Carbon Stocks across a Fifty Year Chronosequence of Rubber Plantations in Tropical China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site and Plot Establishment
2.2. Field Sampling and Measurements
2.3. Statistical Analysis
3. Results
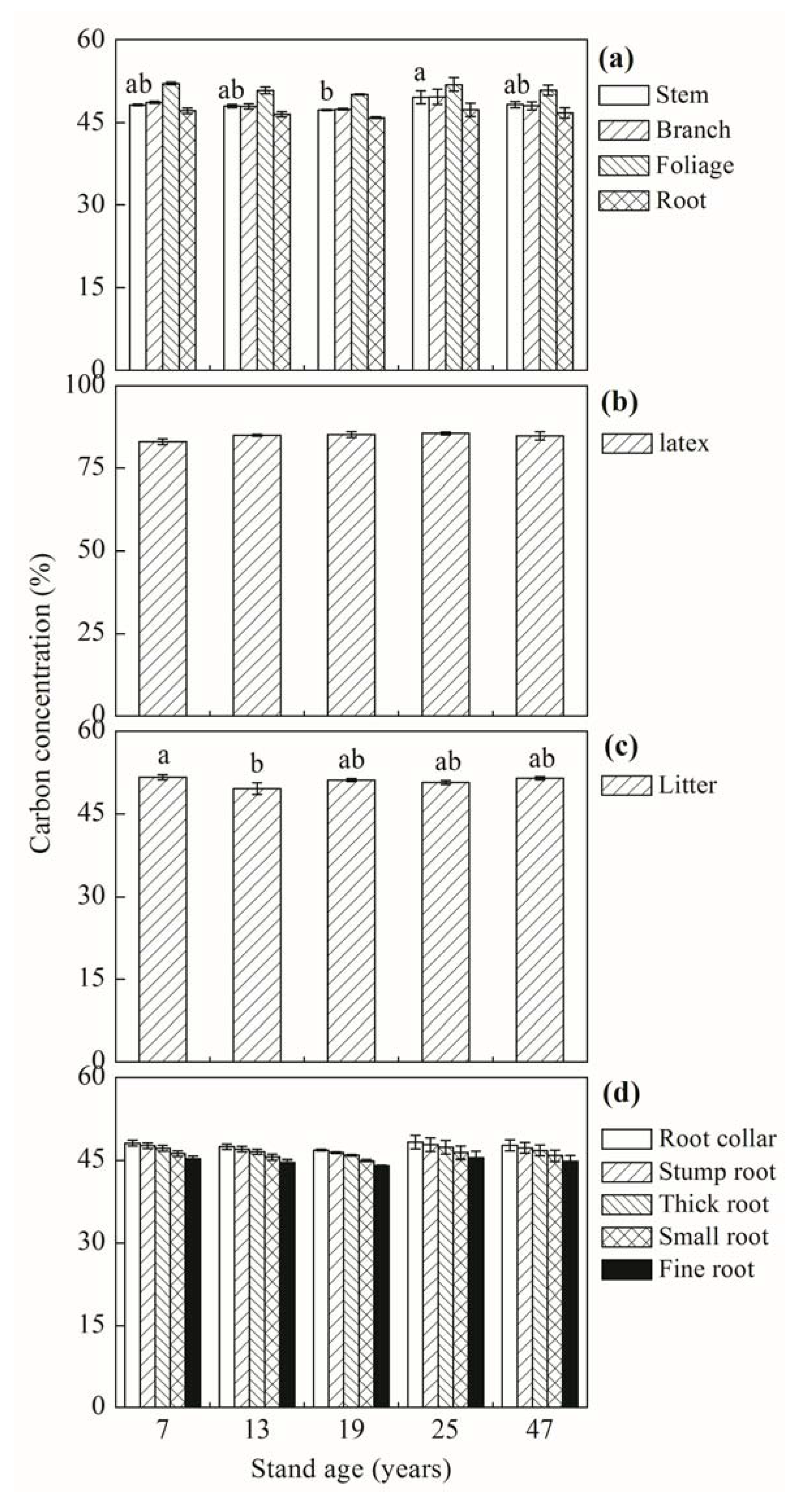
3.1. Biomass C Concentration and Stock
3.2. Biomass NPP and Associated C Sequestration
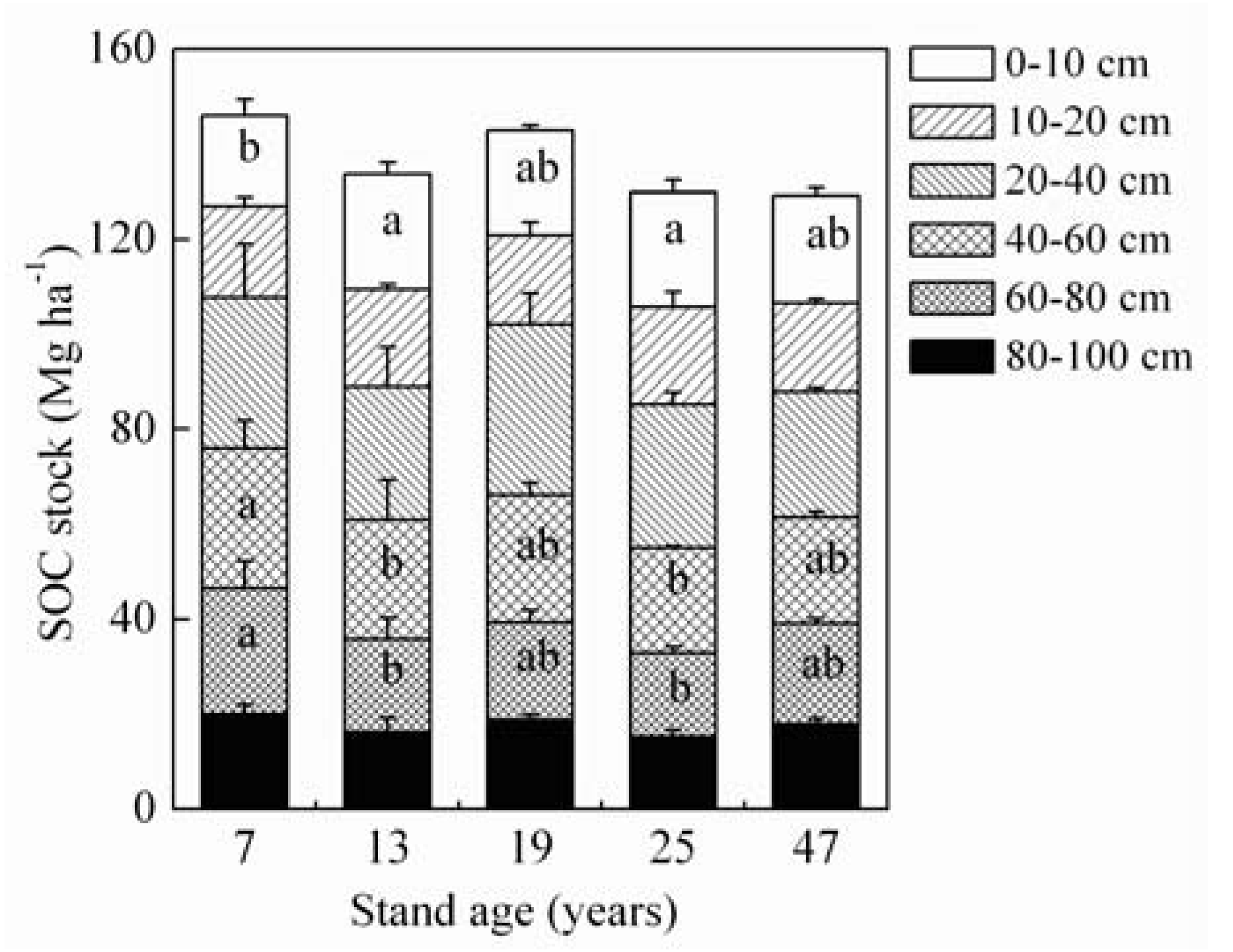
3.3. SOC Concentration and Stock
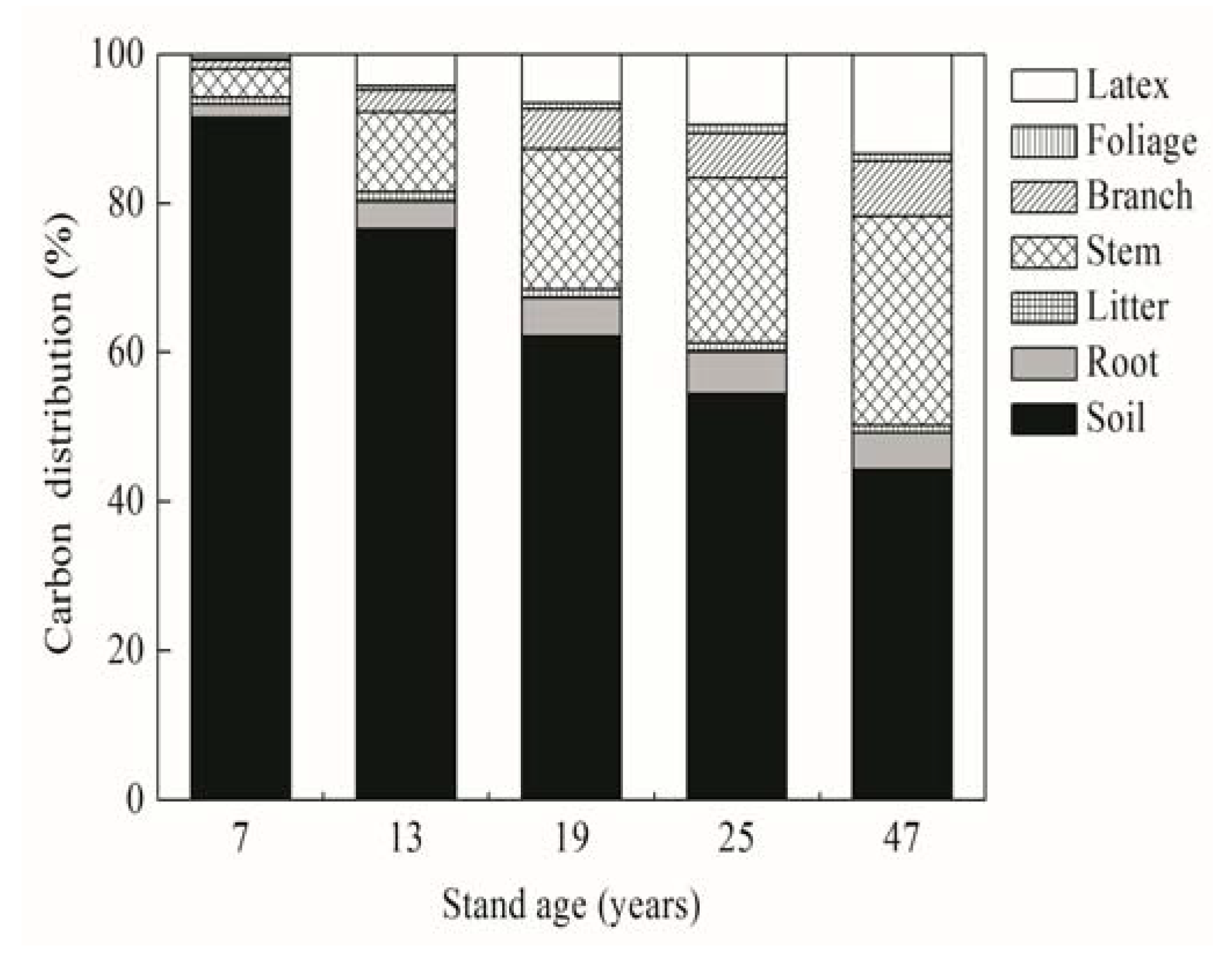
3.4. Ecosystem C Stock and Its Relationship between Main Compartments
4. Discussion
4.1. Biomass C Accumulation
4.2. Soil C Stock
4.3. Ecosystem C Stock
4.4. The Implications of Rubber Plantation on C Sequestration
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cheng, J.Z.; Lee, X.Q.; Theng, B.K.G.; Zhang, L.K.; Fang, B.; Li, F.S. Biomass accumulation and carbon sequestration in an age-sequence of Zanthoxylum bungeanum plantations under the Grain for Green Program in karst regions, Guizhou province. Agric. For. Meteorol. 2015, 203, 88–95. [Google Scholar] [CrossRef]
- Nogueira, E.M.; Yanai, A.M.; Fonseca, F.O.; Fearnside, P.M. Carbon stock loss from deforestation through 2013 in Brazilian Amazonia. Glob. Chang. Biol. 2015, 21, 1271–1292. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.D.; Birdsey, R.A.; Fang, J.Y.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.C.; Potapov, P.V.; Moore, R.; Hancher, M.; Turubanova, S.A.; Tyukavina, A.; Thau, D.; Stehman, S.V.; Goetz, S.J.; Loveland, T.R. High-resolution global maps of 21st-century forest cover change. Science 2013, 342, 850–853. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.; Castella, J.C.; Ziegler, A.D.; Westley, S.B. Rubber plantations expand in mountainous Southeast Asia: What are the consequences for the environment? AsiaPacific Issues 2014, 114, 1–8. [Google Scholar]
- Zhang, L.; Kono, Y.; Kobayashi, S.; Hu, H.B.; Zhou, R.; Qin, Y. The expansion of smallholder rubber farming in Xishuangbanna, China: A case study of two Dai villages. Land Use Policy 2015, 42, 628–634. [Google Scholar] [CrossRef]
- Li, H.M.; Ma, Y.X.; Aide, T.M.; Liu, W.J. Past, present and future land-use in Xishuangbanna, China and the implications for carbon dynamics. For. Ecol. Manag. 2008, 255, 16–22. [Google Scholar] [CrossRef]
