Density Functional Theory Study on Mechanism of Mercury Removal by CeO2 Modified Activated Carbon
Abstract
:1. Introduction
2. Models and Computational Details
2.1. AC and CeO2-AC Models
2.2. Computational Parameters
3. Results and Discussion
3.1. Hg0 Adsorption Mechanism
3.2. HCl Adsorption Mechanism
3.3. HgCl Adsorption Mechanism
3.4. HgCl2 Adsorption Mechanism
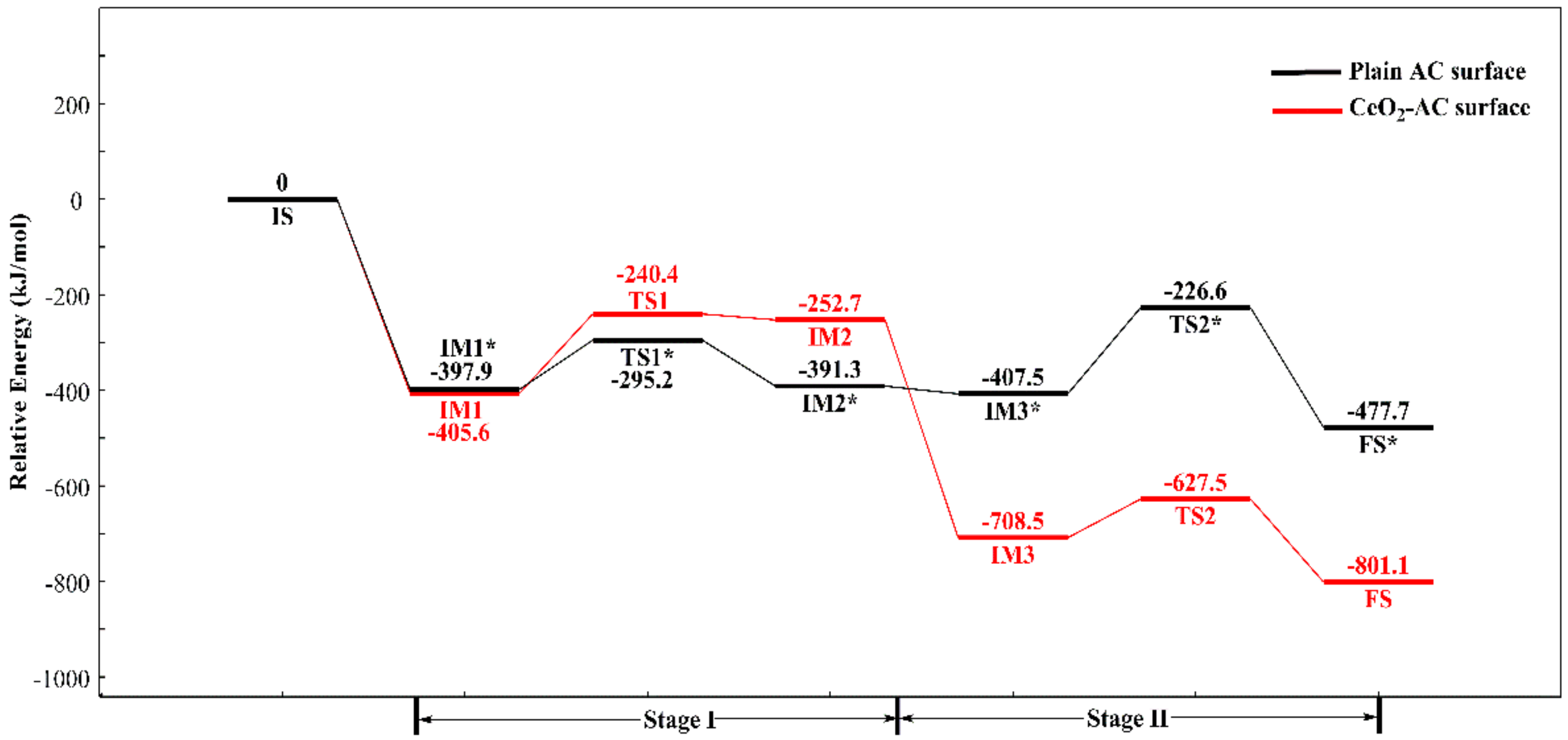
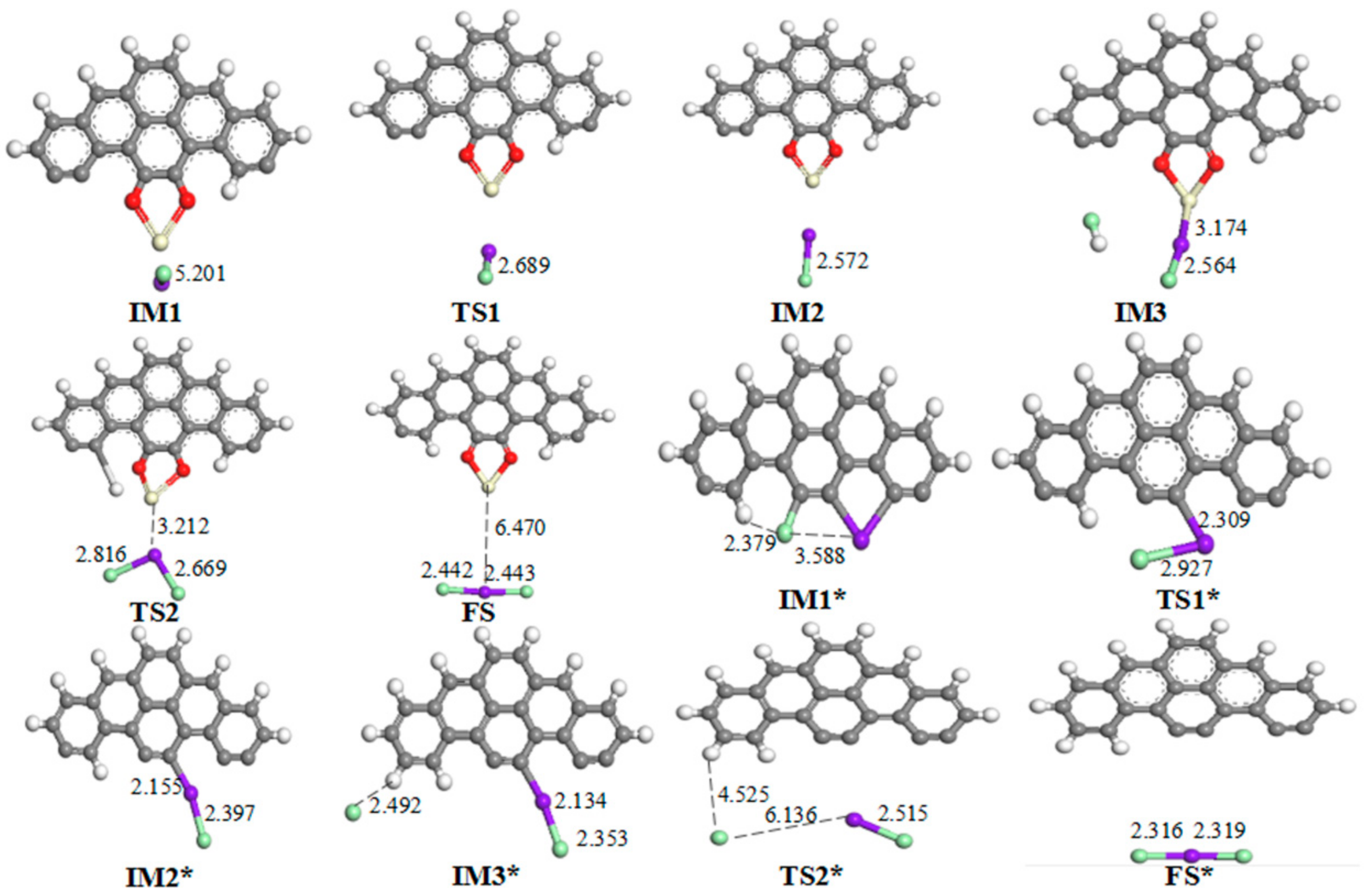
3.5. Hg0 Oxidation Mechanism
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wang, S.X.; Zhang, L.; Wang, L.; Wu, Q.R.; Wang, F.Y.; Hao, J.M. A review of atmospheric mercury emissions, pollution and control in China. Front. Environ. Sci. Eng. 2014, 8, 631–649. [Google Scholar] [CrossRef]
- Streets, D.G.; Lu, Z.F.; Levin, L.; Ter Schure, A.; Sunderland, E.M. Historical releases of mercury to air, land, and water from coal combustion. Sci. Total Environ. 2017, 615, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.C.; Shen, Z.M.; Tang, Q.L.; Yang, B.W.; Fan, M.H. A DFT study of Hg0, adsorption on Co3O4 (110) surface. Chem. Eng. J. 2016, 289, 349–355. [Google Scholar] [CrossRef]
- Liu, Z.J.; Zhang, Z.Y.; Choi, S.K.; Liu, Y.Y. Surface properties and pore structure of anthracite, bituminous coal and lignite. Energies 2018, 11, 1502. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, S.X.; Streets, D.G.; Hao, J.M.; Chan, M.; Jiang, J.K. Trends in anthropogenic mercury emissions in China from 1995 to 2003. Environ. Sci. Technol. 2006, 40, 5312–5318. [Google Scholar] [CrossRef] [PubMed]
