [CrossRef] [PubMed]
- Driffort, V.; Gillet, L.; Bon, E.; Marionneau-Lambot, S.; Oullier, T.; Joulin, V.; Collin, C.; Pagès, J.C.; Jourdan, M.L.; Chevalier, S.; et al. Ranolazine inhibits Nav1.5-mediated breast cancer cell invasiveness and lung colonization. Mol. Cancer 2014, 13, 264. [Google Scholar] [CrossRef] [PubMed]
- Guzel, R.M.; Ogmen, K.; Ilieva, K.M.; Fraser, S.P.; Djamgoz, M.B.A. Colorectal cancer invasiveness in vitro: Predominant contribution of neonatal Nav1.5 under normoxia and hypoxia. J. Cell. Physiol. 2019, 234, 6582–6593. [Google Scholar] [CrossRef] [PubMed]
- House, C.D.; Vaske, C.J.; Schwartz, A.; Obias, V.; Frank, B.; Luu, T.; Sarvazyan, N.; Irby, R.; Strausberg, R.L.; Hales, T.G.; et al. Voltage-gated Na+ channel SCN5A is a key regulator of a gene transcriptional network that controls colon cancer invasion. Cancer Res. 2010, 70, 6957–6967. [Google Scholar] [CrossRef] [PubMed]
- Baptista-Hon, D.T.; Robertson, F.M.; Robertson, G.B.; Owen, S.J.; Rogers, G.W.; Lydon, E.L.; Lee, N.H.; Hales, T.G. Potent inhibition by ropivacaine of metastatic colon cancer SW620 cell invasion and Nav1.5 channel function. Br. J. Anaesth. 2014, 113, i39–i48. [Google Scholar] [CrossRef] [PubMed]
- Diss, J.K.J.; Stewart, D.; Pani, F.; Foster, C.S.; Walker, M.M.; Patel, A.; Djamgoz, M.B.A. A potential novel marker for human prostate cancer: Voltage-gated sodium channel expression in vivo. Prostate Cancer Prostatic Dis. 2005, 8, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, T.; Kubota, N.; Tsutsumi, T.; Oguri, A.; Imuta, H.; Jo, T.; Oonuma, H.; Soma, M.; Meguro, K.; Takano, H.; et al. Eicosapentaenoic acid inhibits voltage-gated sodium channels and invasiveness in prostate cancer cells. Br. J. Pharmacol. 2009, 156, 420–431. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.; Zhang, C.; Wang, Z.; Chen, Y.; Xie, H.; Li, S.; Liu, X.; Liu, Z.; Chen, P. Mechanistic insights into Nav1.7-dependent regulation of rat prostate cancer cell invasiveness revealed by toxin probes and proteomic analysis. FEBS J. 2019, 286, 2549–2561. [Google Scholar] [CrossRef]
- Campbell, T.M.; Main, M.J.; Fitzgerald, E.M. Functional expression of the voltage-gated Na+-channel Nav1.7 is necessary for EGF-mediated invasion in human non-small cell lung cancer cells. J. Cell Sci. 2013, 126, 4939–4949. [Google Scholar] [CrossRef]
- Fulgenzi, G.; Graciotti, L.; Faronato, M.; Soldovieri, M.V.; Miceli, F.; Amoroso, S.; Annunziato, L.; Procopio, A.; Taglialatela, M. Human neoplastic mesothelial cells express voltage-gated sodium channels involved in cell motility. Int. J. Biochem. Cell Biol. 2006, 38, 1146–1159. [Google Scholar] [CrossRef]
- Diaz, D.; Delgadillo, D.M.; Hernandez-Gallegos, E.; Ramirez-Dominguez, M.E.; Hinojosa, L.M.; Ortiz, C.S.; Berumen, J.; Camacho, J.; Gomora, J.C. Functional expression of voltage-gated sodium channels in primary cultures of human cervical cancer. J. Cell. Physiol. 2007, 210, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Plata, E.; Ortiz, C.S.; Marquina-Castillo, B.; Medina-Martinez, I.; Alfaro, A.; Berumen, J.; Rivera, M.; Gomora, J.C. Overexpression of Nav1.6 channels is associated with the invasion capacity of human cervical cancer. Int. J. Cancer 2012, 130, 2013–2023. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Huang, N.; Huang, H.; Sun, L.; Dong, S.; Su, J.; Zhang, J.; Wang, L.; Lin, L.; Shi, M.; et al. Voltage-gated sodium channel Nav 1.7 promotes gastric cancer progression through MACC1-mediated upregulation of NHE1. Int. J. Cancer 2016, 139, 2553–2569. [Google Scholar] [CrossRef]
- Gao, R.; Shen, Y.; Cai, J.; Lei, M.; Wang, Z. Expression of voltage-gated sodium channel alpha subunit in human ovarian cancer. Oncol. Rep. 2010, 23, 1293–1299. [Google Scholar] [PubMed]
- Gao, R.; Cao, T.; Chen, H.; Cai, J.; Lei, M.; Wang, Z. Nav1.5-E3 antibody inhibits cancer progression. Transl. Cancer Res. 2019, 8, 44–50. [Google Scholar] [CrossRef]