- Van Straaten, O.; Corre, M.D.; Wolf, K.; Tchienkoua, M.; Cuellar, E.; Matthews, R.B.; Veldkamp, E. Conversion of lowland tropical forests to tree cash crop plantations loses up to one-half of stored soil organic carbon. Proc. Natl. Acad. Sci. USA 2015, 112, 9956–9960. [Google Scholar] [CrossRef] [PubMed]
- De Blécourt, M.; Brumme, R.; Xu, J.C.; Corre, M.D.; Veldkamp, E. Soil carbon stocks decrease following conversion of secondary forests to rubber (Hevea brasiliensis) plantations. PloS ONE 2013, 8, e69357. [Google Scholar] [CrossRef] [PubMed]
- Kotowska, M.M.; Leuschner, C.; Triadiati, T.; Meriem, S.; Hertel, D. Quantifying above- and belowground biomass carbon loss with forest conversion in tropical lowlands of Sumatra (Indonesia). Glob. Chang. Biol. 2015, 21, 3620–3634. [Google Scholar] [CrossRef] [PubMed]
- De Blécourt, M.; Hänsel, V.M.; Brumme, R.; Corre, M.D.; Veldkamp, E. Soil redistribution by terracing alleviates soil organic carbon losses caused by forest conversion to rubber plantation. For. Ecol. Manag. 2014, 313, 26–33. [Google Scholar] [CrossRef]
- Kobayashi, N.; Kumagai, T.O.; Miyazawa, Y.; Matsumoto, K.; Tateishi, M.; Lim, T.K.; Mudd, R.G.; Ziegler, A.D.; Giambelluca, T.W.; Yin, S. Transpiration characteristics of a rubber plantation in central Cambodia. Tree Physiol. 2014, 34, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Uri, V.; Varik, M.; Aosaar, J.; Kanal, A.; Kukumägi, M.; Lõhmus, K. Biomass production and carbon sequestration in a fertile silver birch (Betula pendula Roth) forest chronosequence. For. Ecol. Manag. 2012, 267, 117–126. [Google Scholar] [CrossRef]
- Li, T.J.; Ren, B.W.; Wang, D.H.; Liu, G.B. Spatial variation in the storages and age-related dynamics of forest carbon sequestration in different climate zones–evidence from Black Locust plantations on the Loess Plateau of China. PLoS ONE 2015, 10, e0121862. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.H.; Luo, Y.Q.; Finzi, A.C. Carbon and nitrogen dynamics during forest stand development: A global synthesis. New Phytol. 2011, 190, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Khasanah, N.; van Noordwijk, M.; Ningsih, H.; Rahayu, S. Carbon neutral? No change in mineral soil carbon stock under oil palm plantations derived from forest or non-forest in Indonesia. Agric. Ecosyst. Environ. 2015, 211, 195–206. [Google Scholar] [CrossRef]
