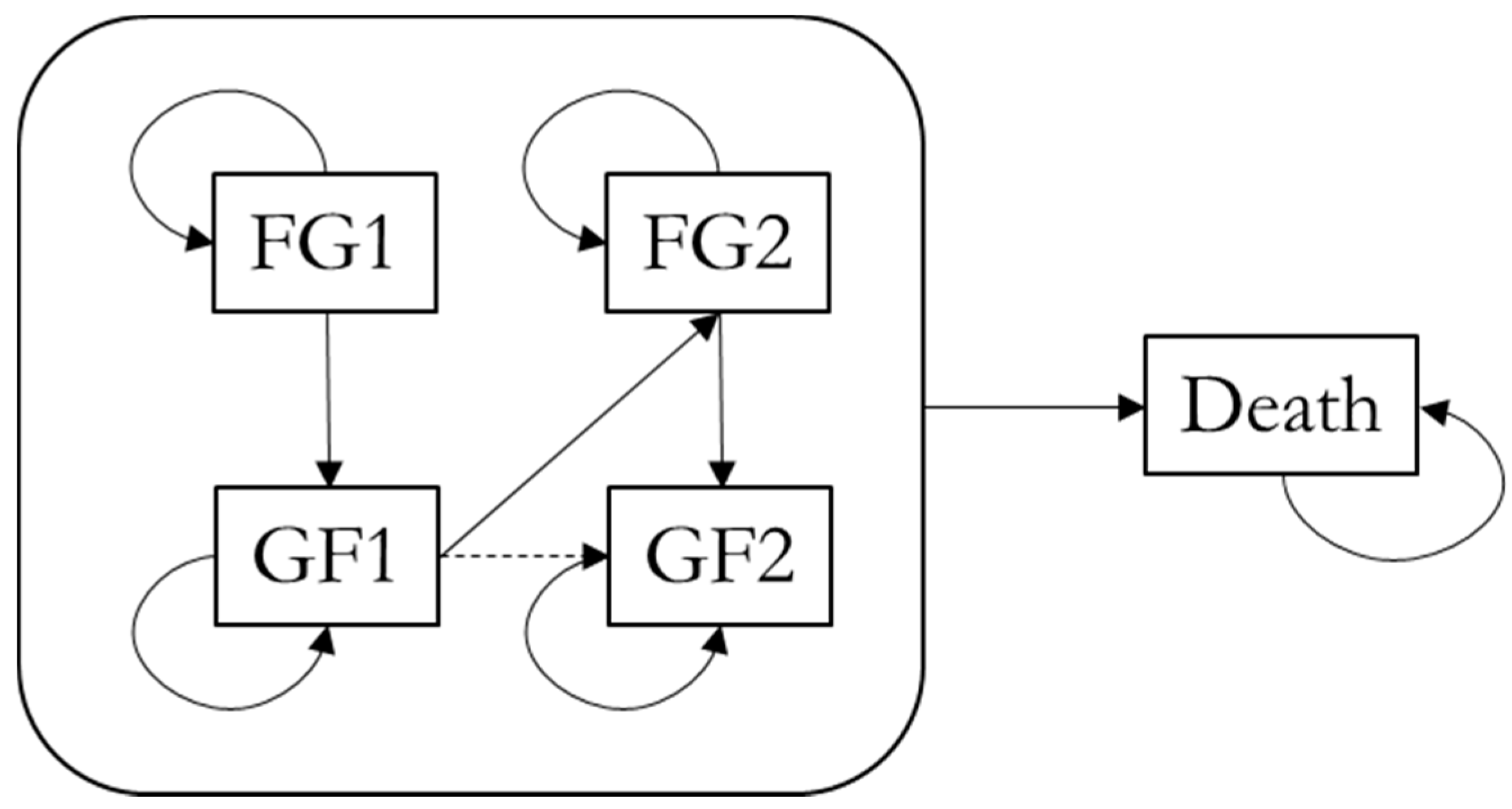
1. Introduction
Chronic kidney disease (CKD) is a disease characterised by a gradual loss in the functioning of the kidneys, occurring over a period of months or years. The worldwide prevalence of CKD is estimated to be 13.4% [
1]. In 2015, CKD led to 1.2 million deaths, which is an increase of 31.7% compared to 2005 [
2]. If the kidneys reach an estimated glomerular filtration rate (eGFR) <15 mL/min, they fail to reach a rate of filtration high enough for the patient to survive. This state is labelled end-stage renal disease (ESRD).