- Li, H.L.; Zhu, L.; Wu, S.K.; Liu, Y.; Shih, K.M. Synergy of CuO and CeO2 combination for mercury oxidation under low-temperature selective catalytic reduction atmosphere. Int. J. Coal Geol. 2017, 170, 69–76. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, Y.; Wu, Y.W.; Han, J.; Zhang, S.L.; Lu, Q.; Yang, Y.P. Mechanism of heterogeneous mercury oxidation by HCl on V2O5(001) surface. Curr. Appl. Phys. 2018, 18, 626–632. [Google Scholar] [CrossRef]
- Zhao, L.; He, Q.S.; Li, L.; Lu, Q.; Dong, C.Q.; Yang, Y.P. Research on the catalytic oxidation of Hg0 by modified SCR catalysts. J. Fuel Chem. Technol. 2015, 43, 628–634. [Google Scholar] [CrossRef]
- Zhou, Z.J.; Liu, X.W.; Zhao, B.; Chen, Z.G.; Shao, H.Z.; Wang, L.L.; Xu, M.H. Effects of existing energy saving and air pollution control devices on mercury removal in coal-fired power plants. Fuel Process. Technol. 2015, 131, 99–108. [Google Scholar] [CrossRef]
- Huang, W.J.; Xu, H.M.; Qu, Z.; Zhao, S.; Chen, W.; Yan, N. Significance of Fe2O3, modified SCR catalyst for gas-phase elemental mercury oxidation in coal-fired flue gas. Fuel Process. Technol. 2016, 149, 23–28. [Google Scholar] [CrossRef]