- Dey, S.K. A preliminary estimation of carbon stock sequestrated through rubber (Hevea brasiliensis) plantation in north eastern region of India. Indian For. 2005, 131, 1429–1436. [Google Scholar]
- Wauters, J.; Coudert, S.; Grallien, E.; Jonard, M.; Ponette, Q. Carbon stock in rubber tree plantations in Western Ghana and Mato Grosso (Brazil). For. Ecol. Manag. 2008, 255, 2347–2361. [Google Scholar] [CrossRef]
- Saengruksawong, C.; Khamyong, S.; Anongrak, N.; Pinthong, J. Growths and carbon stocks of para rubber plantations on Phonpisai Soil Series in Northeastern Thailand. Rubber Thai J. 2012, 1, 1–18. [Google Scholar]
- Maggiotto, S.R.; Oliveira, D.D.; Marur, C.J.; Stivari, S.M.S.; Leclerc, M.; Wagner-Riddle, C. Potential carbon sequestration in rubber tree plantations in the northwestern region of the Paraná State, Brazil. Acta Sci. Agron. 2014, 36, 239–245. [Google Scholar] [CrossRef]
- Nizami, S.M.; Zhang, Y.P.; Sha, L.Q.; Zhao, W.; Zhang, X. Managing carbon sinks in rubber (Hevea brasilensis) plantation by changing rotation length in SW China. PLoS ONE 2014, 9, e115234. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.W.; Pang, J.P.; Chen, M.Y.; Guo, X.M.; Zeng, R. Biomass and its estimation model of rubber plantations in Xishuangbanna, Southwest China. Chin. J. Ecol. 2009, 28, 1942–1948. [Google Scholar]
- Yang, J.C.; Huang, J.H.; Tang, J.W.; Pan, Q.M.; Han, X.G. Carbon sequestration in rubber tree plantations established on former arable lands in Xishuangbanna, SW China. Chin. J. Plant Ecol. 2005, 29, 296–303. [Google Scholar]
- Cotta, M.K.; Jacovine, L.A.G.; Valverde, S.R.; Paiva, H.N.D.; Virgens Filho, A.D.C.; Silva, M.L.D. Economic analysis of the rubber-cocoa intercropping for generation of certified emission reduction. Rev. Árvore 2006, 30, 969–979. (In Portuguese) [Google Scholar] [CrossRef]
- Cheng, C.M.; Wang, R.S.; Jiang, J.S. Variation of soil fertility and carbon sequestration by planting Hevea brasiliensis in Hainan Island, China. J. Environ. Sci. 2007, 19, 348–352. [Google Scholar] [CrossRef]
- Fernandes, T.J.G.; Soares, C.P.B.; Jacovine, L.A.G.; Alvarenga, A.D.P. Quantification of carbon stored in the aerial part and roots of 12-year-old Hevea sp., in the “Zona da Mata Mineira”. Rev. Árvore 2007, 31, 657–665. (In Portuguese) [Google Scholar] [CrossRef]
