Physiological and Psychological Effects of a Forest Therapy Program on Middle-Aged Females
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects
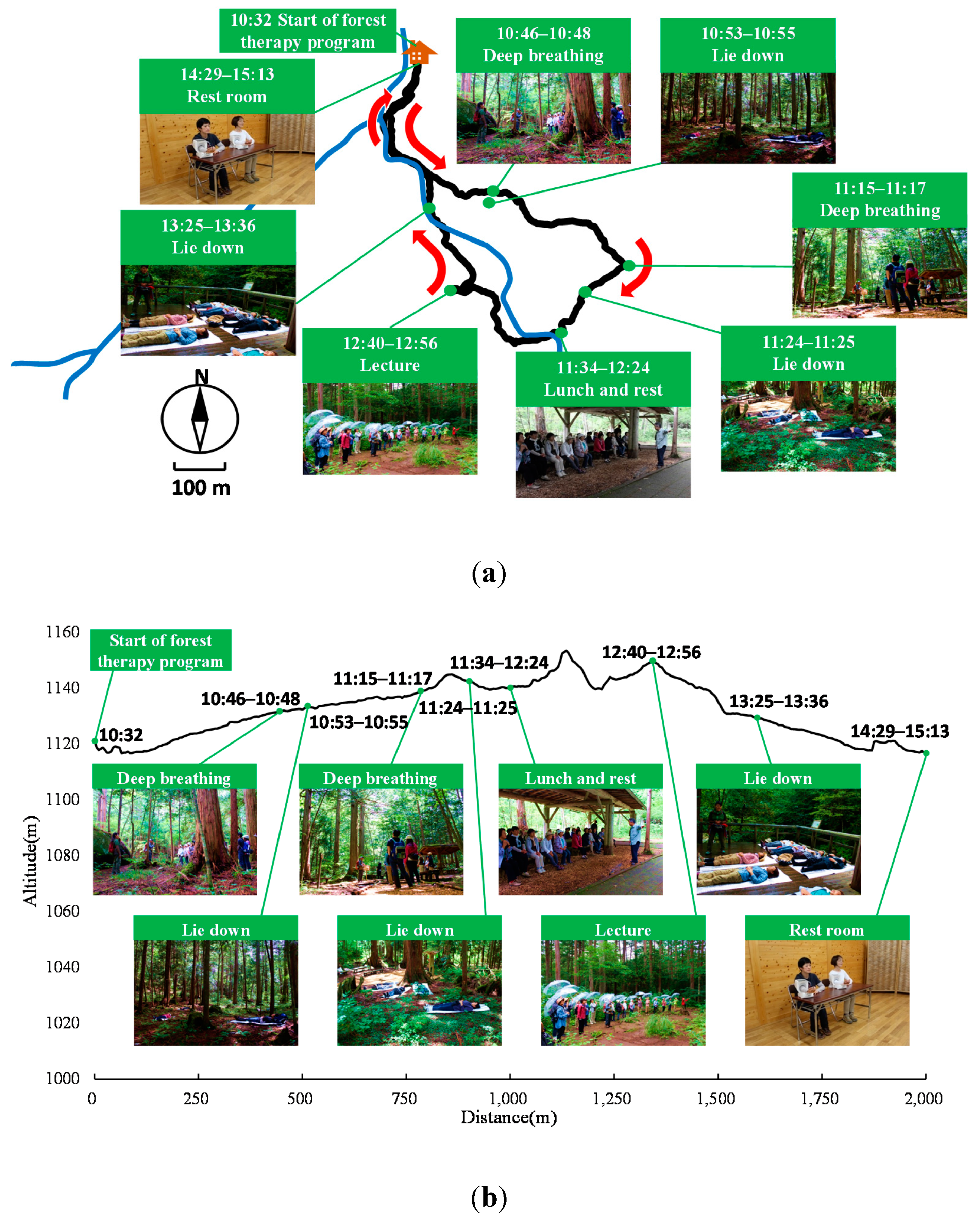
2.2. Experimental Sites
2.3. Physiological Indices
2.4. Psychological Indices
2.5. Experimental Design
| Time | Event | Calorie Consumption (Kcal/min) |
|---|---|---|
| 10:32–10:45 | Stroll (Forest ) | 1.21 |
| 10:46–10:48 | Deep breathing (Forest) | 0 |
| 10:49–10:52 | Stroll (Forest) | 0.15 |
| 10:53–10:55 | Lie down (Forest) | 0 |
| 10:56–11:14 | Stroll (Forest) | 0.65 |
| 11:15–11:17 | Deep breathing (Forest) | 0.10 |
| 11:18–11:23 | Stroll (Forest) | 0.48 |
| 11:24–11:25 | Lie down (Forest) | 0.06 |
| 11:26–11:33 | Stroll (Forest) | 0.52 |
| 11:34–12:24 | Lunch and rest (Resting room) | 0.04 |
| 12:25–12:39 | Stroll (Forest) | 0.92 |
| 12:40–12:56 | Lecture (Forest) | 0.08 |
| 12:57–13:09 | Stroll (Forest ) | 0.66 |
| 13:10–13:24 | Rest (Forest) | 0.01 |
| 13:25–13:36 | Lie down & abdominal breathing (Forest) | 0.00 |
| 13:37–13:59 | Chat (Forest) | 0.01 |
| 14:00–14:28 | Stroll (Forest) | 0.76 |
| 14:29–15:13 | Rest (Resting room) | 0.02 |
2.6. Statistical Analysis
3. Results
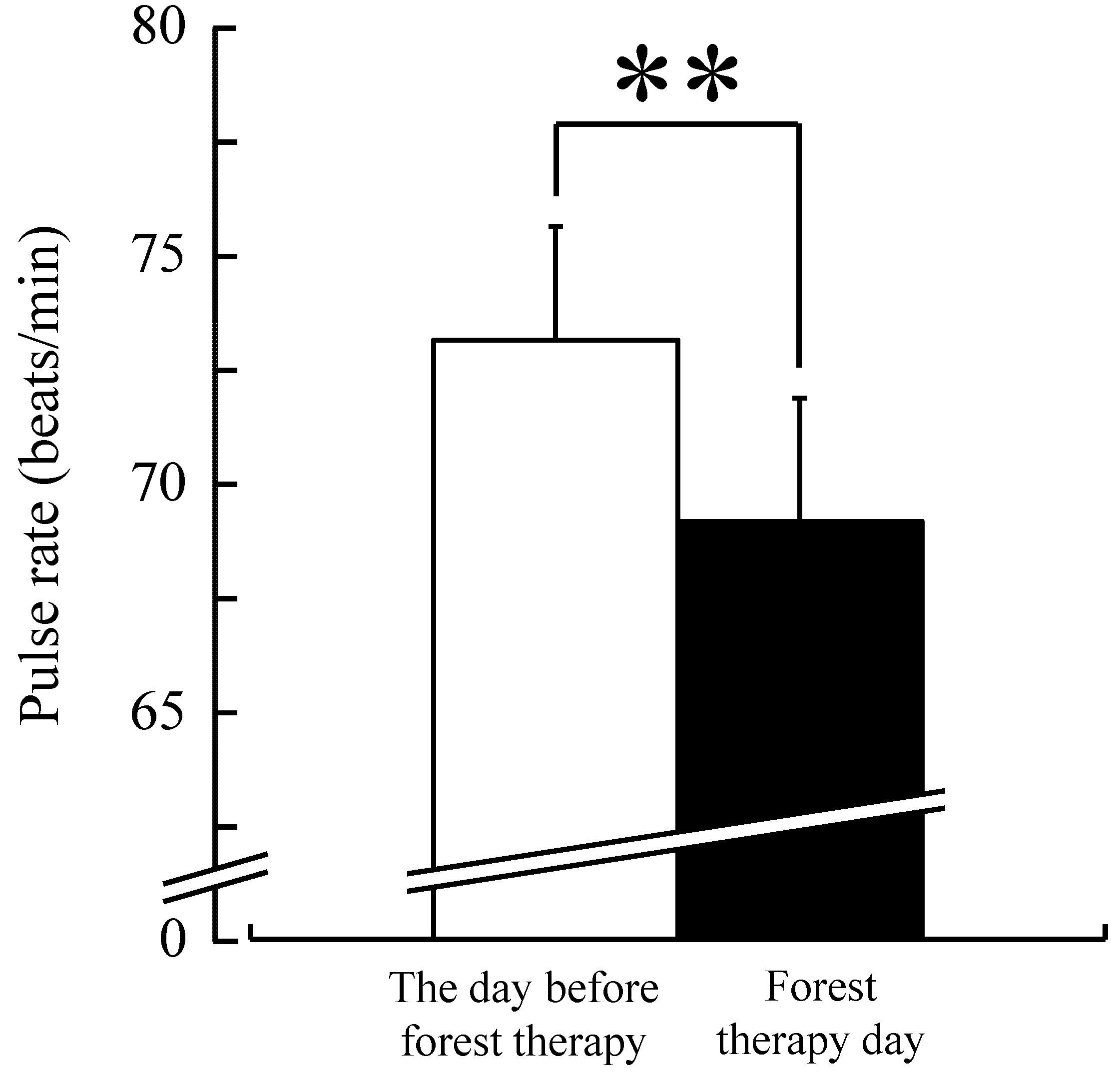
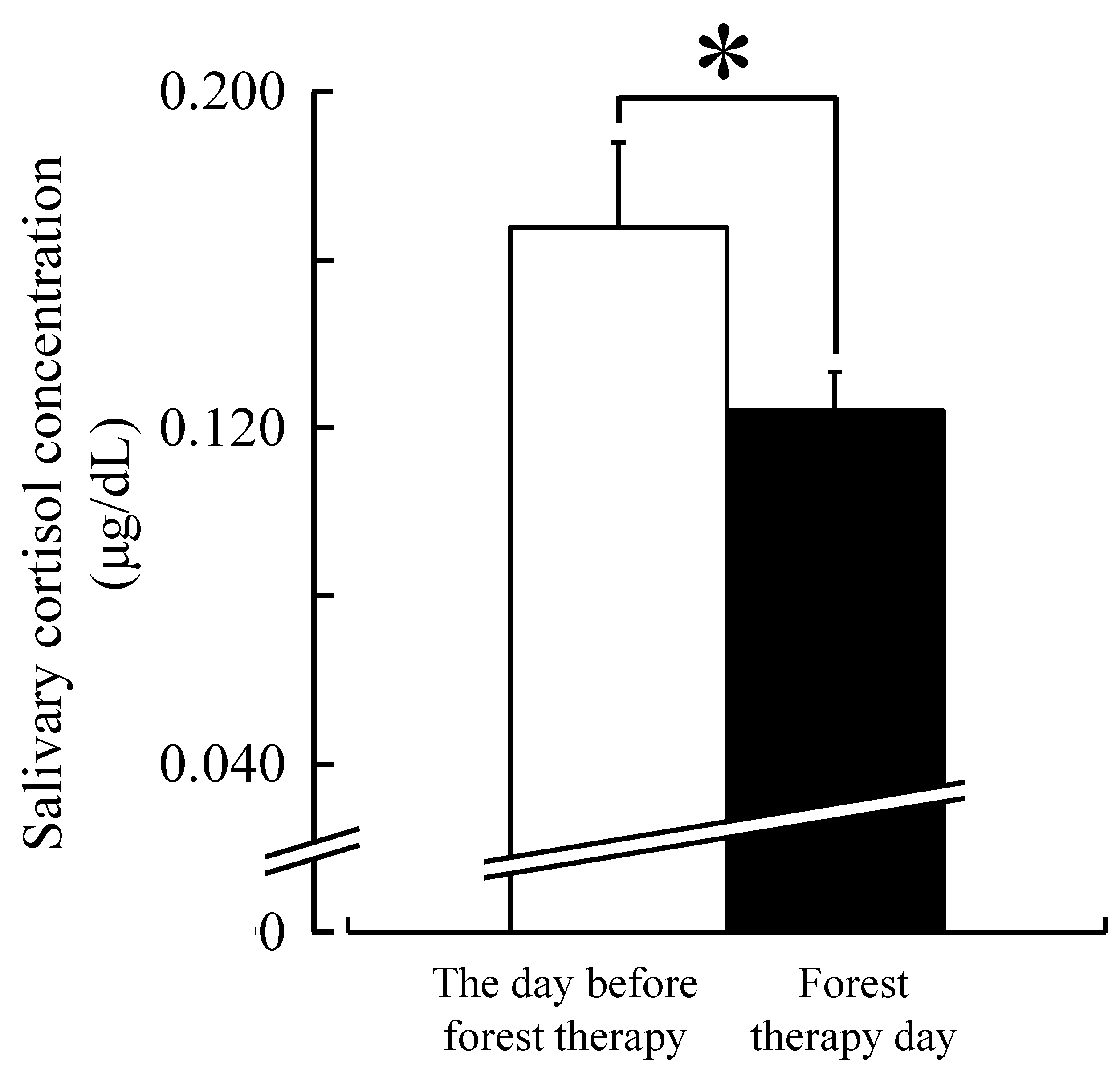
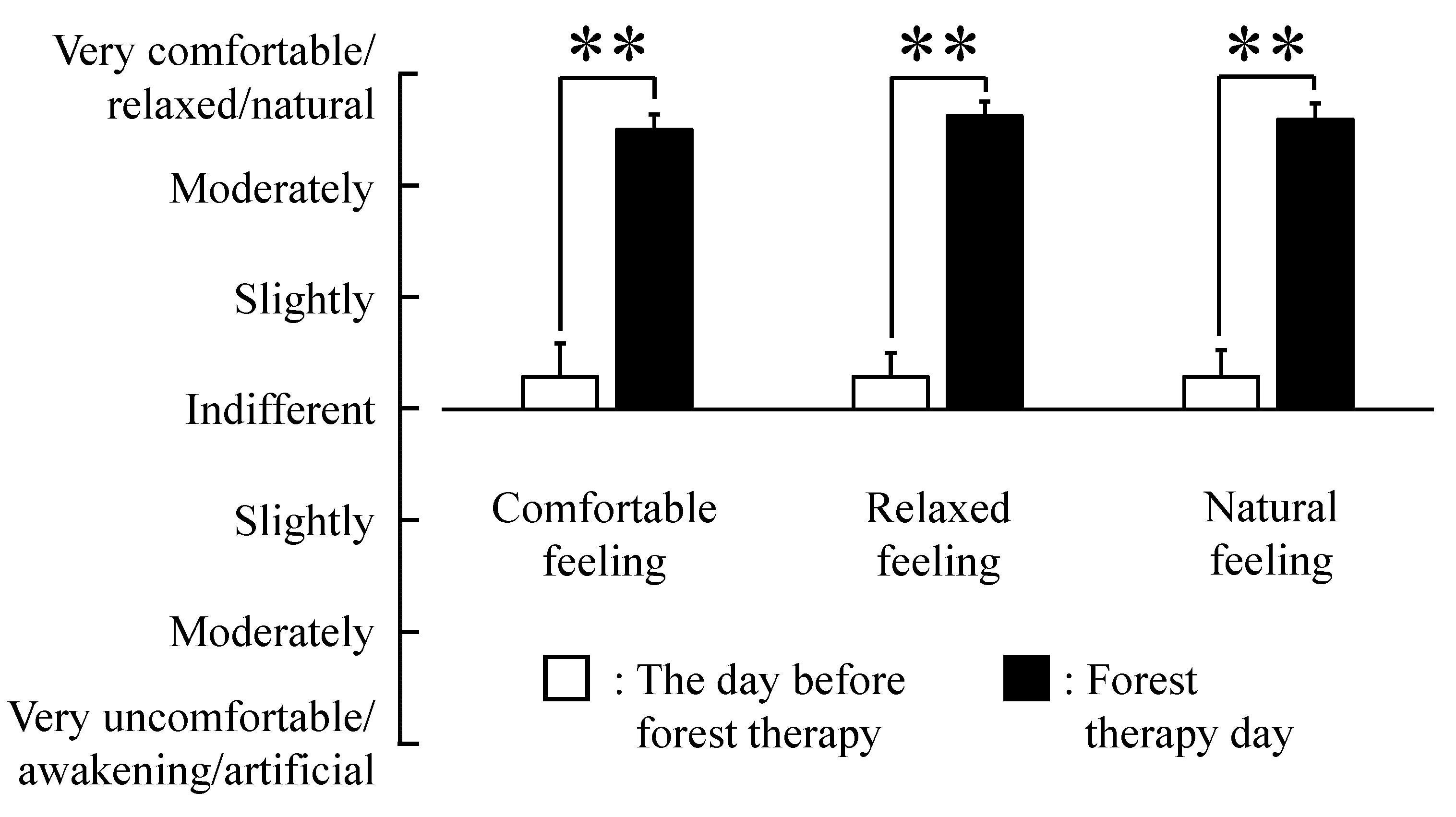
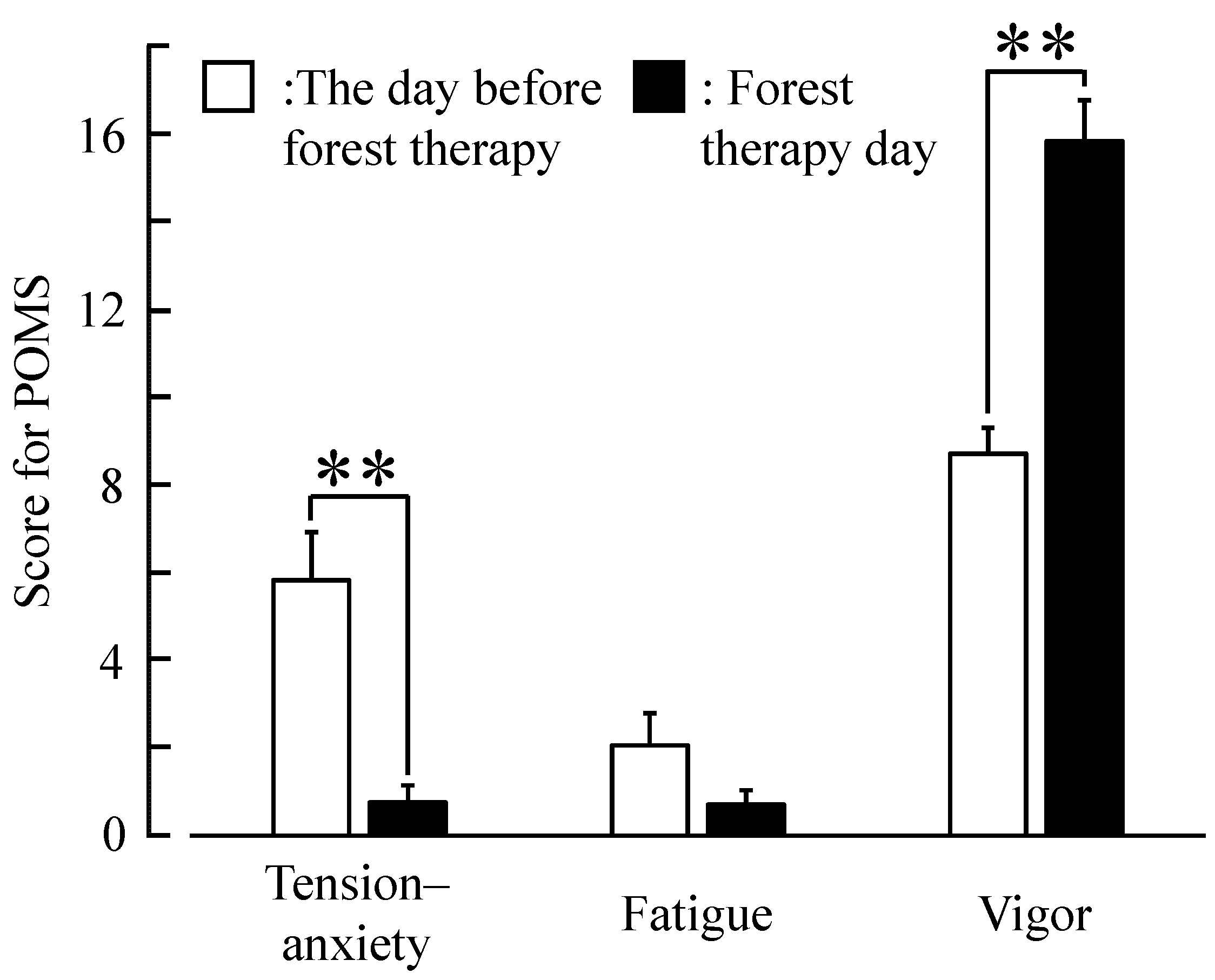
