Growth and Physiological Adaptation of Salix matsudana Koidz. to Periodic Submergence in the Hydro-Fluctuation Zone of the Three Gorges Dam Reservoir of China
Abstract
:1. Introduction
2. Materials and Methods
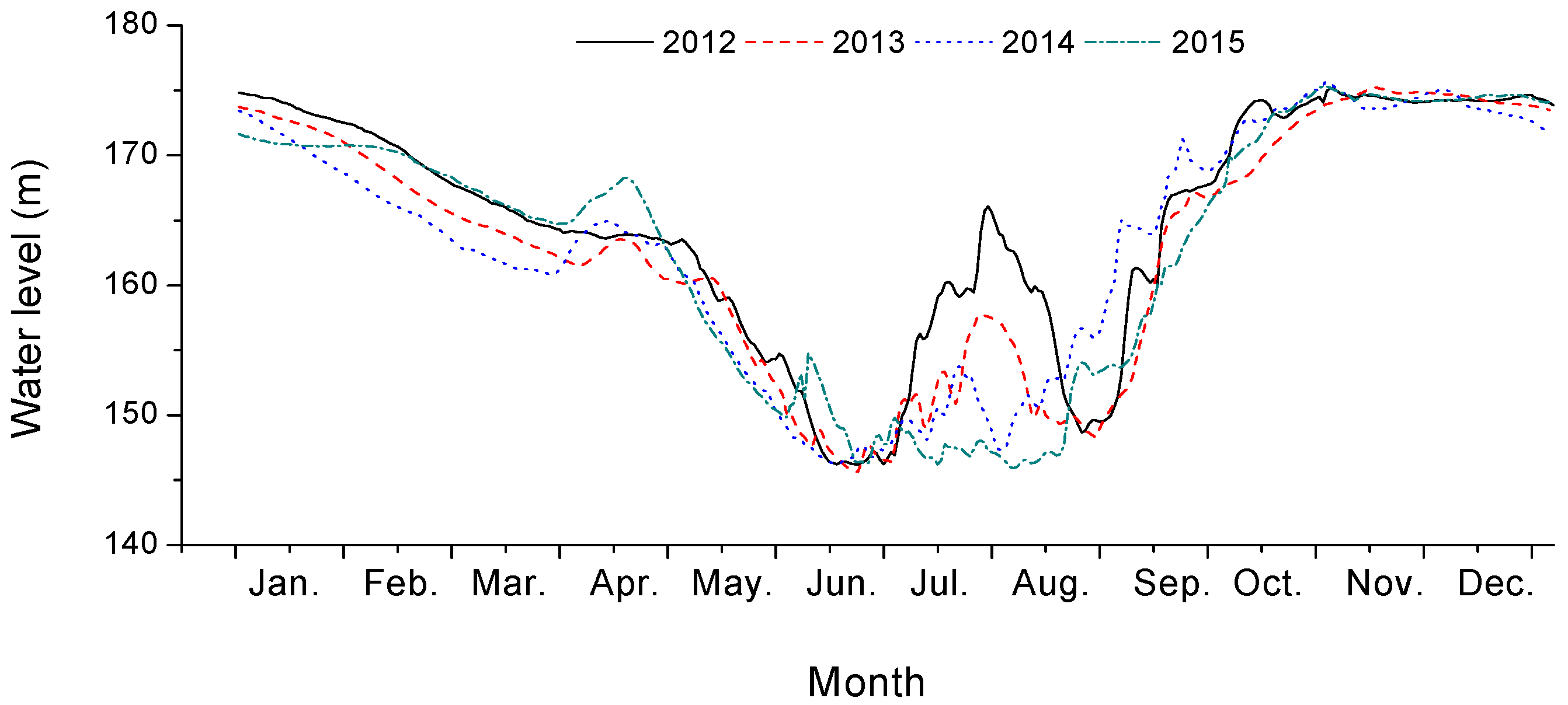
2.1. Research Site Description
2.2. Experimental Design
2.3. Measurement of Physiological Responses
2.4. Measurement of Plant Growth
2.5. Data Analysis
3. Results
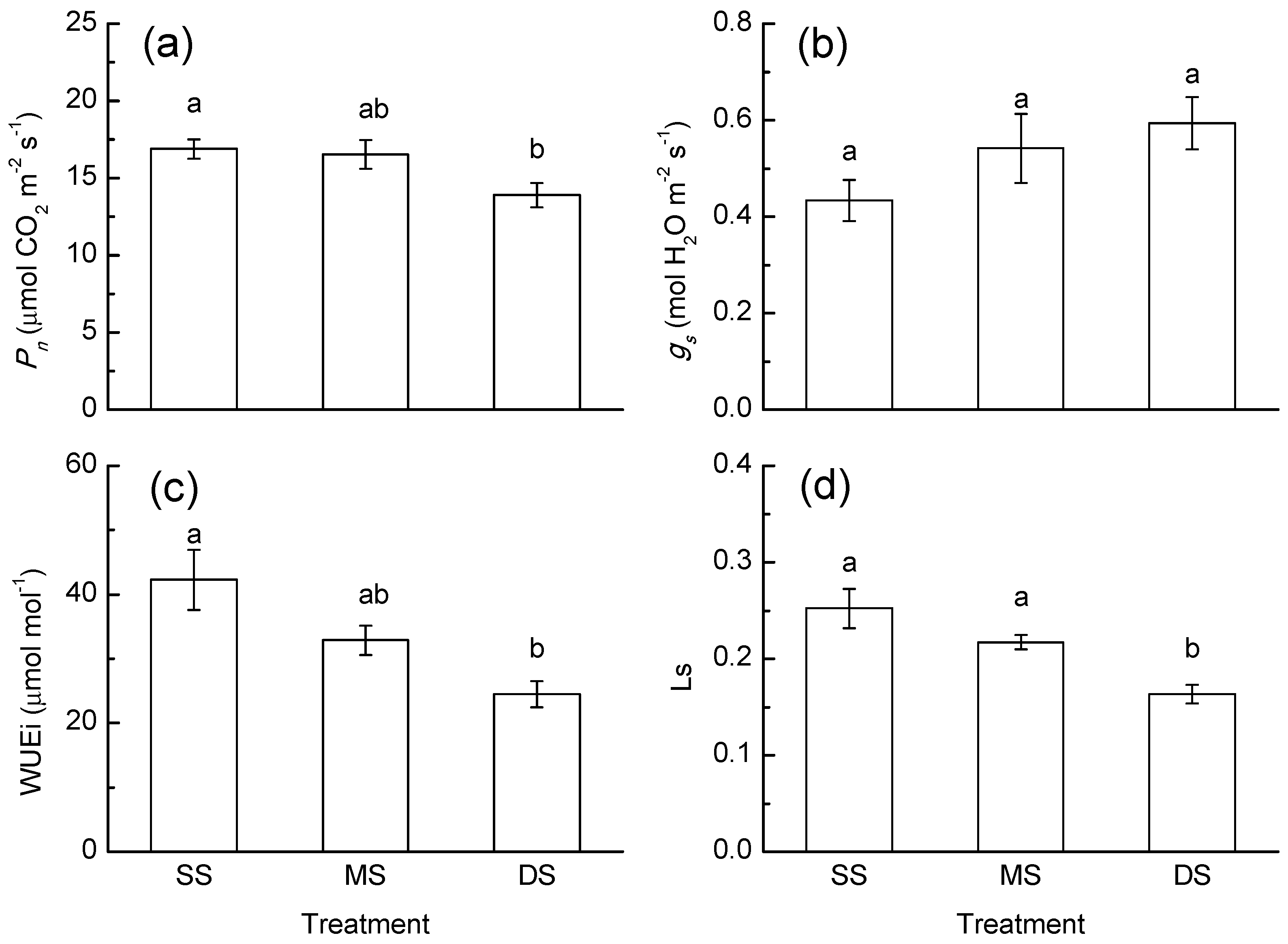
3.1. Gas Exchange Response
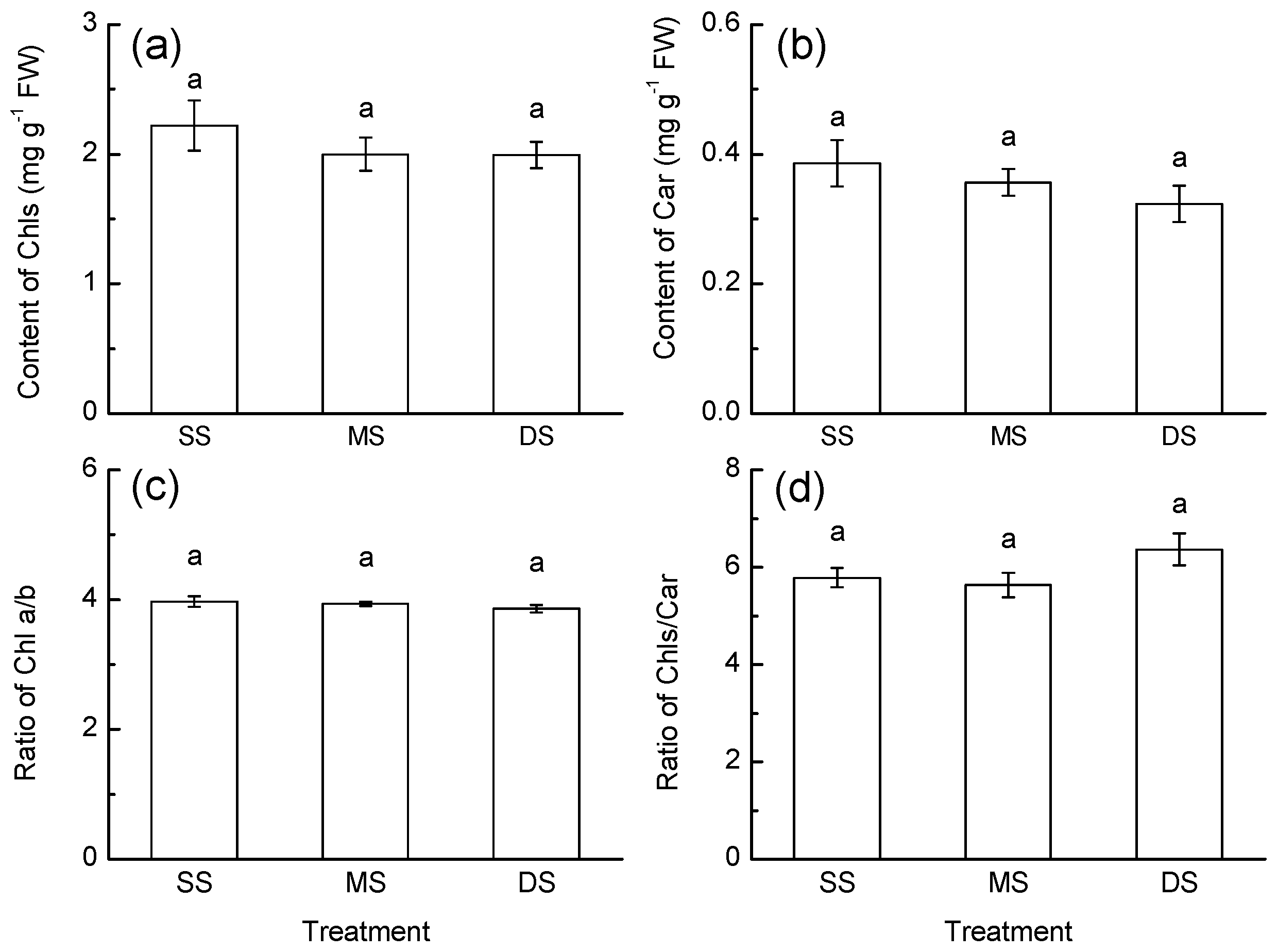
3.2. Pigment Content
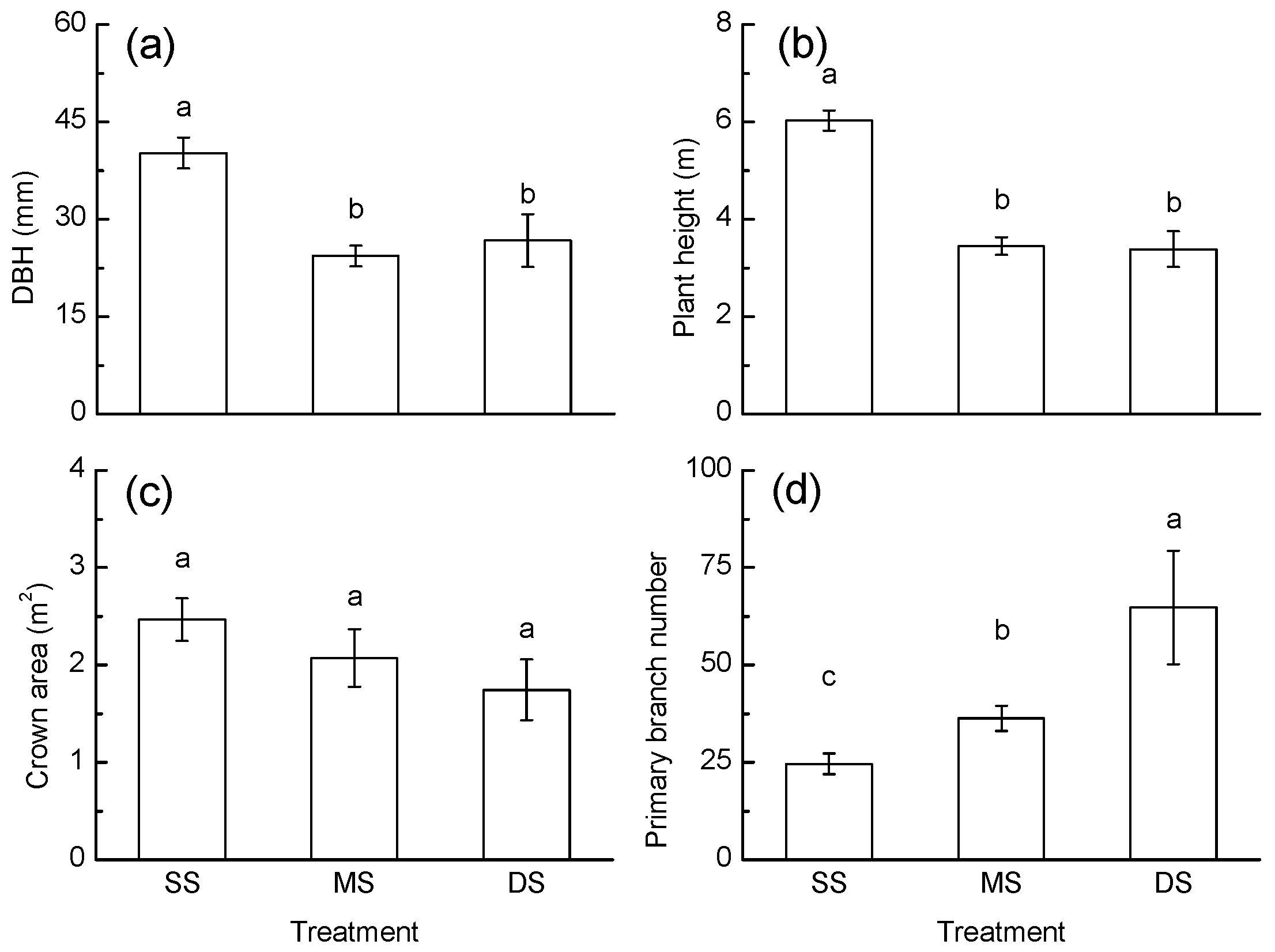
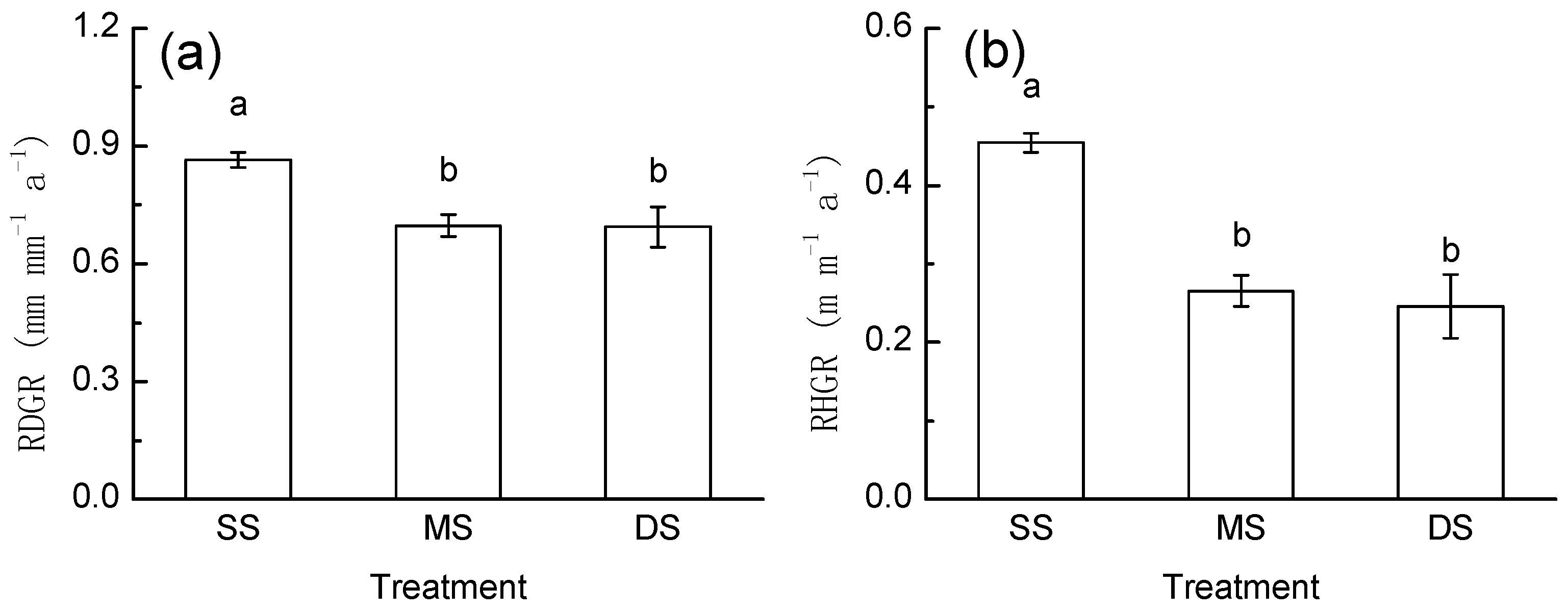
3.3. Growth Response
3.4. Correlation
4. Discussion
4.1. Photosynthesis Response to Periodic Submergence
4.2. Growth Response to Periodic Submergence
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xu, X.B.; Tan, Y.; Yang, G.S.; Li, H.P.; Su, W.Z. Soil erosion in the Three Gorges Reservoir area. Soil Res. 2011, 49, 212–222. [Google Scholar] [CrossRef]
- Ye, C.; Li, S.Y.; Zhang, Y.R.; Zhang, Q.F. Assessing soil heavy metal pollution in the water-level-fluctuation zone of the Three Gorges Reservoir, China. J. Hazard. Mater. 2011, 191, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Yuan, J.; Sun, X.; Zhang, M. Spatial distribution and release of nitrogen in soils in the water fluctuation zone of the Three Gorges Reservoir. J. Food Agric. Environ. 2012, 10, 787–791. [Google Scholar]
- Subklew, G.; Ulrich, J.; Fürst, L.; Höltkemeier, A. Environmental impacts of the Yangtze Three Gorges project: An overview of the Chinese-German research cooperation. J. Earth Sci. 2010, 21, 817–823. [Google Scholar] [CrossRef]
- Fan, D.Y.; Xiong, G.M.; Zhang, A.Y.; Liu, X.; Xie, Z.Q.; Li, Z.J. Effect of water-lever regulation on species selection for ecological restoration practice in the water-level fluctuation zone of Three Gorges Reservoir. Chin. J. Plant Ecol. 2015, 39, 416–432. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, F.; Wu, Y.; Ma, Y.; Guan, S.; Huang, Y. Characteristics of the soil seed bank of planted and natural restored draw-down zones in the Three Gorges Reservoir region. Ecol. Eng. 2017, 103, 127–133. [Google Scholar] [CrossRef]