- Xie, A.; Gallant, B.; Guo, H.; Gonzalez, A.; Clark, M.; Madigan, A.; Feng, F.; Chen, H.D.; Cui, Y.; Dudley, S.C., Jr.; et al. Functional cardiac Na+ channels are expressed in human melanoma cells. Oncol. Lett. 2018, 16, 1689–1695. [Google Scholar] [CrossRef]
- Zhang, J.; Mao, W.; Dai, Y.; Qian, C.; Dong, Y.; Chen, Z.; Meng, L.; Jiang, Z.; Huang, T.; Hu, J.; et al. Voltage-gated sodium channel Nav1.5 promotes proliferation, migration and invasion of oral squamous cell carcinoma. Acta Biochim. Biophys. Sin. 2019, 51, 562–570. [Google Scholar] [CrossRef]
- Xing, D.; Wang, J.; Ou, S.; Wang, Y.; Qiu, B.; Ding, D.; Guo, F.; Gao, Q. Expression of neonatal Nav1.5 in human brain astrocytoma and its effect on proliferation, invasion and apoptosis of astrocytoma cells. Oncol. Rep. 2014, 31, 2692–2700. [Google Scholar] [CrossRef] [Green Version]
- Ou, S.W.; Kameyama, A.; Hao, L.Y.; Horiuchi, M.; Minobe, E.; Wang, W.Y.; Makita, N.; Kameyama, M. Tetrodotoxin-resistant Na+ channels in human neuroblastoma cells are encoded by new variants of Nav1.5/SCN5A. Eur. J. Neurosci. 2005, 22, 793–801. [Google Scholar] [CrossRef]
- Liu, J.; Tan, H.; Yang, W.; Yao, S.; Hong, L. The voltage-gated sodium channel Nav1.7 associated with endometrial cancer. J. Cancer 2019, 10, 4954–4960. [Google Scholar] [CrossRef]
- Roger, S.; Rollin, J.; Barascu, A.; Besson, P.; Raynal, P.I.; Iochmann, S.; Lei, M.; Bougnoux, P.; Gruel, Y.; Le Guennec, J.Y. Voltage-gated sodium channels potentiate the invasive capacities of human non-small-cell lung cancer cell lines. Int. J. Biochem. Cell Biol. 2007, 39, 774–786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Djamgoz, M.B.A. Biophysics of cancer: Cellular excitability (“CELEX”) hypothesis of metastasis. J. Clin. Exper. Oncol. 2014, S1, 005. [Google Scholar] [CrossRef]
- Djamgoz, M.B.A. Bioelectricity of cancer: Voltage-gated ion channels and direct-current electric fields. In The Physiology of Bioelectricity in Development, Tissue Regeneration, and Cancer; Pullar, C., Ed.; Taylor & Francis: London, UK, 2011; pp. 269–294. [Google Scholar]
- Cabello, M.; Ge, H.; Aracil, C.; Moschou, D.; Estrela, P.; Manuel Quero, J.; Pascu, S.I.; Rocha, P.R.F. Extracellular electrophysiology in the prostate cancer cell model PC-3. Sensors 2019, 19, 139. [Google Scholar] [CrossRef] [PubMed]
- Simon, A.; Yang, M.; Marrison, J.L.; James, A.D.; O’Toole, P.J.; Kaye, P.M.; Whittington, M.A.; Chawla, S.; Brackenbury, W.J. Metastatic breast cancer cells induce altered microglial morphology and electrical excitability in vivo. BioRxiv 2019. [Google Scholar] [CrossRef]
- Djamgoz, M.B.A.; Onkal, R. Persistent current blockers of voltage-gated sodium channels: A clinical opportunity for controlling metastatic disease. Recent Pat. Anticancer Drug Discov. 2013, 8, 66–84. [Google Scholar] [CrossRef]
- Brisson, L.; Gillet, L.; Calaghan, S.; Besson, P.; Le Guennec, J.Y.; Roger, S.; Gore, J. Nav1.5 enhances breast cancer cell invasiveness by increasing NHE1-dependent H+ efflux in caveolae. Oncogene 2011, 30, 2070–2076. [Google Scholar] [CrossRef]
- Brisson, L.; Driffort, V.; Benoist, L.; Poet, M.; Counillon, L.; Antelmi, E.; Rubino, R.; Besson, P.; Labbal, F.; Chevalier, S.; et al. Nav1.5 Na+ channels allosterically regulate the NHE-1 exchanger and promote the activity of breast cancer cell invadopodia. J. Cell Sci. 2013, 126, 4835–4842. [Google Scholar] [CrossRef]
- Leanza, L.; Managò, A.; Zoratti, M.; Gulbins, E.; Szabo, I. Pharmacological targeting of ion channels for cancer therapy: In vivo evidences. Biochim. Biophys. Acta 2016, 1863, 1385–1397. [Google Scholar] [CrossRef]
- Shan, B.; Dong, M.; Tang, H.; Wang, N.; Zhang, J.; Yan, C.; Jiao, X.; Zhang, H.; Wang, C. Voltage-gated sodium channels were differentially expressed in human normal prostate, benign prostatic hyperplasia and prostate cancer cells. Oncol. Lett. 2014, 8, 345–350. [Google Scholar] [CrossRef] [Green Version]
- Yang, M.; Kozminski, D.J.; Wold, L.A.; Modak, R.; Calhoun, J.D.; Isom, L.L.; Brackenbury, W.J. Therapeutic potential for phenytoin: Targeting Nav1.5 sodium channels to reduce migration and invasion in metastatic breast cancer. Breast Cancer Res. Treat. 2012, 134, 603–615. [Google Scholar] [CrossRef]