- Fernández-Miranda, N.; Rodriguez, E.; Lopez-Anton, M.A.; Garcia, R.; Martinez-Tarazona, M.R. A New approach for retaining mercury in energy generation processes: Regenerable carbonaceous sorbents. Energies 2017, 10, 1311. [Google Scholar] [CrossRef]
- Gao, X.P.; Zhou, Y.N.; Tan, Y.J.; Cheng, Z.W.; Tang, Z.W.; Jia, J.P. Unveiling adsorption mechanisms of elemental mercury on defective boron nitride monolayer: A computational study. Energy Fuels 2018, 32, 5331–5337. [Google Scholar] [CrossRef]
- Zhao, L.; Wu, Y.; Han, J.; Lu, Q.; Yang, Y.; Zhang, L. Mechanism of mercury adsorption and oxidation by oxygen over the CeO2 (111) surface: A DFT study. Materials 2018, 11, 485. [Google Scholar] [CrossRef] [PubMed]
- Li, H.L.; Wu, C.Y.; Li, Y.; Zhang, J.Y. CeO2-TiO2 catalysts for catalytic oxidation of elemental mercury in low-rank coal combustion flue gas. Environ. Sci. Technol. 2011, 45, 7394–7400. [Google Scholar] [CrossRef] [PubMed]
- Jampaiah, D.; Samuel, J.I.; Ylias, S.; James, T.; Selvakannan, P.R.; Ayman, N.; Benjaram, M.R.; Suresh, B. Ceria-zirconia modified MnOx catalysts for gaseous elemental mercury oxidation and adsorption. Catal. Sci. Technol. 2016, 6, 1792–1803. [Google Scholar] [CrossRef]
- Jampaiah, D.; Katie, T.; Perala, V.; Samuel, J.I.; Ylias, S.; James, T.; Suresh, B.; Benjaram, M.R. Catalytic oxidation and adsorption of elemental mercury over nanostructured CeO2–MnOx catalyst. RSC Adv. 2015, 38, 30331–30341. [Google Scholar] [CrossRef]