- Langer, E.R.; Steward, G.A.; Kimberley, M.O. Vegetation structure, composition and effect of pine plantation harvesting on riparian buffers in New Zealand. For. Ecol. Manag. 2008, 256, 949–957. [Google Scholar] [CrossRef]
- Chazdon, R.L. Beyond deforestation: Restoring forests and ecosystem services on degraded lands. Science 2008, 320, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.D.; Edwards, S.L.; Demarais, S.; Ezell, A.W. Vegetation community responses to different establishment regimes in loblolly pine (Pinus taeda) plantations in southern MS, USA. For. Ecol. Manag. 2009, 257, 553–560. [Google Scholar] [CrossRef]
- Azza, N.; Denny, P.; van de Koppel, J. Floating mats: Their occurrence and influence on shoreline distribution of emergent vegetation. Freshw. Biol. 2006, 51, 1286–1297. [Google Scholar] [CrossRef]
- Deletic, A. Sediment transport in urban runoff over grassed areas. J. Hydrol. 2005, 301, 108–122. [Google Scholar] [CrossRef]
- Bardgett, R.D.; van der Wal, R.; Jonsdottir, I.S.; Quirk, H.; Dutton, S. Temporal variability in plant and soil nitrogen pools in a High-Arctic ecosystem. Soil Biol. Biochem. 2007, 39, 2129–2137. [Google Scholar] [CrossRef]
- Dunchemin, M.; Hogue, R. Reduction in agricultural non-point source pollution in the first year following establishment of an integrated grass/tree filter strip system in Southern Quebec (Canada). Agric. Ecosyst. Environ. 2009, 131, 85–97. [Google Scholar] [CrossRef]
- Mudd, S.M.; Alpaos, A.D.; Morris, J.T. How does vegetation affect sedimentation on tidal marshes? Investigating particle capture and hydrodynamic controls on biologically mediated sedimentation. J. Geophys. Res. 2010, 115, F03029. [Google Scholar] [CrossRef]
- Ye, C.; Cheng, X.L.; Zhang, Y.L.; Wang, Z.X.; Zhang, Q.F. Soil nitrogen dynamics following short-term revegetation in the water level fluctuation zone of the Three Gorges Reservoir, China. Ecol. Eng. 2012, 38, 37–44. [Google Scholar] [CrossRef]
- Li, C.X.; Wei, H.; Geng, Y.H.; Schneider, R. Effects of submergence on photosynthesis and growth of Pterocarya stenoptera (Chinese wingnut) seedlings in the recently-created Three Gorges Reservoir Region of China. Wetl. Ecol. Manag. 2010, 18, 485–494. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.F.; Liu, S.B.; Huang, H.W. Vegetation reconstruction in the water-level-fluctuation zone of the Three Gorges Reservoir. Chin. Bull. Bot. 2005, 22, 513–522. [Google Scholar]