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miyazaki, Y.; Ikei, H.; Song, C. Forest medicine research in Japan. Jpn. J. Hyg. 2014, 69, 123–135. (In Japanese) [Google Scholar] [CrossRef]
- Lee, J.; Park, B.J.; Tsunetsugu, Y.; Kagawa, T.; Miyazaki, Y. The restorative effects of viewing real forest landscapes: Based on a comparison with urban landscapes. Scand. J. Forest. Res. 2009, 24, 227–234. [Google Scholar]
- Tsunetsugu, Y.; Park, B.J.; Ishii, H.; Hirano, H.; Kagawa, T.; Miyazaki, Y. Physiological effects of “Shinrin-yoku” (taking in the atmosphere of the forest) in an old-growth broadleaf forest in Yamagata prefecture, Japan. J. Physiol. Anthropol. 2007, 26, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Tsunetsugu, Y.; Kasetani, T.; Kagawa, T.; Miyazaki, Y. The physiological effects of Shinrin-yoku (taking in the forest atmosphere or forest bathing): Evidence from field experiments in 24 forests across Japan. Environ. Health. Prev. Med. 2010, 15, 18–26. [Google Scholar] [PubMed]
- Park, B.J.; Tsunetsugu, Y.; Lee, J.; Kagawa, T.; Miyazaki, Y. Effect of the forest environment on physiological relaxation using the results of field tests at 35 sites throughout Japan. In Forest Medicine; Li, Q., Ed.; Nova Science Publishers: New York, NY, USA, 2011; pp. 55–65. [Google Scholar]
- Park, B.J.; Kasetani, T.; Morikawa, T.; Tsunetsugu, Y.; Kagawa, T.; Miyazaki, Y. Physiological effects of forest recreation in a young conifer forest in Hinokage Town, Japan. Silva Fennica 2009, 43, 291–301. [Google Scholar] [CrossRef]