- Peng, J.; Ou, Q.; Wu, X.; Zhang, R.; Zhao, Q.; Jiang, W.; Lu, Z.; Wan, D.; Pan, Z.; Fang, Y. Expression of voltage-gated sodium channel Nav1.5 in non-metastatic colon cancer and its associations with estrogen receptor (ER)-β expression and clinical outcomes. Chin. J. Cancer 2017, 36, 89. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Charcas, O.; Espinosa, A.M.; Alfaro, A.; Herrera-Carrillo, Z.; Ramirez-Cordero, B.E.; Cortes-Reynosa, P.; Perez Salazar, E.; Berumen, J.; Gomora, J.C. The invasiveness of human cervical cancer associated to the function of Nav1.6 channels is mediated by MMP-2 activity. Sci. Rep. 2018, 8, 12995. [Google Scholar] [CrossRef] [PubMed]
- Igci, Y.Z.; Bozgeyik, E.; Borazan, E.; Pala, E.; Suner, A.; Ulasli, M.; Gurses, S.A.; Yumrutas, O.; Balik, A.A.; Igci, M. Expression profiling of SCN8A and NDUFC2 genes in colorectal carcinoma. Exp. Oncol. 2015, 37, 77–80. [Google Scholar] [CrossRef]
- Wu, W.K.; Li, G.R.; Wong, H.P.; Hui, M.K.; Tai, E.K.; Lam, E.K.; Shin, V.Y.; Ye, Y.N.; Li, P.; Yang, Y.H.; et al. Involvement of Kv1.1 and Nav1.5 in proliferation of gastric epithelial cells. J. Cell. Physiol. 2006, 207, 437–444. [Google Scholar] [CrossRef]
- Pedersen, S.F.; Stock, C. Ion channels and transporters in cancer: Pathophysiology, regulation, and clinical potential. Cancer Res. 2013, 73, 1658–1661. [Google Scholar] [CrossRef]
- Martin, K.C.; Zukin, R.S. RNA trafficking and local protein synthesis in dendrites: An overview. J. Neurosci. 2006, 26, 7131–7134. [Google Scholar] [CrossRef]
- Pfeiffer, B.E.; Huber, K.M. Current advances in local protein synthesis and synaptic plasticity. J. Neurosci. 2006, 26, 7147–7150. [Google Scholar] [CrossRef]
- Schedel, J.; Distler, O.; Woenckhaus, M.; Gay, R.E.; Simmen, B.; Michel, B.A.; Müller-Ladner, U.; Gay, S. Discrepancy between mRNA and protein expression of tumour suppressor maspin in synovial tissue may contribute to synovial hyperplasia in rheumatoid arthritis. Ann. Rheum. Dis. 2004, 63, 1205–1211. [Google Scholar] [CrossRef] [Green Version]
- Bugan, I.; Kucuk, S.; Karagoz, Z.; Fraser, S.P.; Kaya, H.; Dodson, A.; Foster, C.S.; Altun, S.; Djamgoz, M.B.A. Anti-metastatic effect of ranolazine in an in vivo rat model of prostate cancer, and expression of voltage-gated sodium channel protein in human prostate. Prostate Cancer Prostatic Dis. 2019. [Google Scholar] [CrossRef]
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A. Grading Committee. The 2014 International Society of Urological Pathology (ISUP) consensus Conference on Gleason grading of prostatic carcinoma: Definition of grading patterns and proposal for a new grading system. Am. J. Surg. Pathol. 2016, 40, 244–252. [Google Scholar] [CrossRef]
- Suy, S.; Hansen, T.P.; Auto, H.D.; Kallakury, B.V.; Dailey, V.; Danner, M.; Macarthur, L.; Zhang, Y.; Miessau, M.J.; Collins, S.P.; et al. Expression of voltage-gated sodium channel Nav1.8 in human prostate cancer is associated with high histological grade. J. Clin. Exp. Oncol. 2012, 1. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, S.; Altun, S.; Gumushan, H.; Patel, A.; Djamgoz, M.B.A. Voltage-gated sodium channel activity promotes prostate cancer metastasis in vivo. Cancer Lett. 2012, 323, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Onganer, P.U.; Seckl, M.J.; Djamgoz, M.B.A. Neuronal characteristics of small-cell lung cancer. Br. J. Cancer 2005, 93, 1197–1201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Onkal, R.; Mattis, J.H.; Fraser, S.P.; Diss, J.K.J.; Shao, D.; Okuse, K.; Djamgoz, M.B.A. Alternative splicing of Nav1.5: An electrophysiological comparison of ‘neonatal’ and ‘adult’ isoforms, and critical involvement of a lysine residue. J. Cell. Physiol. 2008, 216, 716–726. [Google Scholar] [CrossRef]
- Chioni, A.M.; Fraser, S.P.; Pani, F.; Foran, P.; Wilkin, G.P.; Diss, J.K.J.; Djamgoz, M.B.A. A novel polyclonal antibody specific for the Nav1.5 voltage-gated Na+ channel ‘neonatal’ splice form. J. Neurosci. Methods 2005, 147, 88–98. [Google Scholar] [CrossRef]