- Li, C.X.; Zhong, Z.C.; Liu, Y. Effect of soil water change on photosynthetic characteristics of Taxodium distichum seedlings in the hydro-fluctuation belt of the Three Gorges Reservoir Area. Acta Ecol. Sin. 2005, 25, 23–28. [Google Scholar] [CrossRef]
- Li, C.X.; Zhong, Z.C.; Geng, Y.H.; Schneider, R. Comparative studies on physiological and biochemical adaptation of Taxodium distichum and Taxodium ascendens seedlings to different soil water regimes. Plant Soil 2010, 329, 481–494. [Google Scholar] [CrossRef]
- Lei, S.T.; Zeng, B.; Xu, S.J.; Su, X.L. Membrane responses of Salix variegata and Cinnamomum camphora to complete submergence in the Three Gorges Reservoir Region. Acta Ecol. Sin. 2012, 32, 227–231. [Google Scholar] [CrossRef]
- Kawano, N.; Ella, E.; Ito, O.; Yamauchi, Y.; Tanaka, K. Metabolic changes in rice seedlings with different submergence tolerance after desubmergence. Environ. Exp. Bot. 2002, 47, 195–203. [Google Scholar] [CrossRef]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Crawford, R.M.M. Seasonal differences in plant responses to flooding and anoxia. Can. J. Bot. 2003, 81, 1224–1226. [Google Scholar] [CrossRef]
- Panda, D.; Sarkar, R.K.; Sharma, S.G. Chlorophyll fluorescence parameters, CO2, photosynthetic rate and regeneration capacity as a result of complete submergence and subsequent re-emergence in rice (Oryza sativa L.). Aquat. Bot. 2008, 88, 127–133. [Google Scholar] [CrossRef]
- Striker, G.G. Time is on our side: The importance of considering a recovery period when assessing flooding tolerance in plants. Ecol. Res. 2012, 27, 983–987. [Google Scholar] [CrossRef]
- Colmer, T.D. Long-distance transport of gases in plants: A perspective on internal aeration and radial oxygen loss from roots. Plant Cell Environ. 2003, 26, 17–36. [Google Scholar] [CrossRef]
- Schaff, S.D.; Pezeshki, S.R.; Shields, F.D., Jr. Effects of soil conditions on survival and growth of black willow cuttings. Environ. Manag. 2003, 31, 748–763. [Google Scholar] [CrossRef] [PubMed]
- Kuzovkina, Y.A.; Quigley, M.F. Willows beyond wetlands: Uses for Salix L. species for environmental projects. Water Air Soil Pollut. 2005, 162, 183–204. [Google Scholar] [CrossRef]