- Sha, L.Q. Carbon Storage and Soil CO2 Efflux of Tropical Seasonal Rain Forest, Rubber Tree Plantation and Paddy Soil in Xishuangbanna. Ph.D. Thesis, Xishuangbanna Tropical Botanical Garden, University of Chinese Academy of Sciences, Beijing, China, 1 June 2008. [Google Scholar]
- Peng, Y. Researches on Carbon Sequestration Function of Plant Subsystem in Rubber (Hevea brasiliensis) Plantation Ecosystem in Western Hainan. Master’s Thesis, Hainan University, Haikou, China, 1 July 2010. [Google Scholar]
- Zheng, Z.B.; Wang, X.; Zhou, Z.D.; Yang, H. Carbon storage and distribution law of the rubber forest ecological system in Danzhou of Hainan. Trop. Agric. Eng. 2010, 34, 45–50. [Google Scholar]
- Egbe, A.E.; Tabot, P.T.; Fonge, B.A.; Bechem, E. Simulation of the impacts of three management regimes on carbon sinks in rubber and oil palm plantation ecosystems of South-Western Cameroon. J. Ecol. Nat. Environ. 2012, 46, 154–162. [Google Scholar]
- Kongsager, R.; Napier, J.; Mertz, O. The carbon sequestration potential of tree crop plantations. Mitig. Adapt. Strateg. Glob. Chang. 2013, 18, 1197–1213. [Google Scholar] [CrossRef]
- Miao, X.L. Study on the Potential for Carbon Sequestration Fartificial PR107 Rubber Forest Ecosystem in Rubber Tree Planting Area of Qiongzhong of Hainan. Master’s Thesis, Hainan University, Haikou, China, 1 July 2013. [Google Scholar]
- Sun, Y. Simulation of Carbon Dynamic Change and Accumulation Process in Rubber Plantation of Xishaungbanna. Ph.D. Thesis, Xishuangbanna Tropical Botanical Garden, University of Chinese Academy of Sciences, Beijing, China, 1 June 2013. [Google Scholar]
- Xiao, Z.W.; Wang, X.H.; Zheng, L.; Wang, X.L.; Gao, L.H.; Tang, J.W. Biomass and its allocation pattern of monoculture and mixed rubber-tree plantations in Xishuangbanna. J. Central South Univ. For. Technol. 2014, 34, 108–116. [Google Scholar]
- Bert, D.; Danjon, F. Carbon concentration variations in the roots, stem and crown of mature Pinus pinaster (Ait.). Forest Ecol. Manag. 2006, 222, 279–295. [Google Scholar] [CrossRef]
- Peichl, M.; Arain, M.A. Above-and belowground ecosystem biomass and carbon pools in an age-sequence of temperate pine plantation forests. Agr. Forest Meteorol. 2006, 140, 51–63. [Google Scholar] [CrossRef]