For patients with ESRD, treatment is limited to renal replacement therapy (RRT) by either dialysis or kidney transplantation. Dialysis is an artificial method of replacing kidney function. The two most commonly used types are haemodialysis (HD) and peritoneal dialysis (PD). Both types of dialysis remove excess water and waste from the blood: one externally, one internally. Both types of dialysis come with a high costs to society and a low quality of life [
3]. Dialysis is estimated to cost between 80,000 and 110,000 euros annually, depending on the type of dialysis [
4]. A kidney transplantation has been shown to be cheaper, while patients survive longer and their quality of life is higher [
5]. The costs for the procedure of kidney transplantation and the first year of care are estimated to be between 60,000 and 80,000 euros, followed by annual maintenance costs of approximately 10,000 euros [
4,
6,
7]. Unfortunately, there are only limited numbers of kidneys available for transplantation and there is a gap between supply and demand [
8,
9].
Once a transplanted kidney fails, a patient will require dialysis or re-transplantation to survive. Since the annual costs for dialysis are higher and the quality of life is lower, every extra day that the transplanted kidney stays functional is beneficial. Prevention of need for re-transplantation by improving graft survival will translate into relief of existing organ shortage. Recent observational studies suggest that a patient’s dietary habits affect the rate of mortality and rate of renal graft failure [
10,
11]. Adhering to the Dietary Approach to Stop Hypertension (DASH) diet has been shown to reduce the risk of death with a functioning graft (DWFG) and renal graft failure [
12]. The DASH is a diet consisting of a low intake of red processed meats, sodium, and sweetened beverages and a high intake of legumes, nuts, low-fat dairy products, fruits, vegetables, and whole grains [
13]. Similar effects have been found for a higher intake of potassium [
14].
The DASH diet and potassium supplementation can be directly adapted into a dietary recommendation for patients. Unfortunately, a key problem for patients is adhering to a diet [
15,
16]. In current practice, patients usually receive dietary advice once, and, without frequent visits to a dietician, the methods to track the food intake of patients are often self-reported dietary surveys, which can be biased and possibly misleading [
17].
We hypothesise that renal transplant recipients (RTR) who adhere to either the DASH diet or potassium supplementation will extend the time until both DWFG and renal graft failure, as well as potentially save costs. The aim of this study is to evaluate the cost-effectiveness and budget impact of higher adherence to either the DASH or potassium supplementation.
4. Discussion
The five-year cost of implementing dietary advice (either DASH or potassium supplementation) in the Dutch population are likely to stay below 10 million euros. Although the investment is quite extensive, savings might outweigh costs: our results show that adhering to the DASH diet and potassium supplementation increases the total amount of QALYs compared to the standard of care. Depending on the adherence, the intervention is either cost-saving or cost-effective. This suggests that from both an economic and healthcare point of view, introducing dietary advice into the Dutch RTR population could be a useful intervention.
To our knowledge, there are no studies looking at the cost-effectiveness of dietary interventions for RTR. There is a multicenter randomised controlled trial study currently in development, which aims to assess the effectiveness of exercise and diet on physical functioning, cardiometabolic health, and weight gain in RTR [
39]. They will also gather data on cost-effectiveness. To address the patients’ diet, each patient in their intervention group will receive 15 months of dietary counselling with a total of 12 sessions. In our model, dietary counselling only took place in the first year with a total of eight sessions. It is unclear whether increasing the duration and number of visits will improve dietary adherence, and therefore the cost-effectiveness and budget impact, since there is no literature available on this subject. Furthermore, there are a limited number of studies looking at the cost-effectiveness of dietary interventions in reducing disease progression in CKD. One study found that a very low-protein diet is cost-effective when compared with a moderate low-protein diet [
40]. Another study found that multidisciplinary care, which includes visiting a dietician, was cost-effective when compared to the normal situation for most patients with CKD [
41]. Even though these models are different from the model used in this study, they still address the fact that it is cost-effective to delay dialysis.