- Yamaci, R.F.; Fraser, S.P.; Battaloglu, E.; Kaya, H.; Erguler, K.; Foster, C.S.; Djamgoz, M.B.A. Neonatal Nav1.5 protein expression in normal adult human tissues and breast cancer. Pathol. Res. Prac. 2017, 213, 900–907. [Google Scholar] [CrossRef]
- Guzel, R.M. Studies of Ionic Mechanisms Associated with Human Cancers. Ph.D. Thesis, Imperial College, London, UK, 2012. [Google Scholar]
- Barshack, I.; Levite, M.; Lang, A.; Fudim, E.; Picard, O.; Ben Horin, S.; Chowers, Y. Functional voltage-gated sodium channels are expressed in human intestinal epithelial cells. Digestion 2008, 77, 108–117. [Google Scholar] [CrossRef]
- Chioni, A.M.; Shao, D.; Grose, R.; Djamgoz, M.B.A. Protein kinase A and regulation of neonatal Nav1.5 expression in human breast cancer cells: Activity-dependent positive feedback and cellular migration. Int. J. Biochem. Cell Bio. 2010, 42, 346–358. [Google Scholar] [CrossRef]
- Fraser, S.P.; Özerlat-Gunduz, I.; Brackenbury, W.J.; Fitzgerald, E.M.; Campbell, T.; Coombes, R.C.; Djamgoz, M.B.A. Regulation of voltage-gated sodium channel expression in cancer: Hormones, growth factors and auto-regulation. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2014, 369, 20130105. [Google Scholar] [CrossRef]
- Zhang, C.; Bosch, M.A.; Qiu, J.; Rønnekleiv, O.K.; Kelly, M.J. 17β-Estradiol increases persistent Na+ current and excitability of AVPV/PeN Kiss1 neurons in female mice. Mol. Endocrinol. 2015, 29, 518–527. [Google Scholar] [CrossRef]
- Bi, R.Y.; Meng, Z.; Zhang, P.; Wang, X.D.; Ding, Y.; Gan, Y.H. Estradiol upregulates voltage-gated sodium channel 1.7 in trigeminal ganglion contributing to hyperalgesia of inflamed TMJ. PLoS ONE 2017, 12, e0178589. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, P.; Fang, J.; Li, Q.; Xiao, H.; Zhou, H.; Tang, B. Simultaneous quantitation of Na+ and K+ in single normal and cancer cells using a new near-infrared fluorescent probe. Anal. Chem. 2015, 87, 6057–6063. [Google Scholar] [CrossRef] [PubMed]
- Leslie, T.K.; James, A.D.; Zaccagna, F.; Grist, J.T.; Deen, S.; Kennerley, A.; Riemer, F.; Kaggie, J.D.; Gallagher, F.A.; Gilbert, F.J.; et al. Sodium homeostasis in the tumour microenvironment. Biochim. Biophys. Acta Rev. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Ouwerkerk, R.; Jacobs, M.A.; Macura, K.J.; Wolff, A.C.; Stearns, V.; Mezban, S.D.; Khouri, N.F.; Bluemke, D.A.; Bottomley, P.A. Elevated tissue sodium concentration in malignant breast lesions detected with non-invasive 23Na MRI. Breast Cancer Res. Treat. 2007, 106, 151–160. [Google Scholar] [CrossRef]
- Broeke, N.C.; Peterson, J.; Lee, J.; Martin, P.R.; Farag, A.; Gomez, J.A.; Moussa, M.; Gaed, M.; Chin, J.; Pautler, S.E.; et al. Characterization of clinical human prostate cancer lesions using 3.0-T sodium MRI registered to Gleason-graded whole-mount histopathology. J. Mag. Res. Imaging 2019, 49, 1409–1419. [Google Scholar] [CrossRef]
- Deen, S.S.; Riemer, F.; McLean, M.A.; Gill, A.B.; Kaggie, J.D.; Grist, J.T.; Crawford, R.; Latimer, J.; Baldwin, P.; Earl, H.M.; et al. Sodium MRI with 3D-cones as a measure of tumour cellularity in high grade serous ovarian cancer. Eur. J. Radiol. Open 2019, 6, 156–162. [Google Scholar] [CrossRef] [Green Version]
- Babsky, A.M.; Ju, S.; Bennett, S.; George, B.; McLennan, G.; Bansal, N. Effect of implantation site and growth of hepatocellular carcinoma on apparent diffusion coefficient of water and sodium MRI. NMR Biomed. 2012, 25, 312–321. [Google Scholar] [CrossRef]
- Babsky, A.M.; Hekmatyar, S.K.; Zhang, H.; Solomon, J.L.; Bansal, N. Application of 23Na MRI to monitor chemotherapeutic response in RIF-1 tumors. Neoplasia 2005, 7, 658–666. [Google Scholar] [CrossRef]
- Schepkin, V.D.; Chenevert, T.L.; Kuszpit, K.; Lee, K.C.; Meyer, C.R.; Johnson, T.D.; Rehemtulla, A.; Ross, B.D. Sodium and proton diffusion MRI as biomarkers for early therapeutic response in subcutaneous tumors. Magn. Reson. Imaging 2006, 24, 273–278. [Google Scholar] [CrossRef] [Green Version]