- Zheng, W.J.; Bao, W.K.; Gu, B.; He, X.; Leng, L. Carbon concentration and its characteristics in terrestrial higher plants. Chin. J. Ecol. 2007, 26, 307–313. [Google Scholar]
- Cao, J.X.; Wang, X.P.; Tian, Y.; Wen, Z.Y.; Zha, T.S. Pattern of carbon allocation across three different stages of stand development of a Chinese pine (Pinus tabulaeformis) forest. Ecol. Res. 2012, 27, 883–892. [Google Scholar] [CrossRef]
- Zhou, Y.R.; Yu, Z.L.; Zhao, S.D. Carbon storage and budget of major Chinese forest types. Acta Phytoecol. Sin. 2000, 24, 518–522. [Google Scholar]
- Wang, F.M.; Xu, X.; Zou, B.; Guo, Z.H.; Li, Z.A.; Zhu, W.X. Biomass accumulation and carbon sequestration in four different aged Casuarina equisetifolia coastal shelterbelt plantations in South China. PLoS ONE 2013, 8, e77449. [Google Scholar]
- Zhang, H.; Song, T.Q.; Wang, K.L.; Du, H.; Yue, Y.M.; Wang, G.X.; Zeng, F.P. Biomass and carbon storage in an age-sequence of Cyclobalanopsis glauca plantations in southwest China. Ecol. Eng. 2014, 73, 184–191. [Google Scholar] [CrossRef]
- Mao, R.; Zeng, D.H.; Hu, Y.L.; Li, L.J.; Yang, D. Soil organic carbon and nitrogen stocks in an age-sequence of poplar stands planted on marginal agricultural land in Northeast China. Plant Soil 2010, 332, 277–287. [Google Scholar] [CrossRef]
- Fonseca, W.; Alice, F.E.; Rey-Benayas, J.M. Carbon accumulation in aboveground and belowground biomass and soil of different age native forest plantations in the humid tropical lowlands of Costa Rica. New For. 2012, 43, 197–211. [Google Scholar] [CrossRef]
- Ming, A.; Jia, H.; Zhao, J.; Tao, Y.; Li, Y. Above-and below-ground carbon stocks in an indigenous tree (Mytilaria laosensis) plantation chronosequence in subtropical China. PLoS ONE 2014, 9, e109730. [Google Scholar] [CrossRef] [PubMed]
- Pregitzer, K.S.; Euskirchen, E.S. Carbon cycling and storage in world forests: Biome patterns related to forest age. Glob. Chang. Biol. 2004, 10, 2052–2077. [Google Scholar] [CrossRef]
- Song, Q.H.; Tan, Z.H.; Zhang, Y.P.; Sha, L.Q.; Deng, X.B.; Zhou, W.J.; Zhao, J.F.; Zhao, J.B.; Zhang, X.; et al. Do the rubber plantations in tropical China act as large carbon sinks? Forest 2014, 7, 42–47. [Google Scholar] [CrossRef]
- Cleveland, C.C.; Taylor, P.; Chadwick, K.D.; Dahlin, K.; Doughty, C.E.; Malhi, Y.; Smith, W.K.; Sullivan, B.W.; Wieder, W.R.; Townsend, A.R. A comparison of plot-based satellite and Earth system model estimates of tropical forest net primary production. Glob. Biogeochem. Cycle 2015, 29, 626–644. [Google Scholar] [CrossRef]