- Tallenthalsell, N.G.; Walker, L.R. Responses of Salix gooddingii and Tamarix ramosissima to flooding. Wetlands 2002, 22, 776–785. [Google Scholar] [CrossRef]
- Higa, M.; Moriyama, T.; Ishikawa, S. Effects of complete submergence on seedling growth and survival of five riparian tree species in the warm-temperate regions of Japan. J. For. Res. 2012, 17, 129–136. [Google Scholar] [CrossRef]
- Kuzovkina, Y.A.; Knee, M.; Quigley, M.F. Effects of soil compaction and flooding on the growth of 12 willow (Salix L.) species. J. Environ. Hortic. 2004, 22, 155–160. [Google Scholar]
- Li, S.; Pezeshki, S.R.; Goodwin, S.; Shields, F.D., Jr. Physiological responses of black willow (Salix nigra) cuttings to a range of soil moisture regimes. Photosynthetica 2004, 42, 585–590. [Google Scholar] [CrossRef]
- Li, S.; Goodwin, S.; Pezeshki, S.R. Photosynthetic gene expression in black willow under various soil moisture regimes. Biol. Plant. 2007, 51, 593–596. [Google Scholar] [CrossRef]
- Waldhoff, D. Leaf structure in trees of central Amazonian floodplain forests (Brazil). Amazoniana 2003, 17, 451–469. [Google Scholar]
- Li, S.; Pezeshki, S.R.; Shields, F.D., Jr. Partial flooding enhances aeration in adventitious roots of black willow (Salix nigra) cuttings. J. Plant Physiol. 2006, 163, 619–628. [Google Scholar] [CrossRef] [PubMed]
- Nakai, A.; Kisanuki, H. Stress responses of Salix gracilistyla, and Salix subfragilis cuttings to repeated flooding and drought. J. For. Res. 2011, 16, 465–472. [Google Scholar] [CrossRef]
- Markus-Michalczyk, H.; Hanelt, D.; Kai, J. Effects of tidal flooding on juvenile willows. Estuar. Coasts 2016, 39, 1–9. [Google Scholar] [CrossRef]
- Zhong, Y.; Liu, Z.X.; Qin, H.W.; Xiong, Y.; Xiang, L.X.; Liu, R.; Yang, Y.; Ma, R. Effects of winter submergence and waterlogging on growth and recovery growth of Salix babylonica. J. South. Agric. 2013, 44, 275–279. [Google Scholar] [CrossRef]
- Zhang, S.S.; Li, R.N.; Wang, W.; Lu, W.Y.; Ren, Y.; Fu, S.L. Tree species selection in the riparian zone of Meishan reservoir. J. Anhui Agric. Univ. 2015, 42, 357–362. [Google Scholar]