- Park, B.J.; Tsunetsugu, Y.; Ishii, H.; Furuhashi, S.; Hirano, H.; Kagawa, T.; Miyazaki, Y. Physiological effects of Shinrin-yoku (taking in the atmosphere of the forest) in a mixed forest in Shinano Town, Japan. Scand. J. Forest. Res. 2008, 23, 278–283. [Google Scholar] [CrossRef]
- Lee, J.; Park, B.J.; Tsunetsugu, Y.; Ohira, T.; Kagawa, T.; Miyazaki, Y. Effect of forest bathing on physiological and psychological responses in young Japanese male subjects. Public Health 2011, 125, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Tsunetsugu, Y.; Takayama, N.; Park, B.J.; Li, Q.; Song, C.; Komatsu, M.; Ikei, H.; Tyrväinen, L.; Kagawa, T.; Miyazaki, Y. Influence of forest therapy on cardiovascular relaxation in young adults. Evid. Based. Complement. Alternat. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Tsunetsugu, Y.; Lee, J.; Park, B.J.; Tyrväinen, L.; Kagawa, T.; Miyazaki, Y. Physiological and psychological effects of viewing urban forest landscapes assessed by multiple measurements. Landsc. Urban Plan. 2013, 113, 90–93. [Google Scholar]
- Tsunetsugu, Y.; Park, B.J.; Miyazaki, Y. Trends in research related to “Shinrin-yoku” (taking in the forest atmosphere or forest bathing) in Japan. Environ. Health. Prev. Med. 2010, 15, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Tsunetsugu, Y.; Kasetani, T.; Hirano, H.; Kagawa, T.; Sato, M.; Miyazaki, Y. Physiological