- Schepkin, V.D.; Lee, K.C.; Kuszpit, K.; Muthuswami, M.; Johnson, T.D.; Chenevert, T.L.; Rehemtulla, A.; Ross, B.D. Proton and sodium MRI assessment of emerging tumor chemotherapeutic resistance. NMR Biomed. 2006, 19, 1035–1042. [Google Scholar] [CrossRef]
- Kline, R.P.; Wu, E.X.; Petrylak, D.P.; Szabolcs, M.; Alderson, P.O.; Weisfeldt, M.L.; Cannon, P.; Katz, J. Rapid in vivo monitoring of chemotherapeutic response using weighted sodium magnetic resonance imaging. Clin. Cancer Res. 2000, 6, 2146–2156. [Google Scholar] [PubMed]
- Jacobs, M.A.; Ouwerkerk, R.; Wolff, A.C.; Gabrielson, E.; Warzecha, H.; Jeter, S.; Bluemke, D.A.; Wahl, R.; Stearns, V. Monitoring of neoadjuvant chemotherapy using multiparametric, 23Na sodium MR, and multimodality (PET/CT/ MRI) imaging in locally advanced breast cancer. Breast Cancer Res. Treat. 2011, 128, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Near, J.; Romagnoli, C.; Bartha, R. Reduced power magnetic resonance spectroscopic imaging of the prostate at 4.0 Tesla. Magn. Reson. Med. 2009, 61, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Barrett, T.; Riemer, F.; McLean, M.A.; Kaggie, J.; Robb, F.; Tropp, J.S.; Warren, A.; Bratt, O.; Shah, N.; Gnanapragasam, V.J.; et al. Quantification of total and intracellular sodium concentration in primary prostate cancer and adjacent normal prostate tissue with magnetic resonance imaging. Invest. Radiol. 2018, 53, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Sişman, A.R.; Sis, B.; Canda, T.; Onvural, B. Electrolytes and trace elements in human breast cyst fluid. Biol. Trace Element Res. 2009, 128, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Rosner, M.H.; Dalkin, A.C. Electrolyte disorders associated with cancer. Adv. Chronic Kidney Dis. 2014, 21, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Berardi, R.; Rinaldi, S.; Caramanti, M.; Grohè, C.; Santoni, M.; Morgese, F.; Torniai, M.; Savini, A.; Fiordoliva, I.; Cascinu, S. Hyponatremia in cancer patients: Time for a new approach. Crit. Rev. Oncol. Hematol. 2016, 102, 15–25. [Google Scholar] [CrossRef]
- Carter, B.C.; Bean, B.P. Sodium entry during action potentials of mammalian central neurons: Incomplete inactivation and reduced metabolic efficiency in fast-spiking neurons. Neuron 2009, 64, 898–909. [Google Scholar] [CrossRef]
- Lee, A.; Fraser, S.P.; Djamgoz, M.B.A. Propranolol inhibits neonatal Nav1.5 activity and invasiveness of MDA-MB-231 breast cancer cells: Effects of combination with ranolazine. J. Cell. Physiol. 2019. [Epub ahead of print]. [Google Scholar] [CrossRef]
- Batcioglu, K.; Uyumlu, A.B.; Satilmis, B.; Yildirim, B.; Yucel, N.; Demirtas, H.; Onkal, R.; Guzel, R.M.; Djamgoz, M.B.A. Oxidative stress in the in vivo DMBA rat model of breast cancer: Suppression by a voltage-gated sodium channel inhibitor (RS100642). Basic Clin. Pharmacol. Toxicol. 2012, 111, 137–141. [Google Scholar] [CrossRef]
- Sheets, M.F.; Fozzard, H.A.; Lipkind, G.M.; Hanck, D.A. Sodium channel molecular conformations and antiarrhythmic drug affinity. Trends Cardiovasc. Med. 2010, 20, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.; Yang, M.; Dowle, A.A.; Thomas, J.R.; Brackenbury, W.J. The sodium channel-blocking antiepileptic drug phenytoin inhibits breast tumour growth and metastasis. Mol. Cancer 2015, 14, 13. [Google Scholar] [CrossRef]
- Chioni, A.M.; Brackenbury, W.J.; Calhoun, J.D.; Isom, L.L.; Djamgoz, M.B.A. A novel adhesion molecule in human breast cancer cells: Voltage-gated Na+ channel beta1 subunit. Int. J. Biochem. Cell Biol. 2009, 41, 1216–1227. [Google Scholar] [CrossRef] [PubMed]
- Diss, J.K.J.; Fraser, S.P.; Walker, M.M.; Patel, A.; Latchman, D.S.; Djamgoz, M.B.A. Beta-subunits of voltage-gated sodium channels in human prostate cancer: Quantitative in vitro and in vivo analyses of mRNA expression. Prostate Cancer and Prostatic Dis. 2008, 11, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.; Millican-Slater, R.; Forrest, L.C.; Brackenbury, W.J. The sodium channel beta1 subunit mediates outgrowth of neurite-like processes on breast cancer cells and promotes tumour growth and metastasis. Int. J. Cancer 2014, 135, 2338–2351. [Google Scholar] [CrossRef]