- Tang, J.L.; Wang, T.; Zhu, B.; Zhao, P.; Xiao, Y.; Wang, R. Tempo-spatial analysis of water quality in tributary bays of the Three Gorges Reservoir region (China). Environ. Sci. Pollut. Res. 2015, 22, 16709–16720. [Google Scholar] [CrossRef] [PubMed]
- Pezeshki, S.R.; Li, S.W.; Shields, F.D., Jr.; Martina, L.T. Factors governing survival of black willow (Salix nigra) cuttings in a streambank restoration project. Ecol. Eng. 2007, 29, 56–65. [Google Scholar] [CrossRef]
- Berry, J.A.; Downton, W.J.S. Environmental regulation of photosynthesis. In Photosynthesis; Govind, J., Ed.; Academic Press: New York, NY, USA, 1982; Volume 2, pp. 263–343. [Google Scholar] [CrossRef]
- Li, C.X.; Geng, Y.H.; Ye, B.; Schneider, R. Response of baldcypress and pondcypress seedlings to multiple stresses and reforestation implication for riparian zone of the Three Gorges Reservoir region of China. Sci. Silv. Sin. 2010, 46, 144–152. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Masaka, K.; Sato, H.; Kon, H.; Fukuchi, M. Demographic and height growth response of native broad-leaved deciduous tree saplings to overhead canopy release in a coastal Pinus thunbergii forest in Hokkaido, northern Japan. J. For. Res. 2012, 17, 421–431. [Google Scholar] [CrossRef]
- Colmer, T.D.; Voesenek, L.A.C.J. Flooding tolerance: Suites of plant traits in variable environments. Funct. Plant Biol. 2009, 36, 665–681. [Google Scholar] [CrossRef]
- Gu, X.; Li, Z.Q.; Jiang, C.D.; Shi, L.; Zhang, H.J.; Xing, Q. Effects of submergence and subsequent re-emergence on photosynthetic characteristics and photoinhibition in Echinodorus amazonicus seedlings. Acta Ecol. Sin. 2009, 29, 6466–6474. [Google Scholar]
- Sharma, P.; Asaeda, T.; Kalibbala, M.; Fujino, T. Morphology, growth and carbohydrate storage of the plant Typha angustifolia at different water depths. Chem. Ecol. 2008, 24, 133–145. [Google Scholar] [CrossRef]
- Sharma, P.; Asaeda, T.; Fujino, T. Effect of water depth on the rhizome dynamics of Typha angustifolia. Wetl. Ecol. Manag. 2008, 16, 43–49. [Google Scholar] [CrossRef]
- Luo, F.L.; Nagel, K.A.; Zeng, B.; Schurr, U.; Matsubara, S. Photosynthetic acclimation is important for post-submergence recovery of photosynthesis and growth in two riparian species. Ann. Bot. 2009, 104, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Glenz, C.; Schlaepfer, R.; Iorgulescu, I.; Kienast, F. Flooding tolerance of central European tree and shrub species. For. Ecol. Manag. 2006, 235, 1–13. [Google Scholar] [CrossRef]