- Hua, X.Y.; Zhou, J.S.; Li, Q.K.; Luo, Z.Y.; Cen, K.F. Gas-phase elemental mercury removal by CeO2 impregnated activated coke. Energy Fuels 2010, 24, 5426–5431. [Google Scholar] [CrossRef]
- Wang, Y.; Li, C.T.; Zhao, L.K.; Xie, Y.E.; Zhang, X.N.; Zeng, G.M.; Wu, H.Y.; Zhang, J. Study on the removal of elemental mercury from simulated flue gas by Fe2O3-CeO2/AC at low temperature. Environ. Sci. Polut. Res. 2016, 23, 5099–5110. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.C.; Han, X.J.; Huang, Z.P.; Hou, Y.Q.; Guo, Y.P.; Wu, M.H. Superior activity of CeO2 modified V2O5/AC catalyst for mercury removal at low temperature. Chem. Eng. J. 2017, 331, 741–749. [Google Scholar] [CrossRef]
- Tian, L.H.; Li, C.T.; Li, Q.; Zeng, G.M.; Gao, Z.; Li, S.H.; Fan, X.P. Removal of elemental mercury by activated carbon impregnated with CeO2. Fuel 2009, 88, 1687–1691. [Google Scholar] [CrossRef]
- Xie, Y.E.; Li, C.T.; Zhao, L.K.; Zhang, J.; Zeng, G.M.; Zhang, X.N.; Zhang, W.; Tao, S.S. Experimental study on Hg0 removal from flue gas over columnar MnOx-CeO2/activated coke. Appl. Surf. Sci. 2015, 333, 59–67. [Google Scholar] [CrossRef]
- Zhang, H.W.; Chen, J.Y.; Zhao, K.; Niu, Q.X.; Wang, L. Removal of vapor-phase elemental mercury from simulated syngas using semi-coke modified by Mn/Ce doping. J. Fuel Chem. Technol. 2016, 4, 394–400. [Google Scholar] [CrossRef]