- Jansson, K.H.; Lynch, J.E.; Lepori-Bui, N.; Czymmek, K.J.; Duncan, R.L.; Sikes, R.A. Overexpression of the VSSC-associated CAM, beta-2, enhances LNCaP cell metastasis associated behavior. Prostate 2012, 72, 1080–1092. [Google Scholar] [CrossRef]
- Adachi, K.; Toyota, M.; Sasaki, Y.; Yamashita, T.; Ishida, S.; Ohe-Toyota, M.; Maruyama, R.; Hinoda, Y.; Saito, T.; Imai, K.; et al. Identification of SCN3B as a novel p53-inducible proapoptotic gene. Oncogene 2004, 23, 7791–7798. [Google Scholar] [CrossRef] [Green Version]
- Bon, E.; Driffort, V.; Gradek, F.; Martinez-Caceres, C.; Anchelin, M.; Pelegrin, P.; Cayuela, M.L.; Marionneau-Lambot, S.; Oullier, T.; Guibon, R.; et al. SCN4B acts as a metastasis-suppressor gene preventing hyperactivation of cell migration in breast cancer. Nat. Commun. 2016, 7, 13648. [Google Scholar] [CrossRef]
- Sanchez-Sandoval, A.L.; Gomora, J.C. Contribution of voltage-gated sodium channel β-subunits to cervical cancer cells metastatic behavior. Cancer Cell Int. 2019, 19, 35. [Google Scholar] [CrossRef]
- Gong, Y.; Yang, J.; Wu, W.; Liu, F.; Su, A.; Li, Z.; Zhu, J.; Wei, T. Preserved SCN4B expression is an independent indicator of favorable recurrence-free survival in classical papillary thyroid cancer. PLoS One 2018, 13, e0197007. [Google Scholar] [CrossRef]
- Martin, F.; Ufodiama, C.; Watt, I.; Bland, M.; Brackenbury, W.J. Therapeutic value of voltage-gated sodium channel inhibitors in breast, colorectal and prostate cancer: A systematic review. Front. Pharmacol. 2015, 6, 273. [Google Scholar] [CrossRef] [PubMed]
- Chamaraux-Tran, T.N.; Mathelin, C.; Aprahamian, M.; Joshi, G.P.; Tomasetto, C.; Diemunsch, P.; Akladios, C. Antitumor effects of lidocaine on human breast cancer cells: An in vitro and in vivo experimental trial. Anticancer Res. 2018, 38, 95–105. [Google Scholar] [PubMed]
- Johnson, M.Z.; Crowley, P.D.; Foley, A.G.; Xue, C.; Connolly, C.; Gallagher, H.C.; Buggy, D.J. Effect of perioperative lidocaine on metastasis after sevoflurane or ketamine-xylazine anaesthesia for breast tumour resection in a murine model. Br. J. Anaesth. 2018, 121, 76–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freeman, J.; Crowley, P.D.; Foley, A.G.; Gallagher, H.C.; Iwasaki, M.; Ma, D.; Buggy, D.J. Effect of perioperative lidocaine, propofol and steroids on pulmonary metastasis in a murine model of breast cancer surgery. Cancers 2019, 11, 613. [Google Scholar] [CrossRef] [PubMed]
- Freeman, J.; Crowley, P.D.; Foley, A.G.; Gallagher, H.C.; Iwasaki, M.; Ma, D.; Buggy, D.J. Effect of perioperative lidocaine and cisplatin on metastasis in a murine model of breast cancer surgery. Anticancer Res. 2018, 38, 5599–5606. [Google Scholar] [CrossRef] [PubMed]
- Xing, W.; Chen, D.T.; Pan, J.H.; Chen, Y.H.; Yan, Y.; Li, Q.; Xue, R.F.; Yuan, Y.F.; Zeng, W.A. Lidocaine induces apoptosis and suppresses tumor growth in human hepatocellular carcinoma cells in vitro and in a xenograft model in vivo. Anesthesiology 2017, 126, 868–881. [Google Scholar] [CrossRef]
- Tada, M.; Imazeki, F.; Fukai, K.; Sakamoto, A.; Arai, M.; Mikata, R.; Tokuhisa, T.; Yokosuka, O. Procaine inhibits the proliferation and DNA methylation in human hepatoma cells. Hepatol. Int. 2007, 1, 355–364. [Google Scholar] [CrossRef] [Green Version]
- Fraser, S.P.; Foo, I.; Djamgoz, M.B.A. Local anaesthetic use in cancer surgery and disease recurrence: Role of voltage-gated sodium channels? Br. J. Anaesth. 2014, 113, 899–902. [Google Scholar] [CrossRef]
- Kim, R. Anesthetic technique and cancer recurrence in oncologic surgery: Unraveling the puzzle. Cancer Metastasis Rev. 2017, 36, 159–177. [Google Scholar] [CrossRef]
- Grandhi, R.K.; Perona, B. Mechanisms of action by which local anesthetics reduce cancer recurrence: A systematic review. Pain Medicine 2019. [Epub ahead of print]. [Google Scholar] [CrossRef]