With this cost-effectiveness analysis, we attempted to determine the total costs and effects of dietary interventions for different rates of adherence to group 1. At a baseline adherence of 0% to group 1 (and therefore the highest adherence rates to group 2 and 3), both the DASH diet and potassium supplementation were dominant; they increased the amount of QALYs and saved costs. This is in line with our expectations, because patients in groups 2 and 3 are less likely to lose a FG. With more patients keeping a FG, the total amount of QALYs is higher, and they have lower annual maintenance costs due to a kidney transplant upkeep being lower than the annual dialysis costs. However, in group 2 and group 3, patients are less likely to die with a FG, resulting in more patients staying alive; these patients require maintenance costs for a longer time, and it could be suspected that this could offset the cost saved due to fewer patients needing dialysis. Our results showed that even though more patients in group 2 and group 3 survived for a longer time, these costs were lower than the costs incurred due to dialysis. Another explanation is that patients who survive longer cause more costs in the future, but due to discounting these costs are relatively low. Contrary to this, the highest number of GFs occurs at the beginning of the model where discounting has a smaller effect.
Even though both treatments were dominant at baseline, we found that the DASH diet resulted in more savings, whereas potassium supplementation resulted in more QALYs. We had expected that an increase in QALYs would result in more savings, as can be seen with the DASH diet. This discrepancy is caused by differences in the transition probabilities of DWFG and GF between the diets. If we compare group 3 of both diets, we notice that the probability of GF is similar but that the probability of DWFG is higher for the DASH diet. This results in lower costs for the DASH diet and more QALYs for potassium supplementation, which seems to explain the fact that the DASH diet results in more savings and potassium supplementation in more QALYs.
It might be expected that the DASH diet and potassium supplementation will result in differences in adherence in practice [
42,
43,
44]. An important reason for this could be related to the fact that it puts a financial burden on the patient as the costs for the DASH diet and potassium supplementation are currently not reimbursed by the Dutch authorities for RTR. This is a commonly seen problem in nutrition economics; the dietary intervention results in a financial burden on the patient whereas the savings mainly occur on the healthcare level. This complicates the interpretation of cost-effectiveness outcomes of dietary interventions. Moreover, although increasing the number of dietician visits from one to eight is expected to increase the adherence, the effect may be disappointing in practice. The DASH diet requires a different approach to cooking which might be challenging for an ageing population.
By dividing the group equally based on their adherence rates, we corrected for the fact that some of the patients do not achieve full adherence after the intervention, and therefore do not achieve maximal health effects. This was further explored by varying the percentage of patients that remained in group 1 after the intervention, that is, when fewer patients follow the diets correctly. Due to the aforementioned reasons, this is a more realistic view of how patients will adhere to the dietary intervention, since dietary adherence has been shown to be difficult to achieve and maintain [
45]. The analyses showed that if the intervention has a positive effect on more than 10% and 20% of the patients in group 1 for the DASH diet and potassium supplementation respectively, the intervention would still be cost-saving.
However, this is based on the assumption that the adherence will remain equal over time. Especially the DASH diet is not easy to maintain [
42]. Moreover, for both the dietary intervention and the potassium supplementation, it was assumed that there was an immediate effect of the intervention, while in reality it could take some time before an effect is achieved. We acknowledge this assumption might not entirely reflect reality and could potentially mitigate the cost-effectiveness results. For future clinical trials or observational cohort studies, it would be interesting to collect data on dietary adherence over time and to model this in a similar way as we did with the survival data.
Next to increasing the number of dietician visits, there are other ways to increase the adherence to diets. Monitoring of food intake has been shown to be a good predictor of dietary adherence, and several types of tools to assist in monitoring have been developed [
46,
47]. Several studies found that patients using a digital monitoring tool were significantly more likely to adhere to their dietary recommendations [
48,
49,
50]. These methods have proven to be successful, but problems often arise due to uncertainty regarding meal size. Recently, an app (LogMeal) was developed based on deep learning and computer vision, which allows easy and objective monitoring of food intake [
24]. The app allows for recognition of food in terms of type of dish, category, and ingredients. The volume of the dish can be estimated, and the micronutrient composition determined. Still, due to the novelty of LogMeal and the limited number of studies regarding long-term dietary adherence, it is unclear how well RTR will adhere to the diet. Our results showed that even with minimal improvements in dietary adherence the treatments remained cost-effective. This can be explained by the relatively low cost of the intervention quickly resulting in a cost-effective treatment.