- Wu, H.Y.; Li, C.T.; Zhao, L.K.; Zhang, J.; Zeng, G.M.; Xie, Y.E.; Zhang, X.N.; Wang, Y. Removal of gaseous elemental mercury by cylindrical activated coke loaded with CoOx-CeO2 from simulated coal combustion flue gas. Energy Fuels 2015, 29, 6747–6757. [Google Scholar] [CrossRef]
- Chen, N.; Yang, R.T. Ab initio molecular orbital calculation on graphite: Selection of molecular system and model chemistry. Carbon 1998, 36, 1061–1070. [Google Scholar] [CrossRef]
- Yang, F.H.; Yang, R.T. Ab initio molecular orbital study of adsorption of atomic hydrogen on graphite: Insight into hydrogen storage in carbon nanotubes. Carbon 2002, 40, 437–444. [Google Scholar] [CrossRef]
- Chen, P.; Gu, M.Y.; Lin, Y.Y.; Yan, D.W.; Huang, Y.Y. Effect of ketone group on heterogeneous reduction of NO by char. J. Feul Chem. Technol. 2018, 46, 521–528. [Google Scholar] [CrossRef]
- Chompoonut, R.; Vinich, P.; Supa, H.; Nawee, K.; Supawadee, N. Complete reaction mechanisms of mercury oxidation on halogenated activated carbon. J. Hazard. Mater. 2016, 310, 253–260. [Google Scholar] [CrossRef]
- Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7764. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rec. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Perdew, J.P.; Burke, K.; Wang, Y. Generalized gradient approximation for the exchange-correlation hole of a many-electron system. Phys. Rev. B. 1996, 54, 16533–16539. [Google Scholar] [CrossRef]