- Exadaktylos, A.K.; Buggy, D.J.; Moriarty, D.C.; Mascha, E.; Sessler, D.I. Can anesthetic technique for primary breast cancer surgery affect recurrence or metastasis? Anesthesiology 2006, 105, 660–664. [Google Scholar] [CrossRef] [PubMed]
- Biki, B.; Mascha, E.; Moriarty, D.C.; Fitzpatrick, J.M.; Sessler, D.I.; Buggy, D.J. Anesthetic technique for radical prostatectomy surgery affects cancer recurrence: A retrospective analysis. Anesthesiology 2008, 109, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Kao, L.T.; Huang, C.; Lin, H.C.; Huang, C.Y. Antiarrhythmic drug usage and prostate cancer: A population-based cohort study. Asian J. Androl. 2018, 20, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Fairhurst, C.; Watt, I.; Martin, F.; Bland, M.; Brackenbury, W.J. Sodium channel-inhibiting drugs and survival of breast, colon and prostate cancer: A population-based study. Sci. Rep. 2015, 5, 16758. [Google Scholar] [CrossRef]
- Fairhurst, C.; Martin, F.; Watt, I.; Doran, T.; Bland, M.; Brackenbury, W.J. Sodium channel-inhibiting drugs and cancer survival: Protocol for a cohort study using the CPRD primary care database. BMJ Open 2016, 6, e011661. [Google Scholar] [CrossRef]
- Takada, M.; Fujimoto, M.; Motomura, H.; Hosomi, K. Inverse association between sodium channel-blocking antiepileptic drug use and cancer: Data mining of spontaneous reporting and claims databases. Int. J. Med. Sci. 2016, 13, 48–59. [Google Scholar] [CrossRef]
- Walker, A.J.; Card, T.; Bates, T.E.; Muir, K. Tricyclic antidepressants and the incidence of certain cancers: A study using the GPRD. Br. J. Cancer 2011, 104, 193–197. [Google Scholar] [CrossRef]
- Reddy, J.P.; Dawood, S.; Mitchell, M.; Debeb, B.G.; Bloom, E.; Gonzalez-Angulo, A.M.; Sulman, E.P.; Buchholz, T.A.; Woodward, W.A. Antiepileptic drug use improves overall survival in breast cancer patients with brain metastases in the setting of whole brain radiotherapy. Radiother. Oncol. 2015, 117, 308–314. [Google Scholar] [CrossRef]
- Minotti, G. Pharmacology at work for cardio-oncology: Ranolazine to treat early cardiotoxicity induced by antitumor drugs. J. Pharmacol. Exp. Ther. 2013, 346, 343–349. [Google Scholar] [CrossRef]
- Menna, P.; Calabrese, V.; Armento, G.; Annibali, O.; Greco, C.; Salvatorelli, E.; Marchesi, F.; Reggiardo, G.; Minotti, G. Pharmacology of cardio-oncology: Chronotropic and lusitropic effects of B-type natriuretic peptide in cancer patients with early diastolic dysfunction induced by anthracycline or nonanthracycline chemotherapy. J. Pharmacol. Exp. Ther. 2018, 366, 158–168. [Google Scholar] [CrossRef]
- Benhaim, L.; Gerger, A.; Bohanes, P.; Paez, D.; Wakatsuki, T.; Yang, D. Gender-specific profiling in SCN1A polymorphisms and time-to-recurrence in patients with stage II/III colorectal cancer treated with adjuvant 5-fluoruracil chemotherapy. Pharmacogenomics J. 2014, 14, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Koltai, T. Voltage-gated sodium channel as a target for metastatic risk reduction with re-purposed drugs. F1000Research 2015, 4, 297. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lu, Z.; Wu, C.; Li, Y.; Kong, Y.; Zhou, R.; Shi, K.; Guo, J.; Li, N.; Liu, J.; et al. Evaluation of the anticancer and anti-metastasis effects of novel synthetic sodium channel blockers in prostate cancer cells in vitro and in vivo. Prostate 2019, 79, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Brackenbury, W.J.; Djamgoz, M.B.A. Activity-dependent regulation of voltage-gated Na+ channel expression in Mat-LyLu rat prostate cancer cell line. J. Physiol. 2006, 573, 343–356. [Google Scholar] [CrossRef] [PubMed]





| Carcinoma | VGSC Subtype(s) | Comment(s) | Reference(s) |
|---|---|---|---|
| Breast | nNav1.5 (and (n)Nav1.7) | Dominant at mRNA level and functional contribution to invasiveness/metastasis verified in vitro and in vivo | [9,10,11,12] |
| Colon | nNav1.5 | Dominant at mRNA level and expressed early in invasiveness; functional contribution to invasiveness verified in vitro and in vivo | [13,14,15] |