effects of Shinrin-yoku (taking in the atmosphere of the forest)—Using salivary cortisol and cerebral activity as indicators. J. Physiol. Anthropol. 2007, 26, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Bratman, G.N.; Hamilton, J.P.; Hahn, K.S.; Daily, G.C.; Gross, J.J. Nature experience reduces rumination and subgenual prefrontal cortex activation. Proc Natl Acad Sci USA 2015, 112, 8567–8572. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Morimoto, K.; Nakadai, A.; Inagaki, H.; Katsumata, M.; Shimizu, T.; Hirata, Y.; Hirata, K.; Suzuki, H.; Miyazaki, Y.; et al. Forest bathing enhances human natural killer activity and expression of anti-cancer proteins. Int. J. Immunopathol. Pharmacol. 2007, 20, 3–8. [Google Scholar] [PubMed]
- Li, Q.; Morimoto, K.; Kobayashi, M.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Suzuki, H.; Li, Y.J.; Wakayama, Y.; et al. Visiting a forest, but not a city, increases human natural killer activity and expression of anti-cancer proteins. Int. J. Immunopathol. Pharmacol. 2008, 21, 117–127. [Google Scholar] [PubMed]
- Li, Q.; Morimoto, K.; Kobayashi, M.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Shimizu, T.; Li, Y.J.; Wakayama, Y. A forest bathing trip increases human natural killer activity and expression of anti-cancer proteins in female subjects. J. Biol. Regul. Homeost. Agents 2008, 22, 45–55. [Google Scholar] [PubMed]