- Delley, B. Hardness conserving semilocal pseudopotentials. Phys. Rev. B. 2002, 66, 155125. [Google Scholar] [CrossRef]
- Halgren, T.A.; Lipscomb, W.N. The synchronous-transit method for determining reaction pathways and locating molecular transition states. Chem. Phys. Lett. 1977, 49, 225–232. [Google Scholar] [CrossRef]
- Orimo, S.; Zuttel, A.; Schlapbach, L.; Majer, G.; Fukunaga, T.; Fujii, H. Hydrogen interaction with carbon nanostructures: Current situation and future prospects. J. Alloy. Compd. 2003, 356, 716–719. [Google Scholar] [CrossRef]
- Zhao, L.K.; Li, C.T.; Wang, Y.; Wu, H.Y.; Gao, L.; Zhang, J.; Zeng, G.M. Simultaneous removal of elemental mercury and NO from simulated flue gas using a CeO2 modified V2O5-WO3/TiO2 catalyst. Catal. Sci. Technol. 2016, 6, 420–430. [Google Scholar] [CrossRef]
- Presto, A.A.; Granite, E.J. Survey of catalysts for oxidation of mercury in flue gas. Environ. Sci. Technol. 2006, 40, 5601–5609. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.K.; Liu, J.; Shen, F.H. Heterogeneous mercury oxidation by HCl over CeO2 catalyst: Density functional theory study. J. Phys. Chem. C 2015, 119, 15047–15055. [Google Scholar] [CrossRef]

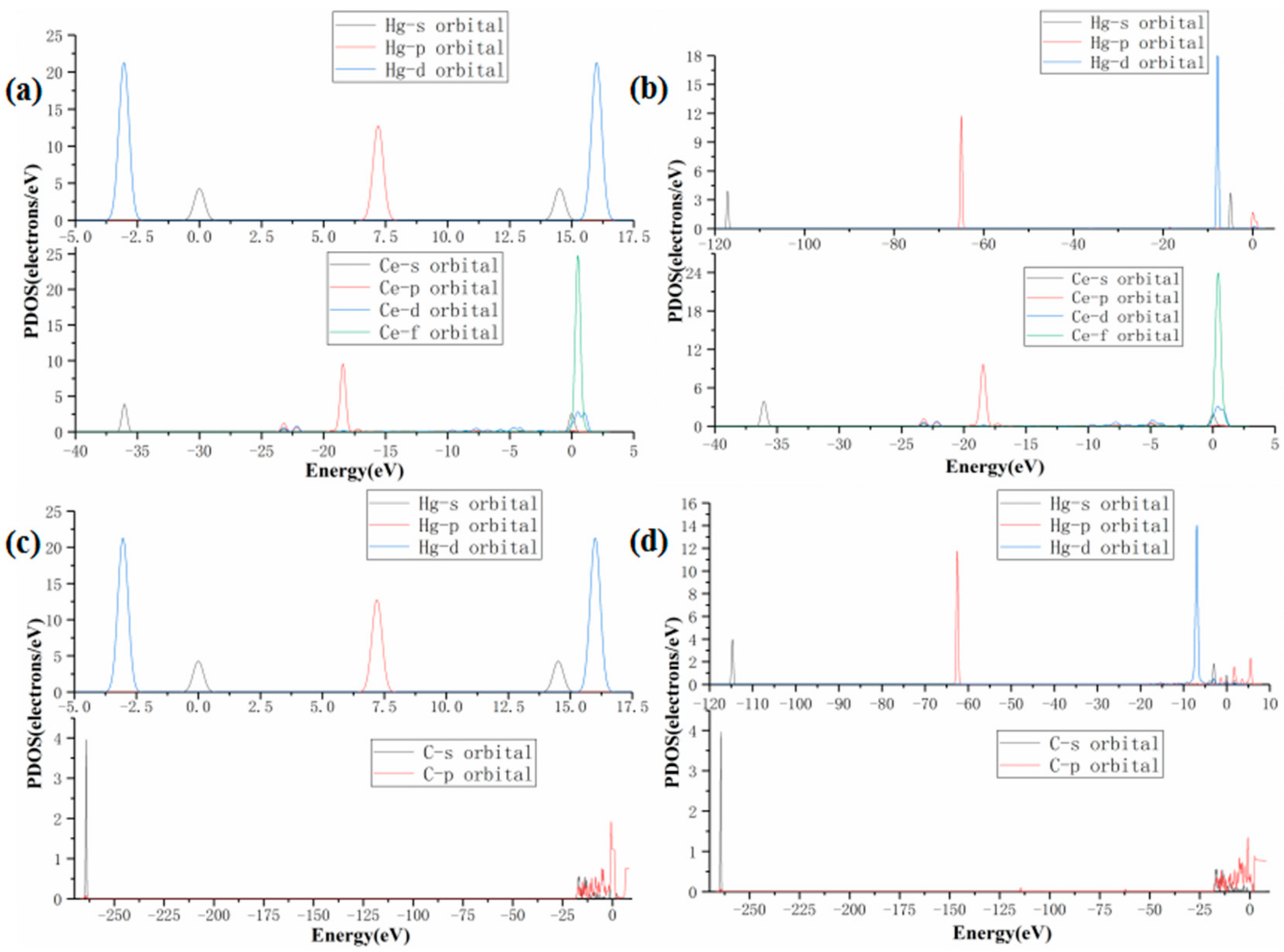
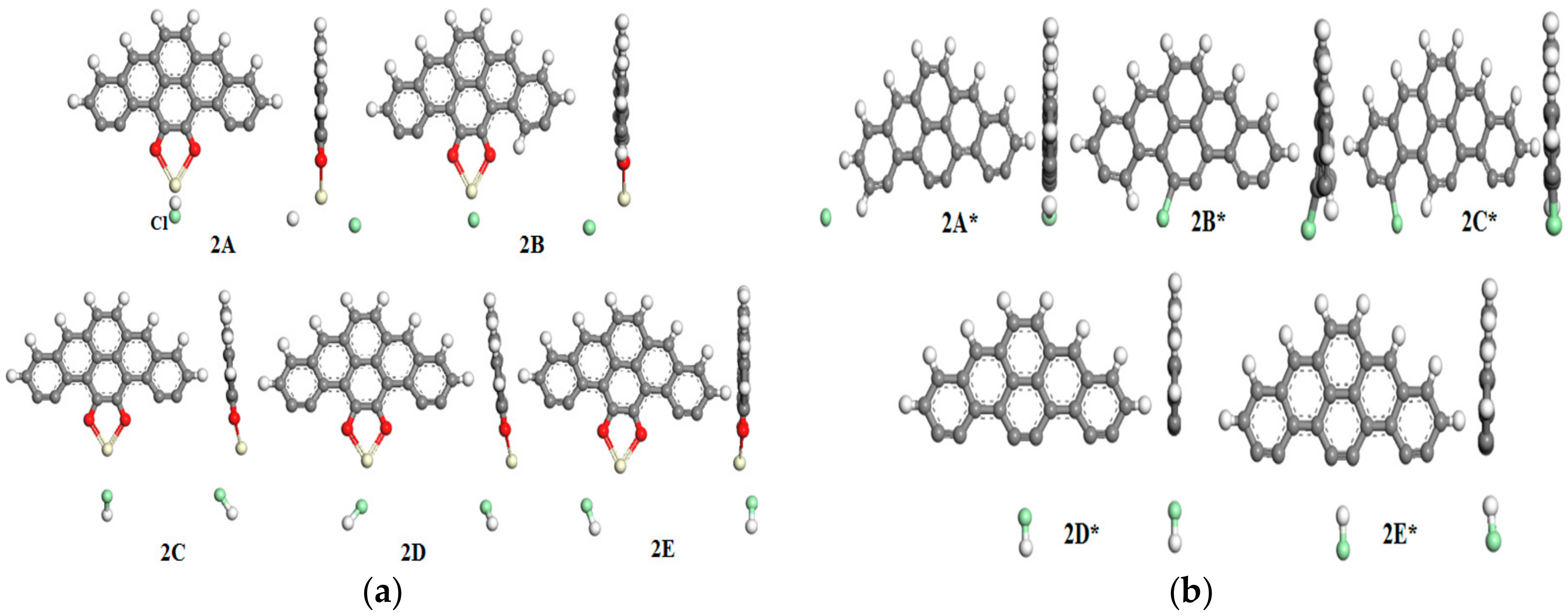



| Bond Type | C–C | C–H | C–O | C≡C | Ce–O | |