- Kissmann, C.; Veiga, E.B.D.; Eichemberg, M.T.; Habermann, G. Morphological effects of flooding on Styrax pohlii and the dynamics of physiological responses during flooding and post-flooding conditions. Aquat. Bot. 2014, 119, 7–14. [Google Scholar] [CrossRef]
- Jing, Y.X.; Li, G.L.; Gu, B.H.; Yang, D.J.; Xiao, L.; Liu, R.X.; Peng, C.L. Leaf gas exchange, chlorophyll fluorescence and growth responses of Melaleuca alternifolia, seedlings to flooding and subsequent recovery. Photosynthetica 2009, 47, 595–601. [Google Scholar] [CrossRef]
- Rodríguez, M.E.; Achinelli, F.G.; Luquez, V.M.C. Leaf traits related to productivity in Populus deltoides during the post-flooding period. Trees 2015, 29, 953–960. [Google Scholar] [CrossRef]
- Parolin, P. Submerged in darkness: Adaptations to prolonged submergence by woody species of the Amazonian floodplains. Ann. Bot. 2009, 103, 359–376. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.F.; Zhao, Y.; Zhang, C.; Tao, X.; Xu, X.N. Growth, leaf gas exchange, and chlorophyll fluorescence responses of two cultivars of Salix integra Thunb. to waterlogging stress. J. Agric. Sci. Technol. 2014, 16, 137–149. [Google Scholar]
- Farquhar, G.D.; Sharkey, T.D. Stomatal conductance and photosynthesis. Annu. Rev. Plant Physiol. 1982, 33, 317–345. [Google Scholar] [CrossRef]
- Herrera, A.; Tezara, W.; Marín, O.; Rengifo, E. Stomatal and non-stomatal limitations of photosynthesis in trees of a tropical seasonally flooded forest. Physiol. Plant 2008, 134, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Larcher, W. Physiological Plant Ecology, 4th ed.; Springer: Berlin, Germany, 2003. [Google Scholar]
- Tracewell, C.A.; Vrettos, J.S.; Bautista, J.A.; Frank, H.A.; Brudvig, G.W. Carotenoid photooxidation in photosystem II. Arch. Biochem. Biophys. 2001, 385, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, C.R.; Britz, S.J. Effect of supplemental ultraviolet radiation on the carotenoid and chlorophyll composition of green-house leaf lettuce (Lactuca sativa L.) cultivars. J. Food Compos. Anal. 2006, 19, 637–644. [Google Scholar] [CrossRef]