- Li, Q.; Kobayashi, M.; Inagaki, H.; Hirata, Y.; Li, Y.J.; Hirata, K.; Shimizu, T.; Suzuki, H.; Katsumata, M.; Wakayama, Y.; et al. A day trip to a forest park increases human natural killer activity and the expression of anti-cancer proteins in male subjects. J. Bio. Regul. Homeost. 2010, 24, 157–165. [Google Scholar]
- Takayama, N.; Korpela, K.; Lee, J.; Morikawa, T.; Tsunetsugu, Y.; Park, B.J.; Li, Q.; Tyrväinen, L.; Miyazaki, Y.; Kagawa, T. Emotional, restorative and vitalizing effects of forest and urban environments at four sites in Japan. Int. J. Environ. Res. Public Health 2014, 11, 7207–7230. [Google Scholar] [PubMed]
- Park, B.J.; Furuya, K.; Kasetani, T.; Takayama, N.; Kagawa, T.; Miyazaki, Y. Relationship between psychological responses and physical environment in forest settings. Landsc. Urban Plan. 2011, 102, 24–32. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Kobayashi, M.; Miura, T.; Taue, M.; Kagawa, T.; Li, Q.; Kumeda, S.; Imai, M.; Miyazaki, Y. Effect of forest walking on autonomic nervous system activity in middle-aged hypertensive individuals. Int. J. Environ. Res. Public Health 2015, 12, 2687–2699. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, H.; Ikei, H.; Song, C.; Kobayashi, M.; Takamatsu, A.; Miura, T.; Taue, M.; Kagawa, T.; Li, Q.; Kumeda, S.; et al. Physiological and psychological effects of forest therapy on middle-age males with high-normal blood pressure. Int. J. Environ. Res. Public Health 2015, 12, 2532–2542. [Google Scholar] [CrossRef] [PubMed]