|---|---|---|---|---|---|---|
| Bond Length (Å) | Conventional AC model | 1.369–1.439 | 1.092 | - | 1.267 | - |
| CeO2 modified AC model | 1.394–1.460 | 1.093 | 1.348 | 1.275 | 2.217 | |
| CeO2-AC Model | AC Model | ||||
|---|---|---|---|---|---|
| Configurations | RHg–Ce (Å) | Eads (kJ/mol) | Configurations | RHg–C (Å) | Eads (kJ/mol) |
| 1A | 3.431 | −46.94 | 1A* | 2.511 | −119.92 |
| 1B | 3.508 | −42.05 | 1B* | 2.462/2.464 | −146.01 |
| CeO2-AC Surface | AC Surface | ||||||
|---|---|---|---|---|---|---|---|
| Configurations | RX–Ce(Å) * | RH–Cl(Å) | Eads(kJ/mol) | Configurations | RX–C(Å) * | RH–Cl(Å) | Eads(kJ/mol) |
| 2A | 2.633/2.157 | 4.036 | −174.27 | 2A* | 1.095 | 2.707 | −149.38 |
| 2B | 2.623 | 5.291 | −371.63 | 2B* | 2.373 | 1.089 | −254.78 |
| 2C | 3.088 | 1.361 | −54.85 | 2C* | 2.257 | 1.088 | −255.40 |
| 2D | 3.071 | 1.362 | −54.97 | 2D* | 3.496/3.501 | 1.291 | −41.20 |
| 2E | 3.226 | 1.343 | −52.63 | 2E* | 2.411/2.385 | 1.304 | −55.79 |
| CeO2-AC Surface | AC Surface | ||||||
|---|---|---|---|---|---|---|---|
| Configurations | RX–Ce (Å) * | RHg–Cl (Å) | Eads (kJ/mol) | Configurations | RHg–C (Å) * | RHg–Cl (Å) | Eads (kJ/mol) |
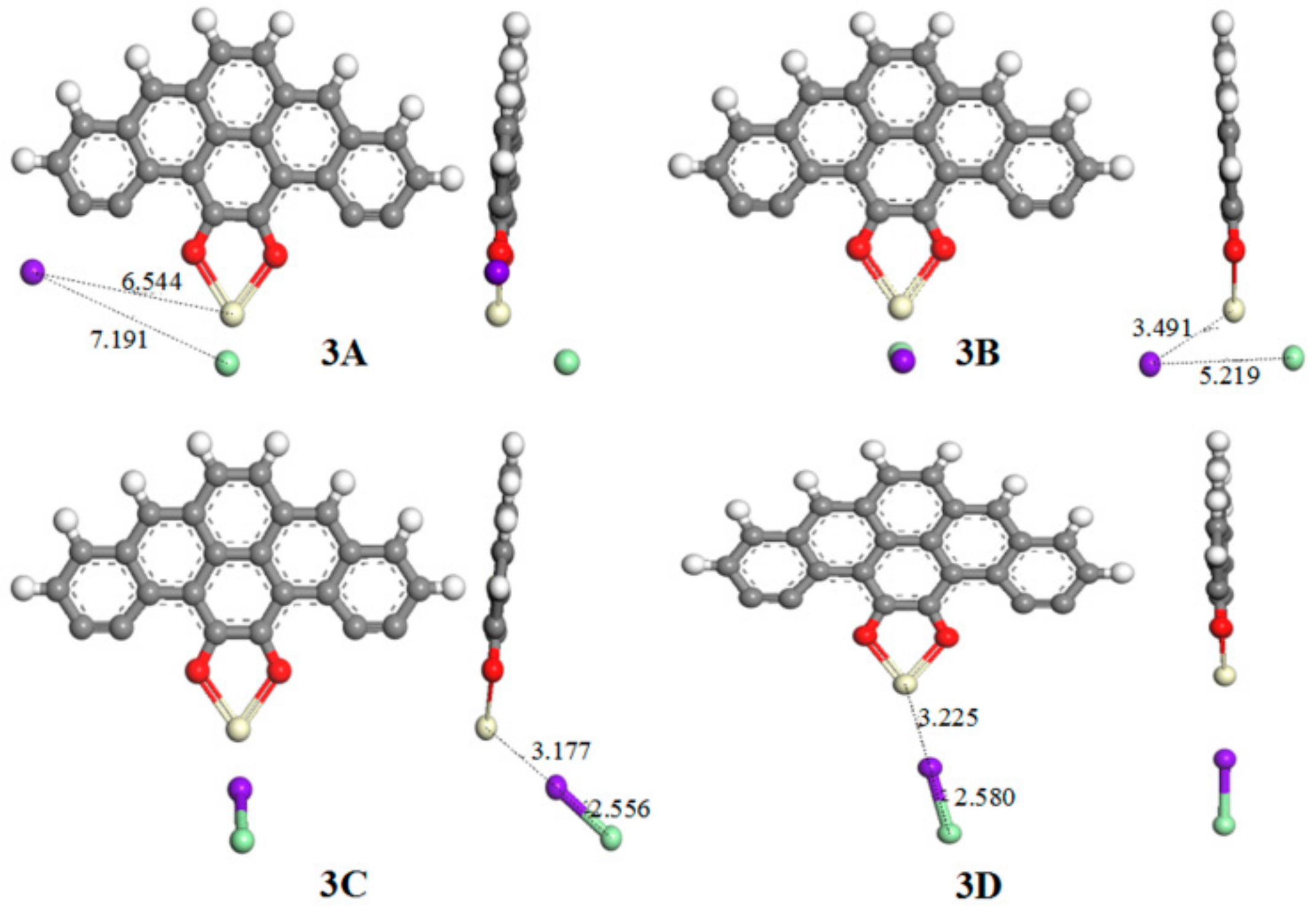
| 3A | 2.623 | 7.191 | −281.58 | 3A* | 2.149 | 2.391 | −258.70 |
| 3B | 2.625 | 5.219 | −294.50 | 3B* | 2.156 | 2.393 | −259.47 |
| 3C | 3.177 | 2.556 | −153.09 | 3C* | 2.323/2.427 | 3.689 | −301.32 |