The PSA showed that the results of our model are robust with 99.7% and 98.8% of simulations being cost-effective for the DASH diet and potassium supplementation, respectively. It is likely that the intervention will either be dominant to the current situation or cost-effective. The DSA showed that for both the DASH diet and potassium supplementation interventions, the parameters with the strongest effect were the probabilities of GF and DWFG, especially for group 1. The change in GF and DWFG probabilities directly affects the number of patients with a FG. Since this is the health state with the highest quality of life and the lowest cost, it is expected that differences in these probabilities will influence the ICER the most. Despite these effects, the interventions stayed either dominant or cost-effective for all parameters.
Evaluating the BIA, we found that the five-year costs of implementing the intervention were relatively low. The main component of the intervention is an increase in visits to the dietician: eight visits of around 30 min each or four hours in total. Dutch health insurance reimburses the first three treatment hours at the dietician for everyone [
51]. This would mean that patients would only have to pay for the last two visits themselves, or they can extend their insurance to also cover this.
4.1. Limitations
To determine the costs of dialysis and transplantation, a study from 1998 was used [
4]. It is likely that over time costs are going to change; however, the study by De Wit et al. is still the most elaborate published costing study regarding renal replacement therapy. It is still being used in recent studies regarding the cost-effectiveness of dialysis and kidney transplantation [
7,
23,
52]. Moreover, based on the knowledge of a Dutch nephrologist, the costs used in our model are still correct.
To determine the transition probabilities in our model, patient level survival data was used. The data was extrapolated and several methods were used to calculate the annual transition probabilities for DWFG and GF [
53]. Based on the advice of a nephrologist, at the end of our extrapolation the probability of dying should be higher. This can be addressed by using long-term survival data of RTR and combining this with the extrapolation data.
In our model, a decision was made against implementing the probability of receiving a pre-emptive re-transplantation. Normally, it is possible that when a patient already has a kidney transplant and this transplant starts failing, instead of first going to dialysis the patient pre-emptively receives a new kidney transplant. However, there is limited data regarding the probability of receiving a pre-emptive re-transplantation. For a normal pre-emptive transplantation in the Netherlands, the probability was 25% in 2017 [
38]. However, it is reasonable to assume that this probability will drop sharply for the second transplant, with one possible reason being that relatives or friends are not able to donate a second kidney. If more data regarding the probability of a pre-emptive re-transplantation were to become available, it would be of interest to implement this in the model.
The number of events/outcomes in the model can be considered limited, which might affect the costs and outcomes of the model. However, we believe including more events would not have changed the general conclusion of the model, but would have increased both the complexity and uncertainty of the model. Since we focussed on the renal graft, for example cardiovascular events were not included in the model. Nevertheless, the DASH diet has shown to reduce the risk of cardiovascular events, and potassium supplementation could also potentially reduce these risks as well [
54,
55]. Therefore, the current outcomes of the model are likely a conservative estimate.
As this model uses retrospective data, the cost-effectiveness is mostly theoretical, since there are no trials comparing dietary adherence with an intervention and control arm.
4.2. Future Perspectives
The next step to be taken is to use RTR long-term survival data to improve the transition probabilities in the model. Doing so will make the model more robust and the results more credible. It is now of interest to look at whether we should focus more on the DASH diet or potassium supplementation. The DASH diet was dominant in all situations, as opposed to potassium supplementation, making it the preferred option. However, the adherence between the two diets could strongly differ: the DASH diet requires a change in diet whereas potassium supplementation only requires a pill. This difference could make the adherence of potassium supplementation much higher, thus making it the preferred option. It is also of interest to look at the combined effect of the DASH diet and potassium supplementation, because these two interventions could theoretically be combined into one dietary recommendation. This most likely would result in even more QALYs gained and costs saved. It is now necessary to investigate in a patient population how well patients will adhere to dietary interventions and to what degree their health outcomes will improve. This model can be used to justify those head-to-head studies, and it can be used to advocate for RTR receiving more visits to the dietician.
Moreover, as mentioned before, the long-term adherence to the DASH diet and potassium supplementation is uncertain. Therefore, a head-to-head trial could be used to investigate dietary adherence. Such trial could also possibly be used to support reimbursement of the DASH diet and potassium supplementation for RTR, so that the financial burden of the diet does not solely come down to the patient. It would be interesting to do a second cost-effectiveness analysis which could include adherence over time data.