- Yang, F.; Wang, Y.; Chan, Z. Perspectives on screening winter-flood-tolerant woody species in the riparian protection forests of the Three Gorges Reservoir. PLoS ONE 2014, 9, e108725. [Google Scholar] [CrossRef] [PubMed]
- Miao, L.F.; Xiao, F.J.; Xu, W.; Yang, F. Reconstruction of wetland zones: Physiological and biochemical responses of Salix variegata to winter submergence-a case study from water level fluctuation zone of the Three Gorges Reservoir. Pol. J. Ecol. 2016, 64, 45–52. [Google Scholar] [CrossRef]
- Mommer, L.; Lenssen, J.P.M.; Huber, H.; Visser, E.J.W.; Kroon, H.D. Ecophysiological determinants of plant performance under flooding: A comparative study of seven plant families. J. Ecol. 2006, 94, 1117–1129. [Google Scholar] [CrossRef]
- Berthelot, J.S.; Saint-Laurent, D.; Gervais-Beaulac, V.; Présent, A. A comparison of the composition and diversity of tree populations along a hydrological gradient in floodplains (southern Québec, Canada). Forests 2015, 6, 929–956. [Google Scholar] [CrossRef]
- Iwanaga, F.; Tanaka, K.; Nakazato, I.; Yamamoto, F. Effects of submergence on growth and survival of saplings of three wetland trees differing in adaptive mechanisms for flood tolerance. For. Syst. 2015, 24, 1–9. [Google Scholar] [CrossRef]
- Voesenek, L.A.C.J.; Colmer, T.D.; Pierik, R.; Millenaar, F.F.; Peeters, A.J.M. How plants cope with complete submergence. New Phytol. 2006, 170, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Vartapetian, B.B.; Generosova, I.P.; Zakhmylova, N.A.; Snkhchyan, A.G. Demonstration of plant adaptation syndrome in plants and possible molecular mechanisms of its realization under conditions of anaerobic stress. Russ. J. Plant Physiol. 2006, 53, 747–755. [Google Scholar] [CrossRef]
- Parolin, P. Submergence tolerance vs. escape from submergence: Two strategies of seedling establishment in Amazonian floodplains. Environ. Exp. Bot. 2002, 48, 177–186. [Google Scholar] [CrossRef]
- Tang, Z.C.; Kozlowski, T.T. Responses of Pinus banksiana and Pinus resinosa seedlings to flooding. Can. J. For. Res. 1983, 13, 633–639. [Google Scholar] [CrossRef]
- Gibbs, J.; Greenway, H. Mechanism of anoxia tolerance in plants. I. Growth, survival and anaerobic catabolism. Funct. Plant Biol. 2003, 30, 1–47. [Google Scholar] [CrossRef]