- NHLBI Obesity Education Initiative. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults; NHLBI Obesity Education Initiative Expert Panel on the Identification, Evaluation, and Treatment of Obesity in Adults (US) National Heart, Lung, and Blood Institute: Baltimore, MD, USA, 1998; p. 228. [Google Scholar]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic; World Health Organization: Geneva, Switzerland, 2000; p. 265. [Google Scholar]
- World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar]
- Osgood, C.E.; Suchi, G.J.; Tannenbaum, P. The Measurement of Meaning; University of Illinois Press: Urbana, IL, USA, 1957. [Google Scholar]
- McNair, D.M.; Lorr, M.; Droppleman, L. Profile of Mood States Manual; Educational and Industrial Testing Service: San Diego, CA, USA, 1992. [Google Scholar]
- Frankenhauser, M. Experimental approach to the study of catecholamines and emotion. In Emotions, Their Parameters and Measurement; Levi, L., Ed.; Raven Press: New York, NY, USA, 1975; p. 209. [Google Scholar]
- Kirschbaum, C.; Hellhammer, D.H. Salivary cortisol in psychoneuroendocrine research: Recent developments and applications. Psychoneuroendocrinology 1994, 19, 313–333. [Google Scholar] [CrossRef]
- Poll, E.M.; Kreitschmann-Andermahr, I.; Langejuergen, Y.; Stanzel, S.; Gilsbach, J.M.; Gressner, A.; Yagmur, E. Saliva collection method affects predictability of serum cortisol. Clin. Chim. Acta. 2007, 382, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, D.B. Oral Bioscience; Churchill Livingstone: London, UK, 1999; p. 136. [Google Scholar]
- Kiernan, M.; Winkleby, M.A. Identifying patients for weight-loss treatment: An empirical evaluation of the NHLBI obesity education initiative expert panel treatment recommendations. Arch. Intern. Med. 2000, 160, 2169–2176. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Ikei, H.; Miyazaki, Y. Elucidation of a physiological adjustment effect in a forest environment: A pilot study. Int. J. Environ. Res. Public Health 2015, 12, 4247–4255. [Google Scholar] [CrossRef] [PubMed]
- Duchesne, A.; Pruessner, J.C. Association between subjective and cortisol stress response depends on the menstrual cycle phase. Psychoneuroendocrinology 2013, 38, 3155–3159. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Shirakawa, T. Characteristics of perceived stress and salivary levels of secretory immunoglobulin A and cortisol in Japanese women with premenstrual syndrome. Nurs. Midwifery Stud. 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.M.; Ziegler, M.G.; Patwardhan, A.R.; Ott, J.B.; Kim, C.S.; Leonelli, F.M.; Knapp, C.F. Gender differences in autonomic cardiovascular regulation: Spectral, hormonal, and hemodynamic indexes. J. Appl. Physiol. 2001, 91, 2611–2618. [Google Scholar] [PubMed]