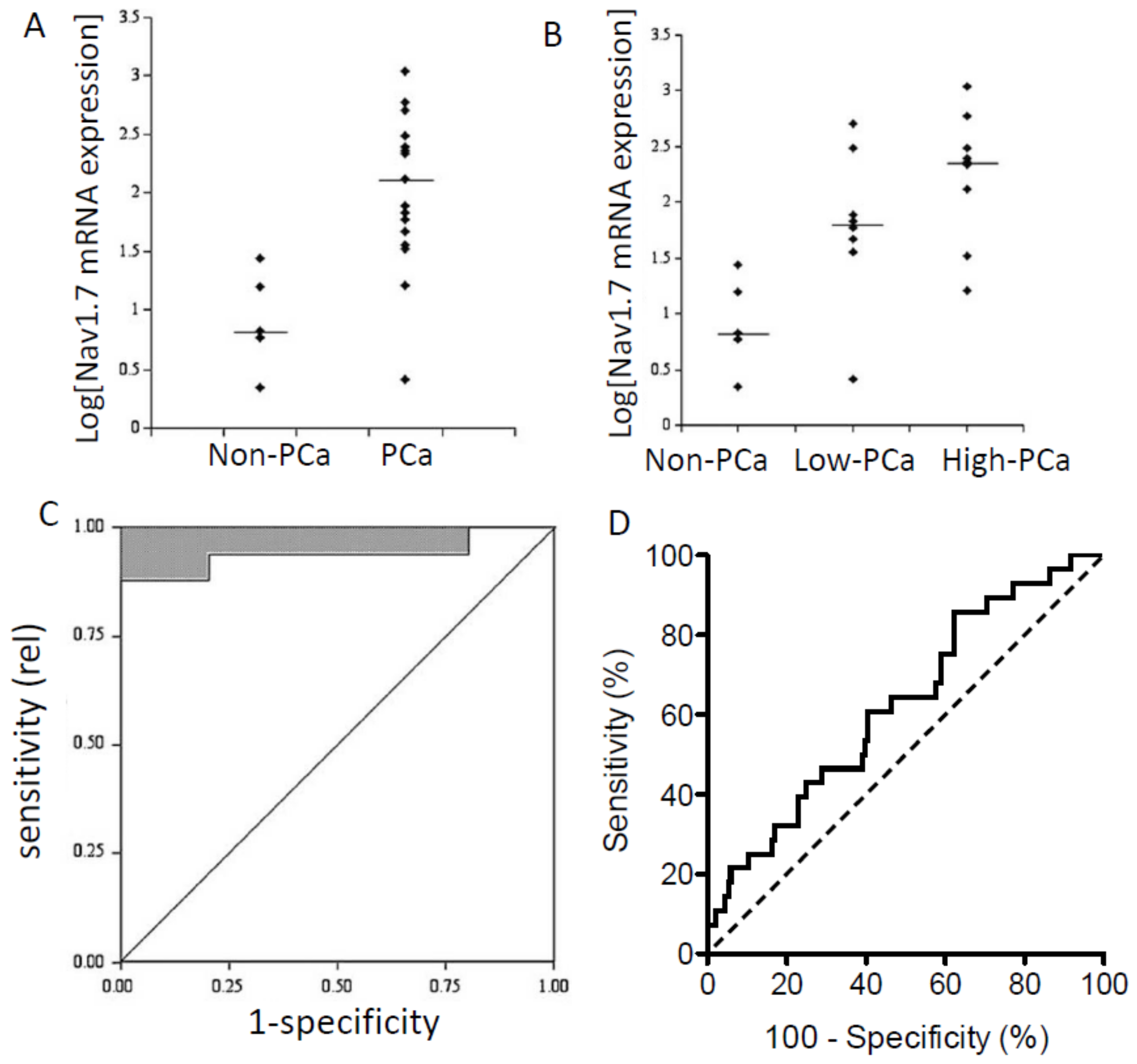
| Prostate | (n)Nav1.7 (and Nav1.6) | Dominant at mRNA level; specific functional contribution to invasiveness tested using peptide toxins | [8,16,17,18] |
| Non-small cell lung carcinoma (NSCLC) | Nav1.7 | Dominant at mRNA level; potentiation of invasiveness demonstrated by use of siRNA | [19] |
| Mesothelioma | Nav1.2, Nav1.6, and Nav1.7 | VGSC activity shown to promote migration in vitro, but the subtype(s) responsible not determined | [20] |
| Cervical | (n)Nav1.6 | Dominant at mRNA level; over-expression potentiated invasiveness | [21,22] |
| Stomach | Nav1.7 | Dominant at mRNA level; silencing suppressed tumour growth in mouse model in vivo | [23] |
| Ovary | Nav1.5 | Dominant at mRNA level; E3 antibody suppressed in vivo growth and in vitro invasiveness | [24,25] |
| Melanoma | Nav1.5 | Expression induced membrane potential depolarization and inhibited Ca2+ uptake | [26] |
| Oral squamous cell carcinoma | Nav1.5 | siRNA confirmed in vitro potentiation of proliferation and invasiveness | [27] |
| Astrocytoma | nNav1.5 | siRNA confirmed in vitro potentiation of proliferation and invasiveness | [28] |
| Neuroblastoma | nNav1.5 | Where ‘neonatal’ Nav1.5 splicing was first described, but role in cancer not investigated | [29] |
| Endometrium | Nav1.7 | Channel block attenuated in vitro cell invasion; expression was associated (i) positively with tumour size and local lymph node metastasis, and (ii) negatively with survival (5–10 years) | [30] |
| VGSC Property | Clinical Consequence(s) | Reference(s) |
|---|---|---|
| Expression much higher in strongly vs. weakly metastatic cancers (by up to several orders of magnitude at mRNA level) | Potential functional diagnostic molecular biomarker | e.g., [9,16,40] |
| Expression is early and upstream of the genes driving invasiveness | Potential functional ‘early’ marker, ideal for diagnostics | [14] |
| Upregulation maintained at protein and functional (signalling) levels | Diagnostics can be extended to conventional immunohistochemistry and even clinical imaging of tissue sodium possibly resulting from channel activity (e.g., 23Na-MRI); expression can be used to determine treatment strategy and efficacy | [9,16,57,65,66] |
| Gene/protein expressed in neonatal splice form in several carcinomas; targetable by antibody | Diagnostics can be made even more specific; antibody can also be used as a drug (i) to block channel activity/metastasis and/or (ii) kill tumour cells (in antibody-drug conjugate mode) | [9,10,56] |
| Activity-dependent regulation (positive feedback)—VGSC blockers suppress both channel activity and expression | VGSC blockage would suppress both activity and expression of the channel, thereby providing long-term benefit to cancer patients taking VGSC drugs | [60,116] |
| Promotes a range of cellular behaviours integral to the metastatic cascade in vitro, as well as metastasis per se in vivo | Therapeutic potential—possible ‘repurposing’ of existing VGSC drugs, INaP blockers (as well as novel antibody), possibly with minimal side effects | [11,12,36,114] |
| Functional expression under the control of steroid hormones and growth factors | Therapeutic potential extended to ‘combination’ treatments | [19,61] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djamgoz, M.B.A.; Fraser, S.P.; Brackenbury, W.J. In Vivo Evidence for Voltage-Gated Sodium Channel Expression in Carcinomas and Potentiation of Metastasis. Cancers 2019, 11, 1675. https://doi.org/10.3390/cancers11111675
Djamgoz MBA, Fraser SP, Brackenbury WJ. In Vivo Evidence for Voltage-Gated Sodium Channel Expression in Carcinomas and Potentiation of Metastasis. Cancers. 2019; 11(11):1675. https://doi.org/10.3390/cancers11111675
Chicago/Turabian StyleDjamgoz, Mustafa B. A., Scott P. Fraser, and William J. Brackenbury. 2019. "In Vivo Evidence for Voltage-Gated Sodium Channel Expression in Carcinomas and Potentiation of Metastasis" Cancers 11, no. 11: 1675. https://doi.org/10.3390/cancers11111675