- Li, X.L.; Luan, C.Y.; Yang, J.; Chen, F.J. Survival and recovery growth of riparian plant Distylium chinense seedlings to complete submergence in the Three Gorges Reservoir region. Procedia Eng. 2012, 28, 85–94. [Google Scholar] [CrossRef]
- Ding, X.; Zou, J.; Li, Y.; Yao, X.; Zou, D.; Zhang, C.; Yang, N.; Niu, Y.; Bian, H.; Deng, J.; et al. Acclimation of Salix triandroides, cuttings to incomplete submergence is reduced by low light. Aquat. Ecol. 2017, 51, 321–330. [Google Scholar] [CrossRef]
- Dionigi, C.P.; Mendelssohn, I.A.; Sullivan, V.I. Effects of soil waterlogging on the energy status and distribution of Salix nigra and S. Exigua (Salicaceae) in the Atchafalaya river basin of Louisiana. Am. J. Bot. 1985, 72, 109–119. [Google Scholar] [CrossRef]
| Treatment | Elevation (Submergence Depth ‡) (m) | Submergence Duration (Exposure Duration §) (day) | ||
|---|---|---|---|---|
| From July 2012 to June 2013 | From July 2013 to June 2014 | From July 2014 to June 2015 | ||
| DS | 165 (10) | 175 (190) | 158 (207) | 217 (148) |
| MS | 170 (5) | 125 (240) | 101 (264) | 141 (224) |
| SS | 175 (0) | 2 (363) | 5 (360) | 8 (357) |
| Indexes | DBH ‡ | TH | CA | PBN |
|---|---|---|---|---|
| Pn | 0.72 *** | 0.588 ** | 0.49 ** | −0.291 |
| gs | 0.158 | −0.048 | 0.298 | 0.095 |
| WUEi | 0.312 | 0.418 * | 0.028 | −0.301 |
| Ls | 0.377 | 0.46 * | 0.095 | −0.352 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Xie, Y.; He, Y.; Li, X.; Yang, W.; Li, C. Growth and Physiological Adaptation of Salix matsudana Koidz. to Periodic Submergence in the Hydro-Fluctuation Zone of the Three Gorges Dam Reservoir of China. Forests 2017, 8, 283. https://doi.org/10.3390/f8080283
Wang C, Xie Y, He Y, Li X, Yang W, Li C. Growth and Physiological Adaptation of Salix matsudana Koidz. to Periodic Submergence in the Hydro-Fluctuation Zone of the Three Gorges Dam Reservoir of China. Forests. 2017; 8(8):283. https://doi.org/10.3390/f8080283
Chicago/Turabian StyleWang, Chaoying, Yingzan Xie, Yanyan He, Xiaoxue Li, Wenhang Yang, and Changxiao Li. 2017. "Growth and Physiological Adaptation of Salix matsudana Koidz. to Periodic Submergence in the Hydro-Fluctuation Zone of the Three Gorges Dam Reservoir of China" Forests 8, no. 8: 283. https://doi.org/10.3390/f8080283