- Peltoniemi, M.; Mäkipää, R.; Liski, J.; Tamminen, P. Changes in soil carbon with stand age–an evaluation of a modelling method with empirical data. Glob. Chang. Biol. 2004, 10, 2078–2091. [Google Scholar] [CrossRef]
- Tang, J.W.; Zhang, J.H.; Song, Q.S.; Cao, M.; Feng, Z.L.; Dang, C.L.; Wu, Z.L. A preliminary study on the biomass of secondary tropical forest in Xishuangbanna. Acta Phytoecol. Sin. 1998, 22, 489–498. [Google Scholar]
- Ziegler, A.D.; Phelps, J.; Yuen, J.Q.; Webb, E.L.; Lawrence, D.; Fox, J.M.; Bruun, T.B.; Leisz, S.J.; Ryan, C.M.; Dressler, W.; et al. Carbon outcomes of major land-cover transitions in SE Asia: great uncertainties and REDD+ policy implications. Glob. Chang. Biol. 2012, 18, 3087–3099. [Google Scholar] [CrossRef]
| Stand Parameter | 7-year-old | 13-year-old | 19-year-old | 25-year-old | 47-year-old |
|---|---|---|---|---|---|
| Longitude | 101°14′45.3″ | 101°14′40.1″ | 101°14′43.5″ | 101°16′28.9″ | 101°16′03.5″ |
| Latitude | 21°56′48.8″ | 21°56′49.6″ | 21°56′34.8″ | 21°54′36.8″ | 21°55′22.9″ |
| Elevation (m) | 580–595 | 600–620 | 560–570 | 570–580 | 550–570 |
| Slope (°) | 30–35 | 25–30 | 25–30 | 15–20 | 0–5 |
| Aspect | Southwest | Southeast | Southeast | Southeast | Southwest |
| Planting spacing (m) | 2.5 × 6 | 2.5 × 6 | 2.5 × 8 | 2.5 × 8 | 2.5 × 8 |
| Average DBH (cm) | 9.3 ± 0.7 d | 15.5 ± 0.8 c | 21.4 ± 1.2 b | 24.9± 0.7 b | 40.4 ± 2.2 a |
| Average height (m) | 7.3 ± 0.9 c | 13.9 ± 1.0 b | 16.3 ± 1.2 ab | 17.2 ± 0.8 a | 21.3 ± 1.1 a |
| Density (plants ha−1) | 667 ± 31 a | 637 ± 26 a | 626 ± 21 a | 510 ± 23 ab | 203 ± 17 b |
| Basal area (m2 ha−1) | 5.1 ± 0.8 c | 11.3 ± 1.0 b | 21.9 ± 1.8 ab | 24.6 ± 0.7 a | 29.5 ± 0.9 a |
| pH | 6.5 ± 0.2 a | 6.4 ± 0.1 a | 6.0 ± 0.1 a | 5.7 ± 0.1 ab | 5.4 ± 0.1 b |
| Clay (%) | 30.3 ± 3.5 a | 28.5 ± 2.3 a | 32.2 ± 2.0 a | 29.1 ± 3.4 a | 26.9 ± 2.9 a |
| Silt (%) | 34.5 ± 2.2 a | 33.0 ± 3.7 a | 33.8 ± 2.7 a | 31.5 ± 0.5 a | 28.0 ± 0.7 b |
| Sand (%) | 35.2 ± 0.4 b | 38.5 ± 1.1 ab | 34.0 ± 0.2 b | 39.4 ± 0.9 a | 45.1 ± 1.8 a |
| Biomass Fraction | 7-year-old | 13-year-old | 19-year-old | 25-year-old | 47-year-old |
|---|---|---|---|---|---|
| Aboveground tree | |||||