| 3D | 3.275 | 2.580 | −139.80 | - | - | - | - |
| CeO2-AC Surface | AC Surface | |||||||
|---|---|---|---|---|---|---|---|---|
| Configurations | RX–Ce (Å) | RHg–Cl (Å) | Eads (kJ/mol) | Configurations | RHg–C (Å) | RHg–Cl (Å) | ∠Cl–Hg–Cl (°) | Eads (kJ/mol) |
| 4A | 2.599 | 5.323/2.672 | −202.70 | 4A* | 2.121 | 2.368/3.468 | - | −369.19 |
| 4B | 2.616 | 5.063/2.586 | −287.46 | 4B* | 6.043/6.119 | 2.356/2.362 | 179.82 | −101.54 |
| 4C | 2.640/2.643 | 3.886/3.857 | −399.03 | 4C* | 3.202/3.258 | 2.367/2.368 | 171.63 | −110.80 |
| - | - | - | - | 4D* | 2.271/2.309 | 2.546/2.515 | 92.65 | −186.98 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Wu, Y.-w.; Han, J.; Wang, H.-x.; Liu, D.-j.; Lu, Q.; Yang, Y.-p. Density Functional Theory Study on Mechanism of Mercury Removal by CeO2 Modified Activated Carbon. Energies 2018, 11, 2872. https://doi.org/10.3390/en11112872
Zhao L, Wu Y-w, Han J, Wang H-x, Liu D-j, Lu Q, Yang Y-p. Density Functional Theory Study on Mechanism of Mercury Removal by CeO2 Modified Activated Carbon. Energies. 2018; 11(11):2872. https://doi.org/10.3390/en11112872
Chicago/Turabian StyleZhao, Li, Yang-wen Wu, Jian Han, Han-xiao Wang, Ding-jia Liu, Qiang Lu, and Yong-ping Yang. 2018. "Density Functional Theory Study on Mechanism of Mercury Removal by CeO2 Modified Activated Carbon" Energies 11, no. 11: 2872. https://doi.org/10.3390/en11112872