- Earnest, C.P.; Lavie, C.J.; Blair, S.N.; Church, T.S. Heart rate variability characteristics in sedentary postmenopausal women following six months of exercise training: The DREW study. PLoS ONE 2008, 3. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochiai, H.; Ikei, H.; Song, C.; Kobayashi, M.; Miura, T.; Kagawa, T.; Li, Q.; Kumeda, S.; Imai, M.; Miyazaki, Y. Physiological and Psychological Effects of a Forest Therapy Program on Middle-Aged Females. Int. J. Environ. Res. Public Health 2015, 12, 15222-15232. https://doi.org/10.3390/ijerph121214984
Ochiai H, Ikei H, Song C, Kobayashi M, Miura T, Kagawa T, Li Q, Kumeda S, Imai M, Miyazaki Y. Physiological and Psychological Effects of a Forest Therapy Program on Middle-Aged Females. International Journal of Environmental Research and Public Health. 2015; 12(12):15222-15232. https://doi.org/10.3390/ijerph121214984
Chicago/Turabian StyleOchiai, Hiroko, Harumi Ikei, Chorong Song, Maiko Kobayashi, Takashi Miura, Takahide Kagawa, Qing Li, Shigeyoshi Kumeda, Michiko Imai, and Yoshifumi Miyazaki. 2015. "Physiological and Psychological Effects of a Forest Therapy Program on Middle-Aged Females" International Journal of Environmental Research and Public Health 12, no. 12: 15222-15232. https://doi.org/10.3390/ijerph121214984