| Stem | 12.86 ± 1.57 (6.20) c | 38.66 ± 1.88 (18.56) c | 92.17 ± 6.77 (43.57) b | 106.56 ± 9.52 (52.88) b | 169.50 ± 16.19 (81.84) a |
| Branch | 3.63 ± 0.44 (1.77) c | 10.74 ± 0.53 (5.15) c | 25.29 ± 1.86 (12.00) b | 29.15 ± 2.60 (14.48) b | 45.54 ± 4.32 (21.87) a |
| Foliage | 0.75 ± 0.09 (0.39) c | 1.87 ± 0.10 (0.95) c | 3.88 ± 0.30 (1.94) b | 4.32 ± 0.37 (2.24) ab | 5.62 ± 0.51 (2.86) a |
| Litter | |||||
| Leaf | 2.50 ± 0.09 (1.29) b | 3.82 ± 0.20 (1.90) a | 4.24 ± 0.30 (2.17) a | 3.77 ± 0.06 (1.92) a | 4.12 ± 0.05 (2.12) a |
| Twig | 0.02 ± 0.00 (0.01) c | 0.40 ± 0.08 (0.20) b | 0.59 ± 0.14 (0.30) ab | 0.82 ± 0.02 (0.42) a | 0.40 ± 0.04 (0.21) b |
| Flower/fruit | 0.10 ± 0.01 (0.06) c | 0.35 ± 0.07 (0.18) b | 0.26 ± 0.07 (0.14) b | 0.92 ± 0.01 (0.47) a | 0.76 ± 0.10 (0.39) a |
| Miscellaneous litter | 0.12 ± 0.01 (0.06) c | 0.15 ± 0.01 (0.08) c | 0.16 ± 0.01 (0.08) bc | 0.23 ± 0.01 (0.12) b | 0.42 ± 0.03 (0.22) a |
| Total litter | 2.74 ± 0.12 (1.42) b | 4.72 ± 0.36 (2.35) a | 5.25 ± 0.53 (2.69) a | 5.74 ± 0.08 (2.93) a | 5.70 ± 0.21 (2.94) a |
| Total AGB | 19.98 ± 2.11 (9.78) c | 55.99 ± 2.87 (27.01) c | 126.59 ± 9.46 (60.20) b | 145.77 ± 12.57 (72.53) b | 226.36 ± 21.23 (109.51) a |
| Root | |||||
| Root collar | 2.41 ± 0.26 (1.16) c | 6.13 ± 0.38 (2.91) b | 7.21 ± 0.61 (3.38) ab | 9.29 ± 0.79 (4.49) a | 9.59 ± 0.83 (4.57) a |
| Stump root | 2.34 ± 0.25 (1.11) c | 4.35 ± 0.27 (2.04) b | 6.20 ± 0.52 (2.87) a | 4.59 ± 0.39 (2.19) b | 4.42 ± 0.38 (2.09) b |
| Thick root | 0.46 ± 0.05 (0.22) c | 1.32 ± 0.08 (0.62) c | 5.15 ± 0.43 (2.37) b | 9.41 ± 0.80 (4.45) a | 8.17 ± 0.71 (3.82) a |
| Small root | 0.44 ± 0.05 (0.21) c | 0.99 ± 0.06 (0.45) c | 4.43 ± 0.37 (1.99) a | 2.54 ± 0.22 (1.18) b | 5.58 ± 0.48 (2.56) a |
| Fine root | 0.47 ± 0.05 (0.21) c | 0.75 ± 0.05 (0.33) c | 2.46 ± 0.21 (1.08) b | 1.69 ± 0.14 (0.77) b | 3.42 ± 0.30 (1.54) a |
| Total BGB | 6.12 ± 0.66 (2.91) c | 13.53 ± 0.84 (6.35) b | 25.45 ± 2.14 (11.69) a | 27.52 ± 2.34 (13.08) a | 31.19 ± 2.69 (14.58) a |
| BGB/AGB | 0.31 ± 0.00 (0.30) a | 0.24 ± 0.00 (0.24) b | 0.20 ± 0.00 (0.19) c | 0.19 ± 0.00 (0.18) d | 0.14 ± 0.00 (0.13) e |
| Latex | 1.10 ± 0.00 (0.91) e | 8.74 ± 0.10 (7.43) d | 17.51 ± 0.28 (14.92) c | 26.47 ± 0.50 (22.66) b | 46.15 ± 0.84 (38.63) a |
| TB | 27.20 ± 2.88 (13.61) c | 78.26 ± 3.72 (40.79) c | 169.55 ± 11.62 (86.82) b | 199.76 ± 14.93 (108.27) b | 303.70 ± 23.71 (162.72) a |
| NPP Fraction | 7-year-old | 13-year-old | 19-year-old | 25-year-old | 47-year-old |
|---|---|---|---|---|---|
| NPPaboveground tree | 2.46 ± 0.30 (1.19) c | 3.94 ± 0.19 (1.90) b,c | 6.39 ± 0.47 (3.03) a | 5.60 ± 0.50 (2.78) a,b | 4.69 ± 0.45 (2.27) a,b |
| NPPlitter | 2.74 ± 0.12 (1.42) b | 4.72 ± 0.36 (2.35) a | 5.25 ± 0.53 (2.69) a | 5.74 ± 0.08 (2.93) a | 5.70 ± 0.21 (2.94) a |
| NPProot | 0.87 ± 0.09 (0.42) b,c | 1.04 ± 0.06 (0.49) a,b | 1.33 ± 0.11 (0.62) a | 1.10 ± 0.09 (0.52) a,b | 0.66 ± 0.05 (0.31) c |
| NPPlatex | 1.10 ± 0.00 (0.91) c | 1.25 ± 0.01 (1.06) b | 1.35 ± 0.02 (1.14) a | 1.39 ± 0.03 (1.19) a | 1.12 ± 0.02 (0.94) c |
| NPPtotal | 7.17 ± 0.51 (3.94) b | 10.95 ± 0.62 (5.79) a | 14.32 ± 1.11 (7.48) a | 13.83 ± 0.67 (7.42) a | 12.18 ± 0.71 (6.46) a |
| Compartments | Regression Equation | R2 | SEE | F-Value | P |
|---|---|---|---|---|---|
| Aboveground tree | y = −0.049x2 + 5.150x − 27.724 | 0.802 | 19.028 | 24.398 | < 0.001 |
| Litter | y = −0.002x2 + 0.154x + 0.547 | 0.841 | 0.271 | 31.703 | < 0.001 |
| Root | y = −0.014x2 + 1.034x − 3.934 | 0.711 | 3.105 | 14.746 | < 0.001 |
| Latex | y = −0.011x2 + 1.533x − 9.878 | 0.995 | 1.090 | 1076.113 | < 0.001 |
| Soil | y = 0.012x2 − 1.015x + 150.766 | 0.181 | 12.590 | 1.329 | 0.300 |
| Total excluding latex C (our study) | y = −0.053x2 + 5.323x + 119.655 | 0.774 | 20.412 | 20.529 | < 0.001 |
| Total excluding latex C (comprehensive) * | y = −0.098x2 + 7.508x + 104.044 | 0.328 | 58.128 | 6.349 | 0.006 |
| Total including latex C (our study) | y = −0.063x2 + 6.856x + 109.78 | 0.869 | 20.343 | 39.612 | < 0.001 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.; Pang, J.; Jepsen, M.R.; Lü, X.; Tang, J. Carbon Stocks across a Fifty Year Chronosequence of Rubber Plantations in Tropical China. Forests 2017, 8, 209. https://doi.org/10.3390/f8060209
Liu C, Pang J, Jepsen MR, Lü X, Tang J. Carbon Stocks across a Fifty Year Chronosequence of Rubber Plantations in Tropical China. Forests. 2017; 8(6):209. https://doi.org/10.3390/f8060209
Chicago/Turabian StyleLiu, Chenggang, Jiaping Pang, Martin Rudbeck Jepsen, Xiaotao Lü, and Jianwei Tang. 2017. "Carbon Stocks across a Fifty Year Chronosequence of Rubber Plantations in Tropical China" Forests 8, no. 6: 209. https://doi.org/10.3390/f8060